research use only
Dexlansoprazole Proton Pump inhibitor
Cat.No.S4099
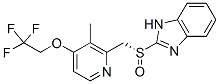
Chemical Structure
Molecular Weight: 369.36
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Sodium Channel Potassium Channel GABA Receptor TRP Channel ATPase |
|---|---|
| Other Proton Pump Inhibitors | Bafilomycin A1 (Baf-A1) Ilaprazole Tenatoprazole Ilaprazole sodium Revaprazan Hydrochloride PAβN dihydrochloride PF-3716556 Ufiprazole |
Solubility
|
In vitro |
DMSO
: 74 mg/mL
(200.34 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 369.36 | Formula | C16H14F3N3O2S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 138530-94-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | T 168390, TAK 390,(R)-Lansoprazole | Smiles | CC1=C(C=CN=C1CS(=O)C2=NC3=CC=CC=C3N2)OCC(F)(F)F | ||
Mechanism of Action
| Features |
A modified release formulation of an enantiomer of lansoprazole.
|
|---|---|
| Targets/IC50/Ki |
Proton pump
H(+),K(+)-ATPase
|
| In vitro |
Dexlansoprazole, constitutes >80% of circulating drug after oral administration of lansoprazole, provides lower clearance and 5-fold greater systemic exposure than the S-enantiomer following oral administration of lansoprazole. This compound MR is a modified release formulation of dexlansoprazole, which employs a novel Dual Delayed Release (DDR) technology that delivers the drug in two discrete phases of release, thereby inhibiting newly activated proton pumps that turn over following initial PPI inactivation of H+,K+-ATPase. This formulation maintains plasma drug concentrations above the threshold level longer than lansoprazole at all doses, resulting in an optimized drug exposure-intragastric pH relationship. It selectively suppresses gastric acid secretion by direct inhibition of the H+K+-ATPase proton pump in the gastric parietal cell, inhibition of this cell membrane enzyme ultimately blocks the final step in acid production. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02442752 | Withdrawn | Pediatric Gastroesophageal Reflux Disease |
Takeda |
June 15 2025 | Phase 1 |
| NCT03316976 | Completed | Healthy Volunteers |
Takeda |
November 22 2017 | Phase 1 |
| NCT03079050 | Completed | GERD|Proton Pump Inhibitor |
American University of Beirut Medical Center|Takeda |
February 27 2017 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































