research use only
Boceprevir HCV Protease inhibitor
Cat.No.S3733
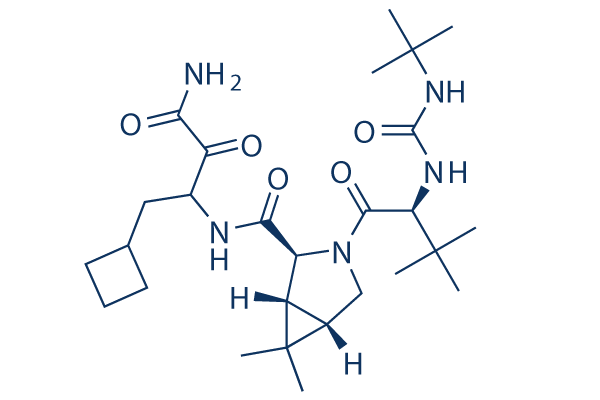
Chemical Structure
Molecular Weight: 519.68
Quality Control
| Related Targets | HDAC Caspase Proteasome Secretase MMP Cysteine Protease Tyrosinase DPP HIV Protease Serine Protease |
|---|---|
| Other HCV Protease Inhibitors | Danoprevir Lomibuvir (VX-222) Asunaprevir Tizoxanide PSI-6206 (GS-331007) 2'-C-Methylcytidine Tegobuvir Herba taxilli Extract Mecarbinate |
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(192.42 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 519.68 | Formula | C27H45N5O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 394730-60-0 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | EBP 520, SCH 503034 | Smiles | CC1(C2C1C(N(C2)C(=O)C(C(C)(C)C)NC(=O)NC(C)(C)C)C(=O)NC(CC3CCC3)C(=O)C(=O)N)C | ||
Mechanism of Action
| Targets/IC50/Ki |
NS3/4A protease
14 nM(Ki)
|
|---|---|
| In vitro |
Treatment with NLRP3 Inflammasome Inhibitor I significantly limits IL-1β release after LPS and ATP challenge. NLRP3 Inflammasome Inhibitor I is not a caspase-1 inhibitor.
|
| In vivo |
The small molecule NLRP3 Inflammasome Inhibitor I, an intermediate substrate in the glyburide synthesis free of the cyclohexylurea moiety, inhibits the formation of the NLRP3 inflammasome in cardiomyocytes and limits the infarct size following myocardial ischemia/reperfusion in the mouse, without affecting glucose metabolism.
|
References |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01909401 | Completed | Liver Transplantation |
Indiana University|Merck Sharp & Dohme LLC |
June 2013 | Early Phase 1 |
| NCT01821625 | Terminated | Thrombocytopenia|Hepatitis C |
University of Texas Southwestern Medical Center|GlaxoSmithKline |
April 2013 | Phase 2|Phase 3 |
| NCT01499498 | Completed | Hepatitis C |
Imperial College London |
December 2012 | Phase 1 |
| NCT01663922 | Completed | Hepatitis C |
St Stephens Aids Trust|University of Liverpool|University of Turin Italy |
August 2012 | Phase 1 |
| NCT01657552 | Completed | Thrombocytopaenia |
GlaxoSmithKline |
August 1 2012 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































