research use only
BMS-986142 BTK inhibitor
Cat.No.S8381
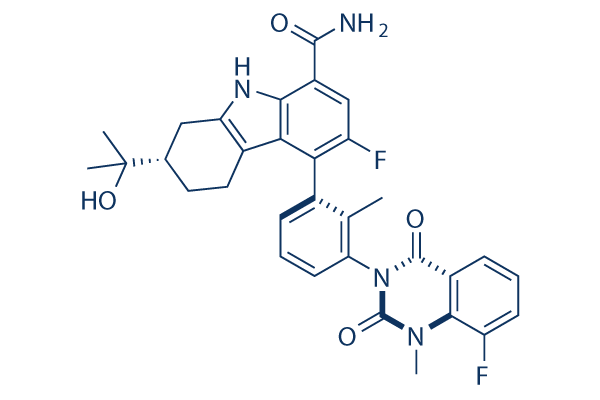
Chemical Structure
Molecular Weight: 572.60
Quality Control
| Related Targets | EGFR VEGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 |
|---|---|
| Other BTK Inhibitors | Catadegbrutinib (BGB-16673) Spebrutinib (AVL-292) tirabrutinib(ONO-4059) hydrochloride LFM-A13 CGI1746 Evobrutinib BMS-935177 CNX-774 Branebrutinib (BMS-986195) Fenebrutinib (GDC-0853) |
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(174.64 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 572.60 | Formula | C32H30F2N4O4 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 1643368-58-4 | -- | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=C(C=CC=C1N2C(=O)C3=C(C(=CC=C3)F)N(C2=O)C)C4=C(C=C(C5=C4C6=C(N5)CC(CC6)C(C)(C)O)C(=O)N)F | ||
Mechanism of Action
| Targets/IC50/Ki |
BTK
(Cell-free assay) 0.5 nM
|
|---|---|
| In vitro |
In B cells stimulated through the B cell receptor (BCR), BMS-986142 potently inhibits signaling and functional end points, including calcium flux (IC50 = 9 nM), production of cytokines, proliferation, and surface expression of the costimulatory molecule CD86 (IC50 = 3−4 nM). In T cells, ITK-catalyzed phosphorylation of PLCγ1 stimulated through the T cell receptor (TCR) is at least 45-fold less potently inhibited by this compound than BTK-dependent signaling end points in B cells, as expected based on the 30-fold selectivity for BTK over ITK in the enzymatic assays. |
| Kinase Assay |
Human Recombinant BTK Enzyme Assay
|
|
To V-bottom 384-well plates were added test compounds, human recombinant BTK (1 nM), fluoresceinated peptide (1.5 μM), ATP (20 μM), and assay buffer (20 mM HEPES at pH 7.4, 10 mM MgCl2, 0.015% Brij 35 surfactant, and 4 mM DTT in 1.6% DMSO), with a final volume of 30 μL. After incubating at room temperature for 60 min, the reaction was terminated by adding 45 μL of 35 mM EDTA to each sample. The reaction mixture was analyzed by electrophoretic separation of the fluorescent substrate and phosphorylated product. Inhibition data were calculated by comparison to control reactions with no enzyme (for 100% inhibition) and controls with no inhibitor (for 0% inhibition). Dose−response curves were generated to determine the concentration required for inhibiting 50% of BTK activity (IC50). This compound was dissolved at 10 mM in DMSO and evaluated at 11 concentrations.
|
|
| In vivo |
In multispecies pharmacokinetic studies, the absolute oral bioavailability of BMS-986142 is 93% in mice, 67% in rats, 33% in cynomolgus monkeys, and 100% in dogs. The total body plasma clearance of this compound is low in all species. The large steady-state volume of distribution observed is indicative of extravascular distribution, in spite of the high protein binding. However, brain penetration is very low in rats (<5% of plasma concentration). This compound blocks the increase in severe proteinuria in a female NZB/W lupus-prone mouse model in vivo. Treatment with this chemical provides robust protection against tubulo-interstitial and glomerular nephritis, as well as inflammatory infiltration. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02638948 | Completed | Rheumatoid Arthritis |
Bristol-Myers Squibb |
February 16 2016 | Phase 2 |
| NCT02257151 | Completed | Healthy Adult |
Bristol-Myers Squibb |
September 2014 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































