research use only
VX-809 (Lumacaftor) CFTR modulator
Cat.No.S1565

Chemical Structure
Molecular Weight: 452.41
Quality Control
| Related Targets | CRM1 CD markers AChR Calcium Channel Sodium Channel Potassium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other CFTR Inhibitors | Tezacaftor (VX-661) VX-445 (Elexacaftor) Vanzacaftor (VX-121) CFTRinh-172 GLPG1837 Galicaftor (ABBV-2222) GlyH-101 FDL169 IOWH032 PPQ-102 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| CFBE41o | Corrector assay | 24 hrs | Corrector activity at CFTR F508-del mutant (unknown origin) expressed in human CFBE41o cells harboring HS-YFP preincubated for 24 hrs followed by forskolin/genistein stimulation for 30 mins by fluorescence assay, EC50 = 2.5704 μM. | 29272749 | ||
| FRT | Corrector assay | 25 mins | Corrector activity at human CFTR F508 deletion mutant expressed in FRT cells incubated for 25 mins with forskolin by YFP-based fluorescence analysis relative to control, EC50 = 2.6 μM. | 26561003 | ||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-12 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| fibroblast cells | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for control Hh wild type fibroblast cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh30 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for RD cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| CFBE41o | Corrector assay | 1 uM | 24 hrs | Corrector activity at CFTR F508-del mutant (unknown origin) expressed in human CFBE41o cells harboring HS-YFP assessed as increase in matured protein levels at cell surface at 1 uM after 24 hrs by electrophoretic mobility assay | 29272749 | |
| HBE | Corrector assay | Corrector activity at CFTR F508del/F508del mutant in primary HBE cells assessed as increase in chloride ion current across apical membrane measured 18 to 24 hrs post compound treatment on basolateral side of cells in presence of channel potentiator GLPG18 | 29251932 | |||
| CFBE41o | Corrector assay | Corrector activity at CFTR F508del mutant (unknown origin) expressed in human CFBE41o cells assessed as increase in fully glycosylated protein by western blot analysis | 26041577 | |||
| CFBE41o | Corrector assay | 1 uM | 24 hrs | Corrector activity at CFTR F508del mutant (unknown origin) expressed in human CFBE41o cells assessed as increase in size of cAMP-dependent current at 1 uM after 24 hrs measured at +100 mV by whole cell patch clamp assay | 26041577 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 90 mg/mL
(198.93 mM)
Ethanol : 13 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 452.41 | Formula | C24H18F2N2O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 936727-05-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | VRT 826809 | Smiles | CC1=C(N=C(C=C1)NC(=O)C2(CC2)C3=CC4=C(C=C3)OC(O4)(F)F)C5=CC(=CC=C5)C(=O)O | ||
Mechanism of Action
| Features |
Higher specificity and efficacy relative to other CFTR defect drugs.
|
|---|---|
| Targets/IC50/Ki |
F508del-CFTR
(Fisher rat thyroid cells) 0.1 μM(EC50)
|
| In vitro |
Lumacaftor (VX-809) acts at the level of the ER to allow a fraction of the F508del-CFTR to adopt a properly folded form, to exit the ER and mobilize to the cell surface for normal functioning. In Fischer rat thyroid (FRT) cells expressing F508del-CFTR, this compound treatment significantly improves F508del-CFTR maturation by 7.1 fold with an EC50 of 0.1 μM, and enhances F508del-CFTR-mediated chloride transport by approximately 5 fold with EC50 of 0.5 μM, while VRT-768 has higher EC50 values of 7.9 μM and 16 μM, respectively. In HEK-293 cells expressing F508del-CFTR, it (3 μM) increases F508del-CFTR exit from the ER by 6 fold, reaching levels comparable to 34% of CFTR. In primary human bronchial epithelial (HBE) cells with F508del-CFTR mutation, the agent increases CFTR maturation and enhances chloride secretion with EC50 of 350 nM and 81 nM, respectively, more efficacious than Corr-4a and VRT-325. F508del-CFTR corrected by it exhibits single-channel open probability of 0.39 similar to normal CFTR of 0.40. Unlike VX-770, it is not a CFTR potentiator, as acute addition has no effect on F508del-CFTR function. In contrast to VRT-325 and Corr-4a, it does not improve the processing of the normal or mutant forms of hERG or P-gp, as well as other disease-causing mislocalized proteins, including α1-antitrypsin Z mutant (E342K-α1-AT) or N370S-β-glucosidase, suggesting that it is specific for CFTR. It in combination with VRT-325 or Corr-4a has additive effect on CFTR-mediated chloride transport in cultured F508del-HBE. |
| Kinase Assay |
F508del-CFTR maturation
|
|
FRT cells stably expressing F508del-CFTR are treated with increasing concentrations of Lumacaftor (VX-809) for 48 hours. After incubation, cells are harvested in ice-cold D-PBS solution (without calcium and magnesium) and pelleted at 1,000 × g at 4 °C. Cell pellets are lysed in 1% Nonidet P-40, 0.5% sodium deoxycholate, 200 mM NaCl, 10 mM Tris, pH 7.8, and 1 mM EDTA plus protease inhibitor mixture (1:250) for 30 minutes on ice. Lysates are spun for 10 minutes at 10,000 × g at 4 °C to pellet nuclei and insoluble material. Approximately 12 μg total protein is heated in Laemmli buffer with 5% β-mercaptoethanol at 37 °C for 5 minutes and loaded onto a 3% to 8% Tris-acetate gel. The gel is transferred to nitrocellulose and processed for Western blotting by using monoclonal CFTR antibody or polyclonal to GAPDH. Blots are developed by enhanced chemiluminescence. Quantification of the relative amounts of bands C and GAPDH is performed by using NIH ImageJ analysis of scanned films.
|
|
| In vivo |
Lumacaftor (VX-809; VRT 826809) is a CFTR modulator that corrects the folding and trafficking of CFTR protein. |
References |
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Immunofluorescence | CFTR / USP13 Cell surface kAE1 / kAE1 |
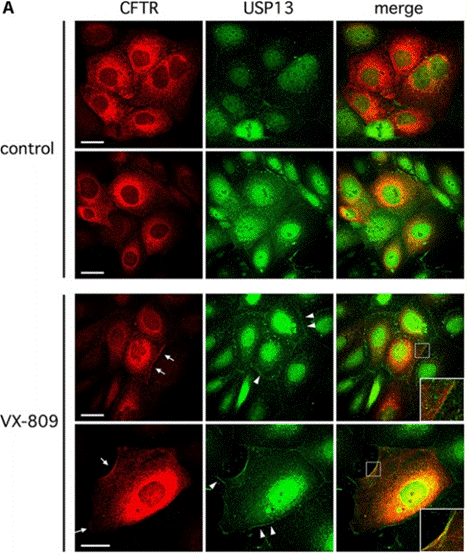
|
30618756 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03512119 | Completed | Cystic Fibrosis Homozygous for Phe 508 Del CFTR|Glucose Intolerance or Newly Diagnosis Diabetes |
University Hospital Strasbourg France |
February 11 2016 | -- |
| NCT02589236 | Completed | Cystic Fibrosis |
Nivalis Therapeutics Inc.|Medidata Solutions |
November 2015 | Phase 2 |
| NCT02514473 | Completed | Cystic Fibrosis |
Vertex Pharmaceuticals Incorporated |
July 2015 | Phase 3 |
| NCT01899105 | Completed | Cystic Fibrosis |
Vertex Pharmaceuticals Incorporated |
July 2013 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































