research use only
Lansoprazole Proton Pump inhibitor
Cat.No.S1354
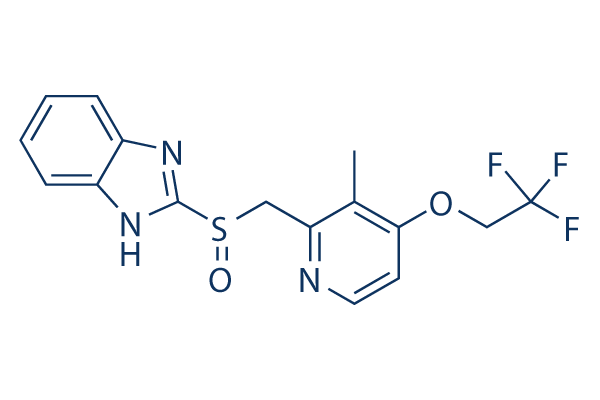
Chemical Structure
Molecular Weight: 369.36
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Sodium Channel Potassium Channel GABA Receptor TRP Channel ATPase |
|---|---|
| Other Proton Pump Inhibitors | Bafilomycin A1 (Baf-A1) Ilaprazole Tenatoprazole Ilaprazole sodium Revaprazan Hydrochloride PF-3716556 Ufiprazole |
Solubility
|
In vitro |
DMSO
: 74 mg/mL
(200.34 mM)
Ethanol : 14 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 369.36 | Formula | C16H14F3N3O2S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 103577-45-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | A-65006, AG-1749 | Smiles | CC1=C(C=CN=C1CS(=O)C2=NC3=CC=CC=C3N2)OCC(F)(F)F | ||
Mechanism of Action
| Targets/IC50/Ki |
Proton pump
H(+),K(+)-ATPase
|
|---|---|
| In vitro |
Lansoprazole inhibits gastric acid secretion via inhibition of gastric hydrogen/potassium adenosine triphosphatase (H+,K(+)-ATPase), an enzyme of the gastric parietal cell membrane that forms part of the proton pump that performs the final step in the acid secretory process. This compound binds covalently to parietal cell H+,K(+)-ATPase, rendering it nonfunctional and inhibiting the secretion of gastric acid. It is a strong anti-secretory agent that acts on gastric H+/K+-adenosine triphosphatase (H+/K+ ATPase) of parietal cells. This chemical inhibits the increased expression of vascular adhesion molecules, the activation of neutrophils, and the production of pro-inflammatory cytokines from activated endothelial cells. It induces several genes, including phase II detoxifying enzyme (NADPH-ubiquinone oxidoreductase, glutathione S-transferase) and antioxidant stress proteins (HO-1, thioredoxin reductase, and superoxide dismutase) in gastric epithelial cells. This compound significantly inhibits the production of CINC-1 from stimulated RGM-1 cells with IL-1β. It up-regulates HO-1 expression throughout Nrf2 in rat gastric epithelial cells, and the up-regulated HO-1 has anti-inflammatory effects.
|
| In vivo |
Lansoprazole inhibits acute inflammatory reactions as well as intestinal mucosal injuries induced by ischemia-reperfusion or indomethacin administration in rats. This compound significantly inhibits intestinal injuries induced by ischemia-reperfusion or indomethacin. This compound administered exogenously prevents the small intestine against ischemia-reperfusion or indomethacin-induced damage, the action being dependent on its anti-inflammatory and anti-oxidative responses.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02442752 | Withdrawn | Pediatric Gastroesophageal Reflux Disease |
Takeda |
June 15 2025 | Phase 1 |
| NCT03316976 | Completed | Healthy Volunteers |
Takeda |
November 22 2017 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































