research use only
Ganciclovir (BW 759) Antiviral Agent
Cat.No.S1878
Chemical Structure
Molecular Weight: 255.23
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV |
|---|---|
| Other CMV Inhibitors | Valaciclovir HCl Bisindolylmaleimide IV Maribavir(1263W94, BW1263W94,GW257406X,GW 1263) Brivudine (BVDU) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| E6SM cell lines | Function assay | Effective concentration required to inhibit Herpes simplex virus-2 (HSV-2) induced cytopathicity by 50% in E6SM cell lines, EC50=1.2 nM | 11495586 | |||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK, IC50=0.0019μM. | 17181158 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 G in HEL cells assessed as reduction of virus-induced cytopathogenicity, MIC=0.0064μM. | 17672445 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.01μM. | 21232828 | |||
| HEL | Antiviral assay | Antiviral activity against HSV-2 G infected in HEL cells assessed as inhibition of viral-induced cytopathogenicity, EC50=0.01μM. | 23795238 | |||
| HEL | Antiviral assay | Antiviral activity against HSV-1 KOS infected in HEL cells assessed as inhibition of viral-induced cytopathogenicity, EC50=0.01μM. | 23795238 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against Herpes simplex virus 1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by microscopic method, EC50=0.01μM. | 30286952 | ||
| HEL | Antiviral assay | 3 days | Antiviral activity against Human herpesvirus 2 strain G infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by microscopic method, EC50=0.01μM. | 30286952 | ||
| HEL | Antiviral assay | Antiviral activity against HSV2 G infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.013μM. | 23099097 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.015μM. | 23099097 | |||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 2.5 uM 1-[6-(triphenylmethoxy)hexyl]thymine, IC50=0.018μM. | 17181158 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS in HEL cells, EC50=0.019μM. | 17181162 | |||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as inhibition of plaque formation measured after 2 to 3 days post infection, EC50=0.019μM. | 29550734 | ||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS in human HEL cells assessed as protection against virus-induced cytopathicity, EC50=0.02μM. | 19281225 | |||
| HEL | Antiviral assay | 4 days | Antiviral activity against HSV1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathogenicity after 4 days, EC50=0.02μM. | 22459876 | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=0.02μM. | 26001344 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.02μM. | 29880251 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.02μM. | 29880251 | |||
| Vero | Antiviral assay | 72 hrs | Antiviral activity against HSV-1 infected in African green monkey Vero cells assessed as inhibition of GFP expression after 72 hrs by GFP-based fluorescence microscopic analysis, EC50=0.025μM. | 31503476 | ||
| HEL | Antiviral assay | Antiviral activity against Human herpesvirus 2 G infected in HEL cells assessed as protection against virus-induced cytopathicity, EC50=0.027μM. | 21565516 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS infected in HEL cells by viral CPE assay, EC50=0.027μM. | 22858222 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 G infected in HEL cells by viral CPE assay, EC50=0.029μM. | 22858222 | |||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against acyclovir-sensitive Herpes simplex virus 1 KOS infected in HEL cells assessed as protection from virus-induced cytopathogenicity after 2 to 3 days by MTT assay, EC50=0.03μM. | 20034711 | ||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as protection from virus-induced cytopathogenicity after 2 to 3 days by MTT assay, EC50=0.03μM. | 20034711 | ||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as protection from virus-induced cytopathogenicity after 2 to 3 days by MTT assay, EC50=0.03μM. | 20034711 | ||
| HEL | Antiviral assay | 4 days | Antiviral activity against Herpes simplex virus 1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 4 days by microscopic analysis, EC50=0.03μM. | 21696963 | ||
| HEL | Antiviral assay | Antiviral activity against Human herpesvirus 1 KOS infected in HEL cells assessed as protection against virus-induced cytopathicity, EC50=0.03μM. | 21565516 | |||
| HEL | Antiviral assay | 4 days | Antiviral activity against Herpes simplex virus-2 G infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 4 days by microscopic analysis, EC50=0.03μM. | 21696963 | ||
| HEL | Antiviral assay | 4 days | Antiviral activity against HSV2 G infected in HEL cells assessed as inhibition of virus-induced cytopathogenicity after 4 days, EC50=0.03μM. | 22459876 | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus-2 G infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=0.03μM. | 23811093 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus-1 KOS infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=0.03μM. | 23811093 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=0.03μM. | 26001344 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as inhibition of virus plaque formation, EC50=0.03μM. | 27639368 | |||
| HELF | Antiviral assay | Antiviral activity against Human herpesvirus 1 KOS strain infected in HELF cells assessed as inhibition of virus-induced cytopathogenicity, EC50=0.03μM. | 30098481 | |||
| HELF | Antiviral assay | Antiviral activity against Human herpesvirus 2 G strain infected in HELF cells assessed as inhibition of virus-induced cytopathogenicity, EC50=0.03μM. | 30098481 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.03μM. | 28829913 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.03μM. | 28757102 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 in HEL cells, EC50=0.032μM. | 17181162 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS in HEL cells assessed as reduction of virus-induced cytopathogenicity, MIC=0.032μM. | 17672445 | |||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as reduction in virus-induced cytopathogenicity, EC50=0.032μM. | 27750154 | |||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 5 uM 1-[6-(triphenylmethoxy)hexyl]thymine, IC50=0.033μM. | 17181158 | |||
| fibroblast cells | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in human foreskin fibroblast cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.04μM. | 19397271 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 G infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.04μM. | 21232828 | |||
| HFF | Antiviral assay | 1 hr | Antiviral activity against Human cytomegalovirus (strain AD169) infected in 1 hr pretreated human HFF cells assessed as reduction in viral-induced cytopathic effect by neutral red uptake assay, EC50=0.04μM. | 21376429 | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.04μM. | 28829913 | |||
| HEL | Antiviral assay | Antiviral activity against wild type Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.04μM. | 28757102 | |||
| Vero | Antiviral assay | 36 hrs | Antiviral activity against HSV 1 infected in African green monkey Vero cells assessed as reduction in plaque formation incubated for 36 hrs by crystal violet staining based assay, EC50=0.04μM. | 32191456 | ||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 10 uM 1-[6-(triphenylmethoxy)hexyl]thymine, IC50=0.041μM. | 17181158 | |||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of plaque formation measured after 2 to 3 days post infection, EC50=0.045μM. | 29550734 | ||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 2.5 uM 1-[6-[1,1-(diphenyl)-1-(4-pyridyl)methoxy]hexyl]thymine, IC50=0.05μM. | 17181158 | |||
| human | Antiviral assay | 7 days | Antiviral activity against HSV2 G infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity after 7 days, EC50=0.05μM. | 21128666 | ||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as inhibition of virus-induced cytopathic effect measured after 2 to 3 days post-infection, EC50=0.051μM. | 29670705 | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 2 G infected in HEL cells assessed as reduction in virus-induced cytopathogenicity, EC50=0.055μM. | 27750154 | |||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect measured after 2 to 3 days post-infection, EC50=0.057μM. | 29670705 | ||
| HEL | Function assay | Compound was tested for anti-viral activity against HSV-1(G) in HEL cells, EC50=0.06μM. | 14643328 | |||
| Hel | Function assay | Effective concentration required to inhibit herpes simplex virus type 1 Kos strain induced cytopathicity in Hel cells, EC50=0.06μM. | 15658858 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS in HEL cells, EC50=0.06μM. | 16392791 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 G in HEL cells, EC50=0.06μM. | 16392791 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against Human herpesvirus 2 strain G infected human HEL cells assessed as reduction of virus-induced cytopathic effect after 3 days by colorimetric formazan-based MTS assay, EC50=0.07μM. | ChEMBL | ||
| HEL | Antiviral assay | Antiviral activity against herpes simplex virus 1 KOS infected in HEL cells assessed as reduction in virus-induced cytopathogenicity by microscopic analysis, EC50=0.07μM. | 30738653 | |||
| HELF | Antiviral assay | 7 days | Antiviral activity against Herpes simplex virus 1 KOS infected in human HELF cells assessed as reduction in virus-induced cytopathogenicity incubated for 7 days by microscopic analysis, EC50=0.07μM. | 31586832 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against HSV1 KOS infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity after 7 days, EC50=0.08μM. | 21128666 | ||
| HEL | Antiviral assay | 3 days | Antiviral activity against Human herpesvirus 2 strain G infected HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by MTT assay, EC50=0.08μM. | ChEMBL | ||
| HEL | Antiviral assay | 3 days | Antiviral activity against Human herpesvirus 1 strain KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by MTT assay, EC50=0.08μM. | ChEMBL | ||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 5 uM 1-[6-[1,1-(diphenyl)-1-(4-pyridyl)methoxy]hexyl]thymine, IC50=0.081μM. | 17181158 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against Human herpesvirus 1 strain KOS infected human HEL cells assessed as reduction of virus-induced cytopathic effect after 3 days by colorimetric formazan-based MTS assay, EC50=0.09μM. | ChEMBL | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus plaque formation, EC50=0.09μM. | 27639368 | |||
| E6SM | Antiviral assay | Antiviral activity against HSV1 KOS-induced cytopathogenicity in E6SM cells, MIC=0.096μM. | 17092728 | |||
| E6SM | Antiviral assay | Antiviral activity against HSV2 G-induced cytopathogenicity in E6SM cells, MIC=0.096μM. | 17092728 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase deficient acyclovir-resistant HSV1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.1μM. | 21232828 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase-deficient ACV-resistant Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=0.1μM. | 28757102 | |||
| OST TK- cells | Cytotoxicity assay | Cytotoxicity against human OST TK- cells expressing HSV1 TK in presence of 10 uM 1-[6-[1,1-(diphenyl)-1-(4-pyridyl)methoxy]hexyl]thymine, IC50=0.11μM. | 17181158 | |||
| HFF | Antiviral assay | 10 days | Antiviral activity against HCMV Towne infected in HFF cells incubated for 10 days by plaque reduction assay, IC50=0.14μM. | 21812420 | ||
| HFF | Function assay | Inhibition of HCMV AD169 replication in HFF cells by cytopathic effect assay, EC50=0.15μM. | 17004726 | |||
| MCA-TK | Cytotoxicity assay | Cytotoxicity against MCA-TK cells, IC50=0.15μM. | 17960926 | |||
| vero | Antiviral assay | Antiviral activity was determined in plaque reduction assay in vero cells against HSV-1 F strain, ID50=0.2μM. | 3871860 | |||
| Vero | Antiviral assay | In vitro antiviral activity was measured against HSV-1(F) in Vero cells, ID50=0.2μM. | 3009811 | |||
| Vero | Antiviral assay | Antiviral activity determined against herpes simplex type 1 (F strain) by plaque reduction in Vero cells, ID50=0.2μM. | 3016263 | |||
| HEL | Antiviral assay | Antiviral activity against HSV2 G in human HEL cells assessed as protection against virus-induced cytopathicity, EC50=0.2μM. | 19281225 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against acyclovir-resistant TK-defecient Herpes simplex virus 1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by microscopic method, EC50=0.2μM. | 30286952 | ||
| fibroblast cells | Function assay | Inhibition of viral cytopathic effect in infected human foreskin fibroblast cell in monolayers of HSV-2 AD Vero cells by 50%, EC50=0.23μM. | 11585457 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus AD169 infected in HEL cells, EC50=0.25μM. | 27128178 | |||
| vero | Antiviral assay | In vitro antiviral and anticellular activity was evaluated against herpes simplex virus HSV-1 (F strain) in vero cells tissue culture, ID50=0.3μM. | 2993615 | |||
| HELF | Antiviral assay | 7 days | Antiviral activity against Herpes simplex virus 2 G infected in human HELF cells assessed as reduction in virus-induced cytopathogenicity incubated for 7 days by microscopic analysis, EC50=0.3μM. | 31586832 | ||
| HS27 | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus infected in human HS27 cells after 7 days by GFP-based fluorescent reduction assay, EC50=0.32μM. | 20047911 | ||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus Davis infected in human HEL cells assessed as reduction in virus plaque formation, EC50=0.38μM. | 23911856 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus Davis 07/1 infected in HEL cells, EC50=0.4μM. | 27128178 | |||
| HEL | Antiviral assay | Antiviral activity against herpes simplex virus 2 G infected in HEL cells assessed as reduction in virus-induced cytopathogenicity by microscopic analysis, EC50=0.4μM. | 30738653 | |||
| HEL | Antiviral assay | Antiviral activity against HSV1 KOS in HEL cells, EC50=0.48μM. | 17181162 | |||
| HFF | Function assay | Inhibitory concentration of the drug against the cytopathic effect for AD169 strain of epstein barr virus-2 (HCMV) in human HFF cells, EC50=0.5μM. | 8394933 | |||
| HEL | Function assay | Inhibition of human CMV DNA synthesis in CMV-infected HEL cells, IC50=0.5μM. | 7752205 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase-deficient acyclovir-resistant Herpes simplex virus 1 KOS infected in HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=0.5μM. | 26001344 | |||
| HELF | Antiviral assay | Antiviral activity against thymidine kinase-deficient acyclovir-resistant Human herpesvirus 1 KOS strain infected in HELF cells assessed as inhibition of virus-induced cytopathogenicity, EC50=0.5μM. | 30098481 | |||
| Vero | Antiviral assay | 36 hrs | Antiviral activity against HSV 2 infected in African green monkey Vero cells assessed as reduction in plaque formation incubated for 36 hrs by crystal violet staining based assay, EC50=0.58μM. | 32191456 | ||
| HEL | Antiviral assay | 5 days | Antiviral activity against Varicella zoster virus OKA infected in human HEL cells assessed as inhibition of virus-induced cytopathicity after 5 days, EC50=0.59μM. | 19339082 | ||
| HEL | Antiviral assay | Antiviral activity against wild type HCMV Davis infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.66μM. | 19226140 | |||
| HEL | Antiviral assay | Antiviral activity against acyclovir-resistant HCMV AD169 clone 1 infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.69μM. | 19226140 | |||
| HEL | Antiviral assay | Antiviral activity against wild type HCMV AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.7μM. | 19226140 | |||
| Vero | Antiviral assay | 36 to 48 hrs | Antiviral activity against Herpes simplex virus 1 F infected in african green monkey Vero cells assessed as plaque reduction after 36 to 48 hrs, EC50=0.7μM. | 17438061 | ||
| HFF | Antiviral assay | 8 to 10 days | Antiviral activity against HCMV TB40 infected in human HFF cells assessed as reduction in plaque formation treated with compound following viral infection and incubated for 8 to 10 days by crystal violet staining based assay, EC50=0.72μM. | 32191456 | ||
| fibroblast cells | Antiviral assay | 1 hour | Antiviral activity against human cytomegalovirus Ad169 (WT) plaque forming unit grown on Human foreskin fibroblast cells upon incubation for 1 hour at 37 degrees C with compound dissolved in DMSO, IC50=0.8μM. | 16134946 | ||
| fibroblast cells | Antiviral assay | 1 hour | Antiviral activity against human cytomegalovirus Ad169 (V823A) plaque forming unit grown on Human foreskin fibroblast cells upon incubation for 1 hour at 37 degrees C with compound dissolved in DMSO, IC50=0.8μM. | 16134946 | ||
| E6SM | Antiviral assay | Antiviral activity against HSV1 TK- KOS ACV-induced cytopathogenicity in E6SM cells, MIC=0.8μM. | 17092728 | |||
| HEL | Antiviral assay | 4 days | Antiviral activity against acyclovir-resistant TK-deficient Herpes simplex virus-1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect after 4 days by microscopic analysis, EC50=0.8μM. | 21696963 | ||
| HEL | Antiviral assay | Antiviral activity against acyclovir-resistant thymidine kinase deficient Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=0.8μM. | 29880251 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase deficient and aciclovir resistant herpes simplex virus 1 KOS infected in HEL cells assessed as reduction in virus-induced cytopathogenicity by microscopic analysis, EC50=0.8μM. | 30738653 | |||
| HELF | Antiviral assay | 7 days | Antiviral activity against Herpes simplex virus 1 TK-KOS-ACV infected in human HELF cells assessed as reduction in virus-induced cytopathogenicity incubated for 7 days by microscopic analysis, EC50=0.8μM. | 31586832 | ||
| HEL | Antiviral assay | Antiviral activity against Herpes simplex virus-2 G infected in human HEL cells assessed as reduction in virus-induced cytopathogenicity, EC50=0.9μM. | 23911856 | |||
| CRFK | Antiviral assay | Antiviral activity against Feline Herpes virus infected in cat CRFK cells assessed as inhibition of virus-induced cytopathic effect by MTS assay, EC50=0.9μM. | 23911856 | |||
| Vero | Antiviral assay | 3 days | Antiviral activity against Herpes simplex virus 1 strain F VR-733 infected in African green monkey Vero cells after 3 days by MTT assay, EC50=0.9μM. | 26460883 | ||
| MRC5 | Antiviral assay | Antiviral activity against HCMV in MRC5 cells by plaque reduction assay, IC50=0.91μM. | 17239594 | |||
| MEF | Antiviral assay | 3 days | Antiviral activity against Mouse Cytomegalovirus Smith (ATCC VR-1399) infected in mouse MEF cells assessed as reduction in plaque formation measured after 3 days by crystal violet staining based assay, EC50=0.96μM. | 32191456 | ||
| HFF | Antiviral assay | 5 days | Antiviral activity against HCMV infected HFF cells after 5 days by dot blot assay, IC50=1μM. | 17329103 | ||
| HEL | Antiviral assay | Antiviral activity against ACV-resistant TK-deficient HSV-1 KOS infected in HEL cells assessed as inhibition of viral-induced cytopathogenicity, EC50=1μM. | 23795238 | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2211 infected in HFF cells by SEAP reporter gene assay, EC50=1.1μM. | 17709468 | |||
| HEL | Antiviral assay | 5 days | Antiviral activity against Varicella zoster virus YS infected in human HEL cells assessed as inhibition of virus induced cytopathicity after 5 days, EC50=1.1μM. | 19339082 | ||
| CRFK | Antiviral assay | Antiviral activity against Feline herpesvirus infected in CRFK cells by viral CPE assay, EC50=1.15μM. | 22858222 | |||
| HEL | Antiviral assay | Antiviral activity against HPMPA-resistant HCMV AD169 clone 2 mutant infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=1.2μM. | 19226140 | |||
| fibroblast cells | Antiviral assay | Antiviral activity against Human cytomegalovirus Towne infected in human foreskin fibroblast cells by plaque reduction assay, EC50=1.2μM. | 19397271 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) wild type expressed in HFF cells was determined by plaque reduction assay, IC50=1.2μM. | 15509176 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) D10 strain (UL89 mutant gene) expressed in HFF cells was determined by plaque reduction assay, IC50=1.2μM. | 15509176 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) r56 strain (UL56 mutant gene) expressed in HFF cells was determined by plaque reduction assay, IC50=1.2μM. | 15509176 | |||
| FRCK | Antiviral assay | Antiviral activity against Feline herpesvirus in feline FRCK cells assessed as inhibition of virus-induced cytopathicity, EC50=1.2μM. | 19281225 | |||
| CRFK | Antiviral assay | 3 days | Antiviral activity against Feline herpesvirus infected in CRFK cells assessed as inhibition of virus-induced cytopathicity after 3 days, EC50=1.2μM. | 23047229 | ||
| vero | Antiviral assay | In vitro antiviral and anticellular activity was evaluated against herpes simplex virus HSV-2 (G strain) in vero cells, ID50=1.3μM. | 2993615 | |||
| fibroblast cells | Antiviral assay | Antiviral activity against human cytomegalovirus (Davis strain) grown on human foreskin fibroblast cells determined by reduction in plaque formation upon incubation at 37 degrees C with the compound dissolved in DMSO, IC50=1.3μM. | 16134946 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2211 in HFF cells by SEAP assay, IC50=1.3μM. | 17043128 | |||
| HEL | Antiviral assay | 7days | Antiviral activity against human cytomegalovirus Davis in HEL cells assessed as reduction of plaque formation after 7days, EC50=1.3μM. | 17539622 | ||
| HFF | Antiviral assay | Antiviral activity against HCMV Davis in HFF cells by plaque reduction assay, IC50=1.3μM. | 17434304 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against HCMV Davis infected in HEL cells assessed as reduction of virus-induced cytopathogenicity after 3 days, EC50=1.3μM. | 17961851 | ||
| HFF | Antiviral assay | Antiviral activity against Human cytomegalovirus infected in HFF cells by plaque reduction assay, IC50=1.3μM. | 20167488 | |||
| HFF | Function assay | Inhibition of HCMV DNA polymerase infected in HFF cells, IC50=1.3μM. | 20403696 | |||
| MRC5 | Function assay | Inhibitory concentration against Davis strain of HCMV in MRC5 cells, IC50=1.4μM. | 11454465 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2241 in HFF cells by SEAP assay, IC50=1.4μM. | 17043128 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2233 in HFF cells by SEAP assay, IC50=1.4μM. | 17043128 | |||
| Vero | Antiviral assay | 3 days | Antiviral activity against Herpes simplex virus 2 strain MS VR-540 infected in African green monkey Vero cells after 3 days by MTT assay, EC50=1.4μM. | 26460883 | ||
| MRC5 | Function assay | Inhibitory concentration against Towne strain of HCMV in MRC5 cells, IC50=1.5μM. | 11454465 | |||
| CCL81 | Antiviral assay | Antiviral activity against HSV1 in CCL81 cells, EC50=1.5μM. | 17188402 | |||
| CCL81 | Antiviral assay | Antiviral activity against HSV2 in CCL81 cells, EC50=1.5μM. | 17188402 | |||
| MRC5 | Function assay | Inhibitory concentration against 2599R strain of HCMV in MRC5 cells, IC50=1.6μM. | 11454465 | |||
| Vero | Antiviral assay | In vitro antiviral activity was measured against HSV-2(G) in Vero cells, ID50=1.6μM. | 3009811 | |||
| HFF | Function assay | In vitro reduction in yield of Towne strain of HCMV in HFF (human fibroblast) cells, IC90=1.6μM. | 9057865 | |||
| HFF | Function assay | Inhibitory concentration against human cytomegalovirus replication in HFF cells was determined by yield reduction assay, IC90=1.6μM. | 9438017 | |||
| HFF | Function assay | Compound was tested for antiviral activity against Towne strain of HCMV in a yield reduction assay using HFF cells, IC90=1.6μM. | 10882374 | |||
| HFF | Function assay | Compound was tested for antiviral activity against human cytomegalovirus (HCMV) by yield reduction assay using HFF cells, IC90=1.6μM. | 10882375 | |||
| HFF | Antiviral assay | Antiviral activity against towne strain HCMV was determined by yield reduction assay in duplicate wells using HFF cells, IC90=1.6μM. | 10882370 | |||
| HFF | Antiviral assay | Antiviral activity against HCMV was determined by yield reduction assay using HFF cells, IC90=1.6μM. | 10882372 | |||
| MRC5 | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus infected in human MRC5 cells after 7 days by GFP-based fluorescent reduction assay, EC50=1.65μM. | 20047911 | ||
| HEL | Antiviral assay | 4 days | Antiviral activity against Cytomegalovirus AD-169 infected in HEL cells assessed as inhibition of virus plaque formation after 4 days, EC50=1.65μM. | 22459876 | ||
| HEL | Antiviral assay | 4 days | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of virus plaque formation after 4 days, EC50=1.65μM. | 22459876 | ||
| HFF | Function assay | Inhibition of HCMV towne replication in HFF cells by plaque reduction assay, EC50=1.8μM. | 17004726 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV AD169 in HFF cells by PRA, IC50=1.9μM. | 17043128 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against HCMV AD169 infected in human HEL cells assessed inhibition of virus-induced cytopathicity after 7 days postinfection, EC50=2μM. | 19226140 | ||
| HFF | Function assay | Inhibition of HCMV Towne replication in HFF cells by plaque reduction assay, EC50=2.1μM. | 15615545 | |||
| HFF | Antiviral assay | 8 days | Antiviral activity against HCMV infected in human HFF cells assessed as reduction in cytopathic effect after 8 days by plaque reduction assay, IC50=2.1μM. | 22607883 | ||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of virus plaque formation, EC50=2.1μM. | 27639368 | |||
| HFF | Antiviral assay | 8 to 10 days | Antiviral activity against HCMV Towne infected in human HFF cells assessed as reduction in plaque formation treated with compound following viral infection and incubated for 8 to 10 days by crystal violet staining based assay, EC50=2.1μM. | 32191456 | ||
| Vero | Antiviral assay | 3 days | Antiviral activity against Human cytomegalovirus Davis VR-807 infected in African green monkey Vero cells after 3 days by MTT assay, EC50=2.14μM. | 26460883 | ||
| HFF | Function assay | Inhibition of HCMV AD169 replication in HFF cells by CPE inhibition assay, EC50=2.2μM. | 15615545 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=2.29μM. | 29880251 | |||
| HEL | Antiviral assay | 5 days | Antiviral activity against TK+ VZV YS in HEL cells assessed as reduction of virus plaque formation after 5 days, EC50=2.3μM. | 17622128 | ||
| HELF | Antiviral assay | 7 days | Antiviral activity against Cytomegalovirus Davis infected in human HELF cells assessed as reduction in virus-induced cytopathogenicity incubated for 7 days by Giemsa staining based microscopic analysis, EC50=2.3μM. | 31586832 | ||
| HEL299 | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus infected in HEL299 cells after 7 days by GFP-based fluorescent reduction assay, EC50=2.39μM. | 20047911 | ||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) C4 strain (UL56 + UL89 mutant gene) expressed in HFF cells was determined by plaque reduction assay, IC50=2.5μM. | 15509176 | |||
| HFF | Antiviral assay | Antiviral activity against Human cytomegalovirus Towne infected in HFF cells by plaque reduction assay, EC50=2.5μM. | 18082410 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus Davis infected in human HEL cells assessed as inhibition of virus-induced cytopathicity after 7 days post infection, EC50=2.5μM. | 19339082 | ||
| HEL | Antiviral assay | 5 days | Antiviral activity against thymidine kinase deficient Varicella zoster virus 07/1 infected in human HEL cells assessed as inhibition of virus-induced cytopathicity after 5 days, EC50=2.5μM. | 19339082 | ||
| HFF | Function assay | Inhibitory concentration against human cytomegalovirus replication in HFF cells was determined by cytopathic effect inhibition assay, IC50=2.6μM. | 9438017 | |||
| HEL | Antiviral assay | Antiviral activity against foscarnet-resistant HCMV AD169 clone C infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=2.6μM. | 19226140 | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2417 with pol A809V mutant infected in HFF cells by SEAP reporter gene assay, EC50=2.6μM. | 17709468 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against CMV Davis in human HEL cells after 7 days, EC50=2.6μM. | 17672445 | ||
| CRFK | Antiviral assay | Antiviral activity against Feline herpesvirus infected in cat CRFK cells assessed as protection from virus-induced cytopathogenicity by MTT assay, EC50=2.6μM. | 20034711 | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2542 with pol T813S mutant infected in HFF cells by SEAP reporter gene assay, EC50=2.7μM. | 17709468 | |||
| HEL | Antiviral assay | Antiviral activity against HCMV Davis in HEL cells assessed as reduction of virus plaque formation, EC50=2.75μM. | 17518459 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=2.96μM. | 28757102 | |||
| BSC-1 | Antiviral assay | Antiviral activity of the compound was evaluated against the Herpes simplex virus type-1 in BSC-1 cells, IC50=3μM. | 2913300 | |||
| MRC5 | Function assay | Inhibitory concentration against AD169 strain of HCMV in MRC5 cells, IC50=3μM. | 11454465 | |||
| HFF | Antiviral assay | Antiviral activity against HCMV Towne in HFF cells by plaque reduction assay, IC50=3μM. | 17161946 | |||
| hhTERT-BJ1 | Antiviral assay | 2 days | Antiviral activity against Human cytomegalovirus Towne infected in human hhTERT-BJ1 cells assessed as inhibition of virus mediated DNA synthesis after 2 days, EC50=3μM. | 18458124 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against acyclovir-resistant thymidine kinase deficient HSV1 KOS infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity after 7 days, EC50=3μM. | 21128666 | ||
| HFF | Antiviral assay | 10 days | Antiviral activity against Human cytomegalovirus Towne infected in HFF cells assessed as reduction in plaque formation after 10 days, IC50=3μM. | 21641218 | ||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus AD-169 infected in human HEL cells assessed as reduction in virus plaque formation, EC50=3.1μM. | 23911856 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus plaque formation, EC50=3.1μM. | 27639368 | |||
| HELF | Antiviral assay | 7 days | Antiviral activity against Cytomegalovirus AD169 infected in human HELF cells assessed as reduction in virus-induced cytopathogenicity incubated for 7 days by Giemsa staining based microscopic analysis, EC50=3.15μM. | 31586832 | ||
| HFF | Antiviral assay | Antiviral activity against ganciclovir-sensitive HCMV1 isolate replication in HFF cells by plaque reduction assay, IC50=3.22μM. | 19010684 | |||
| HFF | Function assay | Compound was tested for antiviral activity against wild-type AD169 strain of HCMV in yield reduction assay using HFF cells, IC90=3.3μM. | 10882374 | |||
| HEL | Antiviral assay | 6 to 7 days | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of virus-induced cytopathic effect measured after 6 to 7 days post-infection, EC50=3.31μM. | 29670705 | ||
| MEF | Function assay | Inhibitory concentration against murine cytomegalovirus replication in MEF cells was determined by plaque reduction assay, IC50=3.4μM. | 9438017 | |||
| BSC-1 | Antiviral assay | Antiviral activity against HSV-1 (herpes simplex virus), determined by ELISA in quadruplicate wells using BSC-1 cells, IC50=3.5μM. | 10882370 | |||
| BSC-1 | Function assay | ELISA assay was performed using BSC-1 cells to determine activity against HSV-1, IC50=3.5μM. | 10882372 | |||
| BSC-1 | Antiviral assay | In vitro antiviral activity against HSV-1 virus in BSC-1 (monkey kidney) cells, IC50=3.5μM. | 9057865 | |||
| HEL | Antiviral assay | Antiviral activity against PMEDAP-resistant HCMV AD169 clone 4 infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=3.5μM. | 19226140 | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2420 with pol G841A mutant infected in HFF cells by SEAP reporter gene assay, EC50=3.5μM. | 17709468 | |||
| BSC-1 | Function assay | Inhibitory concentration required against herpes simplex virus type-1 (HSV-1) expressed in BSC-1 cells was determined by ELISA assay, IC50=3.5μM. | 15509174 | |||
| BSC-1 | Function assay | Inhibitory concentration required against herpes simplex virus type-1 (HSV-1) expressed in BSC-1 cells was determined by ELISA assay, IC50=3.5μM. | 15509175 | |||
| BSC-1 | Function assay | Inhibitory concentration required against herpes simplex virus type-1 (HSV-1) expressed in BSC-1 cells was determined by ELISA assay, IC50=3.5μM. | 15509176 | |||
| HFF | Function assay | Compound was tested for antiviral activity against 2916 AD169 strain of HCMV in yield reduction assay using HFF cells, IC90=3.6μM. | 10882374 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against HCMV Davis infected in human HEL cells assessed inhibition of virus-induced cytopathicity after 7 days postinfection, EC50=3.6μM. | 19226140 | ||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2258 with UL97 C592G mutant infected in HFF cells by SEAP reporter gene assay, EC50=3.9μM. | 17709468 | |||
| HEL | Antiviral assay | 2 to 3 days | Antiviral activity against thymidine kinase-deficient ACV-resistant Herpes simplex virus 1 KOS infected in HEL cells assessed as inhibition of plaque formation measured after 2 to 3 days post infection, EC50=3.91μM. | 29550734 | ||
| MRC-5 | Antiherpesvirus assay | In vitro antiherpesvirus activity against MRC-5 cells infected with HCMV (AD-169 strain), IC50=4μM. | 1323678 | |||
| HEL | Antiviral assay | Antiviral activity against cidofovir HCMV AD169 clone 5 infected in human HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=4μM. | 19226140 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase deficient and acyclovir-resistant HSV1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=4μM. | 23099097 | |||
| HEL | Antiviral assay | Antiviral activity against TK-negative Herpes simplex virus 1 KOS infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=4μM. | 23811093 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase-deficient acyclovir-resistant Herpes simplex virus 1 KOS infected in HEL cells assessed as reduction in virus-induced cytopathogenicity, EC50=4μM. | 27750154 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as reduction in viral cytopathic effect, EC50=4.05μM. | 29407990 | |||
| HFF | Function assay | Compound was tested for antiviral activity against C-4 Towne strain of HCMV in yield reduction assay using HFF cells, IC50=4.1μM. | 10882374 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2291 in HFF cells by PRA, IC50=4.1μM. | 17043128 | |||
| CRFK | Antiviral assay | 4 days | Antiviral activity against Feline herpesvirus infected in cat CRFK cells assessed as inhibition of virus-induced cytopathic effect after 4 days by colorimetric formazan-based MTS assay, EC50=4.1μM. | 22459876 | ||
| CRFK | Antiviral assay | Antiviral activity against feline herpesvirus infected in cat CRFK cells assessed as reduction of virus-induced cytopathogenicity by MTS assay, EC50=4.1μM. | 23811093 | |||
| HEL | Antiviral assay | Antiviral activity against ganciclovir-resistant HCMV AD169 clone 4 infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=4.3μM. | 19226140 | |||
| HEL | Antiviral assay | Antiviral activity against ACV-resistant thymidine kinase deficient Herpes simplex virus 1 KOS infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=4.3μM. | 28829913 | |||
| HFF | Function assay | Compound was tested for antiviral activity against wild-type Towne strain of HCMV in yield reduction assay using HFF cells, IC50=4.5μM. | 10882374 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against Human cytomegalovirus Davis expressed in HEL cells assessed as reduction in plaque formation incubated for 7 days by Giemsa stain based microscopy, EC50=4.72μM. | 26291038 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus AD169 in HEL cells assessed as reduction of plaque formation after 7 days, EC50=4.8μM. | 17539622 | ||
| HEL | Antiviral assay | 3 days | Antiviral activity against HCMV AD169 infected in HEL cells assessed as reduction of virus-induced cytopathogenicity after 3 days, EC50=4.8μM. | 17961851 | ||
| HEL | Antiviral assay | 6 to 7 days | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as inhibition of plaque formation measured after 6 to 7 days post infection, EC50=4.83μM. | 29550734 | ||
| MRC5 | Antiviral assay | Antiviral activity against Human CMV Towne in MRC5 cells by PRA, IC50=4.9μM. | 17043128 | |||
| MRC5 | Antiviral assay | In vitro antiviral activity was measured against HCMV(AD-169) in MRC5 cells, ID50=5μM. | 3009811 | |||
| H-1 | Function assay | Effective concentration required to inhibit Epstein-barr virus replication in H-1 cells in DNA hybridization assay, EC50=5μM. | 15634003 | |||
| H1 | Function assay | Inhibition of EBV replication in H1 cells by DNA hybridization assay, EC50=5μM. | 17004726 | |||
| H1 | Function assay | Inhibition of EBV replication in H1 cells by DNA hybridization assay, EC50=5μM. | 15615545 | |||
| CEM | Cytotoxicity assay | Cytotoxicity against CEM cells, CC50=5μM. | 15615545 | |||
| HFF | Antiviral assay | 5 to 7 days | Antiviral activity against HCMV infected HFF cells after 5 to 7 days by viral yield assay, EC99.9=5μM. | 17329103 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against HCMV AD169 in HEL cells assessed as reduction of virus plaque formation after 7 days, EC50=5.1μM. | 17622128 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against HCMV AD169/Davis infected in human HEL cells assessed as reduction of virus-induced cytopathogenicity after 7 days, EC50=5.2μM. | 21128666 | ||
| AKATA | Antiviral assay | 48 hrs | Antiviral activity against EBV infected in human AKATA cells assessed as reduction in IgG-induced lytic virus replication measured at 48 hrs post-lytic induction by qRT-PCR analysis, EC50=5.2μM. | 32191456 | ||
| MRC5 | Antiviral assay | Antiviral activity against Human CMV T2296 in MRC5 cells by PRA, IC50=5.4μM. | 17043128 | |||
| fibroblast cells | Antiviral assay | Antiviral activity against Murine cytomegalovirus infected in mouse embryonic fibroblast cells by plaque reduction assay, EC50=5.5μM. | 19397271 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2293 in HFF cells by SEAP assay, IC50=5.6μM. | 17043128 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as reduction in viral cytopathic effect, EC50=5.63μM. | 29407990 | |||
| NIH 3T3 | Antiviral assay | 4 to 5 days | Antiviral activity against Murine cytomegalovirus strain Smith infected in mouse NIH 3T3 cells after 4 to 5 days by plaque reduction assay, EC50=5.7μM. | 18458124 | ||
| RG2TK+ | Cytotoxicity assay | 72 hrs | Cytotoxicity against HSV1-tk gene overexpressing RG2TK+ cells after 72 hrs by MTT assay, CC50=5.86μM. | 18800764 | ||
| MRC-5 | Function assay | Concentration required to reduce viral plaque formation by Human Cytomegalovirus (HCMV) (strain AD-169) in MRC-5 cells, IC50=5.9μM. | 10966740 | |||
| HEL | Antiviral assay | Antiviral activity against HCMV Davis infected in HEL cells by viral CPE assay, EC50=5.9μM. | 22858222 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus Davis infected in human HEL cells assessed as reduction of plaque formation, EC50=5.98μM. | 23811093 | |||
| HCMV AD 169 | Antiviral assay | In vitro antiviral and anticellular activity was evaluated against human cytomegalovirus (HCMV AD 169) in (HCMV AD 169) cells, ID50=6μM. | 2993615 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as protection against virus-induced cytopathicity, EC50=6μM. | 21565516 | |||
| HEL | Antiviral assay | 3 days | Antiviral activity against acyclovir-resistant TK-deficient Hepres simplex virus 1 KOS infected in HEL cells assessed as inhibition of virus-induced cytopathic effect after 3 days by MTT assay, EC50=6μM. | ChEMBL | ||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2784 with pol A809V UL97 C592G double mutant infected in HFF cells by SEAP reporter gene assay, EC50=6.1μM. | 17709468 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against Human cytomegalovirus AD169 expressed in HEL cells assessed as reduction in plaque formation incubated for 7 days by Giemsa stain based microscopy, EC50=6.12μM. | 26291038 | ||
| MRC-5 | Function assay | Inhibitory concentration for anti-HCMV activity against AD-169 strain in MRC-5 cells, IC50=6.3μM. | 9836613 | |||
| HEL | Antiviral assay | Antiviral activity against HCMV AD169 in HEL cells assessed as reduction of virus plaque formation, EC50=6.3μM. | 17518459 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus AD-169 infected in HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=6.3μM. | 26001344 | |||
| HEL | Antiviral assay | 6 to 7 days | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of plaque formation measured after 6 to 7 days post infection, EC50=6.46μM. | 29550734 | ||
| HEL | Antiviral assay | 7 days | Antiviral activity against CMV AD169 in human HEL cells after 7 days, EC50=6.5μM. | 17672445 | ||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus Davis infected in HEL cells assessed as reduction in virus plaque formation, EC50=6.5μM. | 27750154 | |||
| HELF | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis strain infected in HELF cells assessed as inhibition of virus-induced cytopathogenicity, EC50=6.52μM. | 30098481 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as protection against virus-induced cytopathicity, EC50=6.54μM. | 21565516 | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV T2798 with pol T813S UL97 C592G double mutant infected in HFF cells by SEAP reporter gene assay, EC50=6.6μM. | 17709468 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=6.79μM. | 28757102 | |||
| HEL | Function assay | Inhibition against DNA of uninfected HEL cells by incorporation of [3H]TdR, IC50=7μM. | 7752205 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2291 in HFF cells by SEAP assay, IC50=7.1μM. | 17043128 | |||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus Davis infected in HEL cells assessed as reduction in virus-induced cytopathicity, EC50=7.1μM. | 22578783 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus Davis infected in HEL cells assessed as reduction of virus-induced cytopathogenicity, EC50=7.1μM. | 26001344 | |||
| CRFK | Antiviral assay | 3 days | Antiviral activity against Felid herpesvirus 1 infected cat CRFK cells assessed as reduction of virus-induced cytopathic effect after 3 days by colorimetric formazan-based MTS assay, EC50=7.3μM. | ChEMBL | ||
| HEL | Antiviral assay | Antiviral activity against human cytomegalovirus Davis infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=7.3μM. | 28829913 | |||
| HFF | Antiviral assay | In vitro antiviral activity against HCMV (Towne strain) in HFF (human fibroblast) cells, IC50=7.4μM. | 9057865 | |||
| HFF | Function assay | Inhibitory concentration against human cytomegalovirus replication in HFF cells was determined by plaque reduction assay, IC50=7.4μM. | 9438017 | |||
| HFF | Function assay | Compound was tested for antiviral activity against Towne strain of HCMV in a plaque reduction assay using HFF cells, IC50=7.4μM. | 10882374 | |||
| HFF | Function assay | Compound was tested for antiviral activity against human cytomegalovirus (HCMV) by plaque reduction assay using HFF cells, IC50=7.4μM. | 10882375 | |||
| HFF | Antiviral assay | Antiviral activity against towne strain HCMV was determined by plaque reduction assay in duplicate wells using HFF cells, IC50=7.4μM. | 10882370 | |||
| HFF | Antiviral assay | Antiviral activity against HCMV was determined by plaque reduction assay using HFF cells, IC50=7.4μM. | 10882372 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) expressed in HFF cells was determined by plaque reduction assay, IC50=7.4μM. | 15509174 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) expressed in HFF cells was determined by plaque reduction assay, IC50=7.4μM. | 15509175 | |||
| HFF | Function assay | Inhibitory concentration required against human cytomegalovirus (HCMV) expressed in HFF cells was determined by plaque reduction assay, IC50=7.4μM. | 15509176 | |||
| HEL | Antiviral assay | Antiviral activity against human cytomegalovirus AD-169 infected in human HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=7.4μM. | 28829913 | |||
| HEL | Antiviral assay | 6 to 7 days | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathic effect measured after 6 to 7 days post-infection, EC50=7.41μM. | 29670705 | ||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as reduction in virus-induced cytopathicity, EC50=7.5μM. | 22578783 | |||
| HFF | Antiviral assay | Antiviral activity was determined by the HCMV plaque assay using HFF cells, IC50=7.7μM. | 9057866 | |||
| HEL | Antiviral assay | Antiviral activity against human cytomegalovirus Davis infected in HEL cells assessed as reduction in viral plaque formation, EC50=7.8μM. | 26443550 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus AD-169 infected in human HEL cells assessed as reduction of plaque formation, EC50=7.87μM. | 23811093 | |||
| CRFK | Antiviral assay | Antiviral activity against Feline herpesvirus infected in CRFK cells assessed as protection against virus-induced cytopathicity, EC50=8.1μM. | 21565516 | |||
| HEL | Antiviral assay | Antiviral activity against human Cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathicity after, EC50=8.2μM. | 23911854 | |||
| HFF | Function assay | In vitro inhibition of human cytomegalovirus in HFF cells by plaque reduction assay, IC50=8.4μM. | ChEMBL | |||
| HFF | Antiviral assay | Antiviral activity against Cytomegalovirus CMV 2817 with pol G841A UL97 C592G double mutant infected in HFF cells by SEAP reporter gene assay, EC50=8.4μM. | 17709468 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against HCMV Davis in HEL cells assessed as reduction of virus plaque formation after 7 days, EC50=8.7μM. | 17622128 | ||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=8.7μM. | 28682067 | |||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase deficient and acyclovir resistant HSV1 KOS in human HEL cells assessed as protection against virus-induced cytopathicity, EC50=9μM. | 19281225 | |||
| HEL | Antiviral assay | Antiviral activity against HCMV AD169 infected in HEL cells by viral CPE assay, EC50=9.1μM. | 22858222 | |||
| MRC-5 | Function assay | Inhibitory activity of compound against human cytomegalovirus (HCMV) in MRC-5 cells, EC50=9.4μM. | 12723951 | |||
| Vero | Antiviral assay | In vitro antiviral activity was measured against HSV-1 (F delta 305) in Vero cells, ID50=10μM. | 3009811 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2293 in HFF cells by PRA, IC50=10μM. | 17043128 | |||
| Vero | Antiviral assay | 3 days | Antiviral activity against Varicella-zoster virus Ellen VR-1367 infected in African green monkey Vero cells after 3 days by MTT assay, EC50=11.1μM. | 26460883 | ||
| HEL | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 infected in HEL cells assessed as inhibition of virus-induced cytopathicity, EC50=11.43μM. | 29880251 | |||
| HELF | Antiviral assay | Antiviral activity against Human cytomegalovirus AD169 strain infected in HELF cells assessed as inhibition of virus-induced cytopathogenicity, EC50=11.75μM. | 30098481 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2311 in HFF cells by PRA, IC50=12.4μM. | 17043128 | |||
| HEL | Antiviral assay | 7 days | Antiviral activity against human cytomegalovirus AD169 infected in human HEL cells assessed as inhibition of virus-induced cytopathicity after 7 days post infection, EC50=12.6μM. | 19339082 | ||
| HEL | Antiviral assay | Antiviral activity against human cytomegalovirus AD169 infected in HEL cells assessed as reduction in viral plaque formation, EC50=13.9μM. | 26443550 | |||
| Vero | Antiviral assay | 36 to 48 hrs | Antiviral activity against Herpes B virus 24105 infected in african green monkey Vero cells assessed as plaque reduction after 36 to 48 hrs, EC50=14.5μM. | 17438061 | ||
| HEL | Antiviral assay | Antiviral activity against thymidine kinase negative and ACV resistant HSV1 KOS infected in HEL cells by viral CPE assay, EC50=14.7μM. | 22858222 | |||
| HEL | Antiviral assay | Antiviral activity against Cytomegalovirus AD169 infected in HEL cells assessed as reduction in virus plaque formation, EC50=14.9μM. | 27750154 | |||
| HEL | Antiviral assay | Antiviral activity against acyclovir-resistant Human herpesvirus 1 KOS infected in HEL cells assessed as protection against virus-induced cytopathicity, EC50=15.7μM. | 21565516 | |||
| HFF | Antiviral assay | Antiviral activity against ganciclovir-resistant HCMV3 isolate replication in HFF cells by plaque reduction assay, IC50=16.4μM. | 19010684 | |||
| HEL | Antiviral assay | Antiviral activity against HCMV Davis strain infected in HEL cells assessed as inhibition of virus-induced cytopathic effect, EC50=16.5μM. | 28682067 | |||
| Vero | Antiviral assay | 36 to 48 hrs | Antiviral activity against Herpes B virus E90-136 infected in african green monkey Vero cells assessed as plaque reduction after 36 to 48 hrs, EC50=19.2μM. | 17438061 | ||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate E2490 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR9 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR4 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR5 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR6 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR2 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A2 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A3 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR11 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A5 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MC1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MP1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against HSV2 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR3 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR8 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR10 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A4 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MR7 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MC2 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MJ1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MJ2 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate MJ3 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against Macacine herpesvirus 1 isolate A6 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | Antiviral activity against HSV1 infected in african green monkey Vero cells by plaque reduction assay, EC50=19.3μM. | 19858259 | |||
| Vero | Antiviral assay | 36 to 48 hrs | Antiviral activity against Herpes B virus 32425 infected in african green monkey Vero cells assessed as plaque reduction after 36 to 48 hrs, EC50=23.5μM. | 17438061 | ||
| HFF | Antiviral assay | 8 to 10 days | Antiviral activity against GCV-resistant HCMV Towne infected in human HFF cells assessed as reduction in plaque formation treated with compound following viral infection and incubated for 8 to 10 days by crystal violet staining based assay, EC50=29.7μM. | 32191456 | ||
| bone marrow cells | Cytotoxicity assay | Cytotoxicity against human bone marrow cells assessed as inhibition of colony forming unit of granulocyte/macrophage, IC50=30μM. | 16814545 | |||
| bone marrow cells | Cytotoxicity assay | 15 days | Cytotoxicity against human bone marrow cells assessed as inhibition of CFU-GM formation after 15 days, CC50=30μM. | 17329103 | ||
| MRC5 | Antiviral assay | Antiviral activity against Human CMV T2287 in MRC5 cells by PRA, IC50=30.1μM. | 17043128 | |||
| HFF | Antiviral assay | Antiviral activity against Human CMV T2311 in HFF cells by SEAP assay, IC50=30.2μM. | 17043128 | |||
| neural precursor cells | Function assay | Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay | 17417631 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| HFF | Antiviral assay | 6 hrs | Antiviral activity against pp28-luciferase expressing HCMV Towne infected in human HFF cells assessed as reduction in viral replication treated with compound immediately after viral infection at MOI of 1 PFU/cell followed by compound washout after 6 hrs a | 32191456 | ||
| HFF | Antiviral assay | 5 uM | Antiviral activity against HCMV Towne infected in human HFF cells assessed as reduction in viral DNA yields in the supernatant at 5 uM treated with compound following viral infection at MOI of 0.1 PFU/cell by US17 real time PCR analysis | 32191456 | ||
| HFF | Antiviral assay | 5 uM | Antiviral activity against HCMV Towne infected in human HFF cells assessed as reduction in viral DNA replication at 5 uM treated with compound following viral infection at MOI of 0.1 PFU/cell by US17 real time PCR analysis | 32191456 | ||
| Vero | Function assay | 10 uM | Inhibition of UL42 gene transcription in HSV-1 infected in African green monkey Vero cells at 10 uM by qRT-PCR analysis | 31503476 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 27 mg/mL
(105.78 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 255.23 | Formula | C9H13N5O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 82410-32-0 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | RS-21592, BW-759, 2'-Nor-2'-deoxyguanosine | Smiles | C1=NC2=C(N1COC(CO)CO)N=C(NC2=O)N | ||
Mechanism of Action
| In vitro |
Ganciclovir is metabolized to the triphosphate form by primarily three cellular enzymes: (1) a deoxyguanosine kinase induced by CMV-infected cells; (2) guanylate kinase; and (3) phosphoglycerate kinase. This compound is sufficient to induce cell death in most bystander cells cocultured with HSV-tk-expressing cells. It significantly reduces DNA synthesis in the transformed cells, whereas it has little effect on DNA synthesis in the nontransformed cells. This chemical exhibits a concentration-dependent reduction in the rate of elongation into mature DNA. It induces cell cycle arrests rather than direct chemical effect on HSVtk-transduced B16F10 melanoma cells. It produces only weak inhibition of DNA synthesis. Ganciclovir-treated cells accumulate in early S-phase and remained there until cell death, suggesting that ganciclovir incorporation in the DNA template is important for cytotoxicity. Its induced apoptosis is due to incorporation of the drug into DNA resulting in replication-dependent formation of DNA double-strand breaks and, at later stages, S and G2/M arrest. The provoked DNA instability is likely to be responsible for the observed initial decline in Bcl-2 level and caspase-9/-3 activation.
|
|---|---|
| In vivo |
Antiviral drug ganciclovir (GCV) inhibits the proliferation of microglia in experimental autoimmune encephalomyelitis (EAE). This compound attenuates neuroinflammation and does not significantly restrain the peripheral immune response.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Cell viability |
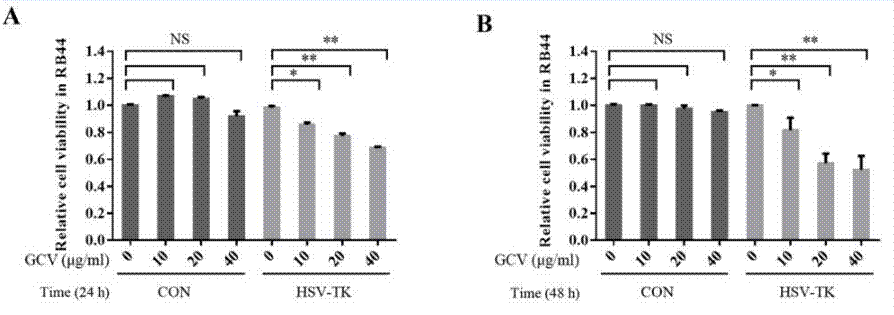
|
29845211 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06407232 | Not yet recruiting | Cytomegalovirus Infections|Kidney Transplant Infection|Pancreas Transplant |
University of Wisconsin Madison|Merck Sharp & Dohme LLC |
May 2024 | Phase 3 |
| NCT06057194 | Not yet recruiting | Infections Cytomegalovirus |
Maimónides Biomedical Research Institute of Córdoba|MERCK SHARP & DOHME DE ESPAÑA S.A. |
October 2023 | Phase 2 |
| NCT04706507 | Recruiting | Acute Respiratory Failure |
Fred Hutchinson Cancer Center|National Heart Lung and Blood Institute (NHLBI) |
June 29 2021 | Phase 3 |
| NCT04478474 | Completed | Cytomegalovirus Infections |
New York Medical College|Merck Sharp & Dohme LLC |
September 15 2020 | -- |
| NCT03698435 | Unknown status | Cytomegalovirus Infections |
University Medical Center Groningen |
May 25 2018 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































