research use only
Tenofovir Disoproxil Fumarate Reverse Transcriptase inhibitor
Cat.No.S1400
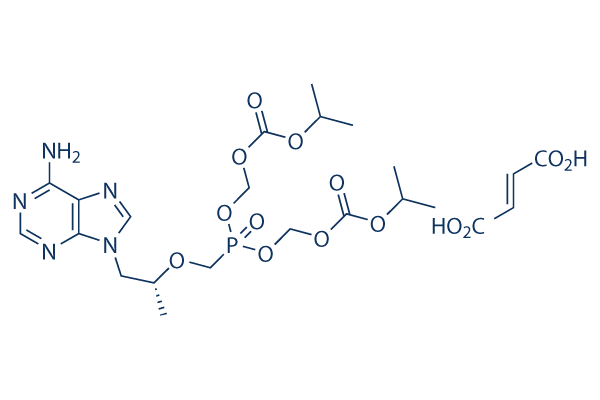
Chemical Structure
Molecular Weight: 635.51
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite HIV HCV Protease |
|---|---|
| Other Reverse Transcriptase Inhibitors | Dapivirine (TMC120) Fangchinoline 3'-Fluoro-3'-deoxythymidine (Alovudine) Salicylanilide Bifendate Ulonivirine Lersivirine (UK-453061) 4-Chloro-2-(trifluoroacetyl)aniline hydrochloride |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MT2 | Function assay | 5 days | Inhibition of virus-induced cytopathic effect in wild type HIV 3a infected MT2 cells after 5 days, EC50=0.015μM | 17562366 | ||
| HepG2 | Cytotoxicity assay | 9 days | Cytotoxicity against human HepG2 cells after 9 days by MTT assay, IC50=2.31μM | 17888662 | ||
| HepG2 | Antiviral assay | 9 days | Antiviral activity against Hepatitis B virus infected human HepG2 cells after 9 days by MTT assay, IC50=5.1μM | 17888662 | ||
| MT-2 | Antiviral assay | Antiviral activity against HIV1 3B infected in human MT-2 cells by two fold dilution method in presence of 10% FBS, EC50=0.0068μM | 19104010 | |||
| MT2 | Antiviral assay | Antiviral activity against HIV1 infected in human MT2 cells assessed as inhibition of viral replication, IC50=0.54μM | 19596885 | |||
| HeLa P4/R5 | Antiviral assay | Antiviral activity against HIV1 infected in human HeLa P4/R5 cells assessed as inhibition of viral replication, IC50=4.7μM | 19596885 | |||
| HeLa P4/R5 | Antiviral assay | Antiviral activity against HIV1 harboring reverse transcriptase K65R mutant infected in human HeLa P4/R5 cells assessed as inhibition of viral replication, IC50=11.4μM | 19596885 | |||
| human bone marrow cells | Cytotoxicity assay | 24 hrs | Cytotoxicity against human bone marrow cells after 24 hrs by BFU-E assay, CC50=0.9μM | 20439609 | ||
| human bone marrow cells | Cytotoxicity assay | 24 hrs | Cytotoxicity against human bone marrow cells after 24 hrs by GM-CFU assay, CC50=1.9μM | 20439609 | ||
| HeLa-T4 | Antiviral assay | 48 hrs | Antiviral activity against single-round HIV1 NLX.Lux-R harboring inactivating mutations in env, vpr and carries firefly luciferase gene in place of nef infected in human HeLa-T4 cells assessed as luciferase activity after 48 hrs by exogenous RT assay, EC50=0.0029μM | 21060108 | ||
| HeLa-T4 | Antiviral assay | 48 hrs | Antiviral activity against single-round HIV1 NLX.Lux-R harboring inactivating mutations in env, vpr and carries firefly luciferase gene in place of nef infected in human HeLa-T4 cells assessed as luciferase activity after 48 hrs by exogenous RT assay, EC95=0.037μM | 21060108 | ||
| HeLaT4 | Antiviral assay | 24 hrs | Antiviral activity against single-round HIV1 NLX.Lux-R harboring inactivating mutations in env, vpr and carries firefly luciferase gene in place of nef assessed as level of infection using human HeLaT4 cells pretreated for 24 hrs followed by exposed to vi, EC50=0.12μM | 21060108 | ||
| HeLaT4 | Antiviral assay | Antiviral activity against single-round HIV1 NLX.Lux-R harboring inactivating mutations in env, vpr and carries firefly luciferase gene in place of nef assessed as level of 2 mins magnetic nanopartials-medated infection in human HeLaT4 cells treated for 1, EC99.6=0.95μM | 21060108 | |||
| HeLaT4 | Cytotoxicity assay | Cytotoxicity against human HeLaT4 cells by WST-1 assay, CC50=34μM | 21060108 | |||
| HeLaT4 | Function assay | 24 hrs | Drug uptake in human HeLaT4 cells assessed as compound persist measured after 3 times washout at 100 time EC95 for HIV1 for 24 hrs | 21060108 | ||
| HeLaT4 | Antiviral assay | 24 hrs | Antiviral activity against single-round HIV1 NLX.Lux-R harboring inactivating mutations in env, vpr and carries firefly luciferase gene in place of nef assessed as level of infection using human HeLaT4 cells pretreated at 100 time EC95 for 24 hrs followed | 21060108 | ||
| PBMC | Antiviral assay | 7 days | Antiviral activity against HIV1 infected in human PBMC assessed as inhibition of viral replication by measuring reverse transcriptase activity in cell supernatant preincubated with cells followed by viral infection measured after 7 days by radioactive inc, EC50=0.0046μM | 27405794 | ||
| HepG2.2.15 | Antiviral assay | 3 days | Antiviral activity against HBV infected in human HepG2.2.15 cells assessed as inhibition of viral DNA in cell supernatant incubated for 3 days in presence of 10% FBS followed by compound treatment in absence of 10% FBS for 3 days by qRT-PCR method, EC50=0.34μM | 27405794 | ||
| HepG2.2.15 | Cytotoxicity assay | 6 days | Cytotoxicity against human HepG2.2.15 cells assessed as reduction in cell viability after 6 days by XTT assay, CC50=29.2μM | 27405794 | ||
| PBMC | Antiviral assay | 30 mins | Antiviral activity against CXCR4-tropic HIV-1 NL4-3 infected in human PHA-stimulated PBMC assessed as inhibition of viral replication by measuring reduction in p24 antigen production preincubated with cells for 30 mins followed by viral infection measured, IC50=0.08μM | 28682067 | ||
| PBMC | Antiviral assay | 30 mins | Antiviral activity against CCR5 tropic HIV1 BaL infected in human PHA-stimulated PBMC assessed as inhibition of viral replication by measuring reduction in p24 antigen production preincubated with cells for 30 mins followed by viral infection measured on , IC50=0.22μM | 28682067 | ||
| MT4 | Antiviral assay | 6 days | Antiviral activity against CXCR4-tropic HIV-1 NL4-3 infected in human MT4 cells assessed as inhibition of virus-induced cytopathic effect after 6 days by XTT dye based assay, EC50=4.89μM | 28682067 | ||
| MT4 | Antiviral assay | 6 days | Antiviral activity against tenofovir-resistant CXCR4-tropic HIV-1 NL4-3 harboring reverse transcriptase K65R mutant infected in human MT4 cells assessed as inhibition of viral replication inhibition of virus-induced cytopathic effect after 6 days by XTT d, EC50=11.3μM | 28682067 | ||
| HepG2.2.15 | Antiviral assay | Antiviral activity against HBV infected in human HepG2.2.15 cells assessed as reduction in cytoplasmic DNA synthesis by reed and munch method, IC50=0.85μM | 31223460 | |||
| HepG2.2.15 | Cytotoxicity assay | Cytotoxicity against human HepG2.2.15 cells infected with HBV, CC50=20.71μM | 31223460 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(157.35 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 635.51 | Formula | C19H30N5O10P.C4H4O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 202138-50-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | GS 1278, Tenofovir DF | Smiles | CC(C)OC(=O)OCOP(=O)(COC(C)CN1C=NC2=C(N=CN=C21)N)OCOC(=O)OC(C)C.C(=CC(=O)O)C(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
HIV reverse transcriptase
(Cell-free assay) |
|---|---|
| In vitro |
Tenofovir is eliminated from systemic circulation renally through a combination of glomerular filtration and active tubular secretion. Tenofovir is not a substrate for human organic cation transporter type 1 (hOCT1) or hOCT2. Tenofovir accumulates to fivefold lower levels in MRP4-overexpressing cells, and its accumulation could be increased by an MRP inhibitor. Tenofovir produces no significant changes in mitochondrial DNA (mtDNA) levels in human hepatoblastoma (HepG2) cells, skeletal muscle cells (SkMCs), or renal proximal tubule epithelial cells. Tenofovir elevates lactate production by less than 20% in HepG2 cells or SkMCs. Tenofovir is efficiently phosphorylated to tenofovir diphosphate (TFV-DP) in both HepG2 cells and primary human hepatocytes. Tenofovir has a 50% effective concentration of 1.1 mM against HBV in cell-based assays, and potency is improved > 50-fold by the addition of bis-isoproxil progroups. Tenofovir has previously demonstrated full activity against lamivudine-resistant HBV in vitro and clinically. Tenofovir inhibits the proliferation of liver-derived HepG2 cells and normal skeletal muscle cells with CC(50) values of 398 μM and 870 μM, respectively. Tenofovir shows substantially weaker effects on proliferation and viability of renal proximal tubule epithelial cells than cidofovir, a related nucleotide analog with the potential to induce renal tubular dysfunction. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05874440 | Recruiting | Chronic Hepatitis b Patients |
Sohag University |
April 15 2023 | -- |
| NCT03576066 | Completed | Chronic Hepatitis B |
Assembly Biosciences |
June 11 2018 | Phase 2 |
| NCT03361956 | Completed | Hepatitis B |
Janssen Sciences Ireland UC |
February 13 2018 | Phase 2 |
| NCT02985996 | Completed | HIV Infections |
Emory University|Centers for Disease Control and Prevention |
February 6 2017 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































