research use only
Prasugrel P2 Receptor antagonist
Cat.No.S1258
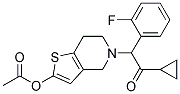
Chemical Structure
Molecular Weight: 373.44
Quality Control
| Related Targets | Adrenergic Receptor AChR 5-HT Receptor COX Calcium Channel Histamine Receptor Dopamine Receptor GABA Receptor TRP Channel Cholinesterase (ChE) |
|---|---|
| Other P2 Receptor Inhibitors | A-438079 Hydrochloride A-804598 AF-353 MRS 2578 A-740003 5-BDBD Aurintricarboxylic acid A-317491 Gefapixant JNJ-47965567 |
Solubility
|
In vitro |
DMSO
: 33 mg/mL
(88.36 mM)
Ethanol : 8 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 373.44 | Formula | C20H20FNO3S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 150322-43-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Effient, Efient, Prasita,CS-747, LY640315,PCR 4099 | Smiles | CC(=O)OC1=CC2=C(S1)CCN(C2)C(C3=CC=CC=C3F)C(=O)C4CC4 | ||
Mechanism of Action
| Targets/IC50/Ki |
P2Y12 receptor
|
|---|---|
| In vitro |
This compound is a novel orally active thienopyridine with faster, higher and more reliable inhibition of platelet aggregation than clopidogrel reflecting its metabolism in vivo to an active metabolite with selective P2Y(12) antagonistic activity. |
| In vivo |
Prasugrel shows platelet inhibition that was 8.2 times more potent than clopidogrel in WT mice. This compound (3 and 10 mg/kg) dose-relatedly and significantly reduces thrombus-mediated cerebral infarction 24 hours after the irradiation in rat models of cerebral and peripheral arterial occlusive diseases. It (0.3-3 mg/kg) reduces incidence, total area, and total number of cerebral infarcts in a dose-related manner 24 hours after the vascular injury in an embolic infarction rats model. This chemical (0.03-3 mg/kg/day) administered from the day before the lauric acid injection for 11 successive days inhibits the progression of the disease in a dose-related manner in rats with lauric acid-induced peripheral arterial occlusive diseases. It administrated in dogs (0.03-0.3 mg/kg/day) and monkeys (0.1 and 0.3 mg/kg/day) once a day for 14 days results in potent, dose-related and cumulative inhibition of ADP-induced platelet aggregation. This compound (0.1-1 mg/kg/day, p.o.) significantly prolongs the time to arterial occlusion and increases the duration of arterial patency in a rat model of electrically-induced arterial thrombosis. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-VEGDFR2 / VEGFR2 / p-STAT3 / STAT3 / p-p65 / p65 / p-AKT / AKT / p-ERK / ERK |
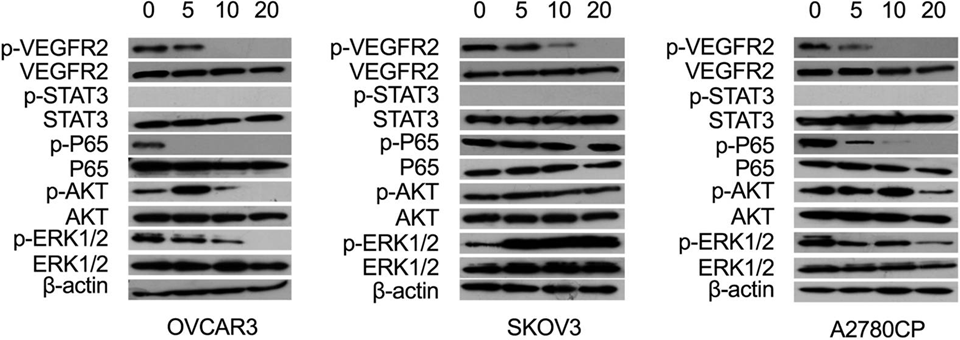
|
28383032 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05774431 | Recruiting | Acute Myocardial Infarction |
University Hospital Heidelberg|AstraZeneca |
March 13 2023 | -- |
| NCT05053828 | Completed | Type2 Diabetes|Antiplatelet Drugs|Myocardial Infarction|Heart Failure |
Tabula Rasa HealthCare |
September 20 2021 | -- |
| NCT04471870 | Completed | Acute Coronary Syndrome |
Chiesi Farmaceutici S.p.A. |
October 23 2020 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































