research use only
Pralatrexate DHFR inhibitor
Cat.No.S1497
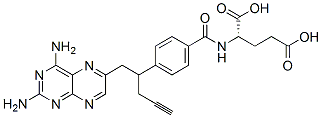
Chemical Structure
Molecular Weight: 477.47
Quality Control
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other DHFR Inhibitors | Calcium Folinate Aminopterin Diaveridine |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human KB cells | Growth inhibition assay | 96 h | Growth inhibition of human KB cells expressing human RFC/FRalpha/PCFT after 96 hrs by CellTiter-blue assay, IC50=0.47 nM | |||
| Chinese hamster R2 cells | Growth inhibition assay | 96 h | Growth inhibition of Chinese hamster R2 cells expressing human PCFT4 after 96 hrs by CellTiter-blue assay, IC50=0.057 μM | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 28 mg/mL
(58.64 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 477.47 | Formula | C23H23N7O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 146464-95-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | NSC 754230 | Smiles | C#CCC(CC1=CN=C2C(=N1)C(=NC(=N2)N)N)C3=CC=C(C=C3)C(=O)NC(CCC(=O)O)C(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
DHFR
|
|---|---|
| In vitro |
Pralatrexate and bortezomib exhibits concentration- and time-dependent cytotoxicity against a broad panel of T-lymphoma cell lines. This compound shows synergism when combined with bortezomib in all cell lines studied. It also induces potent apoptosis and caspase activation when combined with bortezomib across the panel. The agent significantly modulates the expression of p27, NOXA, HH3, and RFC-1 as assessed by Western blot assays. This compound is rationally designed for improved cellular transport via RFC-1, and to have greater intracellular drug retention through the enhanced formation of polyglutamylated conjugates. It is thought to exert its pharmacological effect primarily through inhibition of DHFR, having an IC50 in the picomolar range. This chemical demonstrates superior intracellular transport via the reduced folate carrier, and increased accumulation within cells by enhanced polyglutamylation. It exhibits antitumor activity that is superior to the activity of other antifolates. Its enhanced activity relative to methotrexate (MTX) is due to its much more rapid rate of transport and polyglutamation, the former less important when the carrier is saturated.
|
| In vivo |
Pralatrexate treatment results in treatment-related toxicity in MV522 mice models, as determined by significant weight loss in some animals prior to death; however, remaining mice regains all lost weight by Day 35.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02594267 | Completed | Peripheral T-Cell Lymphoma (PTCL) |
Acrotech Biopharma Inc.|Axis Clinicals Limited |
November 10 2015 | Phase 1 |
| NCT01947140 | Completed | Lymphoid Malignancies|Multiple Myeloma|Lymphoma|Hodgkin Lymphoma|Non-hodgkin Lymphoma |
Jennifer Amengual|Columbia University |
September 9 2013 | Phase 1|Phase 2 |
| NCT01532011 | Completed | Advanced Cancers|Solid Tumors |
M.D. Anderson Cancer Center |
March 2012 | Phase 1 |
| NCT01114282 | Completed | Multiple Myeloma |
Stanford University|National Comprehensive Cancer Network |
August 2010 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
We are just wondering if it is a racemic mixture or a monomer? Will you please let us know?
Answer:
It is the S enantiomer.






































