research use only
Peramivir Trihydrate Neuraminidase inhibitor
Cat.No.S2716
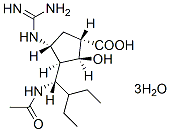
Chemical Structure
Molecular Weight: 382.45
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV |
|---|---|
| Other Neuraminidase Inhibitors | Laninamivir Octanoate Laninamivir |
Solubility
|
In vitro |
5%TFA : 3 mg/mL
DMSO
: 0.2 mg/mL
(0.52 mM)
Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 382.45 | Formula | C15H28N4O4.3H2O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1041434-82-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | BCX-1812, RWJ-270201, S-021812 | Smiles | CCC(CC)C(C1C(CC(C1O)C(=O)O)N=C(N)N)NC(=O)C.O.O.O | ||
Mechanism of Action
| In vivo |
Peramivir protects mice against severe disease outcomes following infection with highly pathogenic avian influenza and multi-dose treatment was efficacious in ferrets. Peramivir combined with Oseltamivir perform better than suboptimal doses of each compound alone to treat influenza infections in mice. Peramivir (30 mg/kg) is effective in promoting the survival of mice infected with systemically replicating H5N1 virus. Peramivir is 30% protective against H1N1 virus in mice at 0.5 mg/kg/d, but ineffective at lower doses when used as monotherapy. Peramivir combined with favipiravir increases the numbers of survivors by 10-50% when the 0.025 mg/kg/d, 0.05 mg/kg/d, and 0.1 mg/kg/d doses of Peramivir are combined with 20 mg/kg/d Favipiravir and when all doses of Peramivir are combined with 40 mg/kg/d Favipiravir. Peramivir combined with favipiravir results in an increase in lifespan, improved body weight and significant reductions in lung hemorrhage score and lung weight in H1N1 infected mice. Peramivir (1 mg/kg/d) combined with Rimantadine (5 mg/kg/d and 10 mg/kg/d) produces weight losses of 1.69 and 0.69, respectively, whereas the combination of 3.0 mg/kg/d Peramivir with 10 and 30 mg/kg/d Rimantadine does not show any weight loss in sub-lethal influenza A (H3N2) virus mouse model.
|
References |
|
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02635724 | Completed | Influenza |
BioCryst Pharmaceuticals |
December 2015 | Phase 3 |
| NCT02369159 | Completed | Influenza |
BioCryst Pharmaceuticals |
March 11 2015 | Phase 3 |
| NCT01224795 | Terminated | Influenza |
BioCryst Pharmaceuticals|Department of Health and Human Services |
October 2010 | Phase 3 |
| NCT01146353 | Withdrawn | Influenza|Renal Failure |
Midwestern University|Northwestern University|BioCryst Pharmaceuticals |
February 2010 | -- |
| NCT01063933 | Withdrawn | Influenza |
National Institute of Allergy and Infectious Diseases (NIAID) |
Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































