research use only
Diacerein Interleukins inhibitor
Cat.No.S4267
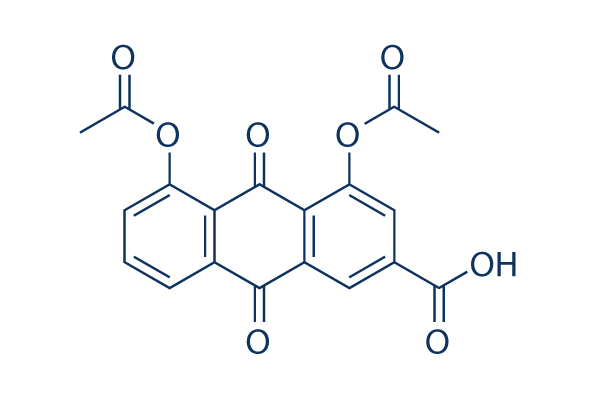
Chemical Structure
Molecular Weight: 368.29
Quality Control
| Related Targets | PD-1/PD-L1 CXCR STING AhR Immunology & Inflammation related CD markers Anti-infection Antioxidant COX Histamine Receptor |
|---|---|
| Other Interleukins Inhibitors | SC144 Ossirene (AS101) Apilimod mesylate Ac-YVAD-cmk EC330 Raleukin Diethyl phosphate Sodium Thiocyanate IQ 3 LMT-28 |
Solubility
|
In vitro |
DMSO
: 18 mg/mL
(48.87 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 368.29 | Formula | C19H12O8 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 13739-02-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC(=O)OC1=CC=CC2=C1C(=O)C3=C(C2=O)C=C(C=C3OC(=O)C)C(=O)O | ||
Mechanism of Action
| In vitro |
Diacerein significantly inhibits LPS-induced IL-1beta production by synovial tissue and cartilage. This compound (1 μM) has a significantly less inhibitory effect on cartilage synthesis than culture media containing LPS only. It decreases NO release in synovial tissue and cartilage media and increases IL-1ra levels in cartilage culture media. This chemical (10 μM) enhances the expression of TGF-beta1 and TGF-beta2 in cultured bovine articular chondrocytes. It reduces, in a dose-dependent manner, the interleukin-1-beta (IL-1beta)-induced MMP-13 production in osteoarthritic subchondral bone. This agent significantly reduces the activity of MMP-13 and cathepsin K in osteoclasts. It effectively blocks the IL-1beta effect on the osteoclast differentiation process and the survival of mature osteoclasts.
|
|---|---|
| In vivo |
Diacerein (100 mg/kg/day) significantly suppresses the paw edema and the increase in serum mucoprotein in the adjuvant-induced arthritic rats. This compound (30 mg/kg/day) combined with Naproxen (3 mg/kg/day) results in significantly greater anti-inflammatory activity than with naproxen alone in the adjuvant-induced arthritic rats. It (100 mg/kg/day) also significantly prevents bone loss and reduces the serum alkaline phosphatase and decreases the excretion of urinary hydroxyproline in the ovariectomized rats. This chemical (25 mg/kg) decreases the thickness of cartilage and subchondral bone in the lesion (middle) zone of the lateral tibial plateau in Merino wethers.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | Cyclin B1 / CDK1 / CDK2 AKT / p-AKT / p38 / p-p38 / p53 p-STAT3 / STAT3 MDM2 / CDC2 |
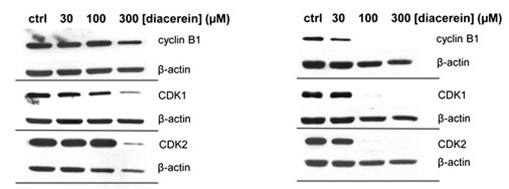
|
26555773 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03472287 | Completed | Epidermolysis Bullosa (EB)|Epidermolysis Bullosa Simplex|Dystrophic Epidermolysis Bullosa|Junctional Epidermolysis Bullosa |
Castle Creek Pharmaceuticals LLC |
May 18 2018 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































