research use only
Rivaroxaban Factor Xa inhibitor
Cat.No.S3002
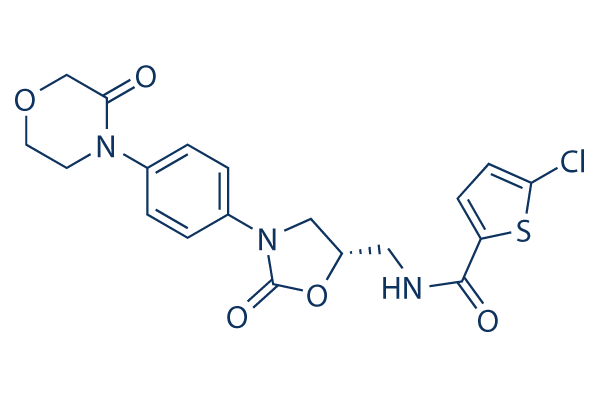
Chemical Structure
Molecular Weight: 435.88
Quality Control
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other Factor Xa Inhibitors | Asundexian |
Solubility
|
In vitro |
DMSO
: 87 mg/mL
(199.59 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 435.88 | Formula | C19H18ClN3O5S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 366789-02-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | BAY 59-7939 | Smiles | C1COCC(=O)N1C2=CC=C(C=C2)N3CC(OC3=O)CNC(=O)C4=CC=C(S4)Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Factor Xa
(Cell-free assay) 0.7 nM
Prothrombinase
(Cell-free assay) 2.1 nM
|
|---|---|
| In vitro |
Rivaroxaban is an oral, direct inhibitor of Factor Xa (FXa), being developed for the prevention and treatment of arterial and venous thrombosis with a Ki of 0.4 nM. This compound also inhibits prothrombinase activity with IC50 of 2.1 nM. It also shows a similar affinity to purified human and rabbit FXa (IC50 0.7 nM and 0.8 nM, respectively), but a lesser potency against purified rat FXa (IC50 3.4 nM). Endogenous human and rabbit FXa in plasma is inhibited to a similar extent by this chemical (IC50 21 nM and 21 nM, respectively), while 14-fold higher concentrations are required in rat plasma (IC50 290 nM). It exhibits high permeability and polarized transport across Caco-2 cells as a substrate of the P-gp, but exhibits no inhibitory effect on P-gp-mediated drug transport up to concentrations of 100 μM in vitro.
|
| Kinase Assay |
Factor Xa Activity
|
|
The activity of Rivaroxaban against purified serine proteases is measured using chromogenic or fluorogenic substrates in 96-well microtiter plates. The enzymes are incubated with this compound or its solvent, dimethyl sulfoxide (DMSO), for 10 minutes. The reactions are initiated by the addition of the substrate, and the color or fluorescence is monitored continuously at 405 nm using a Spectra Rainbow Thermo Reader, or at 630/465 nm using a SPECTRAfluor plus, respectively, for 20 minutes. Enzymatic activity is analyzed in the following buffers (final concentrations): human FXa (0.5 nM), rabbit FXa (2 nM), rat FXa (10 nM), or urokinase (4 nM) in 50 mM Tris–HCl buffer pH 8.3, 150 mM NaCl, and 0.1% bovine serum albumin (BSA); Pefachrome FXa (50–800 μM) or chromozym U (250 μM) with thrombin (0.69 nM), trypsin (2.2 nM), or plasmin (3.2 nM) in 0.1 μM Tris–HCl, pH 8.0, and 20 mM CaCl2; chromozym TH (200 μM), chromozym plasmin (500 μM), or chromozym trypsin (500 μM) with FXIa (1 nM) or APC (10 nM) in 50mM phosphate buffer, pH 7.4, 150 mM NaCl; and S 2366 (150 or 500 μM) with FVIIa (1 nM) and tissue factor (3 nM) in 50 mM Tris–HCl buffer,pH 8.0, 100 mM NaCl, 5 mM CaCl2 and 0.3% BSA, H-D-Phe-Pro-Arg-6-amino-1-naphthalene-benzylsulfonamide-H2O (100 μM) and measured for 3 hours. The FIXaβ/FX assay, comprising FIXaβ (8.8 nM) and FX (9.5 nM) in 50 mM Tris–HCl buffer, pH 7.4, 100 mM NaCl, 5 mM CaCl2 and 0.1% BSA, is started by the addition of I-1100 (50 μM), and measured for 60 minutes. The inhibitory constant (Ki) against FXa is calculated according to the Cheng–Prusoff equation. The IC50 is the amount of inhibitor required to diminish the initial velocity of the control by 50%.
|
|
| In vivo |
Rivaroxaban reduces venous thrombosis in a dose dependent manner (ED50 0.1 mg/kg i.v.) in a rat venous stasis model. This compound reduces arterial thrombus formation in an arteriovenous (AV) shunt in rats (ED50 5.0 mg/kg p.o.) and rabbits (ED50 0.6 mg/kg p.o.). Plasma pharmacokinetics of this chemical are linear across the investigated dose range (1-10 mg/kg in rats, 0.3-3 mg/kg in dogs). Plasma clearance is low: 0.4 L/kg/h in rats and 0.3 L/kg/h in dogs; the volume of distribution (V(ss)) is moderate: 0.3 L/kg in rats, and 0.4 L/kg in dogs. The elimination half-life after oral administration is short in both species (0.9-2.3 hours).
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06193863 | Not yet recruiting | Prevention of Venous Thromboembolism|Congenital Heart Disease|Fontan Procedure|Children |
Bayer|Janssen Research & Development LLC |
May 31 2024 | -- |
| NCT06195540 | Not yet recruiting | Venous Thromboembolism|Deep Vein Thrombosis|Pulmonary Embolism|Lower Limb Trauma|Thromboprophylaxis|Immobilisation |
University Hospital Angers |
May 31 2024 | Phase 3 |
| NCT06314763 | Completed | Drug Drug Interaction Study |
Radboud University Medical Center|Amgen |
November 9 2023 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































