research use only
Albendazole ADC Cytotoxin inhibitor
Cat.No.S1640
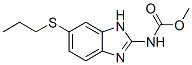
Chemical Structure
Molecular Weight: 265.33
Quality Control
| Related Targets | Akt Wnt/beta-catenin HSP PKC ROCK Microtubule Associated Integrin Bcr-Abl Actin FAK |
|---|---|
| Other ADC Cytotoxin Inhibitors | Triptolide SN-38 Luteolin (+)-Bicuculline Rutin Artemisinin Pinocembrin Psoralen Luteoloside Lycorine hydrochloride |
Solubility
|
In vitro |
DMSO
: 17 mg/mL
(64.07 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 265.33 | Formula | C12H15N3O2S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 54965-21-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | SKF-62979 | Smiles | CCCSC1=CC2=C(C=C1)N=C(N2)NC(=O)OC | ||
Mechanism of Action
| Targets/IC50/Ki |
tubulin
|
|---|---|
| In vitro |
Albendazole is reported to be teratogenic in rats, and is extensively metabolized to the sulfoxide derivative. This compound and its sulfoxide metabolite elicit an accumulation of cells in the mitotic phase of the cell cycle.
|
| In vivo |
Albendazole causes an induction of hepatic activities of CYP1A1-associated ethoxyresorufin O-deethylase (EROD) 65 fold, CYP1A2-associated methoxyresorufin O-demethylase (MROD) 6 fold, CYP2B1-associated penthoxyresorufin O-dealkylase (PROD) 4 fold, CYP2B2-associated benzyloxyresorufin O-dealkylase (BROD) 14 fold, as well as a partial reduction of CYP2E1-associated 4-nitrophenol hydroxylase (4-NPH) activity in the rat. This compound is metabolized to its pharmacologically active sulfoxide metabolites by liver and lung microsomes from sheep and cattle, as well as by cattle intestinal microsomes. This benzimidazole derivative has a broad spectrum of activity against human and animal helminthe parasites. It and its metabolites are extensively distributed to the digestive tract, mainly into the abomasal fluid, after the i.v. and i.r. administrations. This compound and its active albendazole sulphoxide (ABZSO) metabolite are recovered in tapeworms collected from both i.v. and i.r. treated lambs.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06201559 | Completed | Intestinal Diseases |
GlaxoSmithKline |
August 21 2023 | Phase 1 |
| NCT05453045 | Not yet recruiting | Pharmacological Action |
Universidad Nacional de Salta|Fundacion Mundo Sano |
July 18 2022 | Not Applicable |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































