
- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-DYKDDDDK Tag magnetic beads
- Anti-DYKDDDDK Tag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
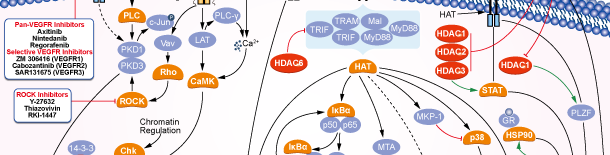
HDAC
Isoform-selective Products
Signaling Pathway
HDAC Products
- All (96)
- HDAC Inhibitors (86)
- HDAC Activator (1)
- New HDAC Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
| S1047 | Vorinostat (SAHA) | Vorinostat (SAHA) is an HDAC inhibitor with IC50 of ~10 nM in a cell-free assay. Vorinostat abrogates productive HPV-18 DNA amplification. |

|
|
| S1053 | Entinostat (MS-275) | Entinostat (MS-275, SNDX-275) strongly inhibits HDAC1 and HDAC3 with IC50 of 0.51 μM and 1.7 μM in cell-free assays, compared with HDACs 4, 6, 8, and 10. Entinostat induces autophagy and apoptosis. Phase 3. |
|
|
| S1030 | Panobinostat (LBH589) | Panobinostat (LBH589, NVP-LBH589) is a novel broad-spectrum HDAC inhibitor with IC50 of 5 nM in a cell-free assay. Panobinostat (LBH589) induces autophagy and apoptosis. Panobinostat effectively disrupts HIV latency in vivo. Phase 3. |
|
|
| S1045 | TSA (Trichostatin A) | TSA (Trichostatin A) is an HDAC inhibitor with IC50 of ~1.8 nM in cell-free assays. |
|
|
| S3020 | Romidepsin | Romidepsin (FK228, Depsipeptide, FR 901228, NSC 630176) is a potent HDAC1 and HDAC2 inhibitor with IC50 of 36 nM and 47 nM in cell-free assays, respectively. Romidepsin (FK228/depsipeptide) controls growth and induces apoptosis in neuroblastoma tumor cells. |
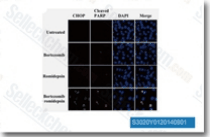
|
|
| S1122 | Mocetinostat (MGCD0103) | Mocetinostat (MGCD0103, MG0103) is a potent HDAC inhibitor with most potency for HDAC1 with IC50 of 0.15 μM in a cell-free assay, 2- to 10- fold selectivity against HDAC2, 3, and 11, and no activity to HDAC4, 5, 6, 7, and 8. Mocetinostat (MGCD0103) induces apoptosis and autophagy. Phase 2. |

|
|
| S7229 | RGFP966 | RGFP966 is an HDAC3 inhibitor with IC50 of 0.08 μM in cell-free assay, exhibits > 200-fold selectivity over other HDAC. |

|
|
| S1085 | Belinostat | Belinostat is a novel HDAC inhibitor with IC50 of 27 nM in a cell-free assay, with activity demonstrated in cisplatin-resistant tumors. Belinostat (PXD101) induces autophagy. |

|
|
| S8049 | Tubastatin A | Tubastatin A is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective against all the other isozymes (1000-fold) except HDAC8 (57-fold). Tubastatin A promotes autophagy and increases apoptosis. |
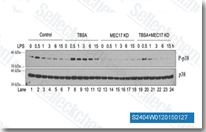
|
|
| S8001 | Ricolinostat (ACY-1215) | Ricolinostat (ACY-1215, Rocilinostat) is a selective HDAC6 inhibitor with IC50 of 5 nM in a cell-free assay. It is >10-fold more selective for HDAC6 than HDAC1/2/3 (class I HDACs) with slight activity against HDAC8, minimal activity against HDAC4/5/7/9/11, Sirtuin1, and Sirtuin2. Ricolinostat (ACY-1215) suppresses cell proliferation and promotes apoptosis. Phase 2. |
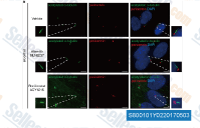
|
|
| S1096 | Quisinostat (JNJ-26481585) 2HCl | Quisinostat (JNJ-26481585) 2HCl is a novel second-generation HDAC inhibitor with highest potency for HDAC1 with IC50 of 0.11 nM in a cell-free assay, modest potent to HDACs 2, 4, 10, and 11; greater than 30-fold selectivity against HDACs 3, 5, 8, and 9 and lowest potency to HDACs 6 and 7. Phase 2. |
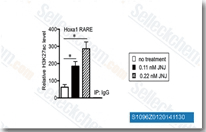
|
|
| S1484 | MC1568 | MC1568 is a selective HDAC inhibitor for maize HD1-A with IC50 of 100 nM in a cell-free assay. It is 34-fold more selective for HD1-A than HD1-B. |
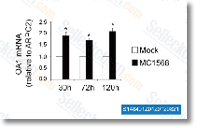
|
|
| S2627 | Tubastatin A HCl | Tubastatin A HCl is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective (1000-fold more) against all other isozymes except HDAC8 (57-fold more). |

|
|
| S2012 | PCI-34051 | PCI-34051 is a potent and specific HDAC8 inhibitor with IC50 of 10 nM in a cell-free assay. It has greater than 200-fold selectivity over HDAC1 and 6, more than 1000-fold selectivity over HDAC2, 3, and 10. PCI-34051 induces caspase-dependent apoptosis. |
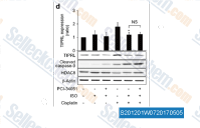
|
|
| S1848 | Curcumin | Curcumin (Diferuloylmethane, Natural Yellow 3, Turmeric yellow) is the principal curcuminoid of the popular Indian spice turmeric, which is a member of the ginger family (Zingiberaceae). It is an inhibitor of p300 histone acetylatransferase(IC50~25 μM)and Histone deacetylase (HDAC); activates Nrf2 pathway and supresses the activation of NF-κB. Curcumin induces mitophagy, autophagy, apoptosis, and cell cycle arrest with antitumor activity. Curcumin reduces renal damage associated with rhabdomyolysis by decreasing ferroptosis-mediated cell death. Curcumin exhibits anti-infective properties against various human pathogens like the influenza virus, hepatitis C virus, HIV and so on. |

|
|
| S2818 | Tacedinaline (CI994) | Tacedinaline (CI994, PD-123654, GOE-5549, Acetyldinaline) is a selective class I HDAC inhibitor with IC50 of 0.9, 0.9, 1.2, and >20 μM for human HDAC 1, 2, 3, and 8, respectively. Phase 3. |

|
|
| S7569 | LMK-235 | LMK-235 is a selective inhibitor of HDAC4 and HDAC5 with IC50 of 11.9 nM and 4.2 nM, respectively. |
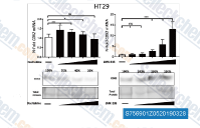
|
|
| S2759 | Fimepinostat (CUDC-907) | CUDC-907 is a dual PI3K and HDAC inhibitor for PI3Kα and HDAC1/2/3/10 with IC50 of 19 nM and 1.7 nM/5 nM/1.8 nM/2.8 nM, respectively. CUDC-907 induces cell cycle arrest and apoptosis in breast cancer cells. Phase 1. |
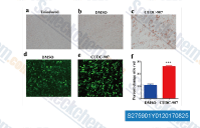
|
|
| S2239 | Tubacin | Tubacin is a highly potent and selective, reversible, cell-permeable HDAC6 inhibitor with an IC50 of 4 nM in a cell-free assay, approximately 350-fold selectivity over HDAC1. Tubacin reduces the replication of the Japanese Encephalitis Virus via the decrease of viral RNA synthesis. |
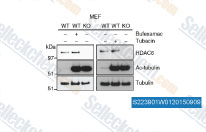
|
|
| S2170 | Givinostat hydrochloride monohydrate | Givinostat (ITF2357) is a potent HDAC inhibitor for maize HD2, HD1B and HD1A with IC50 of 10 nM, 7.5 nM and 16 nM in cell-free assays. Phase 2. |

|
|
| S1168 | Valproic Acid sodium | Valproic Acid sodium is a HDAC inhibitor by selectively inducing proteasomal degradation of HDAC2, used in the treatment of epilepsy, bipolar disorder and prevention of migraine headaches. Valproic acid induces Notch1 signaling in small cell lung cancer (SCLC) cells. Valproic acid is under investigation for treatment of HIV and various cancers. Valproic acid (VPA) induces autophagy and mitophagy by upregulation of BNIP3 and mitochondrial biogenesis by upregulating PGC-1α. |

|
|
| S3944 | VPA (Valproic acid) | VPA (Valproic acid) is a fatty acid with anticonvulsant properties used in the treatment of epilepsy. It is also a histone deacetylase (HDAC) inhibitor and is under investigation for treatment of HIV and various cancers. Valproic acid (VPA) induces autophagy and mitophagy by upregulation of BNIP3 and mitochondrial biogenesis by upregulating PGC-1α. Valproic acid activates Notch-1 signaling. | ||
| S2244 | AR-42 | AR-42 (HDAC-42) is an HDAC inhibitor with IC50 of 30 nM. Phase 1. |
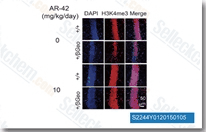
|
|
| S7324 | TMP269 | TMP269 is a potent, selective class IIa HDAC inhibitor with IC50 of 157 nM, 97 nM, 43 nM and 23 nM for HDAC4, HDAC5, HDAC7 and HDAC9, respectively. |
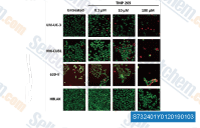
|
|
| S1999 | Sodium butyrate | Sodium butyrate (NaB, Butanoic acid sodium salt), sodium salt of butyric acid, is a histone deacetylase inhibitor and competitively binds to the zinc sites of class I and II histone deacetylases (HDACs). Sodium butyrate (NaB) inhibits cell cycle progression, promotes differentiation, and induces apoptosis and autophagy in several types of cancer cells. |
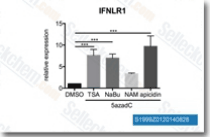
|
|
| S1515 | Pracinostat (SB939) | Pracinostat (SB939) is a potent pan-HDAC inhibitor with IC50 of 40-140 nM with exception for HDAC6. It has no activity against the class III isoenzyme SIRT I. Pracinostat (SB939) induces apoptosis in tumor cells. Phase 2. |
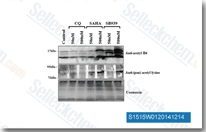
|
|
| S1194 | CUDC-101 | CUDC-101 is a potent multi-targeted inhibitor against HDAC, EGFR and HER2 with IC50 of 4.4 nM, 2.4 nM, and 15.7 nM, and inhibits class I/II HDACs, but not class III, Sir-type HDACs. Phase 1. |
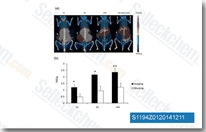
|
|
| S1090 | Abexinostat (PCI-24781) | Abexinostat (PCI-24781, CRA-024781) is a novel pan-HDAC inhibitor mostly targeting HDAC1 with Ki of 7 nM, modest potent to HDACs 2, 3, 6, and 10 and greater than 40-fold selectivity against HDAC8. Phase 1/2. |
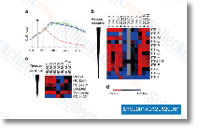
|
|
| S7595 | Santacruzamate A (CAY10683) | Santacruzamate A (CAY10683) is a potent and selective HDAC inhibitor with IC50 of 119 pM for HDAC2, >3600-fold selectivity over other HDACs. |

|
|
| S2341 | (-)-Parthenolide | (-)-Parthenolide, an inhibitor of the Nuclear Factor-κB Pathway, specifically depletes HDAC1 protein without affecting other class I/II HDACs; Also promotes the ubiquitination of MDM2 and activates p53 cellular functions. |
-Parthenolide-S234101W0220161110.gif)
|
|
| S1095 | Dacinostat (LAQ824) | Dacinostat (LAQ824, NVP-LAQ824) is a novel HDAC inhibitor with IC50 of 32 nM and is known to activate the p21 promoter. |

|
|
| S7596 | CAY10603 | CAY10603 (BML-281) is a potent and selective HDAC6 inhibitor with IC50 of 2 pM, >200-fold selectivity over other HDACs. |
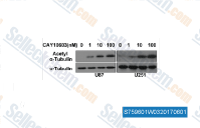
|
|
| S7292 | RG2833 (RGFP109) | RG2833 (RGFP109) is a brain-penetrant HDAC inhibitor with IC50 of 60 nM and 50 nM for HDAC1 and HDAC3 in cell-free assays, respectively. |

|
|
| S7617 | Tasquinimod | Tasquinimod (ABR-215050) is an orally active antiangiogenic agent by allosterically inhibiting HDAC4 signalling. Phase 3. |

|
|
| S4125 | 4-PBA (Sodium Phenylbutyrate) | 4-PBA (Sodium Phenylbutyrate) is a salt of 4-phenylbutyrate (4-PBA) or 4-phenylbutyric acid.Sodium phenylbutyrate is a histone deacetylase inhibitor, used to treat urea cycle disorders. |
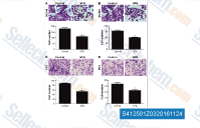
|
|
| S8567 | Tucidinostat (Chidamide) | Tucidinostat (Chidamide, HBI-8000, CS-055) is a low nanomolar inhibitor of HDAC1, 2, 3, and 10, the HDAC isotypes well documented to be associated with the malignant phenotype with IC50 values of 95, 160, 67, 78 nM for HDAC1, 2, 3, 10 respectively. | ||
| S8502 | TMP195 | TMP195 (TFMO 2) is a selective, first-in-class, class IIa HDAC inhibitor with Ki of 59, 60, 26 and 15nM for HDAC4, HDAC5, HDAC7 and HDAC9, respectively. | ||
| S7473 | Nexturastat A | Nexturastat A is a potent and selective HDAC6 inhibitor with IC50 of 5 nM, >190-fold selectivity over other HDACs. |
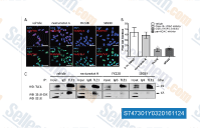
|
|
| S1422 | Droxinostat | Droxinostat (NS 41080) is a selective inhibitor of HDAC, mostly for HDACs 6 and 8 with IC50 of 2.47 μM and 1.46 μM, greater than 8-fold selective against HDAC3 and no inhibition to HDAC1, 2, 4, 5, 7, 9, and 10. |
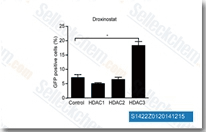
|
|
| S7555 | Domatinostat (4SC-202) | Domatinostat (4SC-202) is a selective class I HDAC inhibitor with IC50 of 1.20 μM, 1.12 μM, and 0.57 μM for HDAC1, HDAC2, and HDAC3, respectively. Also displays inhibitory activity against Lysine specific demethylase 1 (LSD1). Phase 1. | ||
| S8043 | Scriptaid | Scriptaid (GCK 1026) is an inhibitor of HDAC. It shows a greater effect on acetylated H4 than H3. |
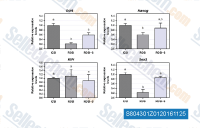
|
|
| S3592 | 4-PBA (4-Phenylbutyric acid) | 4-PBA (4-Phenylbutyric acid) is a histone deacetylase (HDAC) inhibitor and a key epigenetic inducer of anti-HCV hepatic hepcidin. 4-Phenylbutyric acid inhibits LPS-induced inflammation through regulating endoplasmic-reticulum (ER) stress and autophagy in acute lung injury models. | ||
| S2779 | M344 | M344 is a potent HDAC inhibitor with IC50 of 100 nM and able to induce cell differentiation. |

|
|
| S2693 | Resminostat | Resminostat (RAS2410) dose-dependently and selectively inhibits HDAC1/3/6 with IC50 of 42.5 nM/50.1 nM/71.8 nM, less potent to HDAC8 with IC50 of 877 nM. |
|
|
| S8464 | Citarinostat (ACY-241) | Citarinostat (ACY-241, HDAC-IN-2) is an orally available selective HDAC6 inhibitor with IC50 of 2.6 nM and 46 nM for HDAC6 and HDAC3, respectively. It has 13 to 18-fold selectivity towards HDAC6 in comparison to HDAC1-3. | ||
| S7689 | BG45 | BG45 is a class I HDAC inhibitor with IC50 of 289 nM, 2.0 µM, 2.2 µM and >20 µM for HDAC3, HDAC1, HDAC2, and HDAC6 in cell-free assays, respectively. | ||
| S8495 | WT161 | WT161 is a potent, selective, and bioavailable HDAC6 inhibitor with IC50 values of 0.4 nM, 8.35 nM and 15.4 nM for HDAC6, HDAC1 and HDAC2, respectively; shown to have >100-fold selectivity over other HDACs. WT161 induces apoptosis. | ||
| S8648 | ACY-738 | ACY-738 inhibits HDAC6 with low nanomolar potency (IC50=1.7 nM) and a selectivity of 60- to 1500-fold over class I HDACs. | ||
| S7278 | HPOB | HPOB is a potent, selective HDAC6 inhibitor with IC50 of 56 nM, >30-fold selectivity over other HDACs. |
|
|
| S8323 | ITSA-1 (ITSA1) | ITSA-1 (ITSA1) is an HDAC activator via TSA suppression, but shows no activity towards other HDAC inhibitors. | ||
| S0709 | Tubastatin A TFA | Tubastatin A TFA (Tubastatin A trifluoroacetate salt) is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective against all the other isozymes (1000-fold) except HDAC8 (57-fold). Tubastatin A promotes autophagy and increases apoptosis. | ||
| S6738 | TC-H 106 | TC-H 106 (Pimelic Diphenylamide 106) is a slow, tight-binding inhibitor of class I histone deacetylases(HDAC) with Ki value of 148 nM, about 102 nM, 14 nM for HDAC1, HDAC2, HDAC3, respectively. | ||
| S6687 | SIS17 | SIS17 is a mammalian histone deacetylase 11 (HDAC 11)-specific inhibitor with IC50 of 0.83 μM. SIS17 inhibits the demyristoylation of HDAC11 substrate, serine hydroxymethyl transferase 2, without inhibiting other HDACs. | ||
| S1703 | Divalproex Sodium | Divalproex Sodium, consisting of a compound of sodium valproate and valproic acid in a 1:1 molar relationship in an enteric coated form, is a HDAC inhibitor, used in the treatment for epilepsy. |
|
|
| S0864 | ACY-775 | ACY-775 is a potent and selective histone deacetylase 6 (HDAC6) inhibitor with IC50 of 7.5 nM. | ||
| S1073 | BML-210 (CAY10433) | BML-210 (CAY10433) is a small molecule inhibitor of HDAC.BML-210 inhibits the HDAC4-VP16-driven reporter signal in a dose-dependent manner with an apparent IC50 of ∼5 µM. | ||
| S8773 | TH34 | TH34 is a HDAC inhibitor that shows pronounced selectivity for HDACs 6, 8 and 10 over HDACs 1, 2 and 3. In a NanoBRET assay, TH34 strongly binds HDAC6, 8 and 10 with low-micromolar IC50 concentrations (HDAC6: 4.6 µM, HDAC8: 1.9 µM, HDAC10: 7.7 µM). | ||
| S9262 | Raddeanin A | Raddeanin A (Raddeanin R3, NSC382873), a triterpenoid saponin from Anemone raddeana Regel, displays moderate inhibitory activity against histone deacetylases (HDACs) and has high antiangiogenic potency, antitumor activity. | ||
| F0735New | HDAC1 Rabbit mAb | |||
| F0838New | HDAC3 Rabbit mAb | HDAC3,Histone Deacetylase 3 (HDAC3) | ||
| F0948New | HDAC5 Rabbit mAb | HDAC5,Histone Deacetylase 5 (HDAC5) | ||
| F0480New | HDAC4 Rabbit mAb | |||
| F2424New | HDAC9 Rabbit mAb | |||
| F1252New | HDAC7 Rabbit mAb | |||
| F2989New | CTBP2 Rabbit mAb | |||
| F2218New | HDAC8 Rabbit mAb | |||
| F1413New | SIN3A Rabbit mAb | mSin3A,SIN3A | ||
| S3981 | Sinapinic Acid | Sinapinic acid (Sinapic acid) is a small naturally occurring hydroxycinnamic acid which belongs to phenylpropanoid family and commonly used as matrix in MALDI mass spectrometry. Sinapinic acid (Sinapic acid) acts as an inhibitor of HDAC, with IC50 of 2.27 mM, and also inhibits ACE-I activity. | ||
| E5952New | PT 3 | PT 3 is a benzamide-class, potent, and selective inhibitor of Histone Deacetylase 3 (HDAC3) with an IC50 of 245 nM. It crosses the blood-brain barrier and enhances learning and memory in novel object recognition models in mice. | ||
| E8302New | YX968 | YX968 is a potent, rapid, and selective PROTAC dual degrader of HDAC3/8 with DC50 values of 1.7 nM and 6.1 nM, respectively. It promotes apoptosis and kills cancer cells with a high potency in vitro and can be used in cancer research. | ||
| E8292New | GEM144 | GEM144 is a potent and orally active dual inhibitor of DNA polymerase α (POLA1) and HDAC11 . It exhibits potent antiproliferative activity in vitro and antitumor efficacy in vivo by inducing p53 acetylation, p21 activation, G1/S cell cycle arrest, and apoptosis. | ||
| E1086 | KT-531 | KT-531 (KT531) is a potent, selective HDAC6 inhibitor with IC50 of 8.5 nM, displays 39-fold selectivity over other HDAC isoforms. | ||
| E1912New | B102(PARP/HDAC-IN-1) | B102(PARP/HDAC-IN-1) is a dual inhibitor of PARP and HDAC, with IC50 of 19.01 nM, 2.13 nM, 1.69 μM, > 10 μM for PARP1, PARP2, HDAC1, HDAC6, respectively, in an enzymatic assay. It exhibits anti-proliferative and anti-tumor effects. It also induces DNA damage and apoptosis in vitro. | ||
| E1686New | SW-100 | SW-100 is a selective inhibitor of histone deacetylase 6 (HDAC6) with an IC50 of 2.3 nM. It displays 1000-fold selectivity for HDAC6 relative to all other HDAC isozymes. | ||
| E1580New | TNG260 | TNG260 is an inhibitor of CoREST-selective deacetylase (CoreDAC) that inhibits HDAC1 with 10-fold selectivity over HDAC3. It decreases intratumoral neutrophil infiltration and exhibits immune-mediated cell killing. | ||
| E1469 | ITF3756 | ITF3756 is a selective histone deacetylase 6 (HDAC6) inhibitor bearing a pentaheterocyclic scaffold. It can be used in research of autoimmune disorders, neurodegenerative diseases, and cancer. | ||
| E1554 | Tefinostat(CHR-2845) | Tefinostat(CHR-2845) is a monocyte/macrophage-targeted inhibitor of pan-histone deacetylase (HDAC). Tefinostat undergoes cleavage mediated by the intracellular esterase human carboxylesterase-1 (hCE-1) to yield the active acid CHR-2847. In vitro studies reveal a specific heightened sensitivity of monocytoid leukemias to tefinostat. | ||
| S2190 | Pyroxamide (NSC 696085) | Pyroxamide(NSC 696085) is a potent inhibitor of affinity-purified HDAC1 with ID50 of 100 nM. It also induces growth suppression and cell death in human rhabdomyosarcoma in vitro. | ||
| S5810 | UF010 | UF010 is a class I HDAC-selective inhibitor with IC50 values of 0.5 nM, 0.1 nM, 0.06 nM, 1.5 nM, 9.1 nM and 15.3 nM for HDAC1, HDAC2, HDAC3, HDAC8, HDAC6 and HDAC10, respectively. | ||
| S8743 | SKLB-23bb | SKLB-23bb is an orally bioavailable HDAC6-selective inhibitor with IC50 values under 100 nmol/L, against most of the cell lines checked. It also has microtubule-disrupting ability. | ||
| S9275 | Isoguanosine | Isoguanosine (Crotonoside) inhibits FLT3 and HDAC3/6 for the treatment of AML.Isoguanosine is a naturally occurring active isomer of guanosine that is found in the seeds of Croton tiglium. | ||
| S6548 | NKL 22 | NKL 22 is a selective inhibitor of HDAC with IC50 of 78 µM. | ||
| E0391New | BRD-6929 | BRD-6929 is an inhibitor of histone deacetylase 1 (HDAC1) and HDAC2 with an IC50s of 0.04 µM and 0.1 µM, respectively. It exhibits antiproliferative activities against a human cancer cell line (HCT116) and in human mammary epithelial cells (HMEC). | ||
| S5771 | Sulforaphane | Sulforaphane is a naturally occurring isothiocyanate derived from the consumption of cruciferous vegetables, such as broccoli, cabbage, and kale. It is an inducer of Nrf2. Sulforaphane is also an inhibitor of histone deacetylase (HDAC) and NF-κB. Sulforaphane increases heme oxygenase-1 (HO-1) and reduces the levels of reactive oxygen species (ROS). Sulforaphane induces cell cycle arrest and apoptosis., | ||
| S7726 | BRD73954 | BRD73954 is a potent and selective HDAC inhibitor with IC50 of 36 nM and 120 nM for HDAC6 and HDAC8, respectively. | ||
| E0812 | CXD101 | CXD101 (HDAC-IN-4) is a potent, selective and orally active class I histone deacetylase (HDAC) inhibitor with IC50s of 63 nM, 570 nM and 550 nM for HDAC1, HDAC2 and HDAC3, respectively. | ||
| S5905 | Suberohydroxamic acid | Suberohydroxamic acid (suberic bishydroxamic acid) is a competitive HDAC inhibitor with IC50 values of 0.25 and 0.3 μM for HDAC1 and HDAC3 respectively. | ||
| S8962 | BRD3308 | BRD3308 is a potant and highly selective inhibitor of HDAC3 with IC50 of 54 nM, 1.26 μM and 1.34 μM for HDAC3, HDAC1 and HDAC2, respectively. BRD3308 activates HIV-1 transcription. BRD3308 suppresses pancreatic β-cell apoptosis induced by inflammatory cytokines (glucolipotoxic stress) and increases functional insulin release. | ||
| S7593 | Splitomicin | Splitomicin is a selective NAD(+)-dependent histone deacetylase Sir2p inhibitor with IC50 of 60 μM, showing a higher activity in a cell-based assay. | ||
| E7940New | ACY-957 | ACY-957 is a selective chemical inhibitor of HDAC1 and HDAC2 with an IC50 values of 7 nM, 18 nM against HDAC1/2 respectively. It induces hemoglobin protein (HbF) in primary erythroid progenitor cells, including those from sickle cell patients, offering a promising approach for treating sickle cell disease (SCD) and β-thalassemia. | ||
| S5438 | Biphenyl-4-sulfonyl chloride | Biphenyl-4-sulfonyl chloride (p-Phenylbenzenesulfonyl, 4-Phenylbenzenesulfonyl, p-Biphenylsulfonyl) is a HDAC inhibitor with synthetic applications in palladium-catalyzed desulfitative C-arylation. | ||
| S9934 | KA2507 | KA2507, a potent, orally active and selective HDAC6 inhibitor with IC50 of 2.5 nM, shows antitumor activities and immune modulatory effects in preclinical models. | ||
| S2132 | SR-4370 | SR-4370 is a potent and selective inhibitor of class I HDACs with IC50 of 0.13 µM, 0.58 µM, 0.006 µM, 2.3 µM, 3.7 µM for HDAC 1, HDAC 2, HDAC 3, HDAC 8, HDAC 6, respectively.SR-4370 suppresses AR signaling and in vivo prostate tumor growth. | ||
| E8178New | Quisinostat | Quisinostat(JNJ-26481585) is a potent and orally active second-generation inhibitor of pan-Histone Deacetylase (HDAC). It inhibits HDAC1, HDAC2, HDAC4, HDAC10, HDAC11 with IC50s of 0.11 nM , 0.33 nM , 0.64 nM , 0.46 nM, 0.37 nM respectively. It exhibits potent in vivo antitumor activity and potential clinical use in various solid and hematologic malignancies. | ||
| S1313 | GSK3117391 | GSK3117391 (GSK3117391A, HDAC-IN-3) is a potent histone deacetylase (HDAC) inhibitor. | ||
| S8769 | Tinostamustine(EDO-S101) | Tinostamustine(EDO-S101) is a first-in-class alkylating deacetylase inhibitor with IC50 values of 9 nM, 9 nM, 25 nM and 107 nM for HDAC1, HDAC2, HDAC3 and HDAC8 (Class 1 HDACs) respectively and 6 nM, 72 nM for HDAC6 and HDAC10 (Class II HDACs). | ||
| S1047 | Vorinostat (SAHA) | Vorinostat (SAHA) is an HDAC inhibitor with IC50 of ~10 nM in a cell-free assay. Vorinostat abrogates productive HPV-18 DNA amplification. |

|
|
| S1053 | Entinostat (MS-275) | Entinostat (MS-275, SNDX-275) strongly inhibits HDAC1 and HDAC3 with IC50 of 0.51 μM and 1.7 μM in cell-free assays, compared with HDACs 4, 6, 8, and 10. Entinostat induces autophagy and apoptosis. Phase 3. |

|
|
| S1030 | Panobinostat (LBH589) | Panobinostat (LBH589, NVP-LBH589) is a novel broad-spectrum HDAC inhibitor with IC50 of 5 nM in a cell-free assay. Panobinostat (LBH589) induces autophagy and apoptosis. Panobinostat effectively disrupts HIV latency in vivo. Phase 3. |

|
|
| S1045 | TSA (Trichostatin A) | TSA (Trichostatin A) is an HDAC inhibitor with IC50 of ~1.8 nM in cell-free assays. |

|
|
| S3020 | Romidepsin | Romidepsin (FK228, Depsipeptide, FR 901228, NSC 630176) is a potent HDAC1 and HDAC2 inhibitor with IC50 of 36 nM and 47 nM in cell-free assays, respectively. Romidepsin (FK228/depsipeptide) controls growth and induces apoptosis in neuroblastoma tumor cells. |

|
|
| S1122 | Mocetinostat (MGCD0103) | Mocetinostat (MGCD0103, MG0103) is a potent HDAC inhibitor with most potency for HDAC1 with IC50 of 0.15 μM in a cell-free assay, 2- to 10- fold selectivity against HDAC2, 3, and 11, and no activity to HDAC4, 5, 6, 7, and 8. Mocetinostat (MGCD0103) induces apoptosis and autophagy. Phase 2. |

|
|
| S7229 | RGFP966 | RGFP966 is an HDAC3 inhibitor with IC50 of 0.08 μM in cell-free assay, exhibits > 200-fold selectivity over other HDAC. |

|
|
| S1085 | Belinostat | Belinostat is a novel HDAC inhibitor with IC50 of 27 nM in a cell-free assay, with activity demonstrated in cisplatin-resistant tumors. Belinostat (PXD101) induces autophagy. |

|
|
| S8049 | Tubastatin A | Tubastatin A is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective against all the other isozymes (1000-fold) except HDAC8 (57-fold). Tubastatin A promotes autophagy and increases apoptosis. |

|
|
| S8001 | Ricolinostat (ACY-1215) | Ricolinostat (ACY-1215, Rocilinostat) is a selective HDAC6 inhibitor with IC50 of 5 nM in a cell-free assay. It is >10-fold more selective for HDAC6 than HDAC1/2/3 (class I HDACs) with slight activity against HDAC8, minimal activity against HDAC4/5/7/9/11, Sirtuin1, and Sirtuin2. Ricolinostat (ACY-1215) suppresses cell proliferation and promotes apoptosis. Phase 2. |

|
|
| S1096 | Quisinostat (JNJ-26481585) 2HCl | Quisinostat (JNJ-26481585) 2HCl is a novel second-generation HDAC inhibitor with highest potency for HDAC1 with IC50 of 0.11 nM in a cell-free assay, modest potent to HDACs 2, 4, 10, and 11; greater than 30-fold selectivity against HDACs 3, 5, 8, and 9 and lowest potency to HDACs 6 and 7. Phase 2. |

|
|
| S1484 | MC1568 | MC1568 is a selective HDAC inhibitor for maize HD1-A with IC50 of 100 nM in a cell-free assay. It is 34-fold more selective for HD1-A than HD1-B. |

|
|
| S2627 | Tubastatin A HCl | Tubastatin A HCl is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective (1000-fold more) against all other isozymes except HDAC8 (57-fold more). |

|
|
| S2012 | PCI-34051 | PCI-34051 is a potent and specific HDAC8 inhibitor with IC50 of 10 nM in a cell-free assay. It has greater than 200-fold selectivity over HDAC1 and 6, more than 1000-fold selectivity over HDAC2, 3, and 10. PCI-34051 induces caspase-dependent apoptosis. |

|
|
| S1848 | Curcumin | Curcumin (Diferuloylmethane, Natural Yellow 3, Turmeric yellow) is the principal curcuminoid of the popular Indian spice turmeric, which is a member of the ginger family (Zingiberaceae). It is an inhibitor of p300 histone acetylatransferase(IC50~25 μM)and Histone deacetylase (HDAC); activates Nrf2 pathway and supresses the activation of NF-κB. Curcumin induces mitophagy, autophagy, apoptosis, and cell cycle arrest with antitumor activity. Curcumin reduces renal damage associated with rhabdomyolysis by decreasing ferroptosis-mediated cell death. Curcumin exhibits anti-infective properties against various human pathogens like the influenza virus, hepatitis C virus, HIV and so on. |

|
|
| S2818 | Tacedinaline (CI994) | Tacedinaline (CI994, PD-123654, GOE-5549, Acetyldinaline) is a selective class I HDAC inhibitor with IC50 of 0.9, 0.9, 1.2, and >20 μM for human HDAC 1, 2, 3, and 8, respectively. Phase 3. |

|
|
| S7569 | LMK-235 | LMK-235 is a selective inhibitor of HDAC4 and HDAC5 with IC50 of 11.9 nM and 4.2 nM, respectively. |

|
|
| S2759 | Fimepinostat (CUDC-907) | CUDC-907 is a dual PI3K and HDAC inhibitor for PI3Kα and HDAC1/2/3/10 with IC50 of 19 nM and 1.7 nM/5 nM/1.8 nM/2.8 nM, respectively. CUDC-907 induces cell cycle arrest and apoptosis in breast cancer cells. Phase 1. |

|
|
| S2239 | Tubacin | Tubacin is a highly potent and selective, reversible, cell-permeable HDAC6 inhibitor with an IC50 of 4 nM in a cell-free assay, approximately 350-fold selectivity over HDAC1. Tubacin reduces the replication of the Japanese Encephalitis Virus via the decrease of viral RNA synthesis. |

|
|
| S2170 | Givinostat hydrochloride monohydrate | Givinostat (ITF2357) is a potent HDAC inhibitor for maize HD2, HD1B and HD1A with IC50 of 10 nM, 7.5 nM and 16 nM in cell-free assays. Phase 2. |

|
|
| S1168 | Valproic Acid sodium | Valproic Acid sodium is a HDAC inhibitor by selectively inducing proteasomal degradation of HDAC2, used in the treatment of epilepsy, bipolar disorder and prevention of migraine headaches. Valproic acid induces Notch1 signaling in small cell lung cancer (SCLC) cells. Valproic acid is under investigation for treatment of HIV and various cancers. Valproic acid (VPA) induces autophagy and mitophagy by upregulation of BNIP3 and mitochondrial biogenesis by upregulating PGC-1α. |

|
|
| S3944 | VPA (Valproic acid) | VPA (Valproic acid) is a fatty acid with anticonvulsant properties used in the treatment of epilepsy. It is also a histone deacetylase (HDAC) inhibitor and is under investigation for treatment of HIV and various cancers. Valproic acid (VPA) induces autophagy and mitophagy by upregulation of BNIP3 and mitochondrial biogenesis by upregulating PGC-1α. Valproic acid activates Notch-1 signaling. | ||
| S2244 | AR-42 | AR-42 (HDAC-42) is an HDAC inhibitor with IC50 of 30 nM. Phase 1. |

|
|
| S7324 | TMP269 | TMP269 is a potent, selective class IIa HDAC inhibitor with IC50 of 157 nM, 97 nM, 43 nM and 23 nM for HDAC4, HDAC5, HDAC7 and HDAC9, respectively. |

|
|
| S1999 | Sodium butyrate | Sodium butyrate (NaB, Butanoic acid sodium salt), sodium salt of butyric acid, is a histone deacetylase inhibitor and competitively binds to the zinc sites of class I and II histone deacetylases (HDACs). Sodium butyrate (NaB) inhibits cell cycle progression, promotes differentiation, and induces apoptosis and autophagy in several types of cancer cells. |

|
|
| S1515 | Pracinostat (SB939) | Pracinostat (SB939) is a potent pan-HDAC inhibitor with IC50 of 40-140 nM with exception for HDAC6. It has no activity against the class III isoenzyme SIRT I. Pracinostat (SB939) induces apoptosis in tumor cells. Phase 2. |

|
|
| S1194 | CUDC-101 | CUDC-101 is a potent multi-targeted inhibitor against HDAC, EGFR and HER2 with IC50 of 4.4 nM, 2.4 nM, and 15.7 nM, and inhibits class I/II HDACs, but not class III, Sir-type HDACs. Phase 1. |

|
|
| S1090 | Abexinostat (PCI-24781) | Abexinostat (PCI-24781, CRA-024781) is a novel pan-HDAC inhibitor mostly targeting HDAC1 with Ki of 7 nM, modest potent to HDACs 2, 3, 6, and 10 and greater than 40-fold selectivity against HDAC8. Phase 1/2. |

|
|
| S7595 | Santacruzamate A (CAY10683) | Santacruzamate A (CAY10683) is a potent and selective HDAC inhibitor with IC50 of 119 pM for HDAC2, >3600-fold selectivity over other HDACs. |

|
|
| S2341 | (-)-Parthenolide | (-)-Parthenolide, an inhibitor of the Nuclear Factor-κB Pathway, specifically depletes HDAC1 protein without affecting other class I/II HDACs; Also promotes the ubiquitination of MDM2 and activates p53 cellular functions. |
-Parthenolide-S234101W0220161110.gif)
|
|
| S1095 | Dacinostat (LAQ824) | Dacinostat (LAQ824, NVP-LAQ824) is a novel HDAC inhibitor with IC50 of 32 nM and is known to activate the p21 promoter. |

|
|
| S7596 | CAY10603 | CAY10603 (BML-281) is a potent and selective HDAC6 inhibitor with IC50 of 2 pM, >200-fold selectivity over other HDACs. |

|
|
| S7292 | RG2833 (RGFP109) | RG2833 (RGFP109) is a brain-penetrant HDAC inhibitor with IC50 of 60 nM and 50 nM for HDAC1 and HDAC3 in cell-free assays, respectively. |

|
|
| S7617 | Tasquinimod | Tasquinimod (ABR-215050) is an orally active antiangiogenic agent by allosterically inhibiting HDAC4 signalling. Phase 3. |

|
|
| S4125 | 4-PBA (Sodium Phenylbutyrate) | 4-PBA (Sodium Phenylbutyrate) is a salt of 4-phenylbutyrate (4-PBA) or 4-phenylbutyric acid.Sodium phenylbutyrate is a histone deacetylase inhibitor, used to treat urea cycle disorders. |

|
|
| S8567 | Tucidinostat (Chidamide) | Tucidinostat (Chidamide, HBI-8000, CS-055) is a low nanomolar inhibitor of HDAC1, 2, 3, and 10, the HDAC isotypes well documented to be associated with the malignant phenotype with IC50 values of 95, 160, 67, 78 nM for HDAC1, 2, 3, 10 respectively. | ||
| S8502 | TMP195 | TMP195 (TFMO 2) is a selective, first-in-class, class IIa HDAC inhibitor with Ki of 59, 60, 26 and 15nM for HDAC4, HDAC5, HDAC7 and HDAC9, respectively. | ||
| S7473 | Nexturastat A | Nexturastat A is a potent and selective HDAC6 inhibitor with IC50 of 5 nM, >190-fold selectivity over other HDACs. |

|
|
| S1422 | Droxinostat | Droxinostat (NS 41080) is a selective inhibitor of HDAC, mostly for HDACs 6 and 8 with IC50 of 2.47 μM and 1.46 μM, greater than 8-fold selective against HDAC3 and no inhibition to HDAC1, 2, 4, 5, 7, 9, and 10. |

|
|
| S7555 | Domatinostat (4SC-202) | Domatinostat (4SC-202) is a selective class I HDAC inhibitor with IC50 of 1.20 μM, 1.12 μM, and 0.57 μM for HDAC1, HDAC2, and HDAC3, respectively. Also displays inhibitory activity against Lysine specific demethylase 1 (LSD1). Phase 1. | ||
| S8043 | Scriptaid | Scriptaid (GCK 1026) is an inhibitor of HDAC. It shows a greater effect on acetylated H4 than H3. |

|
|
| S3592 | 4-PBA (4-Phenylbutyric acid) | 4-PBA (4-Phenylbutyric acid) is a histone deacetylase (HDAC) inhibitor and a key epigenetic inducer of anti-HCV hepatic hepcidin. 4-Phenylbutyric acid inhibits LPS-induced inflammation through regulating endoplasmic-reticulum (ER) stress and autophagy in acute lung injury models. | ||
| S2779 | M344 | M344 is a potent HDAC inhibitor with IC50 of 100 nM and able to induce cell differentiation. |

|
|
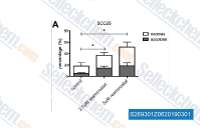
| S2693 | Resminostat | Resminostat (RAS2410) dose-dependently and selectively inhibits HDAC1/3/6 with IC50 of 42.5 nM/50.1 nM/71.8 nM, less potent to HDAC8 with IC50 of 877 nM. |

|
|
| S8464 | Citarinostat (ACY-241) | Citarinostat (ACY-241, HDAC-IN-2) is an orally available selective HDAC6 inhibitor with IC50 of 2.6 nM and 46 nM for HDAC6 and HDAC3, respectively. It has 13 to 18-fold selectivity towards HDAC6 in comparison to HDAC1-3. | ||
| S7689 | BG45 | BG45 is a class I HDAC inhibitor with IC50 of 289 nM, 2.0 µM, 2.2 µM and >20 µM for HDAC3, HDAC1, HDAC2, and HDAC6 in cell-free assays, respectively. | ||
| S8495 | WT161 | WT161 is a potent, selective, and bioavailable HDAC6 inhibitor with IC50 values of 0.4 nM, 8.35 nM and 15.4 nM for HDAC6, HDAC1 and HDAC2, respectively; shown to have >100-fold selectivity over other HDACs. WT161 induces apoptosis. | ||
| S8648 | ACY-738 | ACY-738 inhibits HDAC6 with low nanomolar potency (IC50=1.7 nM) and a selectivity of 60- to 1500-fold over class I HDACs. | ||
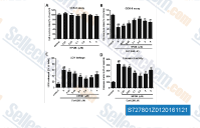
| S7278 | HPOB | HPOB is a potent, selective HDAC6 inhibitor with IC50 of 56 nM, >30-fold selectivity over other HDACs. |

|
|
| S0709 | Tubastatin A TFA | Tubastatin A TFA (Tubastatin A trifluoroacetate salt) is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective against all the other isozymes (1000-fold) except HDAC8 (57-fold). Tubastatin A promotes autophagy and increases apoptosis. | ||
| S6738 | TC-H 106 | TC-H 106 (Pimelic Diphenylamide 106) is a slow, tight-binding inhibitor of class I histone deacetylases(HDAC) with Ki value of 148 nM, about 102 nM, 14 nM for HDAC1, HDAC2, HDAC3, respectively. | ||
| S6687 | SIS17 | SIS17 is a mammalian histone deacetylase 11 (HDAC 11)-specific inhibitor with IC50 of 0.83 μM. SIS17 inhibits the demyristoylation of HDAC11 substrate, serine hydroxymethyl transferase 2, without inhibiting other HDACs. | ||
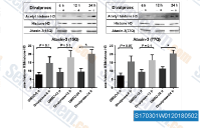
| S1703 | Divalproex Sodium | Divalproex Sodium, consisting of a compound of sodium valproate and valproic acid in a 1:1 molar relationship in an enteric coated form, is a HDAC inhibitor, used in the treatment for epilepsy. |

|
|
| S0864 | ACY-775 | ACY-775 is a potent and selective histone deacetylase 6 (HDAC6) inhibitor with IC50 of 7.5 nM. | ||
| S1073 | BML-210 (CAY10433) | BML-210 (CAY10433) is a small molecule inhibitor of HDAC.BML-210 inhibits the HDAC4-VP16-driven reporter signal in a dose-dependent manner with an apparent IC50 of ∼5 µM. | ||
| S8773 | TH34 | TH34 is a HDAC inhibitor that shows pronounced selectivity for HDACs 6, 8 and 10 over HDACs 1, 2 and 3. In a NanoBRET assay, TH34 strongly binds HDAC6, 8 and 10 with low-micromolar IC50 concentrations (HDAC6: 4.6 µM, HDAC8: 1.9 µM, HDAC10: 7.7 µM). | ||
| S9262 | Raddeanin A | Raddeanin A (Raddeanin R3, NSC382873), a triterpenoid saponin from Anemone raddeana Regel, displays moderate inhibitory activity against histone deacetylases (HDACs) and has high antiangiogenic potency, antitumor activity. | ||
| S3981 | Sinapinic Acid | Sinapinic acid (Sinapic acid) is a small naturally occurring hydroxycinnamic acid which belongs to phenylpropanoid family and commonly used as matrix in MALDI mass spectrometry. Sinapinic acid (Sinapic acid) acts as an inhibitor of HDAC, with IC50 of 2.27 mM, and also inhibits ACE-I activity. | ||
| E5952New | PT 3 | PT 3 is a benzamide-class, potent, and selective inhibitor of Histone Deacetylase 3 (HDAC3) with an IC50 of 245 nM. It crosses the blood-brain barrier and enhances learning and memory in novel object recognition models in mice. | ||
| E8302New | YX968 | YX968 is a potent, rapid, and selective PROTAC dual degrader of HDAC3/8 with DC50 values of 1.7 nM and 6.1 nM, respectively. It promotes apoptosis and kills cancer cells with a high potency in vitro and can be used in cancer research. | ||
| E8292New | GEM144 | GEM144 is a potent and orally active dual inhibitor of DNA polymerase α (POLA1) and HDAC11 . It exhibits potent antiproliferative activity in vitro and antitumor efficacy in vivo by inducing p53 acetylation, p21 activation, G1/S cell cycle arrest, and apoptosis. | ||
| E1086 | KT-531 | KT-531 (KT531) is a potent, selective HDAC6 inhibitor with IC50 of 8.5 nM, displays 39-fold selectivity over other HDAC isoforms. | ||
| E1912New | B102(PARP/HDAC-IN-1) | B102(PARP/HDAC-IN-1) is a dual inhibitor of PARP and HDAC, with IC50 of 19.01 nM, 2.13 nM, 1.69 μM, > 10 μM for PARP1, PARP2, HDAC1, HDAC6, respectively, in an enzymatic assay. It exhibits anti-proliferative and anti-tumor effects. It also induces DNA damage and apoptosis in vitro. | ||
| E1686New | SW-100 | SW-100 is a selective inhibitor of histone deacetylase 6 (HDAC6) with an IC50 of 2.3 nM. It displays 1000-fold selectivity for HDAC6 relative to all other HDAC isozymes. | ||
| E1580New | TNG260 | TNG260 is an inhibitor of CoREST-selective deacetylase (CoreDAC) that inhibits HDAC1 with 10-fold selectivity over HDAC3. It decreases intratumoral neutrophil infiltration and exhibits immune-mediated cell killing. | ||
| E1469 | ITF3756 | ITF3756 is a selective histone deacetylase 6 (HDAC6) inhibitor bearing a pentaheterocyclic scaffold. It can be used in research of autoimmune disorders, neurodegenerative diseases, and cancer. | ||
| E1554 | Tefinostat(CHR-2845) | Tefinostat(CHR-2845) is a monocyte/macrophage-targeted inhibitor of pan-histone deacetylase (HDAC). Tefinostat undergoes cleavage mediated by the intracellular esterase human carboxylesterase-1 (hCE-1) to yield the active acid CHR-2847. In vitro studies reveal a specific heightened sensitivity of monocytoid leukemias to tefinostat. | ||
| S2190 | Pyroxamide (NSC 696085) | Pyroxamide(NSC 696085) is a potent inhibitor of affinity-purified HDAC1 with ID50 of 100 nM. It also induces growth suppression and cell death in human rhabdomyosarcoma in vitro. | ||
| S5810 | UF010 | UF010 is a class I HDAC-selective inhibitor with IC50 values of 0.5 nM, 0.1 nM, 0.06 nM, 1.5 nM, 9.1 nM and 15.3 nM for HDAC1, HDAC2, HDAC3, HDAC8, HDAC6 and HDAC10, respectively. | ||
| S8743 | SKLB-23bb | SKLB-23bb is an orally bioavailable HDAC6-selective inhibitor with IC50 values under 100 nmol/L, against most of the cell lines checked. It also has microtubule-disrupting ability. | ||
| S9275 | Isoguanosine | Isoguanosine (Crotonoside) inhibits FLT3 and HDAC3/6 for the treatment of AML.Isoguanosine is a naturally occurring active isomer of guanosine that is found in the seeds of Croton tiglium. | ||
| S6548 | NKL 22 | NKL 22 is a selective inhibitor of HDAC with IC50 of 78 µM. | ||
| E0391New | BRD-6929 | BRD-6929 is an inhibitor of histone deacetylase 1 (HDAC1) and HDAC2 with an IC50s of 0.04 µM and 0.1 µM, respectively. It exhibits antiproliferative activities against a human cancer cell line (HCT116) and in human mammary epithelial cells (HMEC). | ||
| S5771 | Sulforaphane | Sulforaphane is a naturally occurring isothiocyanate derived from the consumption of cruciferous vegetables, such as broccoli, cabbage, and kale. It is an inducer of Nrf2. Sulforaphane is also an inhibitor of histone deacetylase (HDAC) and NF-κB. Sulforaphane increases heme oxygenase-1 (HO-1) and reduces the levels of reactive oxygen species (ROS). Sulforaphane induces cell cycle arrest and apoptosis., | ||
| S7726 | BRD73954 | BRD73954 is a potent and selective HDAC inhibitor with IC50 of 36 nM and 120 nM for HDAC6 and HDAC8, respectively. | ||
| E0812 | CXD101 | CXD101 (HDAC-IN-4) is a potent, selective and orally active class I histone deacetylase (HDAC) inhibitor with IC50s of 63 nM, 570 nM and 550 nM for HDAC1, HDAC2 and HDAC3, respectively. | ||
| S5905 | Suberohydroxamic acid | Suberohydroxamic acid (suberic bishydroxamic acid) is a competitive HDAC inhibitor with IC50 values of 0.25 and 0.3 μM for HDAC1 and HDAC3 respectively. | ||
| S8962 | BRD3308 | BRD3308 is a potant and highly selective inhibitor of HDAC3 with IC50 of 54 nM, 1.26 μM and 1.34 μM for HDAC3, HDAC1 and HDAC2, respectively. BRD3308 activates HIV-1 transcription. BRD3308 suppresses pancreatic β-cell apoptosis induced by inflammatory cytokines (glucolipotoxic stress) and increases functional insulin release. | ||
| S7593 | Splitomicin | Splitomicin is a selective NAD(+)-dependent histone deacetylase Sir2p inhibitor with IC50 of 60 μM, showing a higher activity in a cell-based assay. | ||
| E7940New | ACY-957 | ACY-957 is a selective chemical inhibitor of HDAC1 and HDAC2 with an IC50 values of 7 nM, 18 nM against HDAC1/2 respectively. It induces hemoglobin protein (HbF) in primary erythroid progenitor cells, including those from sickle cell patients, offering a promising approach for treating sickle cell disease (SCD) and β-thalassemia. | ||
| S5438 | Biphenyl-4-sulfonyl chloride | Biphenyl-4-sulfonyl chloride (p-Phenylbenzenesulfonyl, 4-Phenylbenzenesulfonyl, p-Biphenylsulfonyl) is a HDAC inhibitor with synthetic applications in palladium-catalyzed desulfitative C-arylation. | ||
| S9934 | KA2507 | KA2507, a potent, orally active and selective HDAC6 inhibitor with IC50 of 2.5 nM, shows antitumor activities and immune modulatory effects in preclinical models. | ||
| S2132 | SR-4370 | SR-4370 is a potent and selective inhibitor of class I HDACs with IC50 of 0.13 µM, 0.58 µM, 0.006 µM, 2.3 µM, 3.7 µM for HDAC 1, HDAC 2, HDAC 3, HDAC 8, HDAC 6, respectively.SR-4370 suppresses AR signaling and in vivo prostate tumor growth. | ||
| E8178New | Quisinostat | Quisinostat(JNJ-26481585) is a potent and orally active second-generation inhibitor of pan-Histone Deacetylase (HDAC). It inhibits HDAC1, HDAC2, HDAC4, HDAC10, HDAC11 with IC50s of 0.11 nM , 0.33 nM , 0.64 nM , 0.46 nM, 0.37 nM respectively. It exhibits potent in vivo antitumor activity and potential clinical use in various solid and hematologic malignancies. | ||
| S1313 | GSK3117391 | GSK3117391 (GSK3117391A, HDAC-IN-3) is a potent histone deacetylase (HDAC) inhibitor. | ||
| S8769 | Tinostamustine(EDO-S101) | Tinostamustine(EDO-S101) is a first-in-class alkylating deacetylase inhibitor with IC50 values of 9 nM, 9 nM, 25 nM and 107 nM for HDAC1, HDAC2, HDAC3 and HDAC8 (Class 1 HDACs) respectively and 6 nM, 72 nM for HDAC6 and HDAC10 (Class II HDACs). | ||
| S8323 | ITSA-1 (ITSA1) | ITSA-1 (ITSA1) is an HDAC activator via TSA suppression, but shows no activity towards other HDAC inhibitors. | ||
| F0735New | HDAC1 Rabbit mAb | |||
| F0838New | HDAC3 Rabbit mAb | HDAC3,Histone Deacetylase 3 (HDAC3) | ||
| F0948New | HDAC5 Rabbit mAb | HDAC5,Histone Deacetylase 5 (HDAC5) | ||
| F0480New | HDAC4 Rabbit mAb | |||
| F2424New | HDAC9 Rabbit mAb | |||
| F1252New | HDAC7 Rabbit mAb | |||
| F2989New | CTBP2 Rabbit mAb | |||
| F2218New | HDAC8 Rabbit mAb | |||
| F1413New | SIN3A Rabbit mAb | mSin3A,SIN3A | ||
| E5952New | PT 3 | PT 3 is a benzamide-class, potent, and selective inhibitor of Histone Deacetylase 3 (HDAC3) with an IC50 of 245 nM. It crosses the blood-brain barrier and enhances learning and memory in novel object recognition models in mice. | ||
| E8302New | YX968 | YX968 is a potent, rapid, and selective PROTAC dual degrader of HDAC3/8 with DC50 values of 1.7 nM and 6.1 nM, respectively. It promotes apoptosis and kills cancer cells with a high potency in vitro and can be used in cancer research. | ||
| E8292New | GEM144 | GEM144 is a potent and orally active dual inhibitor of DNA polymerase α (POLA1) and HDAC11 . It exhibits potent antiproliferative activity in vitro and antitumor efficacy in vivo by inducing p53 acetylation, p21 activation, G1/S cell cycle arrest, and apoptosis. | ||
| E1912New | B102(PARP/HDAC-IN-1) | B102(PARP/HDAC-IN-1) is a dual inhibitor of PARP and HDAC, with IC50 of 19.01 nM, 2.13 nM, 1.69 μM, > 10 μM for PARP1, PARP2, HDAC1, HDAC6, respectively, in an enzymatic assay. It exhibits anti-proliferative and anti-tumor effects. It also induces DNA damage and apoptosis in vitro. | ||
| E1686New | SW-100 | SW-100 is a selective inhibitor of histone deacetylase 6 (HDAC6) with an IC50 of 2.3 nM. It displays 1000-fold selectivity for HDAC6 relative to all other HDAC isozymes. | ||
| E1580New | TNG260 | TNG260 is an inhibitor of CoREST-selective deacetylase (CoreDAC) that inhibits HDAC1 with 10-fold selectivity over HDAC3. It decreases intratumoral neutrophil infiltration and exhibits immune-mediated cell killing. | ||
| E0391New | BRD-6929 | BRD-6929 is an inhibitor of histone deacetylase 1 (HDAC1) and HDAC2 with an IC50s of 0.04 µM and 0.1 µM, respectively. It exhibits antiproliferative activities against a human cancer cell line (HCT116) and in human mammary epithelial cells (HMEC). | ||
| E7940New | ACY-957 | ACY-957 is a selective chemical inhibitor of HDAC1 and HDAC2 with an IC50 values of 7 nM, 18 nM against HDAC1/2 respectively. It induces hemoglobin protein (HbF) in primary erythroid progenitor cells, including those from sickle cell patients, offering a promising approach for treating sickle cell disease (SCD) and β-thalassemia. | ||
| E8178New | Quisinostat | Quisinostat(JNJ-26481585) is a potent and orally active second-generation inhibitor of pan-Histone Deacetylase (HDAC). It inhibits HDAC1, HDAC2, HDAC4, HDAC10, HDAC11 with IC50s of 0.11 nM , 0.33 nM , 0.64 nM , 0.46 nM, 0.37 nM respectively. It exhibits potent in vivo antitumor activity and potential clinical use in various solid and hematologic malignancies. |
Choose Selective HDAC Inhibitors
Tags: HDAC inhibition | HDAC cancer | HDAC assay | histone deacetylase activity | HDAC inhibitors cancer | HDAC activity assay | HDAC activation | HDAC inhibitor mechanism | selective HDAC inhibitors | HDAC phosphorylation | HDAC inhibitor drugs | HDAC inhibitors in clinical trials | HDAC inhibitors list | HDAC inhibitor review







































