research use only
Ricolinostat (ACY-1215) HDAC inhibitor
Cat.No.S8001
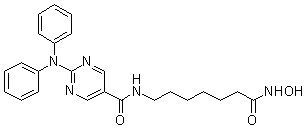
Chemical Structure
Molecular Weight: 433.5
Quality Control
Products Often Used Together with Ricolinostat (ACY-1215)
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| A-172 | Growth Inhibition Assay | 10 nM | 24/48 h | inhibits cell growth time dependently | 26150340 | |
| U87MG | Growth Inhibition Assay | 10 nM | 24/48 h | inhibits cell growth time dependently | 26150340 | |
| Hbl-1 | Growth Inhibition Assay | 48 h | IC50=1.6 μM | 26116270 | ||
| OCI-Ly10 | Growth Inhibition Assay | 48 h | IC50=0.9 μM | 26116270 | ||
| Riva | Growth Inhibition Assay | 48 h | IC50=2.2 μM | 26116270 | ||
| Su-DHL2 | Growth Inhibition Assay | 48 h | IC50=3.3 μM | 26116270 | ||
| OCI-Ly1 | Growth Inhibition Assay | 48 h | IC50=2.4 μM | 26116270 | ||
| OCI-Ly7 | Growth Inhibition Assay | 48 h | IC50=1.2 μM | 26116270 | ||
| Su-DHL4 | Growth Inhibition Assay | 48 h | IC50=4.7 μM | 26116270 | ||
| Su-DHL6 | Growth Inhibition Assay | 48 h | IC50=3.2 μM | 26116270 | ||
| Hbl-2 | Growth Inhibition Assay | 48 h | IC50=1.9 μM | 26116270 | ||
| Jeko-1 | Growth Inhibition Assay | 48 h | IC50=1.5 μM | 26116270 | ||
| Jvm-2 | Growth Inhibition Assay | 48 h | IC50=4.0 μM | 26116270 | ||
| Rec-1 | Growth Inhibition Assay | 48 h | IC50=2.3 μM | 26116270 | ||
| CCL-119 | Growth Inhibition Assay | 48 h | IC50=1.7 μM | 26116270 | ||
| H9 | Growth Inhibition Assay | 48 h | IC50=1.2 μM | 26116270 | ||
| HH | Growth Inhibition Assay | 48 h | IC50=2.5 μM | 26116270 | ||
| Sup-T1 | Growth Inhibition Assay | 48 h | IC50=1.6 μM | 26116270 | ||
| MM.1S | Function Assay | 0-5μM | 6 h | increases acetylated α-tubulin | 22262760 | |
| MM.1S | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 | |
| MM.1R | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 | |
| RPMI8226 | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 | |
| MM.1S | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| OPM1 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| RPMI | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| MM.1R | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| LR5 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| OPM2 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 | |
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.0047 μM. | 28038324 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human N-terminal GST-tagged HDAC6 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence assay, IC50 = 0.009 μM. | 29500130 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal His-tagged HDAC3/N-terminal GST-tagged NCOR2 (395 to 489 residues) expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate additi, IC50 = 0.037 μM. | 29500130 | ||
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant C-terminal FLAG-tagged HDAC2 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.048 μM. | 28038324 | ||
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant C-terminal FLAG-His-tagged HDAC1 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.058 μM. | 28038324 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal His-tagged HDAC2 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence assay, IC50 = 0.066 μM. | 29500130 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal FLAG/His-tagged HDAC1 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence ass, IC50 = 0.1 μM. | 29500130 | ||
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 1 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.29 μM. | 30365892 | ||
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 4 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.54 μM. | 30365892 | ||
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 2 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.58 μM. | 30365892 | ||
| RPMI8226 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RPMI8226 cells after 72 hrs by MTT assay, IC50 = 1.468 μM. | 26443078 | ||
| SEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SEM cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.61 μM. | 30365892 | ||
| SUP-B15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SUP-B15 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.92 μM. | 30365892 | ||
| RPMI18226 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RPMI18226 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.97 μM. | 30365892 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 2.36 μM. | 30365892 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| KCL22 | Cytotoxicity assay | 72 hrs | Cytotoxicity against imatinib-resistant human KCL22 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.38 μM. | 30365892 | ||
| U266 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U266 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.52 μM. | 30365892 | ||
| SUP-B15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against imatinib-resistant human SUP-B15 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.54 μM. | 30365892 | ||
| KCL22 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KCL22 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.75 μM. | 30365892 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 3 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 4.45 μM. | 30365892 | ||
| MV4-11 | Function assay | 1000 nM | 6 hrs | Inhibition of HDAC1/2/3 in human MV4-11 cells assessed as upregulation of histone H3 acetylation at 1000 nM after 6 hrs by Western blot analysis | 26443078 | |
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 | |
| SEM | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human SEM cells at IC50 to 2 times IC50 after 24 to 72 hrs by trypan exclusion method | 30365892 | ||
| HEL | Cell cycle assay | 1 to 10 uM | 48 hrs | Cell cycle arrest in human HEL cells assessed as accumulation at G1 phase at 1 to 10 uM after 48 hrs propidium iodide staining based flow cytometry | 29940115 | |
| SEM | Function assay | 18 hrs | Inhibition of HDAC6 in human SEM cells assessed as decrease in aggresome accumulation at IC50 after 18 hrs by fluorescence microscopic method | 30365892 | ||
| SEM | Function assay | 1.6 uM | 18 hrs | Inhibition of HDAC6 in human SEM cells assessed as decrease in aggresome accumulation at 1.6 uM after 18 hrs by FACS analysis | 30365892 | |
| SH-SY5Y | Function assay | 0.1 to 1 uM | 24 hrs | Inhibition of HDAC6 in human SH-SY5Y cells assessed as increase in acetylation of alpha-tubulin at 0.1 to 1 uM after 24 hrs by Western blot analysis | 30028616 | |
| SH-SY5Y | Function assay | 0.1 to 1 uM | 24 hrs | Inhibition of class 1 HDAC in human SH-SY5Y cells assessed as increase in acetylation of histone H3 at 0.1 to 1 uM after 24 hrs by Western blot analysis | 30028616 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 42 mg/mL
(96.88 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 433.5 | Formula | C24H27N5O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1316214-52-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Rocilinostat | Smiles | C1=CC=C(C=C1)N(C2=CC=CC=C2)C3=NC=C(C=N3)C(=O)NCCCCCCC(=O)NO | ||
Mechanism of Action
| Features |
Induced less cytotoxicity in PHA-stimulated PBMCs from 4 healthy donors compared with the pan-HDAC inhibitor SAHA.
|
|---|---|
| Targets/IC50/Ki |
HDAC6
(Cell-free assay) 4.7 nM
HDAC2
(Cell-free assay) 48 nM
HDAC3
(Cell-free assay) 51 nM
HDAC1
(Cell-free assay) 58 nM
HDAC8
(Cell-free assay) 100 nM
|
| In vitro |
ACY-1215 is a hydroxamic acid derivative. ACY-1215 is 12-, 10-, and 11-fold less active against HDAC1, HDAC2, and HDAC3 (class I HDACs), respectively. ACY-1215 has minimal activity (IC50 > 1μM) against HDAC4, HDAC5, HDAC7, HDAC9, HDAC11, Sirtuin1, and Sirtuin2, and has slight activity against HDAC8 (IC50 = 0.1μM). The IC50 values for ACY-1215 for T-cell toxicity is 2.5μM. ACY-1215 overcomes tumor cell growth and survival conferred by BMSCs and cytokines in the BM milieu. |
| Kinase Assay |
HDAC enzymatic assays
|
|
ACY-1215 is dissolved and subsequently diluted in assay buffer [50 mM HEPES, pH 7.4, 100 mM KCl, 0.001% Tween-20, 0.05% BSA, and 20 μM tris(2-carboxyethyl)phosphine] to 6-fold the final concentration. HDAC enzymes are diluted to 1.5-fold of the final concentration in assay buffer and pre-incubated with ACY-1215 for 10 minutes before the addition of the substrate. The amount of FTS (HDAC1, HDAC2, HDAC3, and HDAC6) or MAZ-1675 (HDAC4, HDAC5, HDAC7, HDAC8, and HDAC9) used for each enzyme is equal to the Michaelis constant (Km), as determined by a titration curve. FTS or MAZ-1675 is diluted in assay buffer to 6-fold the final concentration with 0.3μM sequencing grade trypsin. The substrate/trypsin mix is added to the enzyme/compound mix and the plate is shaken for 60 seconds and then placed into a SpectraMax M5 microtiter plate reader. The enzymatic reaction is monitored for release of 7-amino-4-methoxy-coumarin over 30 minutes, after deacetylation of the lysine side chain in the peptide substrate, and the linear rate of the reaction is calculated.
|
|
| In vivo |
ACY-1215 is readily absorbed by tumor tissue. Moreover, the drug does not accumulate in tumor tissue, as evidenced by the parallel decline of acetylated α-tubulin in blood cells and tumor tissue by 24 hours after dose. |
References |
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | Ac-α-tubulin / Ac-Histone H4 Survivin / P21 / CDC2 / p53 / p-p53(S392) / Cyclin A2 / Cyclin B1 Bax / Bim / Bcl2 / Cleaved caspase-3 / Cleaved caspase-9 / Cleaved PARP PI3K(p85) / AKT / p-AKT(S473) / PRAS40 / Rag C / mTOR / p-mTOR / ERK / p-ERK Ac-β-catenin(K49) / p-β-catenin / β-catenin |
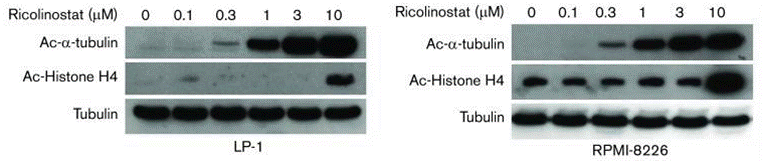
|
31015208 |
| Immunofluorescence | β-tubulin / β-catenin |
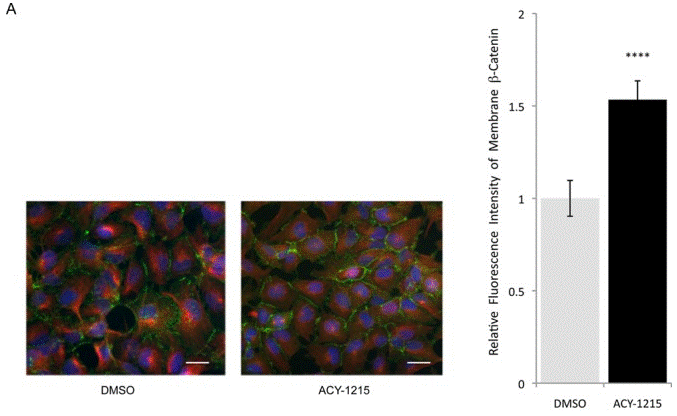
|
25546293 |
| Growth inhibition assay | Cell viability |
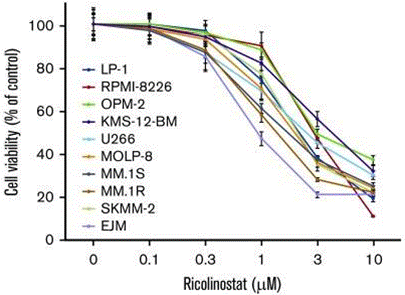
|
31015208 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02632071 | Completed | Metastatic Breast Cancer|Breast Carcinoma |
Columbia University|Acetylon Pharmaceuticals Incorporated|National Cancer Institute (NCI) |
March 1 2016 | Phase 1 |
| NCT01583283 | Completed | Multiple Myeloma |
Celgene |
July 12 2012 | Phase 1 |
| NCT01323751 | Completed | Multiple Myeloma |
Celgene|The Leukemia and Lymphoma Society |
July 2011 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What would you suggest to obtain a clear solution of it?
Answer:
It can be dissolved in 2% DMSO/30% PEG 300/ddH2O at 5 mg/ml clearly, while in 1% DMSO/30% polyethylene glycol/1% Tween 80 at 30 mg/ml it is a suspension for oral administration. Please note that the precipitation will go out from the clear solution after stayed for about half an hour, so it is recommended to prepare the solution just before use.






































