research use only
Quisinostat (JNJ-26481585) Dihydrochloride HDAC inhibitor
Cat.No.S1096
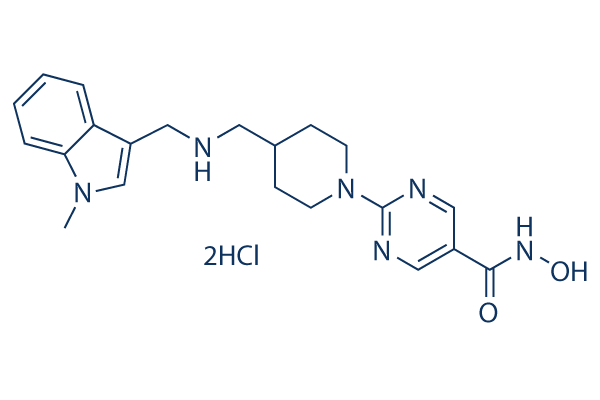
Chemical Structure
Molecular Weight: 467.39
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| A549 cells | Cell viability assay | 24, 48, or 72 h | the IC50 values of cells for 48 and 72 h of quisinostat treatment were 82.4 and 42.0 nM, respectively. | 27423454 | ||
| RH30 cells | Function assay | 15 nM | 24 h and 30 h | GSK690 and JNJ-26481585 cooperated to induce cleavage of the initiator caspase-9 into its active p35 and p37 fragments and of the effector caspase-3 into its active p12 and p17 fragments | 28617441 | |
| RD cells | Function assay | 15 nM | 24 h and 30 h | GSK690 and JNJ-26481585 cooperated to induce cleavage of the initiator caspase-9 into its active p35 and p37 fragments and of the effector caspase-3 into its active p12 and p17 fragments | 28617441 | |
| HepG2 | Cell viability assay | 5, 10, 20, 40, 80, 160 and 320 nM | 24, 48, 72 h | The IC50 values of cells for quisinostat treatment at 48 and 72 h were 81.2 and 30.8 nM, respectively. | 28849080 | |
| HuT78 | Function assay | 10 nM | increases the level of acetylated lysine K10 of histone H3 | 30012418 | ||
| Sf9 | Function assay | Inhibition of full length recombinant human HDAC1 expressed in baculovirus infected Sf9 cells using RHKK-Ac as substrate by fluorescence analysis, IC50 = 0.00011 μM. | 27606546 | |||
| Sf9 | Function assay | Inhibition of full length recombinant human HDAC2 expressed in baculovirus infected Sf9 cells using RHKK-Ac as substrate by fluorescence analysis, IC50 = 0.00033 μM. | 27606546 | |||
| Sf9 | Function assay | Inhibition of full length recombinant human HDAC3/NCOR2 expressed in baculovirus infected Sf9 cells using RHKK-Ac as substrate by fluorescence analysis, IC50 = 0.00486 μM. | 27606546 | |||
| HUT78 | Function assay | 18 hrs | Pro-apoptotic activity in human HUT78 cells after 18 hrs by caspase-Glo 3/7 assay, EC50 = 0.0065 μM. | 30122227 | ||
| BL21 (DE3) | Function assay | Binding affinity to human His-thioredoxin-tagged HDAC8 expressed in Escherichia coli BL21 (DE3) cells by ITC method, Kd = 0.0227 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Binding affinity to Schistosoma mansoni His-tagged HDAC8 expressed in Escherichia coli BL21 (DE3) cells by ITC method, Kd = 0.0284 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Inhibition of human His-thioredoxin-tagged HDAC8 expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.0644 μM. | 30347148 | |||
| Sf9 | Function assay | Inhibition of full length recombinant human HDAC6 expressed in baculovirus infected Sf9 cells using RHKK-Ac as substrate by fluorescence analysis, IC50 = 0.0768 μM. | 27606546 | |||
| BL21 (DE3) | Function assay | Inhibition of human His-thioredoxin-tagged HDAC8 mL6 mutant expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.093 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Inhibition of human His-thioredoxin-tagged HDAC8 mL1/mL6 mutant expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.094 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Inhibition of human His-thioredoxin-tagged HDAC8 mL6/L179I mutant expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.169 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Inhibition of human His-thioredoxin-tagged HDAC8 mL1/mL6/L179I mutant expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.217 μM. | 30347148 | |||
| BL21 (DE3) | Function assay | Inhibition of Schistosoma mansoni His-tagged HDAC8 expressed in Escherichia coli BL21 (DE3) cells using Fluor de Lys (R)-HDAC8 as substrate by fluorometric method, IC50 = 0.3034 μM. | 30347148 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-12 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for DAOY cells | 29435139 | |||
| fibroblast cells | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for control Hh wild type fibroblast cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-SH cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for MG 63 (6-TG R) cells | 29435139 | |||
| fibroblast cells | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for control Hh wild type fibroblast cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh30 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for OHS-50 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for SK-N-SH cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for RD cells | 29435139 | |||
| Daoy | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for Daoy cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for TC32 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for MG 63 (6-TG R) cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB1643 cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SJ-GBM2 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh18 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Saos-2 cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for SJ-GBM2 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for RD cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 79 mg/mL
(169.02 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 467.39 | Formula | C21H28Cl2N6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 875320-31-3 | -- | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1C=C(C2=CC=CC=C21)CNCC3CCN(CC3)C4=NC=C(C=N4)C(=O)NO.Cl.Cl | ||
Mechanism of Action
| Features |
An orally bioavailable, second-generation, hydroxamic acid-based HDAC inhibitor.
|
|---|---|
| Targets/IC50/Ki |
HDAC1
(Cell-free assay) 0.11 nM
HDAC2
(Cell-free assay) 0.33 nM
HDAC11
(Cell-free assay) 0.37 nM
HDAC10
(Cell-free assay) 0.46 nM
HDAC4
(Cell-free assay) 0.64 nM
HDAC5
(Cell-free assay) 3.69 nM
HDAC8
(Cell-free assay) 4.26 nM
HDAC3
(Cell-free assay) 4.86 nM
|
| In vitro |
JNJ-26481585 exhibits broad spectrum antiproliferative activity in solid and hematologic cancer cell lines, such as all lung, breast, colon, prostate, brain, and ovarian tumor cell lines, with IC50 ranging from 3.1-246 nM, which is more potent than R306465, CRA-24781, or mocetinostat in various human cancer cell lines tested. A recent study shows that JNJ-26481585 promotes myeloma cell death at low nanomolar concentrations by resulting in Mcl-1 depletion and Hsp72 induction. |
| Kinase Assay |
HDAC activity assays
|
|
In all cases, full-length HDAC proteins are expressed using baculovirus-infected Sf9 cells. In addition, HDAC3 is coexpressed as a complex with human NCOR2. For assessing activity of HDAC1-containing cellular complexes, immunoprecipitated HDAC1 complexes are incubated with an [3H]acetyl- labeled fragment of histone H4 peptide [biotin-(6-aminohexanoic)Gly-Ala-(acetyl[3H])Lys-Arg-His-Arg-Lys-Val-NH2] in a total volume of 50μL enzyme assay buffer (25mM HEPES (pH 7.4), 1 M sucrose, 0.1 mg/mL BSA and 0.01% (v/v) Triton X-100). Incubation is performed for 45 minutes at 37 °C (immunoprecipitates) or 30 min at room temperature. Before addition of substrate, HDAC inhibitors are added at increasing concentrations and preincubated for 10 minutes at room temperature. After incubation, the reaction is quenched with 35μL stop buffer (1 M HCl and 0.4 M acetic acid). Released [3H]acetic acid is extracted with 800μL ethyl acetate and quantified by scintillation counting. Equal amounts of HDAC1 are immunoprecipitated as indicated by Western blot analysis. HDAC1 activity results are presented as mean ± SD of three independent experiments on a single lysate.
|
|
| In vivo |
In an HDAC1-responsive A2780 ovarian tumor screening model, JNJ-26481585 dosing at its maximal tolerated dose (10 mg/kg i.p. and 40 mg/kg p.o.) for 3 days leads to an HDAC1-regulated fluorescence , which predicts tumor growth inhibition. Furthermore, JNJ-26481585 also shows more potent inhibitory effects on the growth of C170HM2 colorectal liver metastases. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | HDAC1 / HDAC2 / HDAC4 p21 / CDK2 / CDK4 / CDK6 / Cyclin D1 / Cyclin E1 / Cyclin A2 Caspase-3/ Cleaved caspase-3 / caspase-9 / Cleaved caspase-9 / PARP / Cleaved PARP / Bcl-xl / Bcl2 / Bax / Survivin PI3K-p110 / PI3K-p85 / p-AKT / JNK / p-JNK / p-c-Jun |
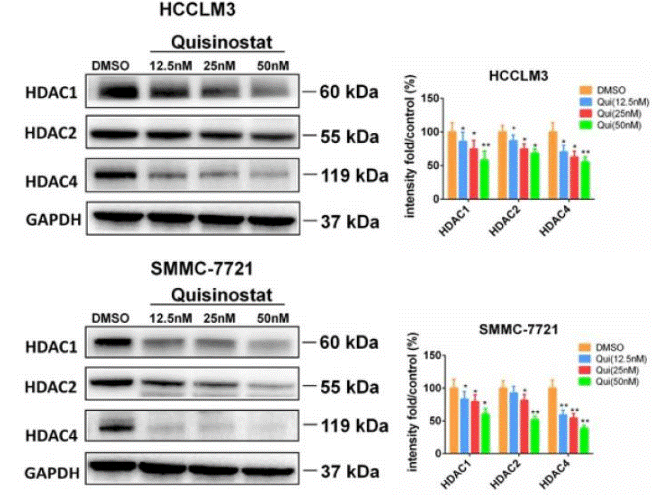
|
30443188 |
| Growth inhibition assay | Cell viability |
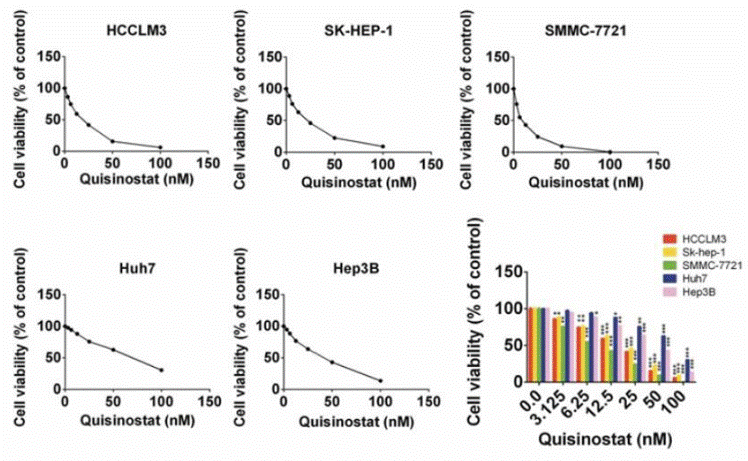
|
30443188 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02948075 | Completed | Ovarian Cancer |
NewVac LLC|Janssen Pharmaceutica N.V. Belgium |
September 2015 | Phase 2 |
| NCT01464112 | Completed | Multiple Myeloma |
Janssen Research & Development LLC |
September 16 2011 | Phase 1 |
| NCT00676728 | Terminated | Advanced or Refractory Leukemia|Myelodysplastic Syndromes |
Johnson & Johnson Pharmaceutical Research & Development L.L.C. |
December 2008 | Phase 1 |
| NCT00677105 | Completed | Lymphoma|Neoplasms |
Johnson & Johnson Pharmaceutical Research & Development L.L.C.|Janssen Pharmaceutica N.V. Belgium |
August 2007 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
I was thinking of resuspending it in 10% hydroxy-propyl-β-cyclodextrin, 25mg/ml mannitol, in sterile water (final pH 8.7). Is this a good vehicle to use? What is the solubility of this compound in such a vehicle?
Answer:
This vehicle can be used for in vivo studies. The following papers also used this vehicle: 1. http://www.nature.com/leu/journal/v23/n10/full/leu2009121a.html; 2. http://cancerres.aacrjournals.org/content/69/13/5307.long (The solvent contains 10% hydroxypropyl-β-cyclodextrin, 0.8% HCl (0.1 N), 0.9 % NaOH (0.1 N), 3.4% mannitol and pyrogen-free water). Its solubility is 2mg/ml.






































