- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Panobinostat (LBH589)
Synonyms: NVP-LBH589
Panobinostat (LBH589, NVP-LBH589) is a novel broad-spectrum HDAC inhibitor with IC50 of 5 nM in a cell-free assay. Panobinostat (LBH589) induces autophagy and apoptosis. Panobinostat effectively disrupts HIV latency in vivo. Phase 3.
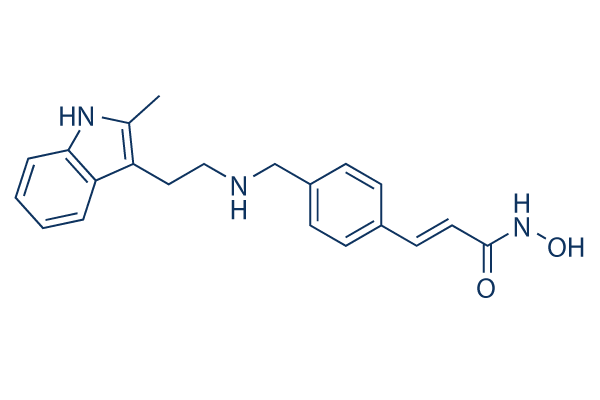
Panobinostat (LBH589) Chemical Structure
CAS: 404950-80-7
Selleck's Panobinostat (LBH589) has been cited by 380 publications
Purity & Quality Control
Batch:
Purity:
99.96%
99.96
Panobinostat (LBH589) Related Products
| Related Targets | HDAC1 HDAC2 HDAC3 HDAC4 HDAC5 HDAC6 HDAC7 HDAC8 HDAC9 HDAC10 HDAC11 HD1 HD2 | Click to Expand |
|---|---|---|
| Related Compound Libraries | Kinase Inhibitor Library FDA-approved Drug Library Natural Product Library Bioactive Compound Library-I Bioactive Compound Library-Ⅱ | Click to Expand |
Signaling Pathway
Choose Selective HDAC Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HT29 | Growth Inhibition Assay | 0-10 μM | 0-4 d | inhibits cell growth in both time- and dose-dependent manner | 26702784 | |
| HepG2 | Growth Inhibition Assay | 0-10 μM | 0-4 d | inhibits cell growth in both time- and dose-dependent manner | 26702784 | |
| HT29 | Function Assay | 50 nM | 24-72 h | induced activation of caspase 3 after 48 h | 26702784 | |
| HepG2 | Function Assay | 50 nM | 24-72 h | induced activation of caspase 3 after 24 h | 26702784 | |
| HCC827 | Growth Inhibition Assay | 5/7.5/10 nM | 72 h | DMSO | enhances the antiproliferative effect of erlotinib | 26675484 |
| A549 | Growth Inhibition Assay | 10/15/20 nM | 72 h | DMSO | enhances the antiproliferative effect of erlotinib | 26675484 |
| NCI-H460 | Growth Inhibition Assay | 10/20/30 nM | 72 h | DMSO | enhances the antiproliferative effect of erlotinib | 26675484 |
| J89GFP | Growth Inhibition Assay | DMSO | EC50=49.85 ± 12.65 nM | 26563568 | ||
| THP89GFP | Growth Inhibition Assay | DMSO | EC50=19.34 ± 6.43 nM | 26563568 | ||
| SK-NEP-1 | Growth Inhibition Assay | 0.01–10.0 μM | 24 h | DMSO | IC50=76.34 nM | 26176219 |
| G401 | Growth Inhibition Assay | 0.01–10.0 μM | 24 h | DMSO | IC50=143.02 nM | 26176219 |
| SK-NEP-1 | Cell Viability Assay | 50 nM | 1–4 d | DMSO | reduces cell survival in a time dependent manner | 26176219 |
| G401 | Cell Viability Assay | 50 nM | 1–4 d | DMSO | reduces cell survival in a time dependent manner | 26176219 |
| SK-NEP-1 | Apoptosis Assay | 50/100 nM | 24 h | DMSO | induces cell apoptosis in a dose-dependent manner | 26176219 |
| G401 | Apoptosis Assay | 50/100 nM | 24 h | DMSO | induces cell apoptosis in a dose-dependent manner | 26176219 |
| SK-NEP-1 | Function Assay | 50/100 nM | 24 h | DMSO | shows the induction of DNA fragmentation | 26176219 |
| G401 | Function Assay | 50/100 nM | 24 h | DMSO | shows the induction of DNA fragmentation | 26176219 |
| SK-NEP-1 | Function Assay | 50/100 nM | 24 h | DMSO | induces cell cycle disorder | 26176219 |
| G401 | Function Assay | 50/100 nM | 24 h | DMSO | induces cell cycle disorder | 26176219 |
| RPMI 8226 | Cell Survival Assay | 2/4/6 nM | 48 h | induces a significant decrease in the cell growth | 26000292 | |
| OPM2 | Cell Survival Assay | 2/4/6 nM | 48 h | induces a significant decrease in the cell growth | 26000292 | |
| U266 | Cell Survival Assay | 2/4/6 nM | 48 h | induces a significant decrease in the cell growth | 26000292 | |
| H929 | Cell Survival Assay | 2/4/6 nM | 48 h | induces a significant decrease in the cell growth | 26000292 | |
| RPMI 8226 | Apoptosis Assay | 4 nM | 24/48 h | induces cell apoptosis in a time-dependent manner | 26000292 | |
| HCC827 | Growth Inhibition Assay | 10 nM | 48 h | DMSO | enhances cisplatin sensitivity | 25944617 |
| NCI-H23 | Growth Inhibition Assay | 10 nM | 48 h | DMSO | enhances cisplatin sensitivity | 25944617 |
| AML3 | Function Assay | 0-1 μM | 24 h | induces DNA fragmentation in a dose-dependent manner | 25612941 | |
| ML-1 | Function Assay | 0-1 μM | 24 h | induces DNA fragmentation in a dose-dependent manner | 25612941 | |
| RPMI-8226vr10 | Function Assay | 0-1 μM | 24 h | induces DNA fragmentation in a dose-dependent manner | 25612941 | |
| ML-1 | Function Assay | 1 μM | 24 h | increases caspase-3 activity 4-fold | 25612941 | |
| RPMI-8226vr10 | Function Assay | 1 μM | 24 h | increases caspase-3 activity 2.5-fold | 25612941 | |
| SK-N-BE (2) | Growth Inhibition Assay | 24 h | IC50=104.0 ± 7.8 nM | 25308916 | ||
| SK-N-BE (2), PAN MK | Growth Inhibition Assay | 24 h | IC50=104.0 ± 7.8 nM | 25308916 | ||
| SK-N-BE (2), MK PAN | Growth Inhibition Assay | 24 h | IC50=382.0 ± 43.2 nM | 25308916 | ||
| SK-N-AS | Growth Inhibition Assay | 24 h | IC50=37.1 ± 2.4 nM | 25308916 | ||
| SK-N-DZ | Growth Inhibition Assay | 24 h | IC50=17.1 ± 0.4 nM | 25308916 | ||
| Caki-1 | Growth Inhibition Assay | 10/25/50 nM | 48 h | inhibits cell growth in a dose dependent manner synergistically with ritonavir | 25279191 | |
| ACHN | Growth Inhibition Assay | 10/25/50 nM | 48 h | inhibits cell growth in a dose dependent manner synergistically with ritonavir | 25279191 | |
| 769-P | Growth Inhibition Assay | 10/25/50 nM | 48 h | inhibits cell growth in a dose dependent manner synergistically with ritonavir | 25279191 | |
| 786-O | Growth Inhibition Assay | 10/25/50 nM | 48 h | inhibits cell growth in a dose dependent manner synergistically with ritonavir | 25279191 | |
| Caki-1 | Apoptosis Assay | 50 nM | 48 h | induces cell apoptosis combined ritonavir | 25279191 | |
| ACHN | Apoptosis Assay | 50 nM | 48 h | induces cell apoptosis combined ritonavir | 25279191 | |
| 769-P | Apoptosis Assay | 50 nM | 48 h | induces cell apoptosis combined ritonavir | 25279191 | |
| 786-O | Apoptosis Assay | 50 nM | 48 h | induces cell apoptosis combined ritonavir | 25279191 | |
| Caki-1 | Growth Inhibition Assay | 25/50 nM | 48 h | DMSO | inhibits cell growth in a dose dependent manner synergistically with bortezomib | 25176354 |
| ACHN | Growth Inhibition Assay | 25/50 nM | 48 h | DMSO | inhibits cell growth in a dose dependent manner synergistically with bortezomib | 25176354 |
| 769-P | Growth Inhibition Assay | 25/50 nM | 48 h | DMSO | inhibits cell growth in a dose dependent manner synergistically with bortezomib | 25176354 |
| Caki-1 | Colony Formation Assay | 50 nM | 7-14 d | DMSO | suppressed colony formation significantly combined with with bortezomib | 25176354 |
| ACHN | Colony Formation Assay | 50 nM | 7-14 d | DMSO | suppressed colony formation significantly combined with with bortezomib | 25176354 |
| 769-P | Colony Formation Assay | 50 nM | 7-14 d | DMSO | suppressed colony formation significantly combined with with bortezomib | 25176354 |
| Caki-1 | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis | 25176354 |
| ACHN | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis | 25176354 |
| 769-P | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis | 25176354 |
| MDA-MB-231 | Morphological Crystal Violet (CV) Assay | 10 nM | 3 d | DMSO | alters cell morphology | 24810497 |
| BT-549 | Morphological Crystal Violet (CV) Assay | 10 nM | 3 d | DMSO | alters cell morphology | 24810497 |
| MCF-7 | Morphological Crystal Violet (CV) Assay | 10 nM | 3 d | DMSO | alters cell morphology | 24810497 |
| MCF-7 | Function Assay | 5-50 nM | 24 h | DMSO | reduced the level of expression of ERα, PR and FoxA1 | 24366407 |
| CTS | Apoptosis Assay | 0–40 nM | 48 h | induces apoptosis in a dose-dependent manner | 24244429 | |
| OCI-AML3 | Apoptosis Assay | 0–40 nM | 48 h | induces apoptosis in a dose-dependent manner | 24244429 | |
| U937 | Apoptosis Assay | 0–40 nM | 48 h | induces apoptosis in a dose-dependent manner | 24244429 | |
| PC3 | Apoptosis Assay | 0-100 nM | 24/48 h | induces apoptosis in a dose-dependent manner | 24163230 | |
| PC3-AR | Apoptosis Assay | 0-100 nM | 24/48 h | induces apoptosis in both time- and dose-dependent manner | 24163230 | |
| PC3 | Growth Inhibition Assay | 0-100 nM | 24/48 h | induces accumulation of subG1 population | 24163230 | |
| PC3-AR | Growth Inhibition Assay | 0-100 nM | 24/48 h | induces cell cycle arrest in the G2M phase | 24163230 | |
| PC3 | Function Assay | 0-100 nM | 24 h | suppresses expression of activated ATM, Akt and Erk1/2 protein | 24163230 | |
| PC3-AR | Function Assay | 0-100 nM | 24 h | suppresses expression of activated ATM, Akt and Erk1/2 protein | 24163230 | |
| OS-RC-2 | Cell Viability Assay | 0-1000 nM | 24/48/72 h | DMSO | decreases cell viability in both time- and dose-dependent manner | 24144737 |
| OS-RC-2 | Growth Inhibition Assay | 50 nM | 48 h | DMSO | induces G2/M arrest | 24144737 |
| OS-RC-2 | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis | 24144737 |
| SK-N-AS | Growth Inhibition Assay | 0–80 nM | 48 h | IC50=27.4 nM | 24098799 | |
| SK-N-DZ | Growth Inhibition Assay | 0–80 nM | 48 h | IC50=21.9 nM | 24098799 | |
| SK-N-SH | Growth Inhibition Assay | 0–80 nM | 48 h | IC50=72.3 nM | 24098799 | |
| SK-N-BE | Growth Inhibition Assay | 0–80 nM | 48 h | IC50=75.4 nM | 24098799 | |
| SK-N-AS | Apoptosis Assay | 0–80 nM | 48 h | potently induced apoptosis in a dose-dependent fashion | 24098799 | |
| SK-N-DZ | Apoptosis Assay | 0–80 nM | 48 h | potently induced apoptosis in a dose-dependent fashion | 24098799 | |
| SK-N-SH | Apoptosis Assay | 0–40 nM | 48 h | potently induced apoptosis in a dose-dependent fashion | 24098799 | |
| SK-N-BE | Apoptosis Assay | 0–40 nM | 48 h | potently induced apoptosis in a dose-dependent fashion | 24098799 | |
| SK-N-AS | Function Assay | 0–80 nM | 48 h | induces a dose-dependent cleavage of caspase 3 and PARP | 24098799 | |
| SK-N-DZ | Function Assay | 0–80 nM | 48 h | induces a dose-dependent cleavage of caspase 3 and PARP | 24098799 | |
| SK-N-SH | Function Assay | 0–40 nM | 48 h | induces a dose-dependent cleavage of caspase 3 and PARP | 24098799 | |
| SK-N-BE | Function Assay | 0–40 nM | 48 h | induces a dose-dependent cleavage of caspase 3 and PARP | 24098799 | |
| HCC-LM3 | Growth Inhibition Assay | 1-1000 nM | 24/48/72 h | DMSO | inhibits cell growth in both time- and dose-dependent manner | 24093956 |
| HepG2 | Growth Inhibition Assay | 1-1000 nM | 24/48/72 h | DMSO | inhibits cell growth in both time- and dose-dependent manner | 24093956 |
| SMMC-7721 | Growth Inhibition Assay | 1-1000 nM | 24/48/72 h | DMSO | inhibits cell growth in both time- and dose-dependent manner | 24093956 |
| HCC-LM3 | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis significantly in a caspase-dependent manner by cleavage of caspases 3, 8 and 9 | 24093956 |
| HepG2 | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis significantly in a caspase-dependent manner by cleavage of caspases 3, 8 and 9 | 24093956 |
| SMMC-7721 | Apoptosis Assay | 50 nM | 48 h | DMSO | induces cell apoptosis significantly in a caspase-dependent manner by cleavage of caspases 3, 8 and 9 | 24093956 |
| HCC-LM3 | Function Assay | 50/100 nM | 24 h | DMSO | decreases the levels of p-STAT3 and p-Akt | 24093956 |
| HepG2 | Function Assay | 50/100 nM | 24 h | DMSO | decreases the levels of p-STAT3 and p-Akt | 24093956 |
| SMMC-7721 | Function Assay | 50/100 nM | 24 h | DMSO | decreases the levels of p-STAT3 and p-Akt | 24093956 |
| HCC-LM3 | Function Assay | 50/100 nM | 24 h | DMSO | downregulates Bcl-xL expression | 24093956 |
| HepG2 | Function Assay | 50/100 nM | 24 h | DMSO | downregulates Bcl-xL expression | 24093956 |
| SMMC-7721 | Function Assay | 50/100 nM | 24 h | DMSO | downregulates Bcl-xL expression | 24093956 |
| FaDu | Growth Inhibition Assay | 100 nM | 8/10/12 h | displayed a significant and prolonged G2/M arrest at 8 and 12 h post release | 24026482 | |
| FaDu | Function Assay | 100 nM | 2/4/8/12 h | induced p21Waf1/Cip1 expression | 24026482 | |
| PC-3 | Growth Inhibition Assay | 0-10 μM | 24/48/72 h | inhibits cell growth in both time- and dose-dependent manner | 23991216 | |
| LNCaP | Growth Inhibition Assay | 0-5 μM | 24/48/72 h | inhibits cell growth in both time- and dose-dependent manner | 23991216 | |
| RWPE-1 | Growth Inhibition Assay | 0-20 μM | 24/48/72 h | inhibits cell growth in both time- and dose-dependent manner | 23991216 | |
| Capan-1 | Function Assay | 25/50/100 nM | 8/24/48 h | DMSO | downregulated Ron mRNA and protein expression and downstream signaling | 23922886 |
| L3.6pl | Function Assay | 25/50/100 nM | 8/24/48 h | DMSO | downregulated Ron mRNA and protein expression and downstream signaling | 23922886 |
| CFPAC-1 | Function Assay | 25/50/100 nM | 8/24/48 h | DMSO | downregulated Ron mRNA and protein expression and downstream signaling | 23922886 |
| Capan-1 | Growth Inhibition Assay | 25/50/100 nM | 48 h | DMSO | reduces cell growth in a dose-dependent manner | 23922886 |
| L3.6pl | Growth Inhibition Assay | 25/50/100 nM | 48 h | DMSO | reduces cell growth in a dose-dependent manner | 23922886 |
| CFPAC-1 | Growth Inhibition Assay | 25/50/100 nM | 48 h | DMSO | reduces cell growth in a dose-dependent manner | 23922886 |
| Capan-1 | Apoptosis Assay | 25/50/100 nM | 48 h | DMSO | induces cell growth in a dose-dependent manner | 23922886 |
| L3.6pl | Apoptosis Assay | 25/50/100 nM | 48 h | DMSO | induces cell growth in a dose-dependent manner | 23922886 |
| CFPAC-1 | Apoptosis Assay | 25/50/100 nM | 48 h | DMSO | induces cell growth in a dose-dependent manner | 23922886 |
| HN22 | Growth Inhibition Assay | 0-20 nM | 24/48 h | DMSO | inhibits cell viability in both time- and dose- dependent manner | 23877235 |
| HSC4 | Growth Inhibition Assay | 0-20 nM | 24/48 h | DMSO | inhibits cell viability in both time- and dose- dependent manner | 23877235 |
| HN22 | Apoptosis Assay | 0-20 nM | 48 h | DMSO | induces cell apoptosis | 23877235 |
| HSC4 | Apoptosis Assay | 0-20 nM | 48 h | DMSO | induces cell apoptosis | 23877235 |
| HN22 | Growth Inhibition Assay | 0-20 nM | 48 h | DMSO | induces G1 phase cell cycle arrest | 23877235 |
| HSC4 | Growth Inhibition Assay | 0-20 nM | 48 h | DMSO | induces G1 phase cell cycle arrest | 23877235 |
| HN22 | Function Assay | 0-20 nM | 48 h | DMSO | suppresses Sp1 expression | 23877235 |
| HSC4 | Function Assay | 0-20 nM | 48 h | DMSO | suppresses Sp1 expression | 23877235 |
| Cal62 | Growth Inhibition Assay | IC50=33 ± 4 nM | 23824064 | |||
| Hth7 | Growth Inhibition Assay | IC50=15 ± 2 nM | 23824064 | |||
| Hth83 | Growth Inhibition Assay | IC50=34 ± 5 nM | 23824064 | |||
| C643 | Growth Inhibition Assay | IC50=71 ± 10 nM | 23824064 | |||
| SW1736 | Growth Inhibition Assay | IC50=35 ± 8 nM | 23824064 | |||
| T241 | Growth Inhibition Assay | IC50=65 ± 7 nM | 23824064 | |||
| T351 | Growth Inhibition Assay | IC50=50 ± 10 nM | 23824064 | |||
| BHP2-7 | Growth Inhibition Assay | IC50=37 ± 6 nM | 23824064 | |||
| T238 | Growth Inhibition Assay | IC50=1,500 ± 200 nM | 23824064 | |||
| HCT8 | Growth Inhibition Assay | 72 h | DMSO | IC50=12.9 ± 1.9 nM | 23299388 | |
| H630 | Growth Inhibition Assay | 72 h | DMSO | IC50=12.4 ± 3.1 nM | 23299388 | |
| cH630 5-FU-res | Growth Inhibition Assay | 72 h | DMSO | IC50=15.5 ± 1.2 nM | 23299388 | |
| HCT116 | Growth Inhibition Assay | 72 h | DMSO | IC50=10.7 ± 2.2 nM | 23299388 | |
| HCT116 p53−/− | Growth Inhibition Assay | 72 h | DMSO | IC50=8.6 ± 1.7 nM | 23299388 | |
| dHCT116 p21−/− | Growth Inhibition Assay | 72 h | DMSO | IC50=5.9 ± 1.3 nM | 23299388 | |
| HT29 | Growth Inhibition Assay | 72 h | DMSO | IC50=16.3 ± 2.3 nM | 23299388 | |
| LoVo | Growth Inhibition Assay | 72 h | DMSO | IC50=5.1 ± 0.6 nM | 23299388 | |
| RKO | Growth Inhibition Assay | 72 h | DMSO | IC50=7.9 ± 2.2 nM | 23299388 | |
| SW480 | Growth Inhibition Assay | 72 h | DMSO | IC50=17.5 ± 0.8 nM | 23299388 | |
| eSW620 | Growth Inhibition Assay | 72 h | DMSO | IC50=9.1 ± 2.1 nM | 23299388 | |
| S2 | Function assay | Inhibition of Plasmodium falciparum HDAC1 expressed in Drosophila melanogaster S2 cells, IC50 = 0.0018 μM. | 19317450 | |||
| COLO205 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human COLO205 cells after 96 hrs by celltiter 96 assay, IC50 = 0.018 μM. | 21634430 | ||
| PC3 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human PC3 cells after 96 hrs by celltiter 96 assay, IC50 = 0.024 μM. | 21634430 | ||
| A2780 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human A2780 cells after 96 hrs by celltiter 96 assay, IC50 = 0.035 μM. | 21634430 | ||
| HCT116 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HCT116 cells after 96 hrs by celltiter 96 assay, IC50 = 0.048 μM. | 21634430 | ||
| HEK293 | Function assay | Inhibition of HDAC3 (unknown origin) expressed in HEK293 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.0021 μM. | 22344701 | |||
| HEK293 | Function assay | Inhibition of HDAC1 (unknown origin) expressed in HEK293 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.0025 μM. | 22344701 | |||
| HEK293 | Function assay | Inhibition of HDAC6 (unknown origin) expressed in HEK293 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.011 μM. | 22344701 | |||
| SF21 | Function assay | Inhibition of flag-tagged HDAC2 (unknown origin) expressed in SF21 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.013 μM. | 22344701 | |||
| HEK293 | Function assay | Inhibition of HDAC4 (unknown origin) expressed in HEK293 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.2 μM. | 22344701 | |||
| SF9 | Function assay | Inhibition of his-strep-tagged HDAC8 (unknown origin) expressed in SF9 cells using [3H]acetylated human histone H4 peptide as substrate by scintillation counting, IC50 = 0.28 μM. | 22344701 | |||
| B16 | Growth inhibition assay | 48 hrs | Growth inhibition of mouse B16 cells incubated for 48 hrs by MTT assay, GI50 = 0.15 μM. | 23009203 | ||
| HeLa | Function assay | Inhibition of HDAC in human HeLa cells using Fluor de Lys as substrate by fluorescence assay, IC50 = 0.03 μM. | 23639537 | |||
| HuH7 | Cytotoxicity assay | 3 days | Cytotoxicity against human HuH7 cells assessed as inhibition of cell viability after 3 days by CellTiter 96 assay, CC50 = 0.0035 μM. | 25490700 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length C-terminal His/FLAG-tagged human recombinant HDAC1 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured a, IC50 = 0.00126 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length C-terminal His-tagged human recombinant HDAC3/NCOR2 (395 to 489 residues) expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substr, IC50 = 0.00227 μM. | 27186676 | ||
| MV4-11 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MV4-11 cells assessed as growth inhibition after 24 hrs by MTT assay, IC50 = 0.00297 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length human recombinant HDAC2 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after 1 hr by fluorescence a, IC50 = 0.00328 μM. | 27186676 | ||
| HCT116 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HCT116 cells assessed as growth inhibition after 24 hrs by MTT assay, IC50 = 0.00336 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length human recombinant HDAC6 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after 1 hr by fluorescence a, IC50 = 0.00416 μM. | 27186676 | ||
| Sf9 | Inhibition of human | 15 mins | Inhibition of human recombinant HDAC10 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after 1 hr by fluorescence assay, IC50 = 0.00445 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length C-terminal His-tagged human recombinant HDAC8 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after , IC50 = 0.00486 μM. | 27186676 | ||
| A2780S | Cytotoxicity assay | 24 hrs | Cytotoxicity against human A2780S cells assessed as growth inhibition after 24 hrs by MTT assay, IC50 = 0.00832 μM. | 27186676 | ||
| A2780S | Function assay | 6 hrs | Inhibition of HDAC6 in human A2780S cells assessed as tubulin acetylation incubated for 6 hrs by cytoblot assay, EC50 = 0.15071 μM. | 27186676 | ||
| A2780S | Function assay | 6 hrs | Inhibition of HDAC1/2/3 in human A2780S cells assessed as histone H3 acetylation incubated for 6 hrs by cytoblot assay, EC50 = 0.1695 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of human recombinant HDAC5 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after 1 hr by fluorescence assay, IC50 = 0.1903 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of N-terminal GST/C-terminal His-tagged human recombinant HDAC4 (627 to 1084 residues) expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrat, IC50 = 0.3378 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of C-terminal His-tagged human recombinant HDAC9 (604 to 1066 residues) expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition meas, IC50 = 0.8878 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of full length human recombinant HDAC11 expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measured after 1 hr by fluorescence , IC50 = 4.112 μM. | 27186676 | ||
| Sf9 | Function assay | 15 mins | Inhibition of N-terminal GST-tagged human recombinant HDAC7 (518 to end residues) expressed in baculovirus infected insect Sf9 cells using Ac-peptide-AMC as substrate assessed as release of AMC preincubated for 15 mins followed by substrate addition measu, IC50 = 4.354 μM. | 27186676 | ||
| Sf9 | Function assay | Inhibition of C-terminal His-tagged and C-terminal FLAG-tagged full length human recombinant HDAC1 expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate , IC50 = 0.001 μM. | 27377864 | |||
| Sf9 | Function assay | 60 mins | Inhibition full length human recombinant HDAC2 expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by fluorescence assay, IC50 = 0.002 μM. | 27377864 | ||
| Sf9 | Function assay | 60 mins | Inhibition of human recombinant HDAC6 expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by fluorescence assay, IC50 = 0.002 μM. | 27377864 | ||
| Sf9 | Function assay | 60 mins | Inhibition of N-terminal GST-tagged full length human recombinant HDAC5 expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by fluorescen, IC50 = 0.092 μM. | 27377864 | ||
| Sf9 | Function assay | 60 mins | Inhibition of C-terminal His-tagged full length human recombinant HDAC8 expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by fluorescen, IC50 = 0.231 μM. | 27377864 | ||
| Sf9 | Function assay | Inhibition of N-terminal GST-tagged and C-terminal His-tagged human recombinant HDAC4 (627 to 1084 residues ) expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as, IC50 = 0.373 μM. | 27377864 | |||
| Sf9 | Function assay | 60 mins | Inhibition of C-terminal His-tagged human recombinant HDAC9 (604 to 1066 residues) expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by, IC50 = 2.68 μM. | 27377864 | ||
| Sf9 | Function assay | 60 mins | Inhibition of N-terminal GST-tagged human recombinant HDAC7 (518 to end residues) expressed in baculovirus coexpressed in fall armyworm Sf9 cells using carboxyfluorescein (FAM)-labeled acetylated/ trifluoroacetylated peptide as substrate after 60 mins by , IC50 = 2.83 μM. | 27377864 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by resazurin assay, IC50 = 0.07 μM. | 28241112 | ||
| NFF | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NFF cells after 72 hrs by SRB assay, IC50 = 0.07 μM. | 28241112 | ||
| HUT78 | Apoptosis assay | 18 hrs | Pro-apoptotic activity in human HUT78 cells after 18 hrs by caspase-Glo 3/7 assay, EC50 = 0.0043 μM. | 30122227 | ||
| NFF | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NFF cells after 72 hrs by sulforhodamine B assay, IC50 = 0.07 μM. | 30245402 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by resazurin dye based assay, IC50 = 0.07 μM. | 30245402 | ||
| M14 | Apoptosis assay | 24 to 48 hrs | Induction of apoptosis human M14 cells assessed as caspase activity after 24 to 48 hrs using DEVD peptide as substrate by ApoTox-Glo triplex assay | 24471466 | ||
| U937 | Function assay | 1 uM | 24 hrs | Inhibition of HDAC6 in human U937 cells assessed as increase of intracellular acetylated alpha-tubulin level at 1 uM after 24 hrs by Western blot analysis | 24694055 | |
| U937 | Function assay | 1 uM | 24 hrs | Inhibition of HDAC1 in human U937 cells assessed as increase of intracellular acetylated histone H3 level at 1 uM after 24 hrs by Western blot analysis | 24694055 | |
| U937 | Function assay | 1 uM | 24 hrs | Inhibition of HDAC2 in human U937 cells assessed as increase of intracellular acetylated histone H3 level at 1 uM after 24 hrs by Western blot analysis | 24694055 | |
| U937 | Function assay | 1 uM | 24 hrs | Inhibition of HDAC3 in human U937 cells assessed as increase of intracellular acetylated histone H4 level at 1 uM after 24 hrs by Western blot analysis | 24694055 | |
| MV4-11 | Function assay | 10 to 1000 nM | 6 hrs | Inhibition of HDAC6 in human MV4-11 cells assessed as upregulation of alpha tubulin acetylation at 10 to 1000 nM after 6 hrs by Western blot analysis | 26443078 | |
| HCT116 | Function assay | 10 to 1000 nM | 6 hrs | Inhibition of HDAC6 in human HCT116 cells assessed as upregulation of alpha tubulin acetylation at 10 to 1000 nM after 6 hrs by Western blot analysis | 26443078 | |
| HCT116 | Function assay | 10 to 1000 nM | 6 hrs | Inhibition of HDAC1/2/3 in human HCT116 cells assessed as upregulation of histone H3 acetylation at 10 to 1000 nM after 6 hrs by Western blot analysis | 26443078 | |
| MV4-11 | Function assay | 10 to 1000 nM | 6 hrs | Inhibition of HDAC1/2/3 in human MV4-11 cells assessed as upregulation of histone H3 acetylation at 10 to 1000 nM after 6 hrs by Western blot analysis | 26443078 | |
| Raji | Cytotoxicity assay | 24 hrs | Cytotoxicity against human Raji cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| Ramos | Cytotoxicity assay | 24 hrs | Cytotoxicity against human Ramos cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| U266 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human U266 cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| RPMI8226 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human RPMI8226 cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| HBL1 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HBL1 cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| MM1S | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MM1S cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| OCI-LY1 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human OCI-LY1 cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| SUDHL4 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human SUDHL4 cells assessed as growth inhibition after 24 hrs by MTT assay | 27186676 | ||
| MV4-11 | Function assay | 50 nM | 24 hrs | Induction of HDAC6 degradation in human MV4-11 cells at 50 nM after 24 hrs by Western blot method | 29589441 | |
| MV4-11 | Function assay | 50 nM | 2 to 24 hrs | Inhibition of HDAC6 in human MV4-11 cells assessed as induction of alpha-tubulin hyperacetylation at 50 nM after 2 to 24 hrs by Western blot method | 29589441 | |
| MV4-11 | Cell cycle arrest assay | 30 to 50 nM | 24 hrs | Cell cycle arrest in human MV4-11 cells assessed as accumulation at sub-G1 phase at 30 to 50 nM after 24 hrs by propidium iodide staining-based flow cytometric method | 29589441 | |
| MV4-11 | Apoptosis assay | 30 nM | 24 to 48 hrs | Induction of apoptosis in human MV4-11 cells at 30 nM after 24 to 48 hrs by Annexin V-PI staining based flow cytometry | 29738953 | |
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Panobinostat (LBH589, NVP-LBH589) is a novel broad-spectrum HDAC inhibitor with IC50 of 5 nM in a cell-free assay. Panobinostat (LBH589) induces autophagy and apoptosis. Panobinostat effectively disrupts HIV latency in vivo. Phase 3. | ||||
|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | LBH589 induces apoptosis among MOLT-4 and Reh cells in a time- and dose-dependent manner. Moreover, LBH589 is more potent in MOLT-4 than in Reh cells. LBH589 markedly prevents the growth of both MOLT-4 and Reh cells in a dose-dependent manner at 48 hours. LBH589 treatment causes a 2- to 3-fold increase in the number of cells in the G2/M phase of the cell cycle compared with the control cells. LBH589 is associated with induction of histone H3K9 and histone H4K8 acetylation as well as decreasing levels of c-Myc expression in a dose-dependent manner. LBH589 treatment also increases the levels of p21 expression. LBH589 treatment also decreases the levels of c-Myc after an initial increase at the lowest dose (10 nM) in Reh cells. In addition, LBH589 gives rise to substantial increases in mRNA levels of proapoptosis and DNA repair genes. LBH589 induces increased levels of acetylated histone H3 and H4 at the GADD45G promoter. [1] Besides, LBH589 inhibits growth of non small cell lung cancer cell lines (such as human H1299, L55 and A549 with IC50 of 5 nM, 11 nM and 30 nM, respectively), mesothelioma (such as human OK-6 and Ok-5 with IC50 of 5 nM and 7 nM, respectively) and small cell lung cancer cell lines (such as human RG-1 and LD-T with IC50 of 4 nM and 5 nM, respectively). [2] | |||
|---|---|---|---|---|
| Cell Research | Cell lines | MOLT-4 cell lines and Reh (pre-B cells) | ||
| Concentrations | 50 nM | |||
| Incubation Time | 48 hours | |||
| Method | Untreated and LBH589-treated cells [human Ph- acute lymphoblastic leukemia MOLT-4 (T cells) and Reh (pre-B cells)] are stained with annexin V and propidium iodide using annexin V-FITC apoptosis detection kit I. The percentage of apoptotic and nonviable cells is determined by flow cytometry. At least 5 × 104 cells are collected with a CyAn ADP Violet cytometer. Percentage apoptosis is calculated considering all the annexin V-positive plus the annexin V/PI-positive cells; percentage loss of cell viability is calculated considering all the annexin V-positive plus the PI-positive and the annexinV/PI-positive cells. | |||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | DNMT1 / EZH2 caspase-8 / cleaved caspase-8 / Sp1 c-Myc / IRF4 Ac-H3 / cleaved caspase-3 / CCND1 / ID1 / ID2 / ID3 / ID4 / Synaptophysin / NeuroD1 RAD51 / BRCA1 / CHK1 / RPL13a H3K9AC / H3K18AC / H3K56AC / H3 / H4K8AC / H4K16AC / H4 / p21 / p27 / cleaved PARP |
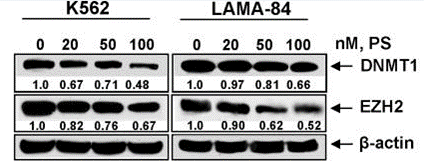
|
19279403 | |
| Immunofluorescence | Synaptophysin / NACM α-tubulin / Acetyl-α-tubulin BiP ATF4 IRE1α / S724-IRE1α |
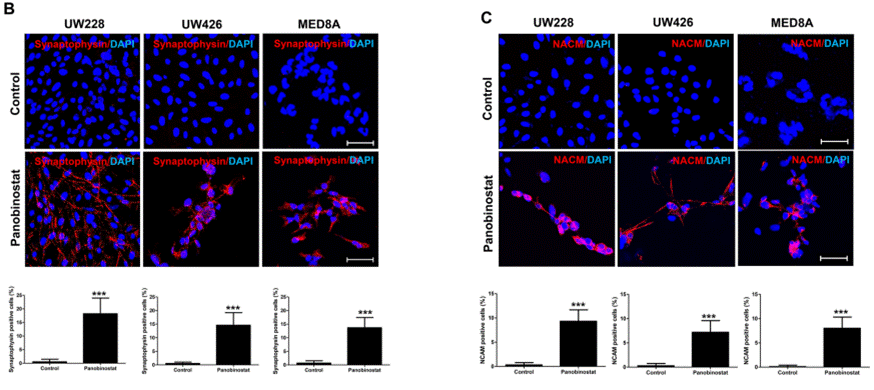
|
28915627 | |
| Growth inhibition assay | Cell viability |
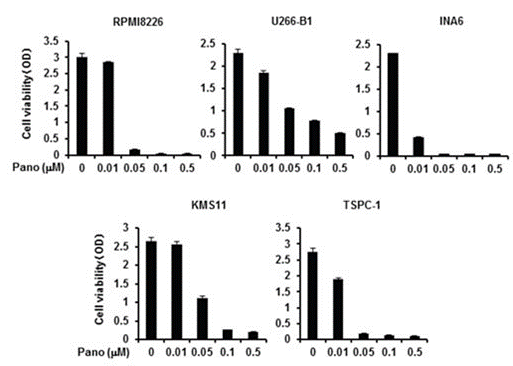
|
27738323 | |
| In Vivo | ||
| In vivo | In lung cancer and mesothelioma animal models, LBH589 markedly decreases tumor growth by 62%. LBH589 is equally effective in immunocompetent and severe combined immunodeficien-cymice, suggesting that the inhibition of tumor growth by LBH589 is not due to direct immunologic effects. Daily LBH589, given i.p. at 20 mg/kg for 5 days per week, leading to an average decrease in growth of 70%. Compared with the corresponding control tumors, LBH589 leads to a 53% decrease for H526-derived tumors, an 81% decrease for BK-T-derived tumors, a 76% decrease for RG-1- derived tumors, and a 70% decrease for H69-derived tumors. In contrast to the lack of tumor regression notes in NSCLC and Meso-derived xenografted tumors that are treated under identical conditions and doses, LBH589 results in dramatic tumor regression in SCLC-derived tumors and RG-1-derived tumor. [2] | |
|---|---|---|
| Animal Research | Animal Models | Severe combined immunodeficiency (SCID) mice with M30 (107 cells) or A549 (5 × 106 cells), H69 (2.5 × 106 cells), BK-T (6.5 × 106), H526 (10 × 106), and RG1 (10 × 106) cells |
| Dosages | 10 mg/kg, 20 mg/kg | |
| Administration | Administered via i.p. injection | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04341311 | Active not recruiting | Diffuse Intrinsic Pontine Glioma|Pediatric Brainstem Glioma|Pediatric Brainstem Gliosarcoma Recurrent|Pediatric Cancer|Pediatric Brain Tumor|Diffuse Glioma |
Dana-Farber Cancer Institute|Celgene|Secura Bio Inc. |
August 10 2020 | Phase 1 |
| NCT03632317 | Withdrawn | Glioma|Diffuse Intrinsic Pontine Glioma |
University of Michigan Rogel Cancer Center |
October 2019 | Phase 2 |
| NCT03982134 | Withdrawn | Melanoma|Non Small Cell Lung Cancer |
Muhammad Furqan|Novartis Pharmaceuticals|University of Iowa |
September 2019 | Phase 1 |
| NCT04326764 | Terminated | Acute Myeloid Leukaemia (AML)|Myelodysplastic Syndromes (MDS) |
Goethe University|Stichting Hemato-Oncologie voor Volwassenen Nederland|Polish Adult Leukemia Group|Schweizerische Arbeitsgemeinschaft für klinische Krebsforschung |
July 24 2018 | Phase 3 |
| NCT03515915 | Unknown status | Patients With Recurrent or Refractory Multiple Myeloma |
University Hospital Montpellier|Poitiers University Hospital |
April 23 2018 | -- |
Chemical Information & Solubility
| Molecular Weight | 349.43 | Formula | C21H23N3O2 |
| CAS No. | 404950-80-7 | SDF | Download Panobinostat (LBH589) SDF |
| Smiles | CC1=C(C2=CC=CC=C2N1)CCNCC3=CC=C(C=C3)C=CC(=O)NO | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 70 mg/mL ( (200.32 mM); Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Frequently Asked Questions
Question 1:
How to reconstitute the compound for in vivo mice study?
Answer:
We recommend the vehicle is 2 % DMSO, 2 % Tween 80, 48%PEG300, 48% water. The compound is first dissolved in DMSO, then add Tween, PEG300, water in sequence.
Tags: buy Panobinostat (LBH589) | Panobinostat (LBH589) supplier | purchase Panobinostat (LBH589) | Panobinostat (LBH589) cost | Panobinostat (LBH589) manufacturer | order Panobinostat (LBH589) | Panobinostat (LBH589) distributor