research use only
Abexinostat (PCI-24781) HDAC inhibitor
Cat.No.S1090
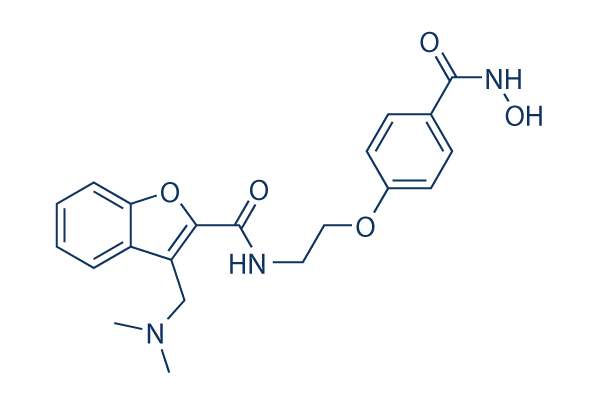
Chemical Structure
Molecular Weight: 397.42
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HeLa | Function assay | 2 hrs | Inhibition of dye-labeled tracer binding to HDAC10 (unknown origin) transfected in human HeLa cells measured after 2 hrs by nano-luciferase reporter gene-based BRET assay, IC50=0.007943μM | 30964290 | ||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB1643 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-MC cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-12 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for LAN-5 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for DAOY cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB-EBc1 cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SJ-GBM2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-37 cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for TC32 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for MG 63 (6-TG R) cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for RD cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh18 cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh30 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Saos-2 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for OHS-50 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-SH cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 80 mg/mL
(201.29 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 397.42 | Formula | C21H23N3O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 783355-60-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | CRA-024781 | Smiles | CN(C)CC1=C(OC2=CC=CC=C21)C(=O)NCCOC3=CC=C(C=C3)C(=O)NO | ||
Mechanism of Action
| Targets/IC50/Ki |
HDAC1
(Cell-free assay) 7 nM(Ki)
HDAC3/SMRT
8.2 nM(Ki)
HDAC6
17 nM(Ki)
HDAC2
(Cell-free assay) 19 nM(Ki)
HDAC10
24 nM
HDAC8
280 nM
|
|---|---|
| In vitro |
Abexinostat (PCI-24781) exhibits potent antitumor activity against a variety of tumor cell lines with GI50 ranging from 0.15 μM to 3.09 μM, and also has an antiproliferative effect on HUVEC endothelial cells with GI50 of 0.43 μM. Treatment with this compound causes dose-dependent accumulation of both acetylated histones and acetylated tubulin in HCT116 or DLD-1 cells, induces expression of p21, and leads to PARP cleavage and accumulation of the γH2AX. Inhibition of HDAC enzymes by PCI-24781 leads to a significant reduction in the transcription of genes specifically associated with HR, including RAD51. Consistent with inhibition of HR, it results in a decreased ability to perform homology directed repair of I-SceI-induced chromosome breaks in transfected CHO cells. It induces S phase depletion, G2 cell cycle arrest, and apoptosis in soft tissue sarcoma (STS) cells, and also causes Rad51 transcriptional repression in STS cells potentially mediated via enhanced E2F1 binding to the Rad51 proximal promoter. Additionally, this compound induces caspase and reactive oxygen species-dependent apoptosis through NF-κB mechanisms in Hodgkin lymphoma and non-Hodgkin lymphoma cell lines.
|
| Kinase Assay |
HDAC Activity
|
|
HDAC activity is measured using a continuous trypsin-coupled assay. For inhibitor characterization, measurements are done in a reaction volume of 100 μL using 96-well assay plates. For each isozyme, the HDAC protein in reaction buffer [50 mM HEPES, 100 mM KCl, 0.001% Tween 20, 5% DMSO (pH 7.4), supplemented with bovine serum albumin at concentrations of 0% (HDAC1), 0.01% (HDAC2, 3, 8, and 10), or 0.05% (HDAC6)] is mixed with Abexinostat (PCI-24781) at various concentrations and allowed to incubate for 15 minutes. Trypsin is added to a final concentration of 50 nM, and acetyl-Gly-Ala-(N-acetyl-Lys)-AMC is added to a final concentration of 25 μM (HDAC1, 3, and 6), 50 μM (HDAC2 and 10), or 100 μM (HDAC8) to initiate the reaction. Negative control reactions are done in the absence of this compound in replicates of eight. Reactions are monitored in a fluorescence plate reader. After a 30-minute lag time, the fluorescence is measured over a 30-minute time frame using an excitation wavelength of 355 nm and a detection wavelength of 460 nm. The increase in fluorescence with time is used as the measure of the reaction rate. Inhibition constants Ki(app) are obtained using the program BatchKi.
|
|
| In vivo |
Abexinostat (PCI-24781) was administered at 200 mg/kg once daily every other day (q.o.d.), which significantly inhibited the growth of HCT116 and DLD-1 xenografts in mice by 69% and 59%, respectively. In the HCT116 model, administration of this compound at 20 mg/kg, 40 mg/kg, 80 mg/kg, or 160 mg/kg once daily for 4 consecutive days followed by 3 days without treatment each week (q.d. × 4 per week) caused inhibition of tumor growth by 48%, 57%, 82.2%, or 80.0%, respectively.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05698524 | Recruiting | Recurrent High Grade Glioma|Anaplastic Astrocytoma|Anaplastic Oligodendroglioma|Glioblastoma|Gliosarcoma |
University of Nebraska|Xynomic Pharmaceuticals Inc. |
June 26 2023 | Phase 1 |
| NCT03934567 | Recruiting | Lymphoma Follicular |
Xynomic Pharmaceuticals Inc. |
April 22 2020 | Phase 2 |
| NCT03939182 | Active not recruiting | Diffuse Large B-cell Lymphoma|Mantle Cell Lymphoma |
Memorial Sloan Kettering Cancer Center|Janssen Scientific Affairs LLC|Xynomic Pharmaceuticals Inc. |
May 29 2019 | Phase 1 |
| NCT03600441 | Active not recruiting | Follicular Lymphoma |
Xynomic Pharmaceuticals Inc. |
August 27 2018 | Phase 2 |
| NCT01543763 | Active not recruiting | Metastatic Solid Tumors |
Pamela Munster|Pharmacyclics LLC.|Novartis|Xynomic Pharmaceuticals Inc.|GlaxoSmithKline Research and Education Foundation for Cardiovascular Disease|University of California San Francisco |
June 25 2012 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































