research use only
CUDC-101 HDAC inhibitor
Cat.No.S1194
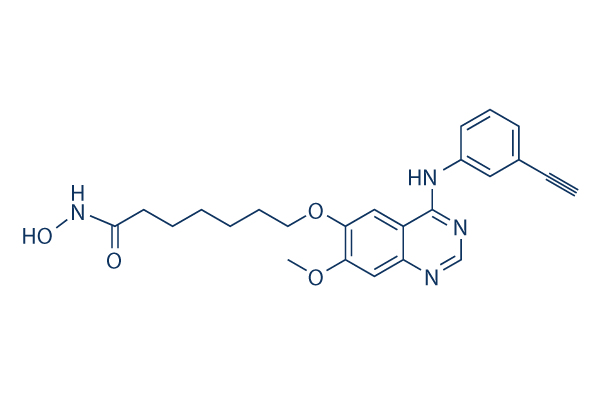
Chemical Structure
Molecular Weight: 434.49
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human SK-BR-3 cells | Proliferation assay | Antiproliferative activity against human SK-BR-3 cells after hrs by ATP content assay, IC50=0.04 μM | ||||
| MDA-MB-231 cells | Proliferation assay | Antiproliferative activity against human MDA-MB-231 cells after hrs by ATP content assay, IC50=0.1 μM | ||||
| human HepG2 cells | Proliferation assay | Antiproliferative activity against human HepG2 cells after hrs by ATP content assay, IC50=0.13 μM | ||||
| human SKHEP1 cells | Proliferation assay | Antiproliferative activity against human SKHEP1 cells after hrs by ATP content assay, IC50=0.22 μM | ||||
| human Hep3B2 cells | Proliferation assay | Antiproliferative activity against human Hep3B2 cells after hrs by ATP content assay, IC50=0.23 μM | ||||
| human BxPC3 cells | Proliferation assay | Antiproliferative activity against human BxPC3 cells after hrs by ATP content assay, IC50=0.27 μM | ||||
| human NCI-H358 cells | Proliferation assay | Antiproliferative activity against human NCI-H358 cells after hrs by ATP content assay, IC50=0.4 μM | ||||
| human MCF7 cells | Proliferation assay | Antiproliferative activity against human MCF7 cells after hrs by ATP content assay, IC50=0.55 μM | ||||
| human HCC827 cells | Proliferation assay | Antiproliferative activity against human HCC827 cells after hrs by ATP content assay, IC50=0.6 μM | ||||
| human H460 cells | Proliferation assay | Antiproliferative activity against human H460 cells after hrs by ATP content assay, IC50=0.7 μM | ||||
| human Capan1 cells | Proliferation assay | Antiproliferative activity against human Capan1 cells after hrs by ATP content assay, IC50=0.8 μM | ||||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 43 mg/mL
(98.96 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 434.49 | Formula | C24H26N4O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1012054-59-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | COC1=C(C=C2C(=C1)N=CN=C2NC3=CC=CC(=C3)C#C)OCCCCCCC(=O)NO | ||
Mechanism of Action
| Targets/IC50/Ki |
EGFR
(Cell-free assay) 2.4 nM
HDAC
(Cell-free assay) 4.4 nM
HDAC1
(Cell-free assay) 4.5 nM
HDAC6
(Cell-free assay) 5.1 nM
HDAC3
(Cell-free assay) 9.1 nM
HDAC5
(Cell-free assay) 11.4 nM
HDAC2
(Cell-free assay) 12.6 nM
HDAC4
(Cell-free assay) 13.2 nM
HER2
(Cell-free assay) 15.7 nM
HDAC10
(Cell-free assay) 26.1 nM
HDAC9
(Cell-free assay) 67.2 nM
HDAC8
(Cell-free assay) 79.8 nM
HDAC7
(Cell-free assay) 373 nM
|
|---|---|
| In vitro |
Specific for class I and class II HDACs, CUDC-101 does not inhibit class III Sir-type HDACs. This compound displays weak activity against other protein kinases including KDR/VEGFR2, Lyn, Lck, Abl-1, FGFR-2, Flt-3, and Ret with IC50 of 0.85 μM, 0.84 μM, 5.91 μM, 2.89 μM, 3.43 μM, 1.5 μM, abd 3.2 μM, respectively. It displays broad antiproliferative activity in many human cancer cell types with IC50 of 0.04-0.80 μM, exhibiting a higher potency and combinations of vorinostat in most cases. This chemical potently inhibits cancer cell lines. It inhibits the resistant EGFR mutant T790M although its effects are incomplete with an Amax of ~60% of peak enzyme activity after inhibition. This compound treatment increases the acetylation of histone H3 and H4, as well as the acetylation of non-histone substrates of HDAC such as p53 and α-tubulin, in a dose-dependant manner in various cancer cell lines. It also suppresses HER3 expression, Met amplification, and AKT reactivation in tumor cells. |
| Kinase Assay |
HDAC, EGFR and HER2 inhibition assays
|
|
The activities of Class I and II HDACs are assessed using the Biomol Color de Lys system. Briefly, HeLa cell nuclear extracts are used as a source of HDACs. Different concentrations of CUDC-101 are added to HeLa cell nuclear extracts in the presence of a colorimetric artificial substrate. Developer is added at the end of the assay and enzyme activity is measured in the Wallac Victor II 1420 microplate reader at 405 nM. EGFR and HER2 kinase activity are measured using HTScan EGF receptor and HER2 kinase assay kits. Briefly, the GST-EGFR fusion protein is incubated with synthetic biotinylated peptide substrate and varying concentrations of this compound in the presence of 400 mM ATP. Phosphorylated substrate is captured with strapavidin-coated 96-well plates. The level of phosphorylation is monitored by antiphospho-tyrosine- and europium-labeled secondary antibodies. The enhancement solution is added at the end of the assay and enzyme activity is measured in the Wallac Victor II 1420 microplate reader at 615 nM.
|
|
| In vivo |
Administration of CUDC-101 at 120 mg/kg/day induces tumor regression in the Hep-G2 liver cancer model, which is more efficacious at its maximum tolerated dose (25 mg/kg/day) and vorinostat at an equimolar concentration dose (72 mg/kg/day). This compound inhibits the growth of -sensitive H358 NSCLC xenografts in a dose-dependent manner. It also shows potent inhibition of tumor growth in the -resistant A549 NSCLC xenograft model. This chemical produces significant tumor regression in the -resistant, HER2-negative, EGFR-overexpressing MDA-MB-468 breast cancer model and the EGFR-overexpressing CAL-27 head and neck squamous cell carcinoma (HNSCC) model. Additionally, it inhibits tumor growth in the K-ras mutant HCT116 colorectal and EGFR/HER2 (neu)-expressing HPAC pancreatic cancer models. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01702285 | Terminated | Cancer |
Curis Inc. |
September 2012 | Phase 1 |
| NCT01384799 | Completed | Head and Neck Cancer |
Curis Inc. |
November 2011 | Phase 1 |
| NCT00728793 | Completed | Tumors |
Curis Inc. |
August 2008 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































