- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Tubastatin A HCl
Tubastatin A HCl is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective (1000-fold more) against all other isozymes except HDAC8 (57-fold more).
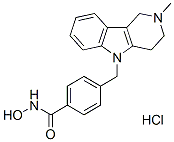
Tubastatin A HCl Chemical Structure
CAS: 1310693-92-5
Selleck's Tubastatin A HCl has been cited by 47 publications
Purity & Quality Control
Batch:
Purity:
99.30%
99.30
Tubastatin A HCl Related Products
| Related Targets | HDAC1 HDAC2 HDAC3 HDAC4 HDAC5 HDAC6 HDAC7 HDAC8 HDAC9 HDAC10 HDAC11 HD1 HD2 | Click to Expand |
|---|---|---|
| Related Compound Libraries | Kinase Inhibitor Library FDA-approved Drug Library Natural Product Library Bioactive Compound Library-I Bioactive Compound Library-Ⅱ | Click to Expand |
Signaling Pathway
Choose Selective HDAC Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| neuron cultures | Kinase assay | 2.5 μM | DMSO | induces α-tubulin hyperacetylation | 20614936 | |
| neuron cultures | Function assay | ~10 μM | DMSO | protects against glutathione depletion-induced oxidative stress | 20614936 | |
| 134/04 | Function assay | 7.5 µM | impairs myotube formation | 22174839 | ||
| C2C12 | Function assay | 7.5 µM | impairs myotube formation | 22174839 | ||
| HaCaT keratinocytes | Function assay | 10 μM | blocks arsenite from inducing Nrf2 protein translation | 22367689 | ||
| JURL-MK1 | Function assay | 10 μM | enhances cell adhesivity to fibronectin | 23022583 | ||
| CML-T1 | Function assay | 10 μM | enhances cell adhesivity to fibronectin | 23022583 | ||
| K562 | Function assay | 10 μM | enhances cell adhesivity to fibronectin | 23022583 | ||
| HL-60 | Function assay | 10 μM | enhances cell adhesivity to fibronectin | 23022583 | ||
| KMCH | Growth inhibitory assay | ~10 μM | decreases proliferation and anchorage-independent growth | 23370327 | ||
| THP-1 | Function assay | ~10 μM | inhibits TNF-α and IL-6 secretion | 23541634 | ||
| RAW 264.7 | Function assay | ~10 μM | attenuates NO production | 23541634 | ||
| HT3 | Function assay | ~5 μM | DMSO | induces the differential α-tubulin acetylation | 23698468 | |
| SiHa | Function assay | ~5 μM | DMSO | induces the differential α-tubulin acetylation | 23698468 | |
| CaSki | Function assay | ~5 μM | DMSO | induces the differential α-tubulin acetylation | 23698468 | |
| SiHa | Function assay | ~5 μM | DMSO | inhibits Thapsigargin- or EGF-induced SOCE activation | 23698468 | |
| CaSki | Function assay | ~5 μM | DMSO | inhibits Thapsigargin- or EGF-induced SOCE activation | 23698468 | |
| MCF-7 | Growth inhibitory assay | 30 μM | DMSO | IC50=15 μM | 23798680 | |
| MCF-7 | Function assay | 30 μM | DMSO | increases the microtubule acetylation level. | 23798680 | |
| MCF-7 | Function assay | 30 μM | DMSO | stabilizes microtubules against cold-induced depolymerization | 23798680 | |
| MCF-7 | Function assay | 15 μM | DMSO | stabilizes microtubules against nocodazole-induced disassembly | 23798680 | |
| MCF-7 | Function assay | 30 μM | DMSO | alteres the assembly dynamics of interphase microtubules | 23798680 | |
| MCF-7 | Function assay | 30 μM | DMSO | increases the binding of HDAC6 with interphase microtubules | 23798680 | |
| PC12 | Function assay | ~3 μM | DMSO | up-regulates anti-oxidative gene expression related to transcription factor XBP1s | 24909686 | |
| PC12 | Growth inhibitory assay | ~3 μM | DMSO | reverse H2O2-induced growth inhibition | 24909686 | |
| HEK293T | Function assay | ~3 μM | DMSO | up-regulated XBP1s protein level | 24909686 | |
| HEK293T | Function assay | ~3 μM | DMSO | delays XBP1s protein degradation via acetylation-mediated proteasomal degradation | 24909686 | |
| Huh7 | Function assay | ~5 μM | DMSO | suppresses proliferation of hepatitis C virus replicon with EC50 = 0.3 μM | 25108326 | |
| SKMEL21 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| SKMEL103 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| SKMEL28 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM164 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM1361a | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM1366 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM793 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM35 | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM983a | Growth inhibitory assay | ~500 nM | DMSO | inhibits cell proliferation | 25957812 | |
| WM793 | Function assay | ~6 μM | DMSO | induce G1 arrest | 25957812 | |
| WM164 | Function assay | ~6 μM | DMSO | induce G1 arrest | 25957812 | |
| WM983a | Function assay | ~6 μM | DMSO | induce G1 arrest | 25957812 | |
| WM164 | Function assay | ~3 μM | DMSO | augments expression of MHC class I and melanoma associated antigens | 25957812 | |
| WM983a | Function assay | ~3 μM | DMSO | augments expression of MHC class I and melanoma associated antigens | 25957812 | |
| IPC298 | Function assay | ~3 μM | DMSO | augments expression of MHC class I and melanoma associated antigens | 25957812 | |
| SKMEL30 | Function assay | ~3 μM | DMSO | augments expression of MHC class I and melanoma associated antigens | 25957812 | |
| TCa83 | Function assay | induces PTEN expression and membrane translocation | 26279303 | |||
| 293T | Function assay | ~2 μg/ml | induces PTEN expression and membrane translocation | 26279303 | ||
| SACC-83 | Function assay | ~2 μg/ml | induces PTEN expression and membrane translocation | 26279303 | ||
| 293T | Function assay | ~2 μg/ml | induces PTEN acetylation at K163 | 26279303 | ||
| U-87 MG | Function assay | ~2 μg/ml | inhibits the migration and invasion | 26279303 | ||
| U-87 MG | Function assay | ~10 μM | inhibits AKT phosphorylation | 26279303 | ||
| U-87 MG | Growth inhibitory assay | ~10 μM | inhibits cell growth | 26279303 | ||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Tubastatin A HCl is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective (1000-fold more) against all other isozymes except HDAC8 (57-fold more). | ||||
|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | Tubastatin A is substantially selective for all 11 HDAC isoforms and maintains over 1000-fold selectivity against all isoforms excluding HDAC8, where it has approximately 57-fold selectivity. In homocysteic acid (HCA) induced neurodegeneration assays, Tubastatin A displays dose-dependent protection against HCA-induced neuronal cell death starting at 5 μM with near complete protection at 10 μM. [1] At 100 ng/mL Tubastatin A increases Foxp3+ T-regulatory cells (Tregs) suppression of T cell proliferation in vitro. [2] Tubastatin A treatment in C2C12 cells would lead to myotube formation impairment when alpha-tubulin is hyperacetylated early in the myogenic process; however, myotube elongation occurs when alpha-tubulin is hyeperacetylated in myotubes. [3] A recent study indicates that Tubastatin A treatment increases cell elasticity as revealed by atomic force microscopy (AFM) tests without exerting drastic changes to the actin microfilament or microtubule networks in mouse ovarian cancer cell lines, MOSE-E and MOSE-L. [4] | |||
|---|---|---|---|---|
| Kinase Assay | Enzyme Inhibition Assays | |||
| Enzyme inhibition assays are performed by the Reaction Biology Corporation, Malvern, PA, using the Reaction Biology HDAC Spectrum platform. (www.reactionbiology.com) The HDAC1, 2, 4, 5, 6, 7, 8, 9, 10, and 11 assays use isolated recombinant human protein; HDAC3/NcoR2 complex is used for the HDAC3 assay. Substrate for HDAC1, 2, 3, 6, 10, and 11 assays is a fluorogenic peptide from p53 residues 379-382 (RHKKAc); substrate for HDAC8 is fluorogenic diacyl peptide based on residues 379-382 of p53 (RHKAcKAc). Acetyl-Lys (trifluoroacetyl)-AMC substrate is used for HDAC4, 5, 7, and 9 assays. Tubastatin A is dissolved in DMSO and tested in 10-dose IC50 mode with 3-fold serial dilution starting at 30 μM. Control Compound Trichostatin A (TSA) is tested in a 10-dose IC50 with 3-fold serial dilution starting at 5 μM. IC50 values are extracted by curve-fitting the dose/response slopes. | ||||
| Cell Research | Cell lines | Primary cortical neuron of fetal Sprague-Dawley rats (embryonic day 17) | ||
| Concentrations | 0-10 μM | |||
| Incubation Time | 24 hours | |||
| Method | Primary cortical neuron cultures are obtained from the cerebral cortex of fetal Sprague-Dawley rats (embryonic day 17) as described previously. All experiments are initiated 24 hours after plating. Under these conditions, the cells are not susceptible to glutamate-mediated excitotoxicity. For cytotoxicity studies, cells are rinsed with warm PBS and then placed in minimum essential medium (Invitrogen) containing 5.5 g/L glucose, 10% fetal calf serum, 2 mM L-glutamine, and 100 μM cystine. Oxidative stress is induced by the addition of the glutamate analogue homocysteate (HCA; 5 mM) to the media. HCA is diluted from 100-fold concentrated solutions that are adjusted to pH 7.5. In combination with HCA, neurons are treated with Tubastatin A at the indicated concentrations. Viability is assessed after 24 hours by MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method. | |||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | EGFR / p-AKT / AKT / p-ERK / ERK |
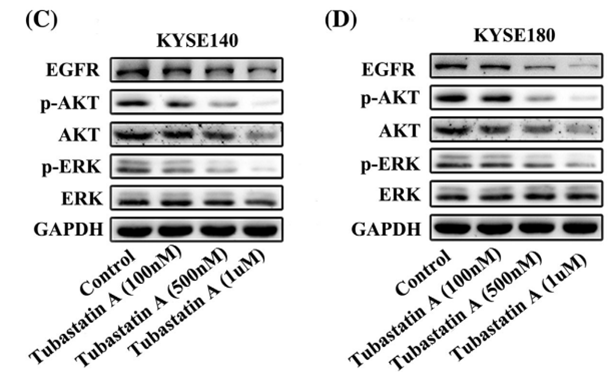
|
29665050 | |
| Immunofluorescence | α-tubulin / Acetylated tubulin HDAC6 |
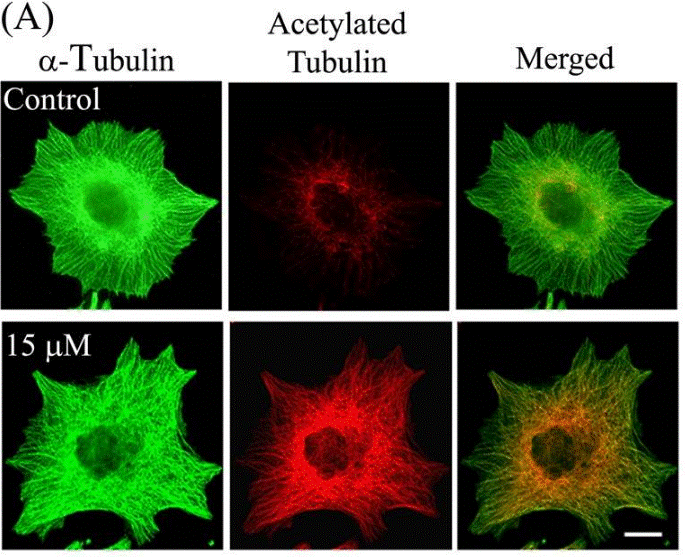
|
23798680 | |
| In Vivo | ||
| In vivo | Daily treatment of Tubastatin A at 0.5mg/kg inhibits HDAC6 to promote Tregs suppressive activity in mouse models of inflammation and autoimmunity, including multiple forms of experimental colitis and fully major histocompatibility complex (MHC)-incompatible cardiac allograft rejection. [2] | |
|---|---|---|
| Animal Research | Animal Models | Na?ve CD45RBhi CD4+ CD25- cells (1 × 106) from WT or HDAC6-/- mice Are injected i.p. into B6/Rag1-/-mice. |
| Dosages | 0.5 mg/kg | |
| Administration | Tubastatin A is injected i.p. daily. | |
Chemical Information & Solubility
| Molecular Weight | 371.86 | Formula | C20H21N3O2.HCl |
| CAS No. | 1310693-92-5 | SDF | Download Tubastatin A HCl SDF |
| Smiles | CN1CCC2=C(C1)C3=CC=CC=C3N2CC4=CC=C(C=C4)C(=O)NO.Cl | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 100 mg/mL ( (268.91 mM); Warmed with 50°C water bath; Ultrasonicated; Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy Tubastatin A HCl | Tubastatin A HCl supplier | purchase Tubastatin A HCl | Tubastatin A HCl cost | Tubastatin A HCl manufacturer | order Tubastatin A HCl | Tubastatin A HCl distributor