- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
research use only
Tubastatin A HDAC inhibitor
Tububastatin A is a potent and selective HDAC6 inhibitor with IC50 of 15 nM in a cell-free assay. It is selective against all the other isozymes (1000-fold) except HDAC8 (57-fold). This compound promotes autophagy and increases apoptosis.
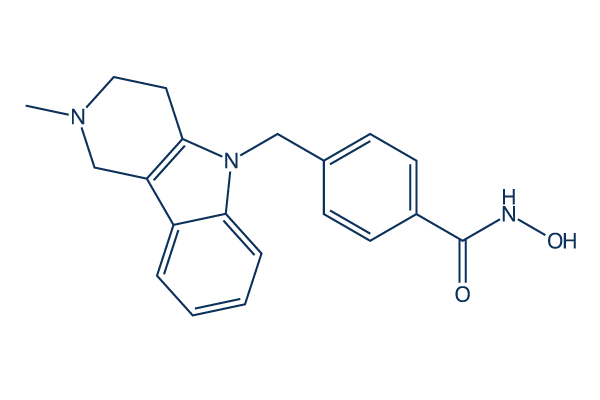
Chemical Structure
Molecular Weight: 335.4
Purity & Quality Control
Batch:
Purity:
99.63%
99.63
Related Products
| Related Targets | HDAC1 HDAC2 HDAC3 HDAC4 HDAC5 HDAC6 HDAC7 HDAC8 HDAC9 HDAC10 HDAC11 HD1 HD2 | Click to Expand |
|---|---|---|
| Related Products | Entinostat (MS-275) TSA (Trichostatin A) Mocetinostat (MGCD0103) RGFP966 Ricolinostat (ACY-1215) Quisinostat (JNJ-26481585) 2HCl MC1568 Tubastatin A HCl PCI-34051 Tacedinaline (CI994) LMK-235 Fimepinostat (CUDC-907) Tubacin Givinostat hydrochloride monohydrate TMP269 AR-42 Sodium butyrate Pracinostat (SB939) Santacruzamate A (CAY10683) Abexinostat (PCI-24781) | Click to Expand |
| Related Compound Libraries | Kinase Inhibitor Library FDA-approved Drug Library Natural Product Library Bioactive Compound Library-I Highly Selective Inhibitor Library | Click to Expand |
Signaling Pathway
Cell Culture and Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Sf9 | Function assay | 2 hrs | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 (1 to 1215 residues) expressed in sf9 cells preincubated with enzyme followed by fluorogenic Arg-His-Lys-Lys(Ac)-AMC substrate addition measured after 2 hrs by fluorescence assay, IC50 = 0.0035 μM. | 27541357 | ||
| Sf9 | Function assay | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 (1 to 1215 residues) expressed in sf9 cells using RHK-K(Ac)-AMC as substrate by fluorescence assay, IC50 = 0.011 μM. | 27541357 | |||
| Sf9 | Function assay | 30 mins | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 30 mins by fluorescence assay, IC50 = 0.013 μM. | 29549837 | ||
| Sf9 | Function assay | 90 mins | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 expressed in Sf9 cells using RHKK(Ac) as substrate after 90 mins by fluorimetric method, IC50 = 0.0137 μM. | 28038324 | ||
| Sf9 | Function assay | 2 hrs | Inhibition of human recombinant HDAC6 expressed in Sf9 cells incubated for 2 hrs using RHKK-Ac fluorogenic substrate, IC50 = 0.015 μM. | 23009203 | ||
| Sf9 | Function assay | Inhibition of human recombinant HDAC6 expressed in baculovirus/sf9 cells using RHKKAc as substrate, IC50 = 0.015 μM. | 23905680 | |||
| Sf9 | Function assay | 90 mins | Inhibition of human recombinant N-terminal GST-tagged full length HDAC6 expressed in insect SF9 cells using fluorogenic ZMAL as substrate after 90 mins by fluorescence-based assay, IC50 = 0.0304 μM. | 30092367 | ||
| HeLaS3 | Function assay | 15 mins | Inhibition of HDAC6 in human HeLaS3 cells preincubated for 15 mins followed by HDAC-Glo substrate addition measured after 30 to 45 mins by ELISA, IC50 = 0.031 μM. | 28337317 | ||
| insect cells | Function assay | 4 hrs | Inhibition of human recombinant HDAC6 expressed in baculovirus infected insect cells using BATCP as substrate after 4 hrs by UHPLC-ESI-MS/MS analysis, IC50 = 0.0349 μM. | 27650925 | ||
| SHSY5Y | Function assay | 8 hrs | Inhibition of HDAC6 in human SHSY5Y cells using BATCP as substrate after 8 hrs by UHPLC-ESI-MS/MS analysis, IC50 = 0.0943 μM. | 27650925 | ||
| SHSY5Y | Function assay | 8 hrs | Inhibition of HDAC in human SHSY5Y cells using MAL as substrate after 8 hrs by UHPLC-ESI-MS/MS analysis, IC50 = 0.1221 μM. | 27650925 | ||
| Sf9 | Function assay | 4 hrs | Inhibition of full length human recombinant C-terminal FLAG/His-tagged HDAC1 expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 4 hrs fluorescence assay, IC50 = 0.718 μM. | 29549837 | ||
| Sf9 | Function assay | Inhibition of human recombinant HDAC8 expressed in baculovirus/sf9 cells using RHKAcKAc as substrate, IC50 = 0.854 μM. | 23905680 | |||
| Sf9 | Function assay | 1 hr | Inhibition of full length human recombinant C-terminal FLAG/His-tagged HDAC1 expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 1 hr fluorescence assay, IC50 = 0.967 μM. | 29549837 | ||
| SHSY5Y | Function assay | 8 hrs | Inhibition of HDAC1 in human SHSY5Y cells using MOCPAC as substrate after 8 hrs by UHPLC-ESI-MS/MS analysis, IC50 = 1.1097 μM. | 27650925 | ||
| Sf9 | Function assay | 30 mins | Inhibition of full length human recombinant C-terminal FLAG/His-tagged HDAC1 expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 30 mins by fluorescence assay, IC50 = 1.54 μM. | 29549837 | ||
| Sf9 | Function assay | 90 mins | Inhibition of human recombinant C-terminal His/FLAG-tagged full length HDAC1 expressed in insect SF9 cells using fluorogenic ZMAL as substrate after 90 mins by fluorescence-based assay, IC50 = 1.91 μM. | 30092367 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by MTT assay, IC50 = 2 μM. | 27541357 | ||
| HCT116 | Antiproliferative assay | Antiproliferative activity against human HCT116 cells, IC50 = 2 μM. | 29945795 | |||
| Sf9 | Function assay | 30 mins | Inhibition of full length human recombinant C-terminal His-tagged HDAC3/N-terminal GST-tagged NCOR2 (95 to 489 residues) expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 30 mins by fluorescence assay, IC50 = 2.26 μM. | 29549837 | ||
| HeLa | Function assay | 6 hrs | Inhibition of HDAC6 in human HeLa cells assessed as reduction in K40 hyperacetylation of alpha-tubulin incubated for 6 hrs by immunofluorescence assay, IC50 = 2.5 μM. | 25454270 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | ||
| HeLaS3 | Function assay | 15 mins | Inhibition of HDAC1 in human HeLaS3 cells preincubated for 15 mins followed by HDAC-Glo substrate addition measured after 30 to 45 mins by ELISA, IC50 = 2.7 μM. | 28337317 | ||
| HeLaS3 | Function assay | 15 mins | Inhibition of HDAC3 in human HeLaS3 cells preincubated for 15 mins followed by HDAC-Glo substrate addition measured after 30 to 45 mins by ELISA, IC50 = 2.9 μM. | 28337317 | ||
| Jurkat | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Jurkat cells assessed as growth inhibition after 72 hrs by MTS assay, IC50 = 3.38 μM. | 24304348 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 3.7 μM. | 27541357 | ||
| MCF7 | Antiproliferative assay | Antiproliferative activity against human MCF7 cells, IC50 = 3.7 μM. | 29945795 | |||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | ||
| HeLaS3 | Function assay | 15 mins | Inhibition of HDAC2 in human HeLaS3 cells preincubated for 15 mins followed by HDAC-Glo substrate addition measured after 30 to 45 mins by ELISA, IC50 = 3.9 μM. | 28337317 | ||
| CAL27 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human CAL27 cells measured after 72 hrs by MTT assay, IC50 = 4.6 μM. | 28581289 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells after 72 hrs by MTT assay, IC50 = 8.6 μM. | 27541357 | ||
| PC3 | Antiproliferative assay | Antiproliferative activity against human PC3 cells, IC50 = 8.6 μM. | 29945795 | |||
| Sf9 | Function assay | 30 mins | Inhibition of full length human recombinant C-terminal His-tagged HDAC2 expressed in baculovirus infected sf9 cells using fluorogenic HDAC substrate 3 after 30 mins by fluorescence assay, IC50 = 9.97 μM. | 29549837 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 10.4 μM. | 27541357 | ||
| MDA-MB-231 | Antiproliferative assay | Antiproliferative activity against human MDA-MB-231 cells, IC50 = 10.4 μM. | 29945795 | |||
| Cal27CisR | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Cal27CisR cells measured after 72 hrs by MTT assay, IC50 = 10.8 μM. | 28581289 | ||
| LNCAP | Cytotoxicity assay | 72 hrs | Cytotoxicity against androgen-dependent human LNCAP cells assessed as growth inhibition after 72 hrs by MTS assay, IC50 = 10.88 μM. | 24304348 | ||
| Cal27CisR | Function assay | 18 hrs | Inhibition of HDAC in human Cal27CisR cells using Boc-Lys(epsilon-Ac)-AMC as substrate preincubated for 18 hrs followed by substrate addition measured after 3 hrs by fluorescence assay, IC50 = 12.1 μM. | 28581289 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTS assay, IC50 = 14.81 μM. | 25899338 | ||
| THLE2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human THLE2 cells after 72 hrs by vialight cell proliferation assay, LC50 = 15.1 μM. | 29549837 | ||
| CAL27 | Function assay | 18 hrs | Inhibition of HDAC in human CAL27 cells using Boc-Lys(epsilon-Ac)-AMC as substrate preincubated for 18 hrs followed by substrate addition measured after 3 hrs by fluorescence assay, IC50 = 16.1 μM. | 28581289 | ||
| Sf9 | Function assay | 2 hrs | Inhibition of human recombinant HDAC1 expressed in Sf9 cells incubated for 2 hrs using RHKK-Ac fluorogenic substrate, IC50 = 16.4 μM. | 23009203 | ||
| Sf9 | Function assay | Inhibition of human recombinant HDAC1 expressed in baculovirus/sf9 cells using RHKKAc as substrate, IC50 = 16.4 μM. | 23905680 | |||
| B16 | Growth inhibition assay | 48 hrs | Growth inhibition of mouse B16 cells incubated for 48 hrs by MTT assay, GI50 = 40.5 μM. | 23009203 | ||
| KB | Function assay | 14 uM | 24 hrs | Inhibition of HDAC1 in human KB cells assessed as increase in histone H4 acetylation at 14 uM after 24 hrs by Western blotting analysis | 25899338 | |
| KMS-12-BM | Function assay | 15 uM | up to 48 hrs | Inhibition of HDAC6 in human KMS-12-BM cells assessed as increase in acetylated tubulin level at 15 uM up to 48 hrs by immunoblot method | 27541357 | |
| MOLM14 | Function assay | 15 uM | up to 48 hrs | Inhibition of HDAC6 in human MOLM14 cells assessed as increase in acetylated tubulin level at 15 uM up to 48 hrs by immunoblot method | 27541357 | |
| U937 | Function assay | 2 uM | 18 hrs | Inhibition of HDAC6 in human U937 cells assessed as increase in alpha-tubulin acetylation at Lys-40 residue at 2 uM after 18 hrs by Western blot method | 28337317 | |
| LNCAP | Function assay | 24 hrs | Inhibition of HDAC6 in human LNCAP cells assessed as inhibition of DHT-induced alpha-tubulin deacetylation by measuring increase in alpha-tubulin acetylation measured after 24 hrs relative to control | 27717544 | ||
| human | Function assay | 24 hrs | Antagonist activity at AR in human LNCAP cells assessed as suppression of DHT-induced AR protein level measured after 24 hrs relative to control | 27717544 | ||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| HEL | Cell cycle assay | 1 to 5 uM | 48 hrs | Cell cycle arrest in human HEL cells assessed as accumulation at G1 phase at 1 to 5 uM after 48 hrs propidium iodide staining based flow cytometry | 29940115 | |
| HeLa | Function assay | 2 uM | 12 hrs | Inhibition of HDAC6 in human HeLa cells assessed as increase in acetyl-tubulin level at 2 uM after 12 hrs by Western blot analysis | 29533873 | |
| HEK293 | Function assay | 10 uM | 24 hrs | Inhibition of HDAC1 in HEK293 cells assessed as increase in histone H3 acetylation at 10 uM after 24 hrs by Western blot method | 28523102 | |
| MV4-11 | Function assay | 200 nM | 24 hrs | Inhibition of HDAC6 in human MV4-11 cells assessed as accumulation of acetylated alpha-tubulin at 200 nM after 24 hrs by Western blot analysis | 29738953 | |
| HCT116 | Cell cycle assay | 5 uM | 48 hrs | Cell cycle arrest in human HCT116 cells assessed as accumulation at sub-G1 phase at 5 uM after 48 hrs by propidium iodide staining-based flow cytometric method | 28038324 | |
| PC12 | Neuroprotective assay | 10 uM | 24 hrs | Neuroprotective activity against H2O2-induced toxicity in rat PC12 cells assessed as cell viability at 10 uM pretreated for 24 hrs followed by H2O2 challenge and measured after 12 hrs by MTT assay relative to control | 30385227 | |
| PC12 | Neuroprotective assay | 5 uM | 24 hrs | Neuroprotective activity against 6-OHDA-induced toxicity in rat PC12 cells assessed as increase in cell viability at 5 uM pretreated for 24 hrs followed by 6-OHDA challenge and measured after 12 hrs by MTT assay | 30385227 | |
| PC12 | Neuroprotective assay | 10 uM | 24 hrs | Neuroprotective activity against 6-OHDA-induced toxicity in rat PC12 cells assessed as increase in cell viability at 10 uM pretreated for 24 hrs followed by 6-OHDA challenge and measured after 12 hrs by MTT assay | 30385227 | |
| PC12 | Cytoprotective assay | 24 hrs | Cytoprotective activity against H2O2-induced damage in rat PC12 cells assessed as decrease in ROS accumulation preincubated for 24 hrs followed by H2O2 challenge measured after 12 hrs by DCFH-DA dye-based fluorescence analysis | 30385227 | ||
| PC12 | Cytoprotective assay | 24 hrs | Cytoprotective activity against H2O2-induced damage in rat PC12 cells assessed as decrease in ROS accumulation preincubated for 24 hrs followed by H2O2 challenge measured after 12 hrs by DCFH-DA dye-based inverted fluorescence microscopic analysis | 30385227 | ||
| PC12 | Neuroprotective assay | 5 to 10 uM | 24 hrs | Neuroprotective activity against 6-OHDA-induced toxicity in rat PC12 cells assessed as increase in cell viability at 5 to 10 uM pretreated for 24 hrs followed by 6-OHDA challenge and measured after 12 hrs coincubated with ebselen by MTT assay | 30385227 | |
| PC12 | Antioxidant assay | 5 uM | 24 hrs | Antioxidant activity against H2O2-induced oxidative stress in rat PC12 cells assessed as decrease in ROS accumulation at 5 uM preincubated for 24 hrs followed by H2O2 challenge and measured after 12 hrs by DCFH-DA dye-based fluorescence analysis | 30385227 | |
| PC12 | Antioxidant assay | 5 uM | 24 hrs | Antioxidant activity against H2O2-induced oxidative stress in rat PC12 cells assessed as decrease in ROS accumulation at 5 uM preincubated for 24 hrs followed by H2O2 challenge and measured after 12 hrs coincubated with ebselen by DCFH-DA dye-based fluore | 30385227 | |
| Click to View More Cell Line Experimental Data | ||||||
Mechanism of Action
| Targets |
|
|---|
In vitro |
||||
| In vitro | Tubastatin A is selective at all isozymes except HDAC8 and maintains over 1000-fold selectivity against all isoforms excluding HDAC8, where it has approximately 57-fold selectivity. This compound preferentially induces α-tubulin hyperacetylation at 2.5 μM. Slight induction of histone hyperacetylation is seen for this chemical at 10 μM. It displays dose-dependent protection against homocysteic acid-induced neuronal cell death starting at 5 μM with near complete protection at 10 μM. [1] This compound (10 μM) induces an increase in acetylated-α-tubulin levels and the restoration of primary cilia expression in the cholangiocarcinoma cell lines (18-fold); and the restoration of primary cilia correlated with downregulated Hedgehog (Hh) and MAPK signaling pathways, as well as decreased cell proliferation rates (in average by 50%) and invasion (by 40%). [2] It shows significant inhibition of TNF-α and IL-6 in LPS stimulated human THP-1 macrophages with an IC50 of 272 nM and 712 nM. This inhibitor inhibits nitric oxide (NO) secretion in murine Raw 264.7 macrophages dose depenndently with an IC50 of 4.2 μM. [3] | |||
|---|---|---|---|---|
| Kinase Assay | HDAC enzymatic assays | |||
| Tubastatin A is dissolved and diluted in assay buffer (50 mM HEPES, pH 7.4, 100 mM KCl, 0.001% Tween-20, 0.05% BSA, and 20 μM tris(2-carboxyethyl)phosphine) to 6-fold of the final concentration. HDAC enzymes are diluted to 1.5-fold of the final concentration in assay buffer and pre-incubated with this compound for 10 minutes before the addition of the substrate. The amount of FTS (HDAC1, HDAC2, HDAC3, and HDAC6) or MAZ-1675 (HDAC4, HDAC5, HDAC7, HDAC8, and HDAC9) used for each enzyme is equal to the Michaelis constant (Km), as determined by a titration curve. FTS or MAZ-1675 is diluted in assay buffer to 6-fold the final concentration with 0.3 μM sequencing grade trypsin. The substrate/trypsin mix is added to the enzyme/compound mix and the plate is shaken for 60 seconds and then placed into a SpectraMax M5 microtiter plate reader. The enzymatic reaction is monitored for release of 7-amino-4-methoxy-coumarin over 30 minutes, after deacetylation of the lysine side chain in the peptide substrate, and the linear rate of the reaction is calculated. | ||||
| Cell Research | Cell lines | Human cholangiocarcinoma cell lines HuCCT-1 | ||
| Concentrations | ~10 μM | |||
| Incubation Time | 21 days | |||
| Method | Anchorage-independent growth is assessed by growing cells in soft agar. About 25,000 cells suspended in 0.4% agar in culture media are layered over a 1% agar layer in a 6-well plate. Media are added twice a week and pictures are taken after 21 days of incubation. The number and size of colonies are analyzed using the Gel-Pro software. | |||
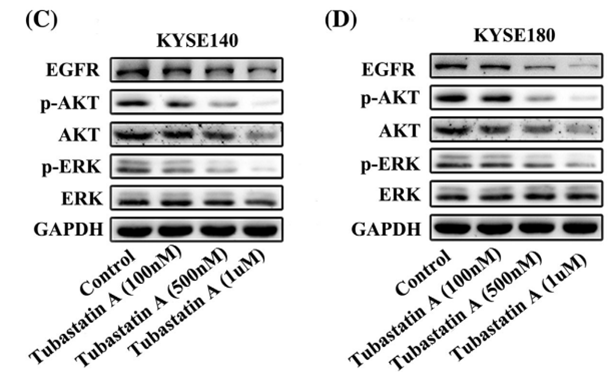
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | EGFR / p-AKT / AKT / p-ERK / ERK |
|
29665050 | |
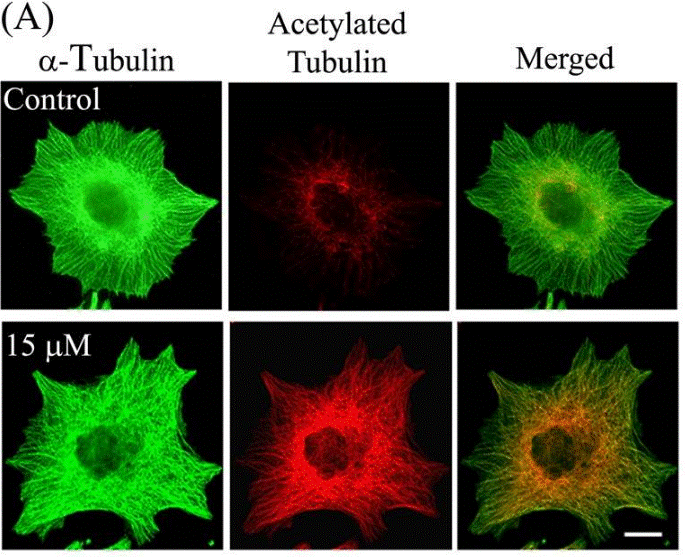
| Immunofluorescence | α-tubulin / Acetylated tubulin HDAC6 |
|
23798680 | |
In Vivo |
||
| In vivo | Tubastatin A reduces the growth of cholangiocarcinoma in vivo. This compound (10 mg/kg) induces a 6-fold lower mean tumor weights in syngeneic rat orthotopic model of cholangiocarcinoma, and reduction of the ratios of tumor weight to liver weight and body weight (5- and 5.6-fold, respectively), as well as a greater frequency of ciliated cholangiocytes compared with controls (29% vs 1.4%). It significantly decreases the amount of PCNA-positive cells in the treated tumors compared with vehicle controls (34% vs 65%). [2] This chemical shows significant inhibition of paw volume at 30 mg/kg i.p. in a Freund's complete adjuvant (FCA) induced animal model of inflammation. It (30 mg/kg i.p.) significant attenuates clinical scores (~ 70%), and IL-6 expression in paw tissues of collagen induced arthritis DBA1 mouse. [3] | |
|---|---|---|
| Animal Research | Animal Models | Rat cholangiocarcinoma xenografts BDEneu |
| Dosages | 10 mg/kg | |
| Administration | i.p. daily | |
References |
|
Chemical Information
| Molecular Weight | 335.4 | Formula | C20H21N3O2 |
| CAS No. | 1252003-15-8 | SDF | Download SDF |
| Synonyms | N/A | ||
| Smiles | CN1CCC2=C(C1)C3=CC=CC=C3N2CC4=CC=C(C=C4)C(=O)NO | ||
Storage and Stability
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 16.7 mg/mL ( (49.79 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Frequently Asked Questions
Question 1:
We are planning to order some tubastatin A but I found out there are two versions of it. One has HCl and one does not. Which one do you recommend for live cell use? Will the HCl containing version significantly change the pH?
Answer:
S8049 and S2627 have same molecular structure. The only difference is S2627 containing HCl and has higher solubility in DMSO (74 mg/mL vs. S8049 9 mg/mL). Since they are the same molecule, the biological function should be similar. I would recommend to use S2627 for cell culture study.
Question 2:
What’s the vehicle do you recommend to dissolve the compound for in vivo experiments?
Answer:
S8049 Tubastatin A can be dissolved in 2% DMSO/30% PEG 300/PBS at 2.5 mg/mL as a clear solution, and it is also a clear solution in 2% DMSO/ corn oil at 2.5 mg/mL. The drug in 2% DMSO/0.5% Tween 80/PBS is a homogeneous suspension at 2.5 mg/mL at first. After stay for a while, the precipitation goes out at the bottom of the tube.