- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
AR-42
Synonyms: HDAC-42
AR-42 (HDAC-42) is an HDAC inhibitor with IC50 of 30 nM. Phase 1.
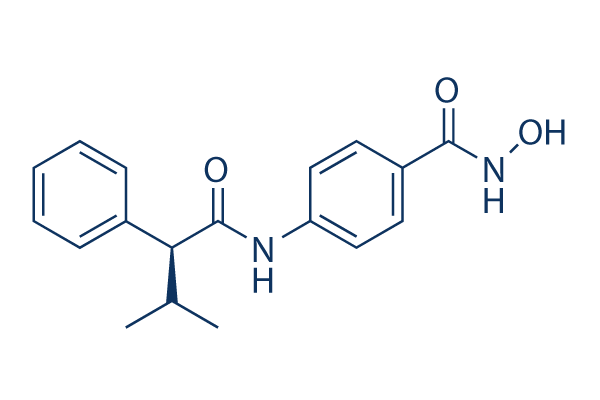
AR-42 Chemical Structure
CAS: 935881-37-1
Selleck's AR-42 has been cited by 25 Publications
6 Customer Reviews
Purity & Quality Control
Batch:
Purity:
99.03%
99.03
AR-42 Related Products
| Related Targets | HDAC1 HDAC2 HDAC3 HDAC4 HDAC5 HDAC6 HDAC7 HDAC8 HDAC9 HDAC10 HDAC11 HD1 HD2 | Click to Expand |
|---|---|---|
| Related Compound Libraries | Kinase Inhibitor Library FDA-approved Drug Library Natural Product Library Bioactive Compound Library-I Bioactive Compound Library-Ⅱ | Click to Expand |
Signaling Pathway
Choose Selective HDAC Inhibitors
Biological Activity
| Description | AR-42 (HDAC-42) is an HDAC inhibitor with IC50 of 30 nM. Phase 1. | ||
|---|---|---|---|
| Features | Greater potency relative to SAHA. | ||
| Targets |
|
| In vitro | ||||
| In vitro | AR-42 treatment induces histone hyperacetylation and p21WAF/CIP1 overexpression, and inhibits the growth of DU-145 cells with IC50 of 0.11 μM. [1] HDAC42 is potent in suppressing the proliferation of U87MG and PC-3 cells, in part, because of its ability to down-regulate Akt signaling. [2] AR-42 inhibits the growth of PC-3 and LNCaP cells with IC50 of 0.48 μM and 0.3 μM, respectively. Compared to SAHA, AR-42 exhibits distinctly superior apoptogenic potency, and causes markedly greater decreases in phospho-Akt, Bcl-xL, and survivin in PC-3 cells. [3] AR-42 treatment induces growth inhibition, cell- cycle arrest, apoptosis, and activation of caspases-3/7 in malignant mast cell lines. AR-42 treatment induces down-regulation of Kit via inhibition of Kit transcription, disassociation between Kit and heat shock protein 90 (HSP90), and up-regulation of HSP70. AR-42 treatment down-regulates the expression of p-Akt, total Akt, phosphorylated STAT3/5 (pSTAT3/5), and total STAT3/5. [6] AR-42 potently inhibits the growth of JeKo-1, Raji, and 697 cells with IC50 of <0.61 μM. AR-42 also sensitizes CLL cells to TNF-Related Apoptosis Inducing Ligand (TRAIL), potentially through reduction of c-FLIP. [7] AR-42 treatment also induces autophagy through downregulation of Akt/mTOR signaling and inducing ER stress in hepatocellular carcinoma (HCC) cells. [8] | |||
|---|---|---|---|---|
| Kinase Assay | In vitro HDAC assay | |||
| HDAC activity is analyzed by using an HDAC assay kit. This assay is based on the ability of DU-145 nuclear extract, which is rich in HDAC activity, to mediate the deacetylation of the biotinylated [3H]-acetyl histone H4 peptide that is bound to streptavidin agarose beads. The release of [3H]-acetate into the supernatant is measured to calculate the HDAC activity. Sodium butyrate (0.25-1 mM) is used as a positive control. | ||||
| Cell Research | Cell lines | DU-145 | ||
| Concentrations | Dissolved in DMSO, final concentrations ~2.5 μM | |||
| Incubation Time | 96 hours | |||
| Method | Cells are exposed to varous concentrations of AR-42 for 96 hours. The medium is removed and replaced by 150 μL of 0.5 mg/mL of MTT in RPMI 1640 medium, and the cells are incubated in the CO2 incubator at 37 °C for 2 hours. Supernatants are removed from the wells, and the reduced MTT dye is solubilized with 200 μL/well of DMSO. Absorbance is determined on a plate reader at 570 nm. |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | gp130 / p-STAT3 / STAT3 / p-AKT / AKT / p-MEK / MEK Cyclin D1 / p21 / p16 / Cyclin A / Cyclin B1 Act-H3 / Act-H3 / Act-tubulin p-Kit / Kit Notch1 / NICD / Nestin / Zeb-1 / BMI-1 |
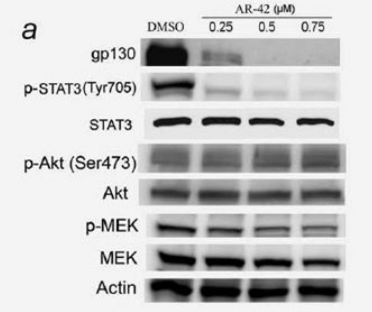
|
20824695 | |
| Growth inhibition assay | Cell viability |
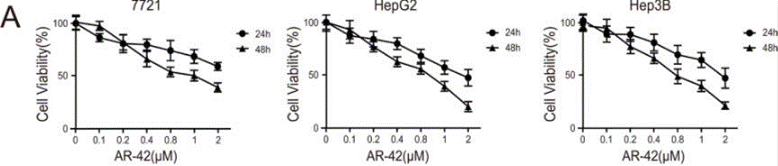
|
26993777 | |
| In Vivo | ||
| In vivo | The growth of PC-3 tumor xenografts is suppressed by 52% and 67% after treatment with AR-42 at 25 mg/kg and 50 mg/kg, respectively, whereas SAHA at 50 mg/kg suppresses growth by 31%. In contrast to mice treated with SAHA, intratumoral levels of phospho-Akt and Bcl-xL are markedly reduced in AR-42 treated mice. [3] In the transgenic adenocarcinoma of the mouse prostate (TRAMP) model, administration of AR-42 not only decreases the severity of prostatic intraepithelial neoplasia (PIN) and completely prevents its progression to poorly differentiated carcinoma, but also shifts tumorigenesis to a more differentiated phenotype, suppressing absolute and relative urogenital tract weights by 86% and 85%, respectively. [5] AR-42 significantly reduces leukocyte counts, and prolongs survival in three separate mouse models of B-cell malignancy without evidence of toxicity. [7] | |
|---|---|---|
| Animal Research | Animal Models | Intact male NCr athymic nude mice inoculated s.c. with PC-3 cells |
| Dosages | ~50 mg/kg/day | |
| Administration | Orally | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02795819 | Terminated | Renal Cell Carcinoma|Soft Tissue Sarcoma|Metastatic Disease |
Virginia Commonwealth University|National Cancer Institute (NCI) |
July 8 2016 | Phase 1 |
| NCT02282917 | Terminated | Vestibular Schwannoma|Meningioma|Acoustic Neuroma|Neurofibromatosis Type 2 |
Massachusetts Eye and Ear Infirmary|Johns Hopkins University|Mayo Clinic|Stanford University|Ohio State University|Nationwide Children''s Hospital |
December 2015 | Early Phase 1 |
Chemical Information & Solubility
| Molecular Weight | 312.36 | Formula | C18H20N2O3 |
| CAS No. | 935881-37-1 | SDF | Download AR-42 SDF |
| Smiles | CC(C)C(C1=CC=CC=C1)C(=O)NC2=CC=C(C=C2)C(=O)NO | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 63 mg/mL ( (201.69 mM); Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Ethanol : 63 mg/mL Water : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy AR-42 | AR-42 supplier | purchase AR-42 | AR-42 cost | AR-42 manufacturer | order AR-42 | AR-42 distributor