research use only
PD-0332991 (Palbociclib) Isethionate CDK4/6 inhibitor
Cat.No.S1579
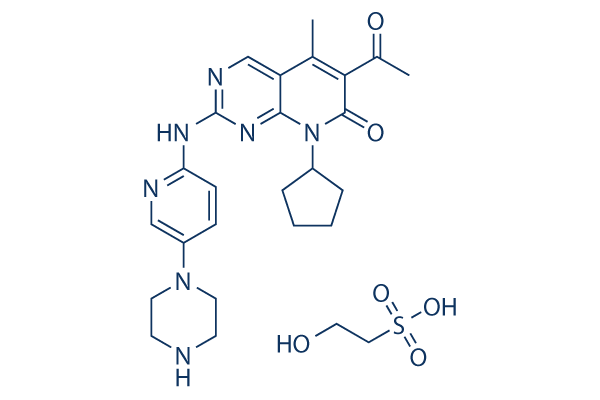
Chemical Structure
Molecular Weight: 573.66
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| H157 | Growth Inhibition Assay | 0-100 μM | 72 h | IC50=9 μM | 26390342 | |
| H2170 | Growth Inhibition Assay | 0-100 μM | 72 h | IC50=5 μM | 26390342 | |
| H520 | Growth Inhibition Assay | 0-100 μM | 72 h | IC50=0.73 μM | 26390342 | |
| H596 | Growth Inhibition Assay | 0-100 μM | 72 h | IC50=12 μM | 26390342 | |
| IMR-32 | Growth Inhibition Assay | 48 h | DMSO | IC50=261 nM | 26225123 | |
| SH-SY5Y | Growth Inhibition Assay | 48 h | DMSO | IC50=676 nM | 26225123 | |
| NGP | Growth Inhibition Assay | 48 h | DMSO | IC50=2.077 μM | 26225123 | |
| SH-EP | Growth Inhibition Assay | 48 h | DMSO | IC50=2.211 μM | 26225123 | |
| IMR-32 | Growth Inhibition Assay | 1 μM | 24 h | DMSO | induces G1 arrest | 26225123 |
| SH-SY5Y | Growth Inhibition Assay | 1 μM | 24 h | DMSO | induces G1 arrest | 26225123 |
| NGP | Growth Inhibition Assay | 1 μM | 24 h | DMSO | induces G1 arrest | 26225123 |
| SH-EP | Growth Inhibition Assay | 1 μM | 24 h | DMSO | induces G1 arrest | 26225123 |
| NGP | Fuction Assay | 0-1 μM | 24 h | DMSO | reduces the expression of TOP2A, CCNE2, and TK1 in a dose-dependent manner | 26225123 |
| IMR-32 | Fuction Assay | 0-1 μM | 24 h | DMSO | reduces the expression of TOP2A, CCNE2, and TK1 in a dose-dependent manner | 26225123 |
| U-CH2 | Growth Inhibition Assay | 0-1 μM | 72 h | IC50=50.2 nM | 26183925 | |
| U-CH3 | Growth Inhibition Assay | 0-1 μM | 72 h | IC50=52 nM | 26183925 | |
| U-CH6 | Growth Inhibition Assay | 0-1 μM | 72 h | IC50=90 nM | 26183925 | |
| U-CH7 | Growth Inhibition Assay | 0-1 μM | 72 h | IC50=98 nM | 26183925 | |
| U-CH11 | Growth Inhibition Assay | 0-1 μM | 72 h | IC50=340 nM | 26183925 | |
| MCF7 | Fuction Assay | 2.5-250 nM | 24 h | reduces Rb phosphorylation | 25991817 | |
| MV4-11 | Apoptosis Assay | 0.5 μM | 24 h | enhances apoptosis induced by sorafenib and AC220 | 25487917 | |
| MOLM13 | Apoptosis Assay | 0.5 μM | 24 h | enhances apoptosis induced by sorafenib and AC220 | 25487917 | |
| MOLM13-ITD alone | Growth Inhibition Assay | IC50=0.089 ± 0.009 μM | 25487917 | |||
| MV4-11-ITD alone | Growth Inhibition Assay | IC50=0.092 ± 0.008 μM | 25487917 | |||
| MOLM13-ITD/D835Y | Growth Inhibition Assay | IC50=0.114 ± 0.011 μM | 25487917 | |||
| MV4-11-ITD/D835V | Growth Inhibition Assay | IC50=0.116 ± 0.011 μM | 25487917 | |||
| MV4-11-ITD/D835V | Growth Inhibition Assay | IC50=0.102 ± 0.006 μM | 25487917 | |||
| MV4-11-ITD/F691L | Growth Inhibition Assay | IC50=0.112 ± 0.015 μM | 25487917 | |||
| MV4-11-ITD/N841K | Growth Inhibition Assay | IC50=0.113 ± 0.002 μM | 25487917 | |||
| U937-FLT3 WT | Growth Inhibition Assay | IC50=0.102 ± 0.013 μM | 25487917 | |||
| P12-ICHIKAWA | Growth Inhibition assay | IC50 = 0.09604 μM | SANGER | |||
| 697 | Growth Inhibition assay | IC50 = 0.14838 μM | SANGER | |||
| NB69 | Growth Inhibition assay | IC50 = 0.1618 μM | SANGER | |||
| EoL-1-cell | Growth Inhibition assay | IC50 = 0.18726 μM | SANGER | |||
| BHT-101 | Growth Inhibition assay | IC50 = 0.19825 μM | SANGER | |||
| SK-NEP-1 | Growth Inhibition assay | IC50 = 0.22002 μM | SANGER | |||
| MHH-NB-11 | Growth Inhibition assay | IC50 = 0.22019 μM | SANGER | |||
| AsPC-1 | Growth Inhibition assay | IC50 = 0.25253 μM | SANGER | |||
| ES1 | Growth Inhibition assay | IC50 = 0.25625 μM | SANGER | |||
| LAMA-84 | Growth Inhibition assay | IC50 = 0.25819 μM | SANGER | |||
| MOLT-16 | Growth Inhibition assay | IC50 = 0.25849 μM | SANGER | |||
| ES7 | Growth Inhibition assay | IC50 = 0.27309 μM | SANGER | |||
| KY821 | Growth Inhibition assay | IC50 = 0.3141 μM | SANGER | |||
| RT-112 | Growth Inhibition assay | IC50 = 0.32105 μM | SANGER | |||
| HL-60 | Growth Inhibition assay | IC50 = 0.34066 μM | SANGER | |||
| MOLT-4 | Growth Inhibition assay | IC50 = 0.34513 μM | SANGER | |||
| KARPAS-45 | Growth Inhibition assay | IC50 = 0.37616 μM | SANGER | |||
| SK-N-AS | Growth Inhibition assay | IC50 = 0.38783 μM | SANGER | |||
| CTB-1 | Growth Inhibition assay | IC50 = 0.40502 μM | SANGER | |||
| NKM-1 | Growth Inhibition assay | IC50 = 0.41189 μM | SANGER | |||
| HTC-C3 | Growth Inhibition assay | IC50 = 0.43295 μM | SANGER | |||
| BE-13 | Growth Inhibition assay | IC50 = 0.44427 μM | SANGER | |||
| KOSC-2 | Growth Inhibition assay | IC50 = 0.4669 μM | SANGER | |||
| NB14 | Growth Inhibition assay | IC50 = 0.48358 μM | SANGER | |||
| CAL-27 | Growth Inhibition assay | IC50 = 0.49459 μM | SANGER | |||
| H9 | Growth Inhibition assay | IC50 = 0.49543 μM | SANGER | |||
| RS4-11 | Growth Inhibition assay | IC50 = 0.50473 μM | SANGER | |||
| PA-1 | Growth Inhibition assay | IC50 = 0.50986 μM | SANGER | |||
| MV-4-11 | Growth Inhibition assay | IC50 = 0.51385 μM | SANGER | |||
| OS-RC-2 | Growth Inhibition assay | IC50 = 0.52162 μM | SANGER | |||
| RPMI-8226 | Growth Inhibition assay | IC50 = 0.52686 μM | SANGER | |||
| HGC-27 | Growth Inhibition assay | IC50 = 0.56499 μM | SANGER | |||
| CHP-212 | Growth Inhibition assay | IC50 = 0.59359 μM | SANGER | |||
| NB10 | Growth Inhibition assay | IC50 = 0.59918 μM | SANGER | |||
| HH | Growth Inhibition assay | IC50 = 0.59943 μM | SANGER | |||
| EW-16 | Growth Inhibition assay | IC50 = 0.60352 μM | SANGER | |||
| ES8 | Growth Inhibition assay | IC50 = 0.60521 μM | SANGER | |||
| HAL-01 | Growth Inhibition assay | IC50 = 0.60567 μM | SANGER | |||
| A204 | Growth Inhibition assay | IC50 = 0.63391 μM | SANGER | |||
| MHH-PREB-1 | Growth Inhibition assay | IC50 = 0.63699 μM | SANGER | |||
| EM-2 | Growth Inhibition assay | IC50 = 0.65064 μM | SANGER | |||
| BV-173 | Growth Inhibition assay | IC50 = 0.65248 μM | SANGER | |||
| ONS-76 | Growth Inhibition assay | IC50 = 0.67782 μM | SANGER | |||
| KM-H2 | Growth Inhibition assay | IC50 = 0.69554 μM | SANGER | |||
| D-263MG | Growth Inhibition assay | IC50 = 0.71712 μM | SANGER | |||
| ES3 | Growth Inhibition assay | IC50 = 0.72893 μM | SANGER | |||
| VA-ES-BJ | Growth Inhibition assay | IC50 = 0.73227 μM | SANGER | |||
| NBsusSR | Growth Inhibition assay | IC50 = 0.74299 μM | SANGER | |||
| NCI-H520 | Growth Inhibition assay | IC50 = 0.74652 μM | SANGER | |||
| ES5 | Growth Inhibition assay | IC50 = 0.7528 μM | SANGER | |||
| T-24 | Growth Inhibition assay | IC50 = 0.77871 μM | SANGER | |||
| SW962 | Growth Inhibition assay | IC50 = 0.80863 μM | SANGER | |||
| EW-3 | Growth Inhibition assay | IC50 = 0.80876 μM | SANGER | |||
| RXF393 | Growth Inhibition assay | IC50 = 0.81279 μM | SANGER | |||
| U251 | Growth Inhibition assay | IC50 = 0.81388 μM | SANGER | |||
| CAMA-1 | Growth Inhibition assay | IC50 = 0.83394 μM | SANGER | |||
| JVM-3 | Growth Inhibition assay | IC50 = 0.85178 μM | SANGER | |||
| COLO-800 | Growth Inhibition assay | IC50 = 0.89778 μM | SANGER | |||
| OVCAR-5 | Growth Inhibition assay | IC50 = 0.9001 μM | SANGER | |||
| LB1047-RCC | Growth Inhibition assay | IC50 = 0.92756 μM | SANGER | |||
| SW954 | Growth Inhibition assay | IC50 = 0.92941 μM | SANGER | |||
| J-RT3-T3-5 | Growth Inhibition assay | IC50 = 0.93606 μM | SANGER | |||
| Mewo | Growth Inhibition assay | IC50 = 0.9366 μM | SANGER | |||
| NCI-H1770 | Growth Inhibition assay | IC50 = 0.94055 μM | SANGER | |||
| HO-1-N-1 | Growth Inhibition assay | IC50 = 0.96281 μM | SANGER | |||
| HSC-3 | Growth Inhibition assay | IC50 = 0.96648 μM | SANGER | |||
| TYK-nu | Growth Inhibition assay | IC50 = 0.99825 μM | SANGER | |||
| KYSE-150 | Growth Inhibition assay | IC50 = 1.00076 μM | SANGER | |||
| SN12C | Growth Inhibition assay | IC50 = 1.00875 μM | SANGER | |||
| MOLT-13 | Growth Inhibition assay | IC50 = 1.01421 μM | SANGER | |||
| TE-11 | Growth Inhibition assay | IC50 = 1.04812 μM | SANGER | |||
| DB | Growth Inhibition assay | IC50 = 1.07273 μM | SANGER | |||
| CAL-39 | Growth Inhibition assay | IC50 = 1.07522 μM | SANGER | |||
| A3-KAW | Growth Inhibition assay | IC50 = 1.08408 μM | SANGER | |||
| CHP-134 | Growth Inhibition assay | IC50 = 1.11807 μM | SANGER | |||
| TGW | Growth Inhibition assay | IC50 = 1.12395 μM | SANGER | |||
| QIMR-WIL | Growth Inhibition assay | IC50 = 1.13134 μM | SANGER | |||
| NCI-SNU-1 | Growth Inhibition assay | IC50 = 1.16354 μM | SANGER | |||
| CGTH-W-1 | Growth Inhibition assay | IC50 = 1.17186 μM | SANGER | |||
| MHH-ES-1 | Growth Inhibition assay | IC50 = 1.17986 μM | SANGER | |||
| LB2241-RCC | Growth Inhibition assay | IC50 = 1.1862 μM | SANGER | |||
| ML-2 | Growth Inhibition assay | IC50 = 1.20734 μM | SANGER | |||
| COR-L23 | Growth Inhibition assay | IC50 = 1.22933 μM | SANGER | |||
| BFTC-905 | Growth Inhibition assay | IC50 = 1.24267 μM | SANGER | |||
| Hs-578-T | Growth Inhibition assay | IC50 = 1.25817 μM | SANGER | |||
| KG-1 | Growth Inhibition assay | IC50 = 1.26686 μM | SANGER | |||
| HEL | Growth Inhibition assay | IC50 = 1.29338 μM | SANGER | |||
| A549 | Growth Inhibition assay | IC50 = 1.29399 μM | SANGER | |||
| COLO-741 | Growth Inhibition assay | IC50 = 1.32089 μM | SANGER | |||
| PC-3 | Growth Inhibition assay | IC50 = 1.35221 μM | SANGER | |||
| HOS | Growth Inhibition assay | IC50 = 1.35296 μM | SANGER | |||
| HT-1080 | Growth Inhibition assay | IC50 = 1.37519 μM | SANGER | |||
| TE-8 | Growth Inhibition assay | IC50 = 1.41774 μM | SANGER | |||
| BHY | Growth Inhibition assay | IC50 = 1.46923 μM | SANGER | |||
| BB65-RCC | Growth Inhibition assay | IC50 = 1.50528 μM | SANGER | |||
| HN | Growth Inhibition assay | IC50 = 1.54071 μM | SANGER | |||
| NCI-H441 | Growth Inhibition assay | IC50 = 1.54907 μM | SANGER | |||
| RPMI-8866 | Growth Inhibition assay | IC50 = 1.58507 μM | SANGER | |||
| CAL-62 | Growth Inhibition assay | IC50 = 1.60862 μM | SANGER | |||
| MG-63 | Growth Inhibition assay | IC50 = 1.61813 μM | SANGER | |||
| SK-LU-1 | Growth Inhibition assay | IC50 = 1.62152 μM | SANGER | |||
| BCPAP | Growth Inhibition assay | IC50 = 1.66457 μM | SANGER | |||
| 22RV1 | Growth Inhibition assay | IC50 = 1.67843 μM | SANGER | |||
| T47D | Growth Inhibition assay | IC50 = 1.68061 μM | SANGER | |||
| MSTO-211H | Growth Inhibition assay | IC50 = 1.69603 μM | SANGER | |||
| DEL | Growth Inhibition assay | IC50 = 1.70273 μM | SANGER | |||
| H4 | Growth Inhibition assay | IC50 = 1.73212 μM | SANGER | |||
| CAL-51 | Growth Inhibition assay | IC50 = 1.74855 μM | SANGER | |||
| ABC-1 | Growth Inhibition assay | IC50 = 1.78582 μM | SANGER | |||
| MZ2-MEL | Growth Inhibition assay | IC50 = 1.79542 μM | SANGER | |||
| YKG-1 | Growth Inhibition assay | IC50 = 1.81061 μM | SANGER | |||
| KM12 | Growth Inhibition assay | IC50 = 1.81602 μM | SANGER | |||
| L-363 | Growth Inhibition assay | IC50 = 1.87412 μM | SANGER | |||
| KU812 | Growth Inhibition assay | IC50 = 1.89282 μM | SANGER | |||
| LOXIMVI | Growth Inhibition assay | IC50 = 1.91228 μM | SANGER | |||
| G-401 | Growth Inhibition assay | IC50 = 1.92428 μM | SANGER | |||
| SW780 | Growth Inhibition assay | IC50 = 1.96246 μM | SANGER | |||
| SW872 | Growth Inhibition assay | IC50 = 1.9833 μM | SANGER | |||
| NB7 | Growth Inhibition assay | IC50 = 1.99323 μM | SANGER | |||
| T98G | Growth Inhibition assay | IC50 = 2.00666 μM | SANGER | |||
| SW1710 | Growth Inhibition assay | IC50 = 2.06945 μM | SANGER | |||
| NCI-H1573 | Growth Inhibition assay | IC50 = 2.07298 μM | SANGER | |||
| KE-37 | Growth Inhibition assay | IC50 = 2.08951 μM | SANGER | |||
| 786-0 | Growth Inhibition assay | IC50 = 2.15439 μM | SANGER | |||
| SAS | Growth Inhibition assay | IC50 = 2.20374 μM | SANGER | |||
| CAL-54 | Growth Inhibition assay | IC50 = 2.20413 μM | SANGER | |||
| SF268 | Growth Inhibition assay | IC50 = 2.23122 μM | SANGER | |||
| SW620 | Growth Inhibition assay | IC50 = 2.26169 μM | SANGER | |||
| MN-60 | Growth Inhibition assay | IC50 = 2.3106 μM | SANGER | |||
| EFO-27 | Growth Inhibition assay | IC50 = 2.32058 μM | SANGER | |||
| NCI-H747 | Growth Inhibition assay | IC50 = 2.32199 μM | SANGER | |||
| HCC2218 | Growth Inhibition assay | IC50 = 2.35374 μM | SANGER | |||
| MIA-PaCa-2 | Growth Inhibition assay | IC50 = 2.36437 μM | SANGER | |||
| SJSA-1 | Growth Inhibition assay | IC50 = 2.37796 μM | SANGER | |||
| RKO | Growth Inhibition assay | IC50 = 2.38496 μM | SANGER | |||
| NB6 | Growth Inhibition assay | IC50 = 2.40374 μM | SANGER | |||
| ES4 | Growth Inhibition assay | IC50 = 2.45422 μM | SANGER | |||
| EGI-1 | Growth Inhibition assay | IC50 = 2.46883 μM | SANGER | |||
| CTV-1 | Growth Inhibition assay | IC50 = 2.52773 μM | SANGER | |||
| NCI-H1355 | Growth Inhibition assay | IC50 = 2.55951 μM | SANGER | |||
| GT3TKB | Growth Inhibition assay | IC50 = 2.59199 μM | SANGER | |||
| SK-HEP-1 | Growth Inhibition assay | IC50 = 2.59266 μM | SANGER | |||
| GAMG | Growth Inhibition assay | IC50 = 2.59394 μM | SANGER | |||
| SK-MES-1 | Growth Inhibition assay | IC50 = 2.61803 μM | SANGER | |||
| RO82-W-1 | Growth Inhibition assay | IC50 = 2.62057 μM | SANGER | |||
| ECC10 | Growth Inhibition assay | IC50 = 2.70206 μM | SANGER | |||
| MCF7 | Growth Inhibition assay | IC50 = 2.71464 μM | SANGER | |||
| D-283MED | Growth Inhibition assay | IC50 = 2.7243 μM | SANGER | |||
| RPMI-7951 | Growth Inhibition assay | IC50 = 2.75694 μM | SANGER | |||
| Ramos-2G6-4C10 | Growth Inhibition assay | IC50 = 2.77099 μM | SANGER | |||
| KGN | Growth Inhibition assay | IC50 = 2.81884 μM | SANGER | |||
| NUGC-3 | Growth Inhibition assay | IC50 = 2.82505 μM | SANGER | |||
| NCI-H292 | Growth Inhibition assay | IC50 = 2.85053 μM | SANGER | |||
| Becker | Growth Inhibition assay | IC50 = 2.95832 μM | SANGER | |||
| NCI-H1299 | Growth Inhibition assay | IC50 = 3.05263 μM | SANGER | |||
| ETK-1 | Growth Inhibition assay | IC50 = 3.0543 μM | SANGER | |||
| TK10 | Growth Inhibition assay | IC50 = 3.20165 μM | SANGER | |||
| VMRC-RCZ | Growth Inhibition assay | IC50 = 3.36488 μM | SANGER | |||
| YH-13 | Growth Inhibition assay | IC50 = 3.44079 μM | SANGER | |||
| DU-145 | Growth Inhibition assay | IC50 = 3.46269 μM | SANGER | |||
| SW1088 | Growth Inhibition assay | IC50 = 3.4747 μM | SANGER | |||
| HOP-92 | Growth Inhibition assay | IC50 = 3.50342 μM | SANGER | |||
| KP-N-YS | Growth Inhibition assay | IC50 = 3.62139 μM | SANGER | |||
| NCI-H460 | Growth Inhibition assay | IC50 = 3.6673 μM | SANGER | |||
| U-2-OS | Growth Inhibition assay | IC50 = 3.72535 μM | SANGER | |||
| A101D | Growth Inhibition assay | IC50 = 3.76936 μM | SANGER | |||
| MDA-MB-231 | Growth Inhibition assay | IC50 = 3.81951 μM | SANGER | |||
| IST-MES1 | Growth Inhibition assay | IC50 = 3.832 μM | SANGER | |||
| COR-L105 | Growth Inhibition assay | IC50 = 4.018 μM | SANGER | |||
| NCI-H1437 | Growth Inhibition assay | IC50 = 4.02302 μM | SANGER | |||
| CAL-85-1 | Growth Inhibition assay | IC50 = 4.02461 μM | SANGER | |||
| MZ1-PC | Growth Inhibition assay | IC50 = 4.18556 μM | SANGER | |||
| VM-CUB-1 | Growth Inhibition assay | IC50 = 4.31284 μM | SANGER | |||
| CHL-1 | Growth Inhibition assay | IC50 = 4.32169 μM | SANGER | |||
| MDA-MB-361 | Growth Inhibition assay | IC50 = 4.33153 μM | SANGER | |||
| NCI-H661 | Growth Inhibition assay | IC50 = 4.50092 μM | SANGER | |||
| EW-11 | Growth Inhibition assay | IC50 = 4.52231 μM | SANGER | |||
| BEN | Growth Inhibition assay | IC50 = 4.52815 μM | SANGER | |||
| BFTC-909 | Growth Inhibition assay | IC50 = 4.56275 μM | SANGER | |||
| NCI-H2087 | Growth Inhibition assay | IC50 = 4.58164 μM | SANGER | |||
| RVH-421 | Growth Inhibition assay | IC50 = 4.6669 μM | SANGER | |||
| P30-OHK | Growth Inhibition assay | IC50 = 4.68008 μM | SANGER | |||
| NCI-H28 | Growth Inhibition assay | IC50 = 4.81661 μM | SANGER | |||
| ES6 | Growth Inhibition assay | IC50 = 4.83016 μM | SANGER | |||
| 769-P | Growth Inhibition assay | IC50 = 4.85926 μM | SANGER | |||
| OE33 | Growth Inhibition assay | IC50 = 4.88161 μM | SANGER | |||
| SW982 | Growth Inhibition assay | IC50 = 4.95061 μM | SANGER | |||
| A388 | Growth Inhibition assay | IC50 = 5.02983 μM | SANGER | |||
| TI-73 | Growth Inhibition assay | IC50 = 5.06194 μM | SANGER | |||
| HCT-116 | Growth Inhibition assay | IC50 = 5.09889 μM | SANGER | |||
| HuP-T3 | Growth Inhibition assay | IC50 = 5.18709 μM | SANGER | |||
| G-402 | Growth Inhibition assay | IC50 = 5.19416 μM | SANGER | |||
| NCI-H1792 | Growth Inhibition assay | IC50 = 5.24622 μM | SANGER | |||
| NCI-H209 | Growth Inhibition assay | IC50 = 5.25942 μM | SANGER | |||
| NCI-H1650 | Growth Inhibition assay | IC50 = 5.30634 μM | SANGER | |||
| LCLC-97TM1 | Growth Inhibition assay | IC50 = 5.31808 μM | SANGER | |||
| S-117 | Growth Inhibition assay | IC50 = 5.36976 μM | SANGER | |||
| GI-ME-N | Growth Inhibition assay | IC50 = 5.39681 μM | SANGER | |||
| NCI-H2122 | Growth Inhibition assay | IC50 = 5.49397 μM | SANGER | |||
| NCI-H1793 | Growth Inhibition assay | IC50 = 5.67593 μM | SANGER | |||
| C2BBe1 | Growth Inhibition assay | IC50 = 5.70088 μM | SANGER | |||
| TE-12 | Growth Inhibition assay | IC50 = 5.80556 μM | SANGER | |||
| LCLC-103H | Growth Inhibition assay | IC50 = 5.917 μM | SANGER | |||
| A673 | Growth Inhibition assay | IC50 = 5.91932 μM | SANGER | |||
| BB30-HNC | Growth Inhibition assay | IC50 = 5.9836 μM | SANGER | |||
| SF295 | Growth Inhibition assay | IC50 = 6.00422 μM | SANGER | |||
| KU-19-19 | Growth Inhibition assay | IC50 = 6.01731 μM | SANGER | |||
| CFPAC-1 | Growth Inhibition assay | IC50 = 6.04443 μM | SANGER | |||
| LoVo | Growth Inhibition assay | IC50 = 6.05063 μM | SANGER | |||
| 8505C | Growth Inhibition assay | IC50 = 6.07573 μM | SANGER | |||
| GMS-10 | Growth Inhibition assay | IC50 = 6.15002 μM | SANGER | |||
| Ca9-22 | Growth Inhibition assay | IC50 = 6.1671 μM | SANGER | |||
| DOK | Growth Inhibition assay | IC50 = 6.22072 μM | SANGER | |||
| FADU | Growth Inhibition assay | IC50 = 6.26039 μM | SANGER | |||
| BxPC-3 | Growth Inhibition assay | IC50 = 6.2732 μM | SANGER | |||
| CAL-33 | Growth Inhibition assay | IC50 = 6.29201 μM | SANGER | |||
| SHP-77 | Growth Inhibition assay | IC50 = 6.31512 μM | SANGER | |||
| LXF-289 | Growth Inhibition assay | IC50 = 6.33455 μM | SANGER | |||
| GB-1 | Growth Inhibition assay | IC50 = 6.382 μM | SANGER | |||
| KS-1 | Growth Inhibition assay | IC50 = 6.38447 μM | SANGER | |||
| D-502MG | Growth Inhibition assay | IC50 = 6.42376 μM | SANGER | |||
| LAN-6 | Growth Inhibition assay | IC50 = 6.51023 μM | SANGER | |||
| H-EMC-SS | Growth Inhibition assay | IC50 = 6.56147 μM | SANGER | |||
| LC-2-ad | Growth Inhibition assay | IC50 = 6.60076 μM | SANGER | |||
| NCI-H1693 | Growth Inhibition assay | IC50 = 6.62215 μM | SANGER | |||
| SK-N-FI | Growth Inhibition assay | IC50 = 6.75044 μM | SANGER | |||
| D-423MG | Growth Inhibition assay | IC50 = 6.76117 μM | SANGER | |||
| KNS-42 | Growth Inhibition assay | IC50 = 6.78197 μM | SANGER | |||
| GCT | Growth Inhibition assay | IC50 = 6.938 μM | SANGER | |||
| DSH1 | Growth Inhibition assay | IC50 = 7.0633 μM | SANGER | |||
| D-247MG | Growth Inhibition assay | IC50 = 7.07881 μM | SANGER | |||
| NCI-SNU-5 | Growth Inhibition assay | IC50 = 7.18371 μM | SANGER | |||
| TE-6 | Growth Inhibition assay | IC50 = 7.20601 μM | SANGER | |||
| NOMO-1 | Growth Inhibition assay | IC50 = 7.22127 μM | SANGER | |||
| NB17 | Growth Inhibition assay | IC50 = 7.30309 μM | SANGER | |||
| EW-22 | Growth Inhibition assay | IC50 = 7.34348 μM | SANGER | |||
| EW-13 | Growth Inhibition assay | IC50 = 7.35162 μM | SANGER | |||
| DOHH-2 | Growth Inhibition assay | IC50 = 7.4402 μM | SANGER | |||
| TGBC1TKB | Growth Inhibition assay | IC50 = 7.49899 μM | SANGER | |||
| GR-ST | Growth Inhibition assay | IC50 = 7.52594 μM | SANGER | |||
| KYSE-520 | Growth Inhibition assay | IC50 = 7.55515 μM | SANGER | |||
| CAPAN-1 | Growth Inhibition assay | IC50 = 7.5951 μM | SANGER | |||
| HCE-4 | Growth Inhibition assay | IC50 = 7.62279 μM | SANGER | |||
| MLMA | Growth Inhibition assay | IC50 = 7.62957 μM | SANGER | |||
| HT-144 | Growth Inhibition assay | IC50 = 7.65368 μM | SANGER | |||
| KYSE-180 | Growth Inhibition assay | IC50 = 7.71169 μM | SANGER | |||
| TE-5 | Growth Inhibition assay | IC50 = 7.95971 μM | SANGER | |||
| IGROV-1 | Growth Inhibition assay | IC50 = 7.98551 μM | SANGER | |||
| NCI-H1581 | Growth Inhibition assay | IC50 = 8.012 μM | SANGER | |||
| SW1990 | Growth Inhibition assay | IC50 = 8.04659 μM | SANGER | |||
| EFM-19 | Growth Inhibition assay | IC50 = 8.08545 μM | SANGER | |||
| IGR-1 | Growth Inhibition assay | IC50 = 8.43023 μM | SANGER | |||
| U-118-MG | Growth Inhibition assay | IC50 = 8.43463 μM | SANGER | |||
| SK-OV-3 | Growth Inhibition assay | IC50 = 8.46765 μM | SANGER | |||
| KNS-62 | Growth Inhibition assay | IC50 = 8.51761 μM | SANGER | |||
| GOTO | Growth Inhibition assay | IC50 = 8.57635 μM | SANGER | |||
| 8305C | Growth Inhibition assay | IC50 = 8.70484 μM | SANGER | |||
| RPMI-2650 | Growth Inhibition assay | IC50 = 8.71955 μM | SANGER | |||
| NEC8 | Growth Inhibition assay | IC50 = 8.74307 μM | SANGER | |||
| KYSE-450 | Growth Inhibition assay | IC50 = 8.86548 μM | SANGER | |||
| RMG-I | Growth Inhibition assay | IC50 = 9.14058 μM | SANGER | |||
| CAKI-1 | Growth Inhibition assay | IC50 = 9.31979 μM | SANGER | |||
| KYSE-510 | Growth Inhibition assay | IC50 = 9.35778 μM | SANGER | |||
| A4-Fuk | Growth Inhibition assay | IC50 = 9.36701 μM | SANGER | |||
| AN3-CA | Growth Inhibition assay | IC50 = 9.45444 μM | SANGER | |||
| SK-N-DZ | Growth Inhibition assay | IC50 = 9.72849 μM | SANGER | |||
| HSC-2 | Growth Inhibition assay | IC50 = 9.76629 μM | SANGER | |||
| EW-1 | Growth Inhibition assay | IC50 = 9.79369 μM | SANGER | |||
| D-566MG | Growth Inhibition assay | IC50 = 9.83664 μM | SANGER | |||
| COLO-792 | Growth Inhibition assay | IC50 = 9.98746 μM | SANGER | |||
| TE-10 | Growth Inhibition assay | IC50 = 10.0396 μM | SANGER | |||
| NCI-H650 | Growth Inhibition assay | IC50 = 10.4286 μM | SANGER | |||
| U-266 | Growth Inhibition assay | IC50 = 10.455 μM | SANGER | |||
| Detroit562 | Growth Inhibition assay | IC50 = 11.0515 μM | SANGER | |||
| NH-12 | Growth Inhibition assay | IC50 = 11.1446 μM | SANGER | |||
| OC-314 | Growth Inhibition assay | IC50 = 11.2842 μM | SANGER | |||
| IST-MEL1 | Growth Inhibition assay | IC50 = 11.5323 μM | SANGER | |||
| KNS-81-FD | Growth Inhibition assay | IC50 = 11.5527 μM | SANGER | |||
| SW1463 | Growth Inhibition assay | IC50 = 11.5989 μM | SANGER | |||
| NCI-H23 | Growth Inhibition assay | IC50 = 11.6553 μM | SANGER | |||
| SK-MEL-2 | Growth Inhibition assay | IC50 = 11.7197 μM | SANGER | |||
| NB13 | Growth Inhibition assay | IC50 = 12.1495 μM | SANGER | |||
| Daoy | Growth Inhibition assay | IC50 = 12.2855 μM | SANGER | |||
| NCI-H1623 | Growth Inhibition assay | IC50 = 12.3801 μM | SANGER | |||
| NMC-G1 | Growth Inhibition assay | IC50 = 12.717 μM | SANGER | |||
| DK-MG | Growth Inhibition assay | IC50 = 12.9482 μM | SANGER | |||
| TCCSUP | Growth Inhibition assay | IC50 = 13.1284 μM | SANGER | |||
| SCC-15 | Growth Inhibition assay | IC50 = 13.2651 μM | SANGER | |||
| NOS-1 | Growth Inhibition assay | IC50 = 13.2893 μM | SANGER | |||
| RH-1 | Growth Inhibition assay | IC50 = 13.3037 μM | SANGER | |||
| SK-MEL-3 | Growth Inhibition assay | IC50 = 13.3728 μM | SANGER | |||
| NB5 | Growth Inhibition assay | IC50 = 13.4067 μM | SANGER | |||
| SNU-387 | Growth Inhibition assay | IC50 = 13.5072 μM | SANGER | |||
| CAL-120 | Growth Inhibition assay | IC50 = 13.6718 μM | SANGER | |||
| LNCaP-Clone-FGC | Growth Inhibition assay | IC50 = 13.7991 μM | SANGER | |||
| ACN | Growth Inhibition assay | IC50 = 14.0288 μM | SANGER | |||
| SK-MEL-30 | Growth Inhibition assay | IC50 = 14.063 μM | SANGER | |||
| COLO-678 | Growth Inhibition assay | IC50 = 14.0822 μM | SANGER | |||
| SCC-9 | Growth Inhibition assay | IC50 = 14.1021 μM | SANGER | |||
| KINGS-1 | Growth Inhibition assay | IC50 = 14.1402 μM | SANGER | |||
| LS-513 | Growth Inhibition assay | IC50 = 14.187 μM | SANGER | |||
| HLE | Growth Inhibition assay | IC50 = 14.3852 μM | SANGER | |||
| SW1573 | Growth Inhibition assay | IC50 = 14.4435 μM | SANGER | |||
| KYSE-140 | Growth Inhibition assay | IC50 = 14.6327 μM | SANGER | |||
| SK-PN-DW | Growth Inhibition assay | IC50 = 14.8001 μM | SANGER | |||
| A253 | Growth Inhibition assay | IC50 = 15.0625 μM | SANGER | |||
| CAL-12T | Growth Inhibition assay | IC50 = 15.4662 μM | SANGER | |||
| COLO-679 | Growth Inhibition assay | IC50 = 15.7683 μM | SANGER | |||
| UACC-257 | Growth Inhibition assay | IC50 = 16.1201 μM | SANGER | |||
| U-87-MG | Growth Inhibition assay | IC50 = 16.3523 μM | SANGER | |||
| HCC1806 | Growth Inhibition assay | IC50 = 16.7071 μM | SANGER | |||
| NCI-H2170 | Growth Inhibition assay | IC50 = 17.2448 μM | SANGER | |||
| AGS | Growth Inhibition assay | IC50 = 17.3808 μM | SANGER | |||
| MEL-HO | Growth Inhibition assay | IC50 = 17.7503 μM | SANGER | |||
| SW48 | Growth Inhibition assay | IC50 = 17.7716 μM | SANGER | |||
| HuP-T4 | Growth Inhibition assay | IC50 = 18.0206 μM | SANGER | |||
| NCI-H720 | Growth Inhibition assay | IC50 = 18.1402 μM | SANGER | |||
| RCC10RGB | Growth Inhibition assay | IC50 = 18.1697 μM | SANGER | |||
| HD-MY-Z | Growth Inhibition assay | IC50 = 18.2254 μM | SANGER | |||
| A427 | Growth Inhibition assay | IC50 = 18.5094 μM | SANGER | |||
| HCC2998 | Growth Inhibition assay | IC50 = 18.6051 μM | SANGER | |||
| EPLC-272H | Growth Inhibition assay | IC50 = 19.0434 μM | SANGER | |||
| C32 | Growth Inhibition assay | IC50 = 19.045 μM | SANGER | |||
| UMC-11 | Growth Inhibition assay | IC50 = 19.2123 μM | SANGER | |||
| CaR-1 | Growth Inhibition assay | IC50 = 19.6804 μM | SANGER | |||
| KYSE-410 | Growth Inhibition assay | IC50 = 19.9139 μM | SANGER | |||
| HuCCT1 | Growth Inhibition assay | IC50 = 20.6293 μM | SANGER | |||
| LB996-RCC | Growth Inhibition assay | IC50 = 20.7168 μM | SANGER | |||
| KYSE-70 | Growth Inhibition assay | IC50 = 20.8059 μM | SANGER | |||
| CAL-72 | Growth Inhibition assay | IC50 = 20.915 μM | SANGER | |||
| Capan-2 | Growth Inhibition assay | IC50 = 21.0413 μM | SANGER | |||
| PANC-08-13 | Growth Inhibition assay | IC50 = 21.2515 μM | SANGER | |||
| SBC-1 | Growth Inhibition assay | IC50 = 21.3081 μM | SANGER | |||
| MFM-223 | Growth Inhibition assay | IC50 = 21.3342 μM | SANGER | |||
| BB49-HNC | Growth Inhibition assay | IC50 = 21.5716 μM | SANGER | |||
| SH-4 | Growth Inhibition assay | IC50 = 21.6618 μM | SANGER | |||
| HuO9 | Growth Inhibition assay | IC50 = 21.9825 μM | SANGER | |||
| AM-38 | Growth Inhibition assay | IC50 = 22.0485 μM | SANGER | |||
| A431 | Growth Inhibition assay | IC50 = 23.2119 μM | SANGER | |||
| YAPC | Growth Inhibition assay | IC50 = 23.2651 μM | SANGER | |||
| LU-139 | Growth Inhibition assay | IC50 = 23.4809 μM | SANGER | |||
| HEC-1 | Growth Inhibition assay | IC50 = 23.4937 μM | SANGER | |||
| SCC-25 | Growth Inhibition assay | IC50 = 24.3006 μM | SANGER | |||
| HT-29 | Growth Inhibition assay | IC50 = 24.3823 μM | SANGER | |||
| PC-14 | Growth Inhibition assay | IC50 = 24.6571 μM | SANGER | |||
| Calu-6 | Growth Inhibition assay | IC50 = 25.5071 μM | SANGER | |||
| SJRH30 | Growth Inhibition assay | IC50 = 25.6496 μM | SANGER | |||
| ChaGo-K-1 | Growth Inhibition assay | IC50 = 26.1629 μM | SANGER | |||
| IA-LM | Growth Inhibition assay | IC50 = 26.3645 μM | SANGER | |||
| GP5d | Growth Inhibition assay | IC50 = 26.4491 μM | SANGER | |||
| NCI-H2291 | Growth Inhibition assay | IC50 = 26.6541 μM | SANGER | |||
| BALL-1 | Growth Inhibition assay | IC50 = 26.9397 μM | SANGER | |||
| HCC1954 | Growth Inhibition assay | IC50 = 26.9808 μM | SANGER | |||
| NCI-H2452 | Growth Inhibition assay | IC50 = 27.4163 μM | SANGER | |||
| LU-99A | Growth Inhibition assay | IC50 = 27.5582 μM | SANGER | |||
| NTERA-S-cl-D1 | Growth Inhibition assay | IC50 = 27.7299 μM | SANGER | |||
| PANC-10-05 | Growth Inhibition assay | IC50 = 27.7775 μM | SANGER | |||
| NCI-H2405 | Growth Inhibition assay | IC50 = 27.9387 μM | SANGER | |||
| MDA-MB-415 | Growth Inhibition assay | IC50 = 28.4137 μM | SANGER | |||
| NCI-H2342 | Growth Inhibition assay | IC50 = 28.5281 μM | SANGER | |||
| TGBC24TKB | Growth Inhibition assay | IC50 = 28.7117 μM | SANGER | |||
| LU-134-A | Growth Inhibition assay | IC50 = 28.9261 μM | SANGER | |||
| SCC-4 | Growth Inhibition assay | IC50 = 31.0495 μM | SANGER | |||
| Saos-2 | Growth Inhibition assay | IC50 = 31.9306 μM | SANGER | |||
| RERF-LC-MS | Growth Inhibition assay | IC50 = 32.8231 μM | SANGER | |||
| M14 | Growth Inhibition assay | IC50 = 32.9764 μM | SANGER | |||
| HPAF-II | Growth Inhibition assay | IC50 = 33.5011 μM | SANGER | |||
| NCI-H1755 | Growth Inhibition assay | IC50 = 34.3305 μM | SANGER | |||
| D-392MG | Growth Inhibition assay | IC50 = 35.8674 μM | SANGER | |||
| A704 | Growth Inhibition assay | IC50 = 36.0427 μM | SANGER | |||
| CP50-MEL-B | Growth Inhibition assay | IC50 = 36.1911 μM | SANGER | |||
| EW-18 | Growth Inhibition assay | IC50 = 36.452 μM | SANGER | |||
| WM-115 | Growth Inhibition assay | IC50 = 36.8099 μM | SANGER | |||
| LU-65 | Growth Inhibition assay | IC50 = 37.1417 μM | SANGER | |||
| NCI-H1563 | Growth Inhibition assay | IC50 = 37.2484 μM | SANGER | |||
| DBTRG-05MG | Growth Inhibition assay | IC50 = 38.0691 μM | SANGER | |||
| NCI-H630 | Growth Inhibition assay | IC50 = 38.4714 μM | SANGER | |||
| NCI-H1155 | Growth Inhibition assay | IC50 = 39.242 μM | SANGER | |||
| OVCAR-3 | Growth Inhibition assay | IC50 = 39.9195 μM | SANGER | |||
| OAW-42 | Growth Inhibition assay | IC50 = 40.4258 μM | SANGER | |||
| JVM-2 | Growth Inhibition assay | IC50 = 41.2415 μM | SANGER | |||
| C3A | Growth Inhibition assay | IC50 = 41.3447 μM | SANGER | |||
| HT55 | Growth Inhibition assay | IC50 = 42.2841 μM | SANGER | |||
| OVCAR-4 | Growth Inhibition assay | IC50 = 42.2974 μM | SANGER | |||
| MEG-01 | Growth Inhibition assay | IC50 = 42.4616 μM | SANGER | |||
| NCI-H82 | Growth Inhibition assay | IC50 = 43.9892 μM | SANGER | |||
| JEG-3 | Growth Inhibition assay | IC50 = 44.947 μM | SANGER | |||
| BPH-1 | Growth Inhibition assay | IC50 = 46.3998 μM | SANGER | |||
| MPP-89 | Growth Inhibition assay | IC50 = 47.2898 μM | SANGER | |||
| ALL-PO | Growth Inhibition assay | IC50 = 47.4188 μM | SANGER | |||
| HT | Growth Inhibition assay | IC50 = 47.492 μM | SANGER | |||
| NCI-H2347 | Growth Inhibition assay | IC50 = 48.0715 μM | SANGER | |||
| A2780 | Growth Inhibition assay | IC50 = 49.4228 μM | SANGER | |||
| KARPAS-299 | Growth Inhibition assay | IC50 = 49.5119 μM | SANGER | |||
| NCI-H1651 | Growth Inhibition assay | IC50 = 49.8821 μM | SANGER | |||
| OCI-AML2 | Growth Inhibition assay | IC50 = 50.2084 μM | SANGER | |||
| OCUB-M | Growth Inhibition assay | IC50 = 50.3047 μM | SANGER | |||
| HuH-7 | Growth Inhibition assay | IC50 = 50.5943 μM | SANGER | |||
| RCM-1 | Growth Inhibition assay | IC50 = 50.9133 μM | SANGER | |||
| FTC-133 | Growth Inhibition assay | IC50 = 51.322 μM | SANGER | |||
| PSN1 | Growth Inhibition assay | IC50 = 51.4335 μM | SANGER | |||
| A498 | Growth Inhibition assay | IC50 = 52.0995 μM | SANGER | |||
| SF126 | Growth Inhibition assay | IC50 = 52.6568 μM | SANGER | |||
| UACC-62 | Growth Inhibition assay | IC50 = 55.2224 μM | SANGER | |||
| PANC-03-27 | Growth Inhibition assay | IC50 = 56.6788 μM | SANGER | |||
| NCI-H727 | Growth Inhibition assay | IC50 = 56.8127 μM | SANGER | |||
| SNU-423 | Growth Inhibition assay | IC50 = 57.2859 μM | SANGER | |||
| LB771-HNC | Growth Inhibition assay | IC50 = 57.8405 μM | SANGER | |||
| HCC1395 | Growth Inhibition assay | IC50 = 58.0464 μM | SANGER | |||
| OAW-28 | Growth Inhibition assay | IC50 = 58.2785 μM | SANGER | |||
| NB12 | Growth Inhibition assay | IC50 = 60.6696 μM | SANGER | |||
| DJM-1 | Growth Inhibition assay | IC50 = 60.7331 μM | SANGER | |||
| MDA-MB-157 | Growth Inhibition assay | IC50 = 61.1649 μM | SANGER | |||
| DU-4475 | Growth Inhibition assay | IC50 = 62.5495 μM | SANGER | |||
| RD | Growth Inhibition assay | IC50 = 62.7387 μM | SANGER | |||
| LS-123 | Growth Inhibition assay | IC50 = 63.2618 μM | SANGER | |||
| NCI-H226 | Growth Inhibition assay | IC50 = 64.22 μM | SANGER | |||
| MS-1 | Growth Inhibition assay | IC50 = 64.8358 μM | SANGER | |||
| no-10 | Growth Inhibition assay | IC50 = 65.7473 μM | SANGER | |||
| SW626 | Growth Inhibition assay | IC50 = 65.7579 μM | SANGER | |||
| DMS-273 | Growth Inhibition assay | IC50 = 66.2296 μM | SANGER | |||
| ESS-1 | Growth Inhibition assay | IC50 = 66.5096 μM | SANGER | |||
| TE-9 | Growth Inhibition assay | IC50 = 66.6316 μM | SANGER | |||
| CAS-1 | Growth Inhibition assay | IC50 = 67.7954 μM | SANGER | |||
| 639-V | Growth Inhibition assay | IC50 = 69.4198 μM | SANGER | |||
| GAK | Growth Inhibition assay | IC50 = 70.2031 μM | SANGER | |||
| NCI-H526 | Growth Inhibition assay | IC50 = 71.0913 μM | SANGER | |||
| DoTc2-4510 | Growth Inhibition assay | IC50 = 71.443 μM | SANGER | |||
| 647-V | Growth Inhibition assay | IC50 = 73.2118 μM | SANGER | |||
| OVCAR-8 | Growth Inhibition assay | IC50 = 73.2367 μM | SANGER | |||
| COLO-680N | Growth Inhibition assay | IC50 = 75.2497 μM | SANGER | |||
| HDLM-2 | Growth Inhibition assay | IC50 = 75.7806 μM | SANGER | |||
| TGBC11TKB | Growth Inhibition assay | IC50 = 79.0192 μM | SANGER | |||
| DMS-114 | Growth Inhibition assay | IC50 = 81.9577 μM | SANGER | |||
| CW-2 | Growth Inhibition assay | IC50 = 82.6818 μM | SANGER | |||
| MDA-MB-175-VII | Growth Inhibition assay | IC50 = 85.3135 μM | SANGER | |||
| HT-1376 | Growth Inhibition assay | IC50 = 85.846 μM | SANGER | |||
| SW1417 | Growth Inhibition assay | IC50 = 88.2906 μM | SANGER | |||
| HCE-T | Growth Inhibition assay | IC50 = 91.7205 μM | SANGER | |||
| OE19 | Growth Inhibition assay | IC50 = 92.7944 μM | SANGER | |||
| NCI-N87 | Growth Inhibition assay | IC50 = 92.8881 μM | SANGER | |||
| SNU-449 | Growth Inhibition assay | IC50 = 93.584 μM | SANGER | |||
| NCI-H1092 | Growth Inhibition assay | IC50 = 96.5851 μM | SANGER | |||
| COLO-684 | Growth Inhibition assay | IC50 = 99.0686 μM | SANGER | |||
| HCC1937 | Growth Inhibition assay | IC50 = 99.9315 μM | SANGER | |||
| EFO-21 | Growth Inhibition assay | IC50 = 110.988 μM | SANGER | |||
| ZR-75-30 | Growth Inhibition assay | IC50 = 114.117 μM | SANGER | |||
| HCC1187 | Growth Inhibition assay | IC50 = 114.179 μM | SANGER | |||
| SW684 | Growth Inhibition assay | IC50 = 114.655 μM | SANGER | |||
| AU565 | Growth Inhibition assay | IC50 = 115.892 μM | SANGER | |||
| HCC38 | Growth Inhibition assay | IC50 = 116.559 μM | SANGER | |||
| LK-2 | Growth Inhibition assay | IC50 = 119.652 μM | SANGER | |||
| LB2518-MEL | Growth Inhibition assay | IC50 = 122.428 μM | SANGER | |||
| COLO-320-HSR | Growth Inhibition assay | IC50 = 126.212 μM | SANGER | |||
| HCC1419 | Growth Inhibition assay | IC50 = 129.227 μM | SANGER | |||
| NCI-H2228 | Growth Inhibition assay | IC50 = 129.321 μM | SANGER | |||
| NCI-H2052 | Growth Inhibition assay | IC50 = 130.358 μM | SANGER | |||
| HCC2157 | Growth Inhibition assay | IC50 = 133.973 μM | SANGER | |||
| HCC1569 | Growth Inhibition assay | IC50 = 135.05 μM | SANGER | |||
| HuO-3N1 | Growth Inhibition assay | IC50 = 135.366 μM | SANGER | |||
| MZ7-mel | Growth Inhibition assay | IC50 = 145.604 μM | SANGER | |||
| SW756 | Growth Inhibition assay | IC50 = 147.143 μM | SANGER | |||
| NCI-H1304 | Growth Inhibition assay | IC50 = 149.222 μM | SANGER | |||
| LN-405 | Growth Inhibition assay | IC50 = 149.732 μM | SANGER | |||
| BT-474 | Growth Inhibition assay | IC50 = 150.969 μM | SANGER | |||
| LS-411N | Growth Inhibition assay | IC50 = 152.319 μM | SANGER | |||
| ATN-1 | Growth Inhibition assay | IC50 = 152.458 μM | SANGER | |||
| SNU-C2B | Growth Inhibition assay | IC50 = 153.444 μM | SANGER | |||
| KYSE-270 | Growth Inhibition assay | IC50 = 155.75 μM | SANGER | |||
| NCI-H1395 | Growth Inhibition assay | IC50 = 156.493 μM | SANGER | |||
| EKVX | Growth Inhibition assay | IC50 = 156.981 μM | SANGER | |||
| HCC70 | Growth Inhibition assay | IC50 = 157.989 μM | SANGER | |||
| NCI-H522 | Growth Inhibition assay | IC50 = 161.368 μM | SANGER | |||
| Ca-Ski | Growth Inhibition assay | IC50 = 161.86 μM | SANGER | |||
| SW1116 | Growth Inhibition assay | IC50 = 163.382 μM | SANGER | |||
| 8-MG-BA | Growth Inhibition assay | IC50 = 165.797 μM | SANGER | |||
| NCI-H1048 | Growth Inhibition assay | IC50 = 172.374 μM | SANGER | |||
| RH-18 | Growth Inhibition assay | IC50 = 172.601 μM | SANGER | |||
| LU-135 | Growth Inhibition assay | IC50 = 173.259 μM | SANGER | |||
| SW948 | Growth Inhibition assay | IC50 = 174.6 μM | SANGER | |||
| LS-1034 | Growth Inhibition assay | IC50 = 183.742 μM | SANGER | |||
| SCH | Growth Inhibition assay | IC50 = 187.022 μM | SANGER | |||
| KP-N-YN | Growth Inhibition assay | IC50 = 192.971 μM | SANGER | |||
| NCI-H69 | Growth Inhibition assay | IC50 = 210.925 μM | SANGER | |||
| SBC-5 | Growth Inhibition assay | IC50 = 211.162 μM | SANGER | |||
| MFE-280 | Growth Inhibition assay | IC50 = 212.067 μM | SANGER | |||
| MC116 | Growth Inhibition assay | IC50 = 212.736 μM | SANGER | |||
| BT-20 | Growth Inhibition assay | IC50 = 216.38 μM | SANGER | |||
| NCI-H1993 | Growth Inhibition assay | IC50 = 217.295 μM | SANGER | |||
| Calu-3 | Growth Inhibition assay | IC50 = 217.37 μM | SANGER | |||
| GCIY | Growth Inhibition assay | IC50 = 221.332 μM | SANGER | |||
| HT-3 | Growth Inhibition assay | IC50 = 222.173 μM | SANGER | |||
| MC-IXC | Growth Inhibition assay | IC50 = 227.215 μM | SANGER | |||
| COR-L88 | Growth Inhibition assay | IC50 = 234.259 μM | SANGER | |||
| SK-MEL-1 | Growth Inhibition assay | IC50 = 235.59 μM | SANGER | |||
| MDA-MB-453 | Growth Inhibition assay | IC50 = 236.33 μM | SANGER | |||
| SiHa | Growth Inhibition assay | IC50 = 247.22 μM | SANGER | |||
| C8166 | Growth Inhibition assay | IC50 = 251.341 μM | SANGER | |||
| SKG-IIIa | Growth Inhibition assay | IC50 = 251.504 μM | SANGER | |||
| HT-1197 | Growth Inhibition assay | IC50 = 268.492 μM | SANGER | |||
| no-11 | Growth Inhibition assay | IC50 = 291.357 μM | SANGER | |||
| ME-180 | Growth Inhibition assay | IC50 = 296.912 μM | SANGER | |||
| NY | Growth Inhibition assay | IC50 = 320.632 μM | SANGER | |||
| C-33-A | Growth Inhibition assay | IC50 = 325.06 μM | SANGER | |||
| NCI-H2029 | Growth Inhibition assay | IC50 = 338.305 μM | SANGER | |||
| IST-SL1 | Growth Inhibition assay | IC50 = 342.105 μM | SANGER | |||
| KLE | Growth Inhibition assay | IC50 = 347.29 μM | SANGER | |||
| SF539 | Growth Inhibition assay | IC50 = 348.01 μM | SANGER | |||
| 5637 | Growth Inhibition assay | IC50 = 360.37 μM | SANGER | |||
| COLO-824 | Growth Inhibition assay | IC50 = 363.58 μM | SANGER | |||
| NCI-H810 | Growth Inhibition assay | IC50 = 363.938 μM | SANGER | |||
| JAR | Growth Inhibition assay | IC50 = 365.01 μM | SANGER | |||
| L-428 | Growth Inhibition assay | IC50 = 369.277 μM | SANGER | |||
| COLO-668 | Growth Inhibition assay | IC50 = 387.033 μM | SANGER | |||
| GI-1 | Growth Inhibition assay | IC50 = 398.59 μM | SANGER | |||
| NCI-H446 | Growth Inhibition assay | IC50 = 399.497 μM | SANGER | |||
| CCRF-CEM | Growth Inhibition assay | IC50 = 403.587 μM | SANGER | |||
| T84 | Growth Inhibition assay | IC50 = 403.686 μM | SANGER | |||
| K-562 | Growth Inhibition assay | IC50 = 432.947 μM | SANGER | |||
| OMC-1 | Growth Inhibition assay | IC50 = 433.814 μM | SANGER | |||
| LB831-BLC | Growth Inhibition assay | IC50 = 434.556 μM | SANGER | |||
| BT-549 | Growth Inhibition assay | IC50 = 435.102 μM | SANGER | |||
| MKN7 | Growth Inhibition assay | IC50 = 439.822 μM | SANGER | |||
| SW1783 | Growth Inhibition assay | IC50 = 449.251 μM | SANGER | |||
| SNB75 | Growth Inhibition assay | IC50 = 486.478 μM | SANGER | |||
| M059J | Growth Inhibition assay | IC50 = 492.923 μM | SANGER | |||
| KURAMOCHI | Growth Inhibition assay | IC50 = 498.612 μM | SANGER | |||
| TE-1 | Growth Inhibition assay | IC50 = 502.729 μM | SANGER | |||
| NCI-H596 | Growth Inhibition assay | IC50 = 530.805 μM | SANGER | |||
| PFSK-1 | Growth Inhibition assay | IC50 = 532.941 μM | SANGER | |||
| MOLM13 | Function assay | 24 hrs | Inhibition of CDK4 in human MOLM13 cells assessed as inhibition of Rb phosphorylation at Ser780 after 24 hrs, IC50 = 0.011 μM. | 24641103 | ||
| COLO205 | Antiproliferative assay | 72 hrs | Antiproliferative activity against Rb-positive human COLO205 cells assessed as incorporation of [3H]thymidine into DNA after 72 hrs by beta-plate counting analysis, IC50 = 0.036 μM. | 24641103 | ||
| MOLM13 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MOLM13 cells harboring FLT3 ITD mutant assessed as incorporation of [3H]thymidine into DNA after 72 hrs by beta-plate counting analysis, IC50 = 0.096 μM. | 24641103 | ||
| MOLM13 | Antiproliferative assay | 72 hrs | Antiproliferative activity against sorafenib-resistant human MOLM13 cells assessed as incorporation of [3H]thymidine into DNA after 72 hrs by beta-plate counting analysis, IC50 = 0.096 μM. | 24641103 | ||
| U937 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human U937 cells assessed as incorporation of [3H]thymidine into DNA after 72 hrs by beta-plate counting analysis, IC50 = 0.14 μM. | 24641103 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK6/cyclin D2 (unknown origin) expressed in baculovirus infected insect cells using GST-fused pRb (792 to 928) as substrate preincubated for 2 mins followed by [gamma-32P]-ATP addition measured after 15 mins by beta plate counting analysis, IC50 = 0.009 μM. | 26115571 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK6/cyclin D2 (unknown origin) expressed in baculovirus infected insect cells using GST-fused pRb (792 to 928) as substrate preincubated for 2 mins followed by [gamma-32P]-ATP addition measured after 15 mins by beta plate counting analysis, IC50 = 0.009 μM. | 26115571 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK4/cyclin D1 (unknown origin) expressed in baculovirus infected insect cells using GST-fused pRb (792 to 928) as substrate preincubated for 2 mins followed by [gamma-32P]-ATP addition measured after 15 mins by beta plate counting analysis, IC50 = 0.011 μM. | 26115571 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK4/cyclin D1 (unknown origin) expressed in baculovirus infected insect cells using GST-fused pRb (792 to 928) as substrate preincubated for 2 mins followed by [gamma-32P]-ATP addition measured after 15 mins by beta plate counting analysis, IC50 = 0.011 μM. | 26115571 | ||
| MDA-MB-435 | Antiproliferative assay | 24 hrs | Antiproliferative activity against Rb-positive human MDA-MB-435 cells assessed as inhibition of [14C]-thymidine incorporation into DNA preincubated for 24 hrs followed by [14C]-thymidine addition measured after 72 hrs by beta plate counting analysis, IC50 = 0.16 μM. | 26115571 | ||
| sf9 | Function assay | Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATP, IC50 = 9.2 μM. | 26851505 | |||
| sf9 | Function assay | Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATP, IC50 = 9.2 μM. | 26851505 | |||
| insect cell | Function assay | 2 mins | Inhibition of CDK4/cyclin D1 (unknown origin) expressed in baculovirus infected insect cells using GST-tagged pRB (792 to 928 residues) as substrate preincubated for 2 mins followed by [gamma32P]ATP addition measured after 15 mins by beta counting method, IC50 = 0.011 μM. | 27171036 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK4/cyclin D1 (unknown origin) expressed in baculovirus infected insect cells using GST-tagged pRB (792 to 928 residues) as substrate preincubated for 2 mins followed by [gamma32P]ATP addition measured after 15 mins by beta counting method, IC50 = 0.011 μM. | 27171036 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK6/cyclin D2 (unknown origin) expressed in baculovirus infected insect cells using GST-tagged pRB (792 to 928 residues) as substrate preincubated for 2 mins followed by [gamma32P]ATP addition measured after 15 mins by beta counting method, IC50 = 0.015 μM. | 27171036 | ||
| insect cell | Function assay | 2 mins | Inhibition of CDK6/cyclin D2 (unknown origin) expressed in baculovirus infected insect cells using GST-tagged pRB (792 to 928 residues) as substrate preincubated for 2 mins followed by [gamma32P]ATP addition measured after 15 mins by beta counting method, IC50 = 0.015 μM. | 27171036 | ||
| MDA-MB-453 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-453 cells assessed as reduction in cell viability after 48 hrs by CCK8 assay, IC50 = 0.137 μM. | 27448913 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability after 48 hrs by CCK8 assay, IC50 = 0.24 μM. | 27448913 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as reduction in cell viability after 48 hrs by CCK8 assay, IC50 = 0.58 μM. | 27448913 | ||
| MCF7 | Antitumor assay | 100 mg/kg/day | 14 days | Antitumor activity against human estrogen-independent human MCF7 cells xenografted in RNU rat assessed as tumor growth inhibition at 100 mg/kg/day, ig administered for 14 days measured on day 15 | 27448913 | |
| A2780 | Growth inhibition assay | 72 hrs | Growth inhibition of human A2780 cells after 72 hrs by resazurin or MTT assay, GI50 = 0.032 μM. | 28156111 | ||
| MV4-11 | Growth inhibition assay | 72 hrs | Growth inhibition of human MV4-11 cells after 72 hrs by resazurin assay, GI50 = 0.05 μM. | 28156111 | ||
| MOLM13 | Growth inhibition assay | 72 hrs | Growth inhibition of human MOLM13 cells after 72 hrs by resazurin or MTT assay, GI50 = 0.062 μM. | 28156111 | ||
| NB4 | Growth inhibition assay | 72 hrs | Growth inhibition of human NB4 cells after 72 hrs by resazurin or MTT assay, GI50 = 0.066 μM. | 28156111 | ||
| PC3 | Growth inhibition assay | 72 hrs | Growth inhibition of human PC3 cells after 72 hrs by resazurin or MTT assay, GI50 = 0.071 μM. | 28156111 | ||
| MDA-MB-453 | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-453 cells after 72 hrs by MTT assay, GI50 = 0.326 μM. | 28156111 | ||
| MCF7 | Growth inhibition assay | 72 hrs | Growth inhibition of human MCF7 cells after 72 hrs by resazurin or MTT assay, GI50 = 0.557 μM. | 28156111 | ||
| M229 | Growth inhibition assay | 72 hrs | Growth inhibition of human M229 cells after 72 hrs by resazurin or MTT assay, GI50 = 1.22 μM. | 28156111 | ||
| M238 | Growth inhibition assay | 72 hrs | Growth inhibition of human M238 cells after 72 hrs by resazurin or MTT assay, GI50 = 1.97 μM. | 28156111 | ||
| M249 | Growth inhibition assay | 72 hrs | Growth inhibition of human M249 cells after 72 hrs by resazurin or MTT assay, GI50 = 1.99 μM. | 28156111 | ||
| M249R | Growth inhibition assay | 72 hrs | Growth inhibition of human M249R cells after 72 hrs by resazurin or MTT assay, GI50 = 2.96 μM. | 28156111 | ||
| C4-2B | Growth inhibition assay | 72 hrs | Growth inhibition of human C4-2B cells after 72 hrs by resazurin or MTT assay, GI50 = 4.15 μM. | 28156111 | ||
| DU145 | Growth inhibition assay | 72 hrs | Growth inhibition of human DU145 cells after 72 hrs by resazurin or MTT assay, GI50 = 5.792 μM. | 28156111 | ||
| MDA-MB-468 | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-468 cells after 72 hrs by resazurin or MTT assay, GI50 = 5.96 μM. | 28156111 | ||
| BPH1 | Growth inhibition assay | 72 hrs | Growth inhibition of nontransformed human BPH1 cells after 72 hrs by resazurin or MTT assay, GI50 = 7.442 μM. | 28156111 | ||
| K562 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human K562 cells after 96 hrs by Cell-Titer Blue assay, IC50 = 2 μM. | 24417566 | ||
| DU145 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human DU145 cells after 96 hrs by Cell-Titer Blue assay, IC50 = 7.5 μM. | 24417566 | ||
| MDA-MB-231 | Cell cycle arrest assay | 0.5 to 1 uM | 24 hrs | Cell cycle arrest in human MDA-MB-231 cells assessed as accumulation at G1 phase at 0.5 to 1 uM after 24 hrs by propidium iodide staining-based flow cytometric analysis | 24417566 | |
| MCF7 | Cell cycle arrest assay | 0.5 to 1 uM | 24 hrs | Cell cycle arrest in human MCF7 cells assessed as accumulation at G1 phase at 0.5 to 1 uM after 24 hrs by propidium iodide staining-based flow cytometric analysis | 24417566 | |
| MDA-MB-231 | Function assay | 0.5 to 5 uM | 24 hrs | Inhibition of recombinant CDK4/cyclinD1 in human MDA-MB-231 cells assessed as inhibition of retinoblastoma phosphorylation at Ser780 at 0.5 to 5 uM after 24 hrs by Western blotting analysis | 24417566 | |
| MDA-MB-231 | Function assay | 0.5 to 5 uM | 24 hrs | Inhibition of recombinant CDK4/cyclinD1 in human MDA-MB-231 cells assessed as inhibition of retinoblastoma phosphorylation at Ser780 at 0.5 to 5 uM after 24 hrs by Western blotting analysis | 24417566 | |
| MCF7 | Function assay | 0.5 to 5 uM | 24 hrs | Inhibition of recombinant CDK4/cyclinD1 in human MCF7 cells assessed as inhibition of retinoblastoma phosphorylation at Ser780 at 0.5 to 5 uM after 24 hrs by Western blotting analysis | 24417566 | |
| MCF7 | Function assay | 0.5 to 5 uM | 24 hrs | Inhibition of recombinant CDK4/cyclinD1 in human MCF7 cells assessed as inhibition of retinoblastoma phosphorylation at Ser780 at 0.5 to 5 uM after 24 hrs by Western blotting analysis | 24417566 | |
| MV4-11 | Function assay | 12 to 24 hrs | Inhibition of CDK4/6 in human MV4-11 cells assessed as reduction in Rb phosphorylation at Ser780 at GI50 after 12 to 24 hrs by Western blot analysis | 28156111 | ||
| MV4-11 | Function assay | 12 to 24 hrs | Inhibition of CDK4/6 in human MV4-11 cells assessed as reduction in Rb phosphorylation at Ser780 at GI50 after 12 to 24 hrs by Western blot analysis | 28156111 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Water : 100 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 573.66 | Formula | C24H29N7O2.C2H6O4S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 827022-33-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | PD0332991 Isethionate | Smiles | CC1=C(C(=O)N(C2=NC(=NC=C12)NC3=NC=C(C=C3)N4CCNCC4)C5CCCC5)C(=O)C.C(CS(=O)(=O)O)O | ||
Mechanism of Action
| Features |
The 1st specific inhibitor for CDK4/6 to show promise in multiple cancers.
|
|---|---|
| Targets/IC50/Ki |
CDK4/CyclinD3
(Cell-free assay) 9 nM
CDK4/CyclinD1
(Cell-free assay) 11 nM
CDK6/CyclinD2
(Cell-free assay) 15 nM
|
| In vitro |
PD 0332991 exhibits absolute selectivity for CDK4/6 with little or no activity against other CDKs. PD 0332991 is effective at reducing Rb phosphorylation at Ser780 and Ser795 in MDA-MB-435 breast carcinoma cells with IC50 of 66 nM and 63 nM, respectively. PD 0332991 is a potent inhibitor of cell growth and suppresses DNA replication by preventing cells from entering S phase. PD 0332991 inhibits thymidine incorporation into the DNA of Rb-positive human breast (such as MDA-MB-435, MCF-7), colon (H1299), and lung carcinomas (Colo-205) as well as human leukemias (CRRF-CEM and K562), with IC50 values ranging from 0.04-0.17 μM. PD 0332991 significant increases the percentage of MDA-MB-453 in G1 period. PD 0332991 inhibits phosphorylation of Rb in cycling CD138+ primary bone marrow myeloma cells, nontransformed primary B cells, MM1.S and CAG HMCLs cells line with IC50 of <0.1 μM, 0.05 μM, and 60-70 nM, respectively. PD 0332991 treatment also induces G1 arrest of CD138+ primary bone marrow myeloma and nontransformed primary B cells. PD 0332991 induces G1 arrest in MM1.S with IC50 of ~0.05 μM. PD 0332991 preferentially inhibits proliferation of luminal estrogen receptor-positive (including HER2-positive) human breast cancer cell lines. PD 0332991 increases gene expression of pRb and cyclin D1 and decreases gene expression of CDKN2A (p16) in most sensitive lines. PD 0332991 enhances sensitivity to tamoxifen in cell lines with conditioned resistance to ER blockade.
|
| Kinase Assay |
CDK activity assays
|
|
CDK assays for IC50 determinations and kinetic evaluation are performed in 96-well filter plates. All CDK-cyclin kinase complexes are expressed in insect cells through baculovirus infection and purified. The substrate for the assays is a fragment (amino acids 792–928) of pRb fused to GST (GST•RB-Cterm). The total reaction volume is 0.1 mL containing a final concentration of 20 mM Tris-HCl, pH 7.4, 50 mM NaCl, 1 mM dithiothreitol, 10 mM MgCl2 25 μM ATP (for CDK4-cyclin D1, CDK6-cyclin D2, and CDK6-cyclin D3) containing 0.25 μCi of [γ-32P]ATP, 20 ng of enzyme, 1 μg of GST•RB-Cterm, and appropriate dilutions of inhibitor. All components except the [γ-32P]ATP are added to the wells, and placed on a plate mixer for 2 min. The reaction is started by adding the [γ-32P]ATP, and incubated at 25℃ for 15 min. The reaction is terminated by addition of 0.1 mL of 20% trichloroacetic acid, and the plate is kept at 4 ℃ for at least 1 hr to allow the substrate to precipitate. The wells are then washed five times with 0.2 mL of 10% trichloroacetic acid, and radioactive incorporation is determined with a β plate counter.
|
|
| In vivo |
PD 0332991(150 mg/kg) produces rapid Colo-205 colon carcinoma xenografts regressions and a corresponding tumor growth delay. PD 0332991 (150 mg/kg) induces complete tumor stasis and cell kill in MDA-MB-435 breast carcinoma. PD 0332991 (150 mg/kg) also induces significant tumor regression in mice bearing the SF-295 glioblastoma xenografts, and in ZR-75-1 breast and PC-3 prostate tumor models (complete suppression of tumor growth). PD 0332991 (150 mg/kg) suppresses Rb Ser780 phosphorylation in MDA-MB-435 breast carcinoma over the full 24-hour period. PD 0332991 (150 mg/kg) down-regulates expression of four E2F-regulated genes CDC2, CCNE2, TK1, and TOP2A in Colo-205 carcinoma xenografts.
PD 0332991 also rapidly inhibits myeloma tumor growth.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
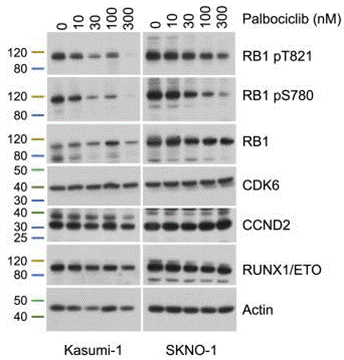
| Western blot | RB1 (pT821) / RB1 (pS780) pAMPKα(T172) / AMPKα pULK1(S317) / ULK1 c-Jun / p-c-Jun / NFκB(p50) p38 / p-P38 / JNK / p-JNK |
|
30300583 |
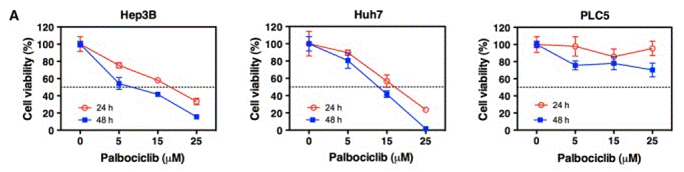
| Growth inhibition assay | Cell viability IC50 |
|
28453226 |
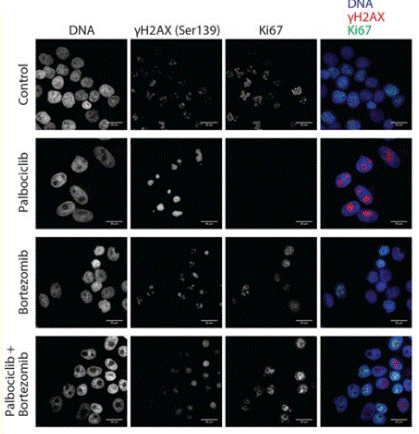
| Immunofluorescence | γH2AX(Ser139) LC3B |
|
29669860 |
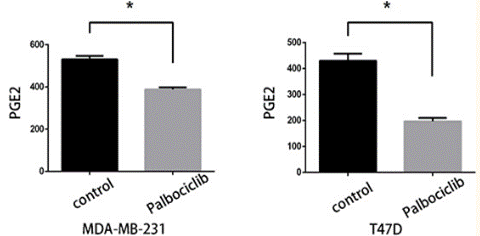
| ELISA | PGE2 |
|
26540629 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05665361 | Recruiting | Advanced Clear Cell Renal Carcinoma (Ccrcc)|Papillary Renal Cell Carcinoma (Prcc) |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
April 24 2024 | Phase 1|Phase 2 |
| NCT06307457 | Completed | Breast Cancer |
Pfizer |
March 11 2024 | -- |
| NCT05709158 | Not yet recruiting | Breast Cancer |
Pfizer |
March 1 2024 | -- |
| NCT05912933 | Not yet recruiting | Breast Cancer |
Pfizer |
March 1 2024 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What is the best vehicle for it for in vitro and in vivo?
Answer:
It can be used with water for vitro use and saline for vivo use.






































