- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
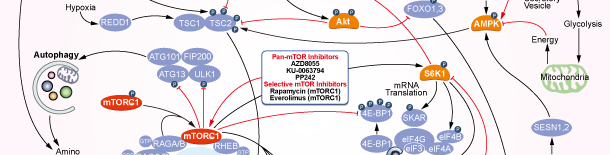
mTOR
mTOR Products
- All (64)
- mTOR Inhibitors (56)
- mTOR Activators (4)
- mTOR Modulators (3)
- New mTOR Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
| S1039 | Rapamycin (Sirolimus) | Rapamycin (Sirolimus) is a specific mTOR inhibitor with IC50 of ~0.1 nM HEK293 cells.Rapamycin binds to FKBP12 and specifically acts as an allosteric inhibitor of mTORC1. Rapamycin is an autophagy activator and an immunosuppressant. |
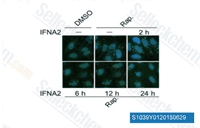
|
|
| S1120 | Everolimus | Everolimus is an mTOR inhibitor of FKBP12 with IC50 of 1.6-2.4 nM in a cell-free assay. Everolimus induces cell apoptosis and autophagy and inhibits tumor cells proliferation. |

|
|
| S1009 | Dactolisib (BEZ235) | Dactolisib (BEZ235, NVP-BEZ235) is a dual ATP-competitive PI3K and mTOR inhibitor for p110α/γ/δ/β and mTOR(p70S6K) with IC50 of 4 nM /5 nM /7 nM /75 nM /6 nM in cell-free assays, respectively. Inhibits ATR with IC50 of 21 nM in 3T3TopBP1-ER cell. Dactolisib induces autophagy and suppresses HIV-1 replication. Phase 2. |
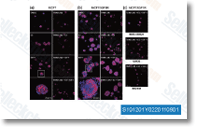
|
|
| S1555 | AZD8055 | AZD8055 is a novel ATP-competitive mTOR inhibitor with IC50 of 0.8 nM in MDA-MB-468 cells with excellent selectivity (∼1,000-fold) against PI3K isoforms and ATM/DNA-PK. AZD8055 induces caspase-dependent apoptosis and also induces autophagy. Phase 1. |
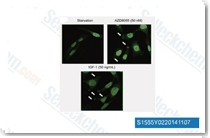
|
|
| S2638 | NU7441 (KU-57788) | NU7441 (KU-57788) is a highly potent and selective DNA-PK inhibitor with IC50 of 14 nM and also inhibits mTOR and PI3K with IC50 of 1.7 μM and 5 μM in cell-free assays, respectively. It reduces the frequency of NHEJ while increasing the rate of HDR following Cas9-mediated DNA cleavage. |

|
|
| S2827 | Torin 1 | Torin 1 is a potent inhibitor of mTORC1/2 with IC50 of 2 nM/10 nM in cell-free assays; exhibits 1000-fold selectivity for mTOR than PI3K. |
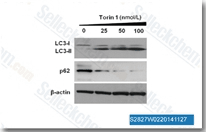
|
|
| S1022 | Ridaforolimus (Deforolimus, MK-8669) | Ridaforolimus (Deforolimus, MK-8669, AP23573) is a selective mTOR inhibitor with IC50 of 0.2 nM in HT-1080 cell line; while not classified as a prodrug, mTOR inhibition and FKBP12 binding is similar to rapamycin. Phase 3. |
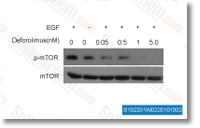
|
|
| S2811 | Sapanisertib (MLN0128) | Sapanisertib (MLN0128, INK 128, TAK-228) is a potent and selective mTOR inhibitor with IC50 of 1 nM in cell-free assays; >200-fold less potent to class I PI3K isoforms, superior in blocking mTORC1/2 and sensitive to pro-invasion genes (vs Rapamycin). Phase 1. |
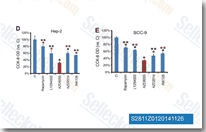
|
|
| S1038 | PI-103 | PI-103 is a multi-targeted PI3K inhibitor for p110α/β/δ/γ with IC50 of 2 nM/3 nM/3 nM/15 nM in cell-free assays, less potent to mTOR/DNA-PK with IC50 of 30 nM/23 nM. PI-103 induces apoptosis in murine T-cell Lymphoma. |

|
|
| S1044 | Temsirolimus | Temsirolimus is a specific mTOR inhibitor with IC50 of 1.76 μM in a cell-free assay. Temsirolimus induces autophagy and apoptosis. |
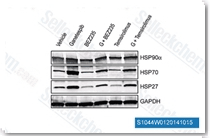
|
|
| S2218 | Torkinib (PP242) | Torkinib (PP242) is a selective mTOR inhibitor with IC50 of 8 nM in cell-free assays; targets both mTOR complexes with >10- and 100-fold selectivity for mTOR than PI3Kδ or PI3Kα/β/γ, respectively. Torkinib (PP242) induces mitophagy and apoptosis. |
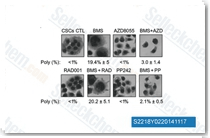
|
|
| S2783 | Vistusertib (AZD2014) | Vistusertib (AZD2014) is a novel mTOR inhibitor with IC50 of 2.8 nM in a cell-free assay; highly selective against multiple PI3K isoforms (α/β/γ/δ). AZD2014 showed no or weak binding to the majority of kinases when tested at 1 μM. AZD2014 induces proliferation suppression, apoptosis, cell cycle arrest, and autophagy in HCC cells with antitumor activity. |
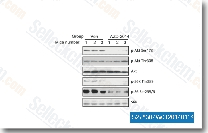
|
|
| S7811 | MHY1485 | MHY1485 is a potent, and cell-permeable mTOR activator, and also potently inhibits autophagy. |

|
|
| S1226 | KU-0063794 | KU-0063794 is a potent and highly specific dual-mTOR inhibitor of mTORC1 and mTORC2 with IC50 of ~10 nM in cell-free assays; no effect on PI3Ks. |

|
|
| S2817 | Torin 2 | Torin 2 is a potent and selective mTOR inhibitor with IC50 of 0.25 nM in p53−/− MEFs cell line; 800-fold greater selectivity for mTOR than PI3K and improved pharmacokinetic properties. Inhibition of ATM/ATR/DNA-PK with EC50 of 28 nM/35 nM/118 nM,in PC3 cell lines respectively. Torin 2 decreases cell viability and induces autophagy and apoptosis. |
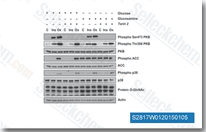
|
|
| S2658 | Omipalisib (GSK2126458) | Omipalisib (GSK2126458, GSK458) is a highly selective and potent inhibitor of p110α/β/δ/γ, mTORC1/2 with Ki of 0.019 nM/0.13 nM/0.024 nM/0.06 nM and 0.18 nM/0.3 nM in cell-free assays, respectively. Omipalisib induces autophagy. Phase 1. |
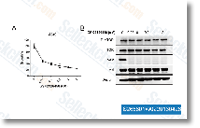
|
|
| S2743 | PF-04691502 | PF-04691502 (PF4691502) is an ATP-competitive PI3K(α/β/δ/γ)/mTOR dual inhibitor with Ki of 1.8 nM/2.1 nM/1.6 nM/1.9 nM and 16 nM in cell-free assays, little activity against either Vps34, AKT, PDK1, p70S6K, MEK, ERK, p38, or JNK. PF-04691502 induces apoptosis. Phase 2. |
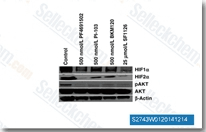
|
|
| S2624 | OSI-027 | OSI-027 (ASP4786, CERC 006, AEVI-006) is a selective and potent dual inhibitor of mTORC1 and mTORC2 with IC50 of 22 nM and 65 nM in cell-free assays, and more than 100-fold selectivity observed for mTOR than PI3Kα, PI3Kβ, PI3Kγ or DNA-PK. OSI-027 induces autophagy in cancer cells. |
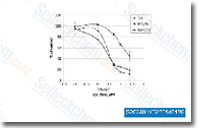
|
|
| S2696 | Apitolisib (GDC-0980) | Apitolisib (GDC-0980, RG7422, GNE 390) is a potent, class I PI3K inhibitor for PI3Kα/β/δ/γ with IC50 of 5 nM/27 nM/7 nM/14 nM in cell-free assays, respectively. Also a mTOR inhibitor with Ki of 17 nM in a cell-free assay, and highly selective versus other PIKK family kinases. Apitolisib activates autophagy and apoptosis simultaneously in pancreatic cancer cells. Phase 2. |
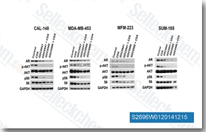
|
|
| S2628 | Gedatolisib (PKI-587) | Gedatolisib (PF-05212384, PKI-587) is a highly potent dual inhibitor of PI3Kα, PI3Kγ and mTOR with IC50 of 0.4 nM, 5.4 nM and 1.6 nM in cell-free assays, respectively. Phase 2. |
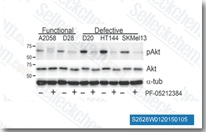
|
|
| S7369 | 4EGI-1 | 4EGI-1 is a competitive eIF4E/eIF4G interaction inhibitor by binding to eIF4E with KD of 25 μM. 4EGI-1 specifically inhibits the function of mTOR by blocking the activation of 4E-BP1. 4EGI-1 induces apoptosis. |
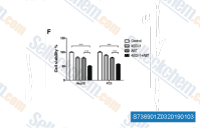
|
|
| S1360 | GSK1059615 | GSK1059615 is a dual inhibitor of PI3Kα/β/δ/γ (reversible) and mTOR with IC50 of 0.4 nM/0.6 nM/2 nM/5 nM and 12 nM, respectively. Phase 1. |

|
|
| S1847 | Clemastine fumarate | Clemastine fumarate is a selective histamine H1 receptor antagonist with IC50 of 3 nM. Clemastine stimulates autophagy via mTOR pathway. |
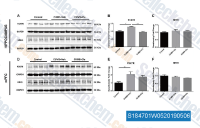
|
|
| S1266 | WYE-354 | WYE-354 is a potent, specific and ATP-competitive inhibitor of mTOR with IC50 of 5 nM, blocks mTORC1/P-S6K(T389) and mTORC2/P-AKT(S473) not P-AKT(T308), selective for mTOR than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |

|
|
| S7646 | Voxtalisib (XL765) | Voxtalisib (SAR245409, XL765) is a dual inhibitor of mTOR/PI3K, mostly for p110γ with IC50 of 9 nM; also inhibits DNA-PK and mTOR. Phase 1/2. |
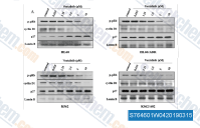
|
|
| S1627 | Nitazoxanide | Nitazoxanide is a synthetic nitrothiazolyl-salicylamide derivative and an antiprotozoal agent(IC50 for canine influenza virus ranges from 0.17 to 0.21 μM). Nitazoxanide modulates autophagy and inhibits mTORC1 signaling. | ||
| S7891 | CC-115 | CC-115 is a dual inhibitor of DNA-dependent protein kinase (DNA-PK) and mammalian target of rapamycin (mTOR) with IC50 values of 0.013 μM and 0.021 μM, respectively. It has potential antineoplastic activity. | ||
| S2661 | WYE-125132 (WYE-132) | WYE-125132 (WYE-132) is a highly potent, ATP-competitive mTOR inhibitor with IC50 of 0.19 nM; highly selective for mTOR versus PI3Ks or PI3K-related kinases hSMG1 and ATR. |
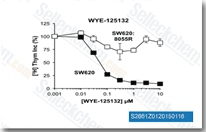
|
|
| S2749 | BGT226 (NVP-BGT226) maleate | BGT226 (NVP-BGT226) maleate is a novel class I PI3K/mTOR inhibitor for PI3Kα/β/γ with IC50 of 4 nM/63 nM/38 nM. Phase 1/2. |
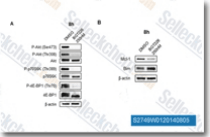
|
|
| S8317 | 3BDO | 3BDO, a butyrolactone derivative, could target FKBP1A and activate the mTOR signaling pathway. It inhibits autophagy in HUVECs. 3BDO inhibits oxLDL-induced apoptosis. | ||
| S2238 | Palomid 529 (P529) | Palomid 529 (P529, SG 00529) inhibits both the mTORC1 and mTORC2 complexes, reduces phosphorylation of pAktS473, pGSK3βS9, and pS6. Phase 1. |
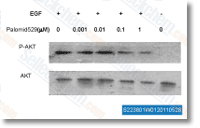
|
|
| S8322 | Samotolisib (LY3023414) | Samotolisib (LY3023414, GTPL8918) is an oral ATP competitive inhibitor of the class I PI3K isoforms, mTOR and DNA-PK. |
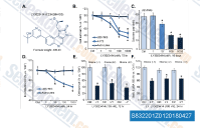
|
|
| S1523 | Voxtalisib (XL765) Analogue | Voxtalisib (SAR245409, XL765) Analogue is a dual inhibitor of mTOR/PI3K, mostly for p110γ with IC50 of 9 nM; also inhibits DNA-PK and mTOR. Phase 1/2. |
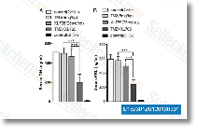
|
|
| S8050 | ETP-46464 | ETP-46464 is a potent and selective inhibitor of ATR with IC50 of 25 nM. | ||
| S2622 | PP121 | PP-121 is a multi-targeted inhibitor of PDGFR, Hck, mTOR, VEGFR2, Src and Abl with IC50 of 2 nM, 8 nM, 10 nM, 12 nM, 14 nM and 18 nM, also inhibits DNA-PK with IC50 of 60 nM. |
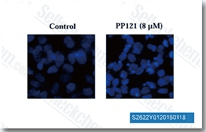
|
|
| S3901 | Astragaloside IV | Astragaloside IV (AST-IV, AS-IV) is a bioactive saponin first isolated from the dried plant roots of the genus Astragalus, which is used in traditional Chinese medicine. It has various effect on the cardiovascular, immune, digestive, and nervous systems. AS-IV suppresses activation of p-Akt, p-mTOR, p-NF-κB and p-Erk1/2. | ||
| S7091 | Zotarolimus (ABT-578) | Zotarolimus (ABT-578,A 179578) is an analogue of rapamycin, and inhibits FKBP-12 binding with IC50 of 2.8 nM. | ||
| S8738 | Bimiralisib (PQR309) | Bimiralisib (PQR309) is a novel brain-penetrant dual PI3K/mTOR inhibitor with in vitro and in vivo antilymphoma activity. It displays excellent selectivity versus PI3K-related lipid kinases, protein kinases and unrelated targets. | ||
| S2396 | Salidroside | Salidroside (Rhodioloside), a phenylpropanoid glycoside isolated from Rhodiola rosea, has been reported to have a broad spectrum of pharmacological properties. Salidroside is a prolyl endopeptidase inhibitor. Salidroside alleviates cachexia symptoms in mouse models of cancer cachexia via activating mTOR signalling. Salidroside protects dopaminergic neurons by enhancing PINK1/Parkin-mediated mitophagy. | ||
| S2689 | WAY-600 | WAY-600 is a potent, ATP-competitive and selective inhibitor of mTOR with IC50 of 9 nM; blocks mTORC1/P-S6K(T389) and mTORC2/P-AKT(S473) but not P-AKT(T308); selective for mTOR than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |
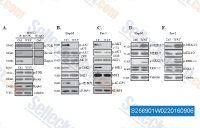
|
|
| S8040 | GDC-0349 | GDC-0349 (RG-7603) is a potent and selective ATP-competitive inhibitor of mTOR with Ki of 3.8 nM, 790-fold inhibitory effect against PI3Kα and other 266 kinases. Phase 1. |
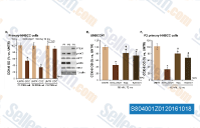
|
|
| S8163 | Paxalisib (GDC-0084) | Paxalisib (GDC-0084, RG7666) is a brain penetrant inhibitor of PI3K and mTOR with Kiapp values of 2 nM, 46 nM, 3 nM, 10 nM and 70 nM for PI3Kα, PI3Kβ, PI3Kδ, PI3Kγ and mTOR. | ||
| S8589 | SF2523 | SF2523 is a highly selective and potent inhibitor of PI3K with IC50 values of 34 nM, 158 nM, 9 nM, 241 nM and 280 nM for PI3Kα, PI3Kγ, DNA-PK, BRD4 and mTOR, respectively. | ||
| S2406 | Chrysophanic Acid | Chrysophanic Acid (Chrysophanol) a natural anthraquinone isolated from Dianella longifolia, is a EGFR/mTOR pathway inhibitor. |
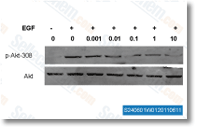
|
|
| S7886 | Onatasertib (CC 223) | Onatasertib (CC 223) is a potent, selective, and orally bioavailable mTOR inhibitor with IC50 of 16 nM, >200-fold selectivity over the related PI3K-α. Phase 1/2. |
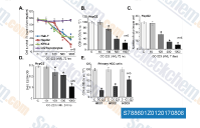
|
|
| S2668 | WYE-687 | WYE-687 is an ATP-competitive and selective inhibitor of mTOR with IC50 of 7 nM; blocks mTORC1/pS6K(T389) and mTORC2/P-AKT(S473) but no effect observed on P-AKT(T308). Selectivity for mTOR is greater than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |
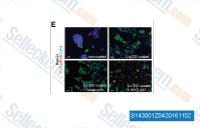
|
|
| S7035 | XL388 | XL388 is a highly potent, selective, ATP-competitive inhibitor of mTOR with IC50 of 9.9 nM, 1000-fold selectivity over the closely related PI3K kinases. | ||
| S6516 | GNE-477 | GNE-477 is a potent and efficacious dual PI3K/mTOR inhibitor with an IC50 of 4 nM for PI3Kα and Kiapp of 21 nM for mTOR. | ||
| S6517 | GNE-493 | GNE-493 (compound 5) is a potent, selective and orally available dual inhibitor of pan-PI3-kinase and mTOR with IC50 of 3.4 nM, 12 nM, 16 nM, 16 nM and 32 nM for PI3Kα, PI3Kβ, PI3Kδ, PI3Kγ and mTOR, respectively. | ||
| E1151 | JR-AB2-011 | JR-AB2-011 is a potent inhibitor of mTOR-mLST8-mSIN1-Rictor complex (mTORC2) which can block mTORC2 signaling and Rictor association with mTOR at lower effective concentrations. | ||
| S9514 | Rotundic acid | Rotundic acid (Rutundic acid), a natural compound, exhibit cytotoxic activities toward human hepatocellular carcinoma (HepG2), malignant melanoma (A375), SCLC (NCI-H446), breast cancer (MCF-7), and colon cancer (HT-29) cell lines.RA induces cell cycle arrest, DNA damage, and apoptosis by modulating the AKT/mTOR and MAPK pathways. | ||
| S8267New | RapaLink-1 | RapaLink-1, a third-generation bivalent inhibitor of mTOR, combines Rapamycin with MLN0128 using an inert chemical linker. It demonstrates efficacy against resistant mutants of malignant brain tumors and inhibits cancer cell progression by binding to mesenchymal markers. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| S6506 | Compound 401 | Compound 401 is a synthetic inhibitor of DNA-PK(IC50=0.28 μM) and mTOR (IC50=5.3 μM). It has no inhibition on p110α/p85α PI3K (>100 μM) and blocks the phosphorylation of S6 kinase 1 Thr389 and Akt Ser473 in COS7 cells. | ||
| E2375 | PI3K/mTOR Inhibitor-2 | PI3K/mTOR Inhibitor-2 is a potent dual pan-PI3K/mTOR inhibitor with antitumor activity. | ||
| E2012 | Ginkgolide K | Ginkgolide K, isolated from Ginkgo biloba, induces protective autophagy through the AMPK/mTOR/ULK1 signaling pathway, and possesses neuroprotective activity. | ||
| S8298 | CZ415 | CZ415, a potent ATP-competitive mTOR inhibitor with very good cell permeability. | ||
| E0795 | COH-SR4 | COH-SR4 suppresses adipogenesis in 3T3-L1 cells through indirect activation of AMPK and downstream modulation of the Mammalian target of rapamycin complex 1 (mTORC1) signaling pathway, which blocks important regulators involved in protein synthesis, cell cycle progression, and expression of key transcription factors and their target adipogenic genes involved in lipid synthesis. | ||
| S3753 | L-Leucine | Leucine ((S)-Leucine, Leu) is one of nine essential amino acids in humans which is important for protein synthesis and many metabolic functions. It contributes to regulation of blood-sugar levels; growth and repair of muscle and bone tissue; growth hormone production; and wound healing. L-Leucine is an essential branched-chain amino acid (BCAA), which activates the mTOR signaling pathway. | ||
| E0115 | MTI-31 | MTI-31 (LXI-15029) is a novel mTORC1/mTORC2 inhibitor with Kd of 0.2 nM in mTOR binding assays and displays >5,000-fold selectivity over PIK3CA, PIK3CB and PIK3G. | ||
| S0179 | mTOR inhibitor-1 | mTOR inhibitor-1 is a novel mTOR pathway inhibitor which can suppress cells proliferation and inducing autophagy. | ||
| S9611 | ABTL-0812 | ABTL0812 (α-Hydroxylinoleic acid, LP-10218, SCLN-0812) inhibits Akt/mTOR axis by inducing the overexpression of TRIB3 and activating autophagy in lung squamous carcinoma cell lines. ABTL0812 also induces AMPK activation and ROS accumulation. | ||
| S8784 | PQR620 | PQR620 is a novel, selective, orally bioavailable and brain penetrant dual TORC1/2 inhibitor. PQR620 has anti-tumor activity across 56 lymphoma models with a median IC50 of 250 nM after 72 h of exposure. | ||
| S7614 | MHY-1685 | MHY-1685 is a novel mTOR inhibitor, which attenuates senescence by modulating autophagy through the inhibition of mTOR in human cardiac stem cells. | ||
| S1039 | Rapamycin (Sirolimus) | Rapamycin (Sirolimus) is a specific mTOR inhibitor with IC50 of ~0.1 nM HEK293 cells.Rapamycin binds to FKBP12 and specifically acts as an allosteric inhibitor of mTORC1. Rapamycin is an autophagy activator and an immunosuppressant. |

|
|
| S1120 | Everolimus | Everolimus is an mTOR inhibitor of FKBP12 with IC50 of 1.6-2.4 nM in a cell-free assay. Everolimus induces cell apoptosis and autophagy and inhibits tumor cells proliferation. |

|
|
| S1009 | Dactolisib (BEZ235) | Dactolisib (BEZ235, NVP-BEZ235) is a dual ATP-competitive PI3K and mTOR inhibitor for p110α/γ/δ/β and mTOR(p70S6K) with IC50 of 4 nM /5 nM /7 nM /75 nM /6 nM in cell-free assays, respectively. Inhibits ATR with IC50 of 21 nM in 3T3TopBP1-ER cell. Dactolisib induces autophagy and suppresses HIV-1 replication. Phase 2. |

|
|
| S1555 | AZD8055 | AZD8055 is a novel ATP-competitive mTOR inhibitor with IC50 of 0.8 nM in MDA-MB-468 cells with excellent selectivity (∼1,000-fold) against PI3K isoforms and ATM/DNA-PK. AZD8055 induces caspase-dependent apoptosis and also induces autophagy. Phase 1. |

|
|
| S2638 | NU7441 (KU-57788) | NU7441 (KU-57788) is a highly potent and selective DNA-PK inhibitor with IC50 of 14 nM and also inhibits mTOR and PI3K with IC50 of 1.7 μM and 5 μM in cell-free assays, respectively. It reduces the frequency of NHEJ while increasing the rate of HDR following Cas9-mediated DNA cleavage. |

|
|
| S2827 | Torin 1 | Torin 1 is a potent inhibitor of mTORC1/2 with IC50 of 2 nM/10 nM in cell-free assays; exhibits 1000-fold selectivity for mTOR than PI3K. |

|
|
| S1022 | Ridaforolimus (Deforolimus, MK-8669) | Ridaforolimus (Deforolimus, MK-8669, AP23573) is a selective mTOR inhibitor with IC50 of 0.2 nM in HT-1080 cell line; while not classified as a prodrug, mTOR inhibition and FKBP12 binding is similar to rapamycin. Phase 3. |

|
|
| S2811 | Sapanisertib (MLN0128) | Sapanisertib (MLN0128, INK 128, TAK-228) is a potent and selective mTOR inhibitor with IC50 of 1 nM in cell-free assays; >200-fold less potent to class I PI3K isoforms, superior in blocking mTORC1/2 and sensitive to pro-invasion genes (vs Rapamycin). Phase 1. |

|
|
| S1038 | PI-103 | PI-103 is a multi-targeted PI3K inhibitor for p110α/β/δ/γ with IC50 of 2 nM/3 nM/3 nM/15 nM in cell-free assays, less potent to mTOR/DNA-PK with IC50 of 30 nM/23 nM. PI-103 induces apoptosis in murine T-cell Lymphoma. |

|
|
| S1044 | Temsirolimus | Temsirolimus is a specific mTOR inhibitor with IC50 of 1.76 μM in a cell-free assay. Temsirolimus induces autophagy and apoptosis. |

|
|
| S2218 | Torkinib (PP242) | Torkinib (PP242) is a selective mTOR inhibitor with IC50 of 8 nM in cell-free assays; targets both mTOR complexes with >10- and 100-fold selectivity for mTOR than PI3Kδ or PI3Kα/β/γ, respectively. Torkinib (PP242) induces mitophagy and apoptosis. |

|
|
| S2783 | Vistusertib (AZD2014) | Vistusertib (AZD2014) is a novel mTOR inhibitor with IC50 of 2.8 nM in a cell-free assay; highly selective against multiple PI3K isoforms (α/β/γ/δ). AZD2014 showed no or weak binding to the majority of kinases when tested at 1 μM. AZD2014 induces proliferation suppression, apoptosis, cell cycle arrest, and autophagy in HCC cells with antitumor activity. |

|
|
| S1226 | KU-0063794 | KU-0063794 is a potent and highly specific dual-mTOR inhibitor of mTORC1 and mTORC2 with IC50 of ~10 nM in cell-free assays; no effect on PI3Ks. |

|
|
| S2817 | Torin 2 | Torin 2 is a potent and selective mTOR inhibitor with IC50 of 0.25 nM in p53−/− MEFs cell line; 800-fold greater selectivity for mTOR than PI3K and improved pharmacokinetic properties. Inhibition of ATM/ATR/DNA-PK with EC50 of 28 nM/35 nM/118 nM,in PC3 cell lines respectively. Torin 2 decreases cell viability and induces autophagy and apoptosis. |

|
|
| S2658 | Omipalisib (GSK2126458) | Omipalisib (GSK2126458, GSK458) is a highly selective and potent inhibitor of p110α/β/δ/γ, mTORC1/2 with Ki of 0.019 nM/0.13 nM/0.024 nM/0.06 nM and 0.18 nM/0.3 nM in cell-free assays, respectively. Omipalisib induces autophagy. Phase 1. |

|
|
| S2743 | PF-04691502 | PF-04691502 (PF4691502) is an ATP-competitive PI3K(α/β/δ/γ)/mTOR dual inhibitor with Ki of 1.8 nM/2.1 nM/1.6 nM/1.9 nM and 16 nM in cell-free assays, little activity against either Vps34, AKT, PDK1, p70S6K, MEK, ERK, p38, or JNK. PF-04691502 induces apoptosis. Phase 2. |

|
|
| S2624 | OSI-027 | OSI-027 (ASP4786, CERC 006, AEVI-006) is a selective and potent dual inhibitor of mTORC1 and mTORC2 with IC50 of 22 nM and 65 nM in cell-free assays, and more than 100-fold selectivity observed for mTOR than PI3Kα, PI3Kβ, PI3Kγ or DNA-PK. OSI-027 induces autophagy in cancer cells. |

|
|
| S2696 | Apitolisib (GDC-0980) | Apitolisib (GDC-0980, RG7422, GNE 390) is a potent, class I PI3K inhibitor for PI3Kα/β/δ/γ with IC50 of 5 nM/27 nM/7 nM/14 nM in cell-free assays, respectively. Also a mTOR inhibitor with Ki of 17 nM in a cell-free assay, and highly selective versus other PIKK family kinases. Apitolisib activates autophagy and apoptosis simultaneously in pancreatic cancer cells. Phase 2. |

|
|
| S2628 | Gedatolisib (PKI-587) | Gedatolisib (PF-05212384, PKI-587) is a highly potent dual inhibitor of PI3Kα, PI3Kγ and mTOR with IC50 of 0.4 nM, 5.4 nM and 1.6 nM in cell-free assays, respectively. Phase 2. |

|
|
| S7369 | 4EGI-1 | 4EGI-1 is a competitive eIF4E/eIF4G interaction inhibitor by binding to eIF4E with KD of 25 μM. 4EGI-1 specifically inhibits the function of mTOR by blocking the activation of 4E-BP1. 4EGI-1 induces apoptosis. |

|
|
| S1360 | GSK1059615 | GSK1059615 is a dual inhibitor of PI3Kα/β/δ/γ (reversible) and mTOR with IC50 of 0.4 nM/0.6 nM/2 nM/5 nM and 12 nM, respectively. Phase 1. |

|
|
| S1266 | WYE-354 | WYE-354 is a potent, specific and ATP-competitive inhibitor of mTOR with IC50 of 5 nM, blocks mTORC1/P-S6K(T389) and mTORC2/P-AKT(S473) not P-AKT(T308), selective for mTOR than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |

|
|
| S7646 | Voxtalisib (XL765) | Voxtalisib (SAR245409, XL765) is a dual inhibitor of mTOR/PI3K, mostly for p110γ with IC50 of 9 nM; also inhibits DNA-PK and mTOR. Phase 1/2. |

|
|
| S1627 | Nitazoxanide | Nitazoxanide is a synthetic nitrothiazolyl-salicylamide derivative and an antiprotozoal agent(IC50 for canine influenza virus ranges from 0.17 to 0.21 μM). Nitazoxanide modulates autophagy and inhibits mTORC1 signaling. | ||
| S7891 | CC-115 | CC-115 is a dual inhibitor of DNA-dependent protein kinase (DNA-PK) and mammalian target of rapamycin (mTOR) with IC50 values of 0.013 μM and 0.021 μM, respectively. It has potential antineoplastic activity. | ||
| S2661 | WYE-125132 (WYE-132) | WYE-125132 (WYE-132) is a highly potent, ATP-competitive mTOR inhibitor with IC50 of 0.19 nM; highly selective for mTOR versus PI3Ks or PI3K-related kinases hSMG1 and ATR. |

|
|
| S2749 | BGT226 (NVP-BGT226) maleate | BGT226 (NVP-BGT226) maleate is a novel class I PI3K/mTOR inhibitor for PI3Kα/β/γ with IC50 of 4 nM/63 nM/38 nM. Phase 1/2. |

|
|
| S2238 | Palomid 529 (P529) | Palomid 529 (P529, SG 00529) inhibits both the mTORC1 and mTORC2 complexes, reduces phosphorylation of pAktS473, pGSK3βS9, and pS6. Phase 1. |

|
|
| S8322 | Samotolisib (LY3023414) | Samotolisib (LY3023414, GTPL8918) is an oral ATP competitive inhibitor of the class I PI3K isoforms, mTOR and DNA-PK. |

|
|
| S1523 | Voxtalisib (XL765) Analogue | Voxtalisib (SAR245409, XL765) Analogue is a dual inhibitor of mTOR/PI3K, mostly for p110γ with IC50 of 9 nM; also inhibits DNA-PK and mTOR. Phase 1/2. |

|
|
| S8050 | ETP-46464 | ETP-46464 is a potent and selective inhibitor of ATR with IC50 of 25 nM. | ||
| S2622 | PP121 | PP-121 is a multi-targeted inhibitor of PDGFR, Hck, mTOR, VEGFR2, Src and Abl with IC50 of 2 nM, 8 nM, 10 nM, 12 nM, 14 nM and 18 nM, also inhibits DNA-PK with IC50 of 60 nM. |

|
|
| S3901 | Astragaloside IV | Astragaloside IV (AST-IV, AS-IV) is a bioactive saponin first isolated from the dried plant roots of the genus Astragalus, which is used in traditional Chinese medicine. It has various effect on the cardiovascular, immune, digestive, and nervous systems. AS-IV suppresses activation of p-Akt, p-mTOR, p-NF-κB and p-Erk1/2. | ||
| S7091 | Zotarolimus (ABT-578) | Zotarolimus (ABT-578,A 179578) is an analogue of rapamycin, and inhibits FKBP-12 binding with IC50 of 2.8 nM. | ||
| S8738 | Bimiralisib (PQR309) | Bimiralisib (PQR309) is a novel brain-penetrant dual PI3K/mTOR inhibitor with in vitro and in vivo antilymphoma activity. It displays excellent selectivity versus PI3K-related lipid kinases, protein kinases and unrelated targets. | ||
| S2689 | WAY-600 | WAY-600 is a potent, ATP-competitive and selective inhibitor of mTOR with IC50 of 9 nM; blocks mTORC1/P-S6K(T389) and mTORC2/P-AKT(S473) but not P-AKT(T308); selective for mTOR than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |

|
|
| S8040 | GDC-0349 | GDC-0349 (RG-7603) is a potent and selective ATP-competitive inhibitor of mTOR with Ki of 3.8 nM, 790-fold inhibitory effect against PI3Kα and other 266 kinases. Phase 1. |

|
|
| S8163 | Paxalisib (GDC-0084) | Paxalisib (GDC-0084, RG7666) is a brain penetrant inhibitor of PI3K and mTOR with Kiapp values of 2 nM, 46 nM, 3 nM, 10 nM and 70 nM for PI3Kα, PI3Kβ, PI3Kδ, PI3Kγ and mTOR. | ||
| S8589 | SF2523 | SF2523 is a highly selective and potent inhibitor of PI3K with IC50 values of 34 nM, 158 nM, 9 nM, 241 nM and 280 nM for PI3Kα, PI3Kγ, DNA-PK, BRD4 and mTOR, respectively. | ||
| S2406 | Chrysophanic Acid | Chrysophanic Acid (Chrysophanol) a natural anthraquinone isolated from Dianella longifolia, is a EGFR/mTOR pathway inhibitor. |

|
|
| S7886 | Onatasertib (CC 223) | Onatasertib (CC 223) is a potent, selective, and orally bioavailable mTOR inhibitor with IC50 of 16 nM, >200-fold selectivity over the related PI3K-α. Phase 1/2. |

|
|
| S2668 | WYE-687 | WYE-687 is an ATP-competitive and selective inhibitor of mTOR with IC50 of 7 nM; blocks mTORC1/pS6K(T389) and mTORC2/P-AKT(S473) but no effect observed on P-AKT(T308). Selectivity for mTOR is greater than PI3Kα (>100-fold) and PI3Kγ (>500-fold). |

|
|
| S7035 | XL388 | XL388 is a highly potent, selective, ATP-competitive inhibitor of mTOR with IC50 of 9.9 nM, 1000-fold selectivity over the closely related PI3K kinases. | ||
| S6516 | GNE-477 | GNE-477 is a potent and efficacious dual PI3K/mTOR inhibitor with an IC50 of 4 nM for PI3Kα and Kiapp of 21 nM for mTOR. | ||
| S6517 | GNE-493 | GNE-493 (compound 5) is a potent, selective and orally available dual inhibitor of pan-PI3-kinase and mTOR with IC50 of 3.4 nM, 12 nM, 16 nM, 16 nM and 32 nM for PI3Kα, PI3Kβ, PI3Kδ, PI3Kγ and mTOR, respectively. | ||
| E1151 | JR-AB2-011 | JR-AB2-011 is a potent inhibitor of mTOR-mLST8-mSIN1-Rictor complex (mTORC2) which can block mTORC2 signaling and Rictor association with mTOR at lower effective concentrations. | ||
| S8267New | RapaLink-1 | RapaLink-1, a third-generation bivalent inhibitor of mTOR, combines Rapamycin with MLN0128 using an inert chemical linker. It demonstrates efficacy against resistant mutants of malignant brain tumors and inhibits cancer cell progression by binding to mesenchymal markers. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| S6506 | Compound 401 | Compound 401 is a synthetic inhibitor of DNA-PK(IC50=0.28 μM) and mTOR (IC50=5.3 μM). It has no inhibition on p110α/p85α PI3K (>100 μM) and blocks the phosphorylation of S6 kinase 1 Thr389 and Akt Ser473 in COS7 cells. | ||
| E2375 | PI3K/mTOR Inhibitor-2 | PI3K/mTOR Inhibitor-2 is a potent dual pan-PI3K/mTOR inhibitor with antitumor activity. | ||
| S8298 | CZ415 | CZ415, a potent ATP-competitive mTOR inhibitor with very good cell permeability. | ||
| E0115 | MTI-31 | MTI-31 (LXI-15029) is a novel mTORC1/mTORC2 inhibitor with Kd of 0.2 nM in mTOR binding assays and displays >5,000-fold selectivity over PIK3CA, PIK3CB and PIK3G. | ||
| S0179 | mTOR inhibitor-1 | mTOR inhibitor-1 is a novel mTOR pathway inhibitor which can suppress cells proliferation and inducing autophagy. | ||
| S9611 | ABTL-0812 | ABTL0812 (α-Hydroxylinoleic acid, LP-10218, SCLN-0812) inhibits Akt/mTOR axis by inducing the overexpression of TRIB3 and activating autophagy in lung squamous carcinoma cell lines. ABTL0812 also induces AMPK activation and ROS accumulation. | ||
| S8784 | PQR620 | PQR620 is a novel, selective, orally bioavailable and brain penetrant dual TORC1/2 inhibitor. PQR620 has anti-tumor activity across 56 lymphoma models with a median IC50 of 250 nM after 72 h of exposure. | ||
| S7614 | MHY-1685 | MHY-1685 is a novel mTOR inhibitor, which attenuates senescence by modulating autophagy through the inhibition of mTOR in human cardiac stem cells. | ||
| S7811 | MHY1485 | MHY1485 is a potent, and cell-permeable mTOR activator, and also potently inhibits autophagy. |

|
|
| S8317 | 3BDO | 3BDO, a butyrolactone derivative, could target FKBP1A and activate the mTOR signaling pathway. It inhibits autophagy in HUVECs. 3BDO inhibits oxLDL-induced apoptosis. | ||
| S2396 | Salidroside | Salidroside (Rhodioloside), a phenylpropanoid glycoside isolated from Rhodiola rosea, has been reported to have a broad spectrum of pharmacological properties. Salidroside is a prolyl endopeptidase inhibitor. Salidroside alleviates cachexia symptoms in mouse models of cancer cachexia via activating mTOR signalling. Salidroside protects dopaminergic neurons by enhancing PINK1/Parkin-mediated mitophagy. | ||
| S3753 | L-Leucine | Leucine ((S)-Leucine, Leu) is one of nine essential amino acids in humans which is important for protein synthesis and many metabolic functions. It contributes to regulation of blood-sugar levels; growth and repair of muscle and bone tissue; growth hormone production; and wound healing. L-Leucine is an essential branched-chain amino acid (BCAA), which activates the mTOR signaling pathway. | ||
| S1847 | Clemastine fumarate | Clemastine fumarate is a selective histamine H1 receptor antagonist with IC50 of 3 nM. Clemastine stimulates autophagy via mTOR pathway. |

|
|
| S9514 | Rotundic acid | Rotundic acid (Rutundic acid), a natural compound, exhibit cytotoxic activities toward human hepatocellular carcinoma (HepG2), malignant melanoma (A375), SCLC (NCI-H446), breast cancer (MCF-7), and colon cancer (HT-29) cell lines.RA induces cell cycle arrest, DNA damage, and apoptosis by modulating the AKT/mTOR and MAPK pathways. | ||
| E0795 | COH-SR4 | COH-SR4 suppresses adipogenesis in 3T3-L1 cells through indirect activation of AMPK and downstream modulation of the Mammalian target of rapamycin complex 1 (mTORC1) signaling pathway, which blocks important regulators involved in protein synthesis, cell cycle progression, and expression of key transcription factors and their target adipogenic genes involved in lipid synthesis. | ||
| S8267New | RapaLink-1 | RapaLink-1, a third-generation bivalent inhibitor of mTOR, combines Rapamycin with MLN0128 using an inert chemical linker. It demonstrates efficacy against resistant mutants of malignant brain tumors and inhibits cancer cell progression by binding to mesenchymal markers. |
Choose Selective mTOR Inhibitors
Tags: mTOR inhibition | mTOR cancer | mTOR activation | mTOR target | mTOR tumor | mTOR phosphorylation | mTOR activity | mTOR inhibitor drugs | mTOR kinase assay | mTOR inhibitor cancer | mTOR inhibitor therapy | mTOR signaling pathway | mTOR inhibitor review