research use only
Nitazoxanide Influenza Virus inhibitor
Cat.No.S1627
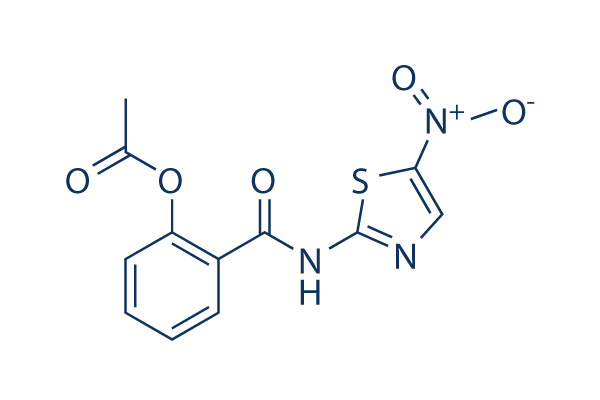
Chemical Structure
Molecular Weight: 307.28
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV |
|---|---|
| Other Influenza Virus Inhibitors | Arbidol HCl Nucleozin Cetylpyridinium chloride monohydrate (-)-Arctigenin Chebulinic acid Adamantane D-Pinitol |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HepG2(2.2.15) | Antiviral assay | Antiviral activity against Hepatitis B virus infected in human HepG2(2.2.15) cells assessed as decrease in extracellular viral DNA measured 24 hrs after last dose, EC50=0.12μM | 21553812 | |||
| HepG2(2.2.15) | Antiviral assay | Antiviral activity against HBV infected in human HepG2(2.2.15) cells assessed as inhibition of extracellular viral DNA level, IC50=0.12μM | 28383274 | |||
| Ava5 | Antiviral assay | 3 days | Antiviral activity against HCV genotype 1b infected in Ava5 cells assessed as inhibition of viral replication after 3 days by blot hybridization analysis, EC50=0.21μM | 22059983 | ||
| Huh7.5 | Antiviral assay | 3 days | Antiviral activity against HCV genotype 1a infected in Huh7.5 cells assessed as inhibition of viral replication after 3 days by blot hybridization analysis, EC50=0.33μM | 22059983 | ||
| HepG2(2.2.15) | Antiviral assay | Antiviral activity against Hepatitis B virus infected in human HepG2(2.2.15) cells assessed as decrease in intracellular viral DNA measured 24 hrs after last dose, EC50=0.59μM | 21553812 | |||
| HepG2(2.2.15) | Antiviral assay | Antiviral activity against Hepatitis B virus infected in human HepG2(2.2.15) cells assessed as decrease in extracellular viral DNA measured 24 hrs after last dose, EC90=0.83μM | 21553812 | |||
| Ava5 | Antiviral assay | 3 days | Antiviral activity against HCV genotype 1b infected in Ava5 cells assessed as inhibition of viral replication after 3 days by blot hybridization analysis, EC90=0.93μM | 22059983 | ||
| Huh7.5 | Antiviral assay | 3 days | Antiviral activity against HCV genotype 1a infected in Huh7.5 cells assessed as inhibition of viral replication after 3 days by blot hybridization analysis, EC90=1.1μM | 22059983 | ||
| BHK-21 | Antiviral assay | Antiviral activity against Chikungunya virus 0611aTw infected in BHK-21 cells by RT-qPCR analysis, EC50=1.96μM | 28689975 | |||
| HepG2(2.2.15) | Antiviral assay | Antiviral activity against Hepatitis B virus infected in human HepG2(2.2.15) cells assessed as decrease in intracellular viral DNA measured 24 hrs after last dose, EC90=2.1μM | 21553812 | |||
| HCT8 | Antiparasitic assay | 48 hrs | Antiparasitic activity against Cryptosporidium parvum SPL infected in human HCT8 cells incubated for 48 hrs by FITC/DAPI staining based fluorescence assay, EC50=2.3μM | 29469575 | ||
| Vero E6 | Function assay | 48 hrs | IC50 for antiviral activity against SARS-CoV-2 in the Vero E6 cell line at 48 h by immunofluorescence-based assay (detecting the viral NP protein in the nucleus of the Vero E6 cells), IC50=2.81838μM | 32353859 | ||
| HCT8 | Antiparasitic assay | 48 hrs | Antiparasitic activity against Cryptosporidium parvum BGF infected in human HCT8 cells incubated for 48 hrs by FITC/DAPI staining based fluorescence assay, EC50=2.9μM | 29469575 | ||
| BHK-21 | Antiviral assay | Antiviral activity against Chikungunya virus infected in BHK-21 cells by RT-qPCR analysis, EC50=2.96μM | 28689975 | |||
| U2OS | Antiviral assay | Antiviral activity against Chikungunya virus infected in human U2OS cells by RT-qPCR analysis, EC50=3.01μM | 28689975 | |||
| HCT8 | Antiparasitic assay | Antiparasitic activity against Cryptosporidium parvum in HCT8 cells, IC50=3.25μM | 16480281 | |||
| HCT-8 | Antimicrobial assay | Antimicrobial activity against Cryptosporidium parvum infected in human HCT-8 cells, IC50=3.8μM | 18591280 | |||
| BHK-21 | Antiviral assay | Antiviral activity against Chikungunya virus 0810bTw infected in BHK-21 cells by RT-qPCR analysis, EC50=4.95μM | 28689975 | |||
| Vero | Cytotoxicity assay | 48 hrs | Cytotoxicity against african green monkey Vero cells assessed as cell viability after 48 hrs by WST-1 assay, IC50=10.74μM | 23787289 | ||
| BHK21 | Cytotoxicity assay | 16 hrs | Cytotoxicity against BHK21 cells assessed as reduction in cell viability after 16 hrs by CCK-8 assay, CC50=25μM | 28689975 | ||
| U2OS | Cytotoxicity assay | 16 hrs | Cytotoxicity against human U2OS cells assessed as reduction in cell viability after 16 hrs by CCK-8 assay, CC50=25μM | 28689975 | ||
| Ava5 | Cytotoxicity assay | 3 days | Cytotoxicity against human Ava5 cells after 3 days by neutral red dye assay, CC50=35μM | 22059983 | ||
| Huh7.5 | Cytotoxicity assay | 3 days | Cytotoxicity against human Huh7.5 cells after 3 days by neutral red dye assay, CC50=49μM | 22059983 | ||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 62 mg/mL
(201.77 mM)
Ethanol : 62 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 307.28 | Formula | C12H9N3O5S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 55981-09-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | NTZ, NSC 697855 | Smiles | CC(=O)OC1=CC=CC=C1C(=O)NC2=NC=C(S2)[N+](=O)[O-] | ||
Mechanism of Action
| Targets/IC50/Ki |
PFOR
mTORC1
|
|---|---|
| In vitro |
Nitazoxanide reduces parasite growth in cell culture by more than 90% with little evidence of drug-associated cytotoxicity. This compound is a new thiazolide antiparasitic agent that shows excellent in vitro activity against a wide variety of protozoa and helminths. It and its metabolite tizoxanide are more active in vitro than metronidazole against G. intestinalis, E. histolytica and T. vaginalis. This agent exhibits potent inhibition of both HBV and HCV replication. It potentiates the effect of subsequent treatment with this compound plus IFN, but not this chemical plus 2'CmeC, in HCV replicon-containing cells. This chemical induces reductions in several HBV proteins (HBsAg, HBeAg, HBcAg) produced by 2.2.15 cells, but does not affect HBV RNA transcription. It exhibits IC50, and IC90 values of 0.017 and 0.776 mg/mL respectively, against E. histolytica, 0.004 and 0.067 mg/mL against G. intestinalis, and 0.034 and 2.046 mg/mL against T. vaginalis. This compound is more toxic than metronidazole and albendazole against E. histolytica. |
| In vivo |
Nitazoxanide is partially effective at reducing the parasite burden in a gnotobiotic piglet diarrhea model when given orally for 11 days at 250 mg/kg/day but not at 125 mg/kg/day. This compound induces a drug-related diarrhea in piglets that might have influenced its therapeutic efficacy. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06049901 | Recruiting | Metastatic Colorectal Cancer |
Tanta University |
March 1 2023 | Phase 3 |
| NCT05701423 | Recruiting | Multiple Sclerosis |
Biogen |
February 8 2023 | -- |
| NCT05368935 | Completed | Renal Impairment|Renal Disease|Kidney Disease |
Genfit |
April 25 2022 | Phase 1 |
| NCT05116826 | Completed | Moderate Hepatic Impairment|Severe Hepatic Impairment|Liver Diseases |
Genfit |
November 5 2021 | Phase 1 |
| NCT03656068 | Completed | Non-alcoholic Steatohepatitis|Fatty Liver|Fibrosis Liver|Compensated Cirrhosis |
Pinnacle Clinical Research PLLC |
December 4 2018 | Phase 2 |
| NCT02684240 | Completed | Tuberculosis |
Weill Medical College of Cornell University |
February 2016 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































