research use only
CAL-101 (Idelalisib) PI3Kδ inhibitor
Cat.No.S2226
Chemical Structure
Molecular Weight: 415.42
Quality Control
| Related Targets | Akt mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other PI3K Inhibitors | GDC-0077 (Inavolisib) SAR405 Quercetin (Sophoretin) LY294002 XL147 analogue Tersolisib (STX-478) Buparlisib (BKM120) 740 Y-P (PDGFR 740Y-P) GO-203 TFA Eganelisib (IPI-549) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MEC1 | Growth Inhibition Assay | DMSO | IC50=20.4 μM | 25999352 | ||
| CLL PBMCs | Growth Inhibition Assay | DMSO | IC50=2.9 nM | 25917267 | ||
| U266 | Growth Inhibition Assay | 40 μM | 48 h | 79.5% inhibition rate | 25339332 | |
| K562 | Function Assay | 1 μM | 3 h | Inhibition of Akt phosphorylation | 25014775 | |
| K562 | Function Assay | 1 μM | 3 h | Inhibition of P70S6K phosphorylation | 25014775 | |
| K562 | Function Assay | 1 μM | 3 h | Inhibition of GSK3 phosphorylation | 25014775 | |
| K562 | Growth Inhibition Assay | 1 μM | 72 h | Inhibition of proliferation | 25014775 | |
| Primary AML cell | Function Assay | 1 μM | 3 h | Inhibition of Akt phosphorylation | 25014775 | |
| Primary AML cell | Function Assay | 1 μM | 3 h | Inhibition of P70S6K phosphorylation | 25014775 | |
| Primary AML cell | Function Assay | 1 μM | 3 h | Inhibition of GSK3 phosphorylation | 25014775 | |
| Primary AML cell | Growth Inhibition Assay | 1 μM | 3 h | Suppression of rRNA synthesis | 25014775 | |
| Microglia | Function Assay | 5 μM | 10 h | DMSO | Decrease of TNFa secretion from LPS-stimulated p110δD910A/D910A microglia | 24625684 |
| Primary CLL cell | Function Assay | 1 μM | 15 min | DMSO | Blocks BCR-induced LCP1 serine-5 activation | 24009233 |
| JEKO-1 | Function Assay | 1 μM | 72 h | Inhibition of Akt phosphorylation in IgM-stimulated JEKO-1 | 23341541 | |
| Granta-519 | Function Assay | 1 μM | 2 h | Inhibition of Akt(t308) phosphorylation | 23341541 | |
| Granta-519 | Function Assay | 1 μM | 2 h | Inhibition of Akt(s473) phosphorylation | 23341541 | |
| JEKO-1 | Growth Inhibition Assay | 10 μM | 72 h | Inhibition of proliferation slightly | 23341541 | |
| JEKO-1 | Growth Inhibition Assay | 5 μM | 72 h | does not induce cell cycle arrest or apoptosis | 23676220 | |
| MAVER-1 | Growth Inhibition Assay | 5 μM | 72 h | does not induce cell cycle arrest or apoptosis | 23676220 | |
| MINO | Growth Inhibition Assay | 5 μM | 72 h | does not induce cell cycle arrest or apoptosis | 23676220 | |
| SP53 | Growth Inhibition Assay | 0.1 μM | 72 h | does not induce cell cycle arrest or apoptosis | 23676220 | |
| HH | Growth Inhibition Assay | 10 μM | 72 h | DMSO | Induction of apoptosis slightly | 22801959 |
| Myla | Growth Inhibition Assay | 10 μM | 72 h | DMSO | does not induce apoptosis | 22801959 |
| SR786 | Growth Inhibition Assay | 10 μM | 72 h | DMSO | does not induce apoptosis | 22801959 |
| HuT78 | Growth Inhibition Assay | 10 μM | 72 h | DMSO | does not induce apoptosis | 22801959 |
| MJ | Growth Inhibition Assay | 10 μM | 72 h | DMSO | does not induce apoptosis | 22801959 |
| DERL7 | Growth Inhibition Assay | 10 μM | 72 h | DMSO | does not induce apoptosis | 22801959 |
| L1236 | Function Assay | 10 μM | 2 h | Inhibition of Akt phosphorylation | 22210877 | |
| L428 | Function Assay | 10 μM | 2 h | Inhibition of Akt phosphorylation | 22210877 | |
| L591 | Function Assay | 10 μM | 2 h | Inhibition of Akt phosphorylation | 22210877 | |
| KMH-2 | Function Assay | 10 μM | 2 h | Inhibition of Akt phosphorylation | 22210877 | |
| L1236 | Function Assay | 5 μM | 24 h | Blocks secretion of the CCL5 | 22210877 | |
| L591 | Function Assay | 5 μM | 24 h | Blocks secretion of the CCL5 | 22210877 | |
| L1236 | Apoptosis Assay | 5 μM | 24 h | Induction of apoptosis | 22210877 | |
| L591 | Apoptosis Assay | 5 μM | 24 h | Induction of apoptosis | 22210877 | |
| U-87MG | Function Assay | 100 nM | 24 h | DMSO | Inhibition of cell migration | 22079609 |
| SW1783 | Function Assay | 100 nM | 24 h | DMSO | Inhibition of cell migration | 22079609 |
| U-87MG | Function Assay | 5 μM | 24 h | DMSO | Inhibition of Akt phosphorylation substantially | 22079609 |
| SW1783 | Function Assay | 5 μM | 24 h | DMSO | Inhibition of Akt phosphorylation substantially | 22079609 |
| U-373MG | Function Assay | 5 μM | 24 h | DMSO | Inhibition of Akt phosphorylation substantially | 22079609 |
| SK-MG3 | Function Assay | 5 μM | 24 h | DMSO | Inhibition of Akt phosphorylation substantially | 22079609 |
| SU-DHL-5 | Function Assay | 1 μM | 24 h | DMSO | Induction of apoptosis | 20959606 |
| WSU-NHL | Function Assay | 1 μM | 24 h | DMSO | Induction of apoptosis | 20959606 |
| CCRF-SB | Function Assay | 1 μM | 24 h | DMSO | Induction of apoptosis | 20959606 |
| INA-6 | Function Assay | 5 μM | 6 h | Inhibition of PI3K/Akt and ERK pathway | 20505158 | |
| LB | Function Assay | 5 μM | 6 h | Inhibition of PI4K/Akt and ERK pathway | 20505158 | |
| B-cells | Function assay | Inhibition of PI3Kdelta in B-cells by proliferation assay, IC50 = 0.0061 μM. | 22924688 | |||
| MOLM14 | Antiproliferative assay | 3 days | Antiproliferative activity against human MOLM14 cells after 3 days by CellTiter-Glo assay, IC50 = 3.6 μM. | 27774127 | ||
| MV4-11 | Antiproliferative assay | 3 days | Antiproliferative activity against human MV4-11 cells after 3 days by CellTiter-Glo assay, IC50 = 6.3 μM. | 27774127 | ||
| Jurkat | Antiproliferative assay | 3 days | Antiproliferative activity against human Jurkat cells after 3 days by CellTiter-Glo assay, IC50 = 7.9 μM. | 27774127 | ||
| Loucy | Antiproliferative assay | 3 days | Antiproliferative activity against human Loucy cells after 3 days by CellTiter-Glo assay, IC50 = 8.4 μM. | 27774127 | ||
| MOLT4 | Antiproliferative assay | 3 days | Antiproliferative activity against human MOLT4 cells after 3 days by CellTiter-Glo assay, IC50 = 10.6 μM. | 27774127 | ||
| insect cells | Function assay | Inhibition of recombinant human full length His-tagged PI3Kgamma expressed in insect cells, IC50 = 0.089 μM. | 27846451 | |||
| SUDHL6 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human SUDHL6 cells measured after 72 hrs by alamar blue assay, IC50 = 0.1176 μM. | 27846451 | ||
| insect cells | Function assay | Inhibition of recombinant human full length His-tagged PI3Kbeta expressed in insect cells, IC50 = 0.565 μM. | 27846451 | |||
| SU-DHL4 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human SU-DHL4 cells measured after 72 hrs by alamar blue assay, IC50 = 1.6 μM. | 27846451 | ||
| Pfeiffer | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Pfeiffer cells measured after 72 hrs by alamar blue assay, IC50 = 6.8 μM. | 27846451 | ||
| KARPAS422 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KARPAS422 cells measured after 72 hrs by alamar blue assay, IC50 = 8.1 μM. | 27846451 | ||
| Sf21 | Function assay | 30 mins | Inhibition of N-terminal His6-tagged recombinant full-length human PI3K p110beta/untagged recombinant full length human p85alpha expressed in baculovirus infected Sf21 insect cells using PIP2/ATP as substrate after 30 mins by TR-FRET assay, IC50 = 3.7 μM. | 28106991 | ||
| Sf21 | Function assay | 30 mins | Inhibition of N-terminal His6-tagged recombinant full-length human PI3K p110beta/untagged recombinant full length human p85alpha expressed in baculovirus infected Sf21 insect cells using PIP2/ATP as substrate after 30 mins by TR-FRET assay, IC50 = 3.7 μM. | 28106991 | ||
| RPMI8266 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human RPMI8266 cells after 72 hrs by MTT assay, IC50 = 0.00549 μM. | 28325601 | ||
| Raji | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Raji cells after 72 hrs by MTT assay, IC50 = 0.00995 μM. | 28325601 | ||
| KARPAS422 | Growth inhibition assay | Growth inhibition of human KARPAS422 cells by CCK8 assay, GI50 = 0.68 μM. | 28835805 | |||
| Pfeiffer | Growth inhibition assay | Growth inhibition of human Pfeiffer cells by CCK8 assay, GI50 = 0.74 μM. | 28835805 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| SUDHL6 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human SUDHL6 cells after 72 hrs by CCK8 assay, IC50 = 0.65 μM. | 29534936 | ||
| RPMI8226 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human RPMI8226 cells after 72 hrs by MTT assay, IC50 = 5.49 μM. | 29534936 | ||
| Raji | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Raji cells after 72 hrs by MTT assay, IC50 = 9.95 μM. | 29534936 | ||
| SU-DHL6 | Growth inhibition assay | Growth inhibition of human SU-DHL6 cells by CellTiter-Glo assay, GI50 = 0.042 μM. | 29601991 | |||
| MOLM13 | Growth inhibition assay | 72 hrs | Growth inhibition of human MOLM13 cells after 72 hrs by CellTiter-Glo luminescent assay, GI50 = 1.7 μM. | 30053721 | ||
| MOLM14 | Growth inhibition assay | 72 hrs | Growth inhibition of human MOLM14 cells after 72 hrs by CellTiter-Glo luminescent assay, GI50 = 6.4 μM. | 30053721 | ||
| MOLM14 | Antitumor assay | 100 mg/kg | 14 days | Antitumor activity against human MOLM14 cells xenografted in nu/nu mouse assessed as tumor growth inhibition at 100 mg/kg, po administered daily via gavage dosed for 14 days and measured daily during compound dosing relative to control | 30053721 | |
| MOLM14 | Antitumor assay | 100 mg/kg | 14 days | Antitumor activity against human MOLM14 cells xenografted in nu/nu mouse assessed as induction of apoptosis at 100 mg/kg, po administered daily via gavage dosed for 14 days and measured daily during compound dosing by TUNEL based assay | 30053721 | |
| MOLM14 | Antitumor assay | 100 mg/kg | 14 days | Antitumor activity against human MOLM14 cells xenografted in nu/nu mouse assessed as inhibition of tumor proliferation 100 mg/kg, po administered daily via gavage dosed for 14 days and measured daily during compound dosing by Ki-67 labelling based immunoh | 30053721 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 83 mg/mL
(199.79 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 415.42 | Formula | C22H18FN7O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 870281-82-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | GS-1101 | Smiles | CCC(C1=NC2=C(C(=CC=C2)F)C(=O)N1C3=CC=CC=C3)NC4=NC=NC5=C4NC=N5 | ||
Mechanism of Action
| Targets/IC50/Ki |
p110δ
(Cell-free assay) 2.5 nM
p110γ
(Cell-free assay) 89 nM
|
|---|---|
| In vitro |
Idelalisib (CAL-101) is not sensitive to other PI3K class I subunits including p110α, p110β, and p110γ. It specifically blocks FcϵR1 p110δ-mediated CD63 expression with an EC50 of 8 nM in primary basophil. This compound exhibits greater activity in B-cell acute lymphoblastic leukemia (B-ALL) and chronic lymphocytic leukemia (CLL) cells compared with acute myeloid leukemia (AML) and myeloproliferative neoplasm (MPN) cells. It produces the reduction in pAktS473, pAktT308, and the downstream target S6 in SU-DHL-5, KARPAS-422 and CCRF-SB cells with EC50 of 0.1 to 1.0 μM. It induces selective cytotoxicity in CLL cells independent of IgVH mutational status or interphase cytogenetics, primarily through a caspase-dependent mechanism. The compound induces cytotoxicity preferentially to CLL cells compared with normal B cells, without producing cytotoxicity in other hematopoietic cells, compared to LY294002. It lacks direct cytotoxic potential to T cells and nature killer (NK) cells. It can inhibit production of inflammatory cytokines, such as IL-6, IL-10, TNF-α, and IFN-γ, and activation-induced cytokines, such as CD40L. It also antagonizes CD40L-mediated CLL cell survival. It induces an accumulation of cells in G1 and a decrease in the S-phase population in L1236 and L591 cells, which indicates this compound as a novel strategy for the treatment of hodgkin lymphoma (HL). |
| Kinase Assay |
PI3K assay
|
|
PI3K assay is preformed on whole-cell lysates from CLL or normal B cells. A PI3K ELISA assay is performed. Briefly, whole-cell extracts are added to a mixture of PI(4,5)P2 substrate and reaction buffer containing adenosine triphosphate (ATP) and allowed to incubate at room temperature. The reaction is stopped by adding PI(3,4,5)P3 detector mixed with EDTA (ethylenediaminetetraacetic acid) and allowed to incubate at room temperature for 1 hour. After this time, the mixture is transferred from each well to a PI3K ELISA plate and allowed to incubate 1 hour. Plates are washed and then incubated with secondary detector for 30 minutes. Plates are washed again, and 3,3′,5,5′-tetramethylbenzidine solution is added for 5 minutes at which time H2SO4 is added to stop all reactions. Plates are read at 450 nm on a Labsystems 96-well plate reader.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | PUMA / p53 Bim / Bcl-xl / Bid / Mcl-1 p-p65 p-AKT / AKT Cleaved caspase 3 / Cleaved caspase 9 Mcl-1 / Bcl-2 / Bid / Bcl-xl / Noxa / Bak / Bax p-FoxO3a / FoxO3a Akt(T308) / PDK1(S241) / GSK-3β(S9) |
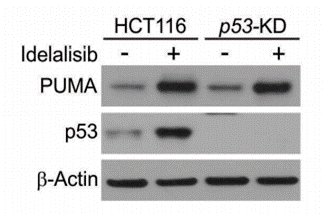
|
28008149 |
| Growth inhibition assay | Cell viability Cell viability |
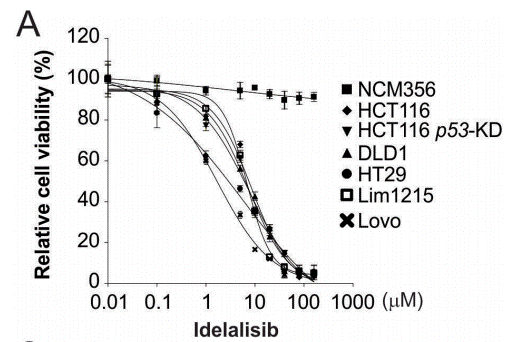
|
28008149 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03582098 | Completed | Chronic Lymphocytic Leukaemia |
Gilead Sciences |
September 12 2018 | -- |
| NCT03151057 | Terminated | B Cells-Tumors|B Cell Chronic Lymphocytic Leukemia|Follicular Lymphoma|Mantle Cell Lymphoma|Large B-Cell Diffuse Lymphoma of Bone (Diagnosis) |
Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins|Gilead Sciences |
July 31 2018 | Phase 1 |
| NCT03568929 | Completed | Follicular Non-Hodgkin''s Lymphoma Refractory |
Gilead Sciences |
May 25 2018 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What is the recommended dose and the route of administration for it in mouse studies?
Answer:
According to the following paper, it can be used by I.V. administration at the concentration of 40 mg/kg. https://www.ncbi.nlm.nih.gov/pubmed/24625684






































