- Bioactive Compounds
- By Signaling Pathways
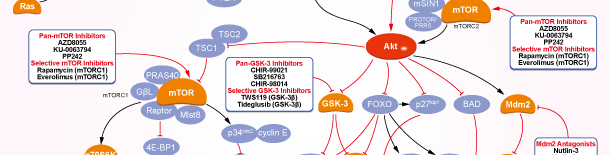
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-DYKDDDDK Tag magnetic beads
- Anti-DYKDDDDK Tag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Akt
Home PI3K/Akt/mTOR -Cytoskeletal Signaling Akt
Akt Products
- All (85)
- Akt Inhibitors (63)
- Akt Activators (7)
- Akt Modulators (6)
- New Akt Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
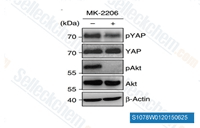
| S1078 | MK-2206 2HCl | MK-2206 2HCl is a highly selective inhibitor of Akt1/2/3 with IC50 of 8 nM/12 nM/65 nM in cell-free assays, respectively; no inhibitory activities against 250 other protein kinases observed. MK-2206 2HCl induces autophagy and apoptosis in cancer cells. Phase 2. |
|
|
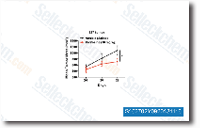
| S1037 | Perifosine | Perifosine is a novel Akt inhibitor with IC50 of 4.7 μM in MM.1S cells, targets pleckstrin homology domain of Akt. Phase 3. |
|
|
| S7863 | SC79 | SC79 is a brain-penetrable Akt phosphorylation activator and an inhibitor of Akt-PH domain translocation. |

|
|
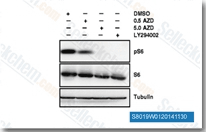
| S8019 | Capivasertib (AZD5363) | Capivasertib (AZD5363) potently inhibits all isoforms of Akt(Akt1/Akt2/Akt3) with IC50 of 3 nM/8 nM/8 nM in cell-free assays, similar to P70S6K/PKA and lower activity towards ROCK1/2. Phase 2. |
|
|
| S1113 | GSK690693 | GSK690693 is a pan-Akt inhibitor targeting Akt1/2/3 with IC50 of 2 nM/13 nM/9 nM in cell-free assays, also sensitive to the AGC kinase family: PKA, PrkX and PKC isozymes. GSK690693 also potently inhibits AMPK and DAPK3 from the CAMK family with IC50 of 50 nM and 81 nM, respectively. GSK690693 affects Unc-51-like autophagy activating kinase 1 (ULK1) activity, robustly inhibits STING-dependent IRF3 activation. Phase 1. |
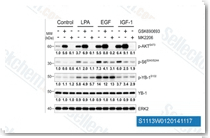
|
|
| S2808 | Ipatasertib (GDC-0068) | Ipatasertib (GDC-0068, RG7440) is a highly selective pan-Akt inhibitor targeting Akt1/2/3 with IC50 of 5 nM/18 nM/8 nM in cell-free assays, 620-fold selectivity over PKA. Phase 2. |
-S280804W1120150906.gif)
|
|
| S1117 | Triciribine (API-2) | Triciribine (API-2) is a DNA synthesis inhibitor, also inhibits Akt in PC3 cell line and HIV-1 in CEM-SS, H9, H9IIIB, U1 cells with IC50 of 130 nM and 20 nM, respectively; does not inhibit PI3K/PDK1; 5000-fold less active in cells lacking adenosine kinase. Phase 1/2. |
|
|
| S2743 | PF-04691502 | PF-04691502 (PF4691502) is an ATP-competitive PI3K(α/β/δ/γ)/mTOR dual inhibitor with Ki of 1.8 nM/2.1 nM/1.6 nM/1.9 nM and 16 nM in cell-free assays, little activity against either Vps34, AKT, PDK1, p70S6K, MEK, ERK, p38, or JNK. PF-04691502 induces apoptosis. Phase 2. |
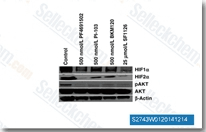
|
|
| S1362 | Rigosertib (ON-01910) | Rigosertib (ON-01910) is a non-ATP-competitive inhibitor of PLK1 with IC50 of 9 nM in a cell-free assay. It shows 30-fold greater selectivity against Plk2 and no activity to Plk3. Rigosertib inhibits PI3K/Akt pathway and activates oxidative stress signals. Rigosertib induces apoptosis in various cancer cells. Phase 3. |
|
|
| S2635 | CCT128930 | CCT128930 is a potent, ATP-competitive and selective inhibitor of Akt2 with IC50 of 6 nM in a cell-free assay, 28-fold greater selectivity for Akt2 than the closely related PKA kinase. CCT128930 induces cell cycle arrest, DNA damage, and autophagy independent of Akt inhibition. High dose of CCT128930 triggers cell apoptosis in HepG2 cells. |

|
|
| S2310 | Honokiol | Honokiol is the active principle of magnolia extract that inhibits Akt-phosphorylation and promotes ERK1/2 phosphorylation. Honokiol causes G0/G1 phase arrest, induces apoptosis, and autophagy via the ROS/ERK1/2 signaling pathway. Honokiol inhibits hepatitis C virus (HCV) infection. Phase 3. |

|
|
| S7521 | Afuresertib (GSK2110183) | Afuresertib (GSK2110183) is a potent, orally bioavailable Akt inhibitor with Ki of 0.08 nM, 2 nM, and 2.6 nM for Akt1, Akt2, and Akt3, respectively. Phase 2. |

|
|
| S2670 | A-674563 HCl | A-674563 HCl is an Akt1 inhibitor with Ki of 11 nM in cell-free assays, modest potent to PKA and >30-fold selective for Akt1 over PKC. |
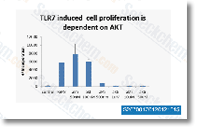
|
|
| S1558 | AT7867 | AT7867 is a potent ATP-competitive inhibitor of Akt1/2/3 and p70S6K/PKA with IC50 of 32 nM/17 nM/47 nM and 85 nM/20 nM in cell-free assays, respectively; little activity outside the AGC kinase family. |
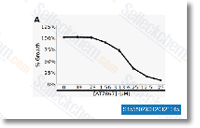
|
|
| S2335 | Oridonin | Oridonin, a diterpenoid purified from Rabdosia rubescens, is a traditional agent with antitumor, anti-bacterial and anti-inflammatory effects. Oridonin inhibits AKT1 and AKT2 kinase activity with IC50 of 8.4 μM and 8.9 μM, respectively. |
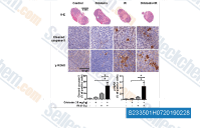
|
|
| S7776 | Akti-1/2 | Akti-1/2 (Akt Inhibitor VIII) is a highly selective Akt1/Akt2 inhibitor with IC50 of 58 nM/210 nM, respectively, about 36-fold selectivity for Akt1 over Akt3. Akti-1/2 induces apoptosis. |

|
|
| S1556 | PHT-427 | PHT-427 (CS-0223) is a dual Akt and PDPK1 inhibitor (high affinity binding for the PH domains of Akt and PDPK1) with Ki of 2.7 μM and 5.2 μM, respectively. |

|
|
| S3056 | Miltefosine | Miltefosine (Hexadecylphosphocholine) inhibits PI3K/Akt activity with ED50 of 17.2 μM and 8.1 μM in carcinoma cell lines A431 and HeLa, first oral drug for Visceral leishmaniasis, effective against both promastigotes and amastigotes. |
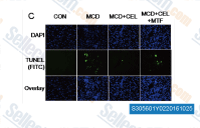
|
|
| S8339 | Miransertib (ARQ 092) HCl | Miransertib (ARQ 092) HCl is a novel, orally bioavailable and selective AKT pathway inhibitor exhibiting a manageable safety profile among patients with advanced solid tumors. |
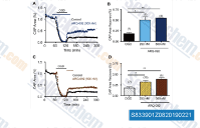
|
|
| S7563 | AT13148 | AT13148 is an oral, ATP-competitive, multi-AGC kinase inhibitor with IC50 of 38 nM/402 nM/50 nM, 8 nM, 3 nM, and 6 nM/4 nM for Akt1/2/3, p70S6K, PKA, and ROCKI/II, respectively. Phase 1. |

|
|
| S7492 | Uprosertib (GSK2141795) | Uprosertib (GSK2141795, GSK795) is a selective, ATP-competitive, and orally bioavailable Akt inhibitor with IC50 of 180 nM, 328 nM, and 38 nM for Akt 1, 2 and 3, respectively. Phase 2. |
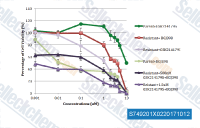
|
|
| S6811 | Miransertib (ARQ-092) | Miransertib (ARQ-092) is a potent, selective and orally bioavailable allosteric inhibitor of Akt with IC50s of 2.7 nM, 14 nM and 8.1 nM for Akt1, Akt2, Akt3, respectively. | ||
| S5313 | SC66 | SC66 is an allosteric inhibitor which displays a dual-inhibitory function toward AKT activity with IC50 values of 0.77, 2.85 and 0.47 μg/ml in HepG2, Huh7 and Hep3B cells after 72 h treatment, respectively. | ||
| S5144 | Neferine | Neferine ((R)-1,2-Dimethoxyaporphine), a natural component of Nelumbo nucifera, has antitumor efficiency. Neferine induces apoptosis in renal cancer cells. Neferine prevents autophagy through activation of Akt/mTOR pathway and Nrf2 in muscle cells. Neferine strongly inhibits NF-κB activation. Neferine possesses a number of therapeutic effects such as anti-diabetic, anti-aging, anti-microbial, anti-thrombotic, anti-arrhythmic, anti-inflammatory and even anti-HIV. | ||
| S3901 | Astragaloside IV | Astragaloside IV (AST-IV, AS-IV) is a bioactive saponin first isolated from the dried plant roots of the genus Astragalus, which is used in traditional Chinese medicine. It has various effect on the cardiovascular, immune, digestive, and nervous systems. AS-IV suppresses activation of p-Akt, p-mTOR, p-NF-κB and p-Erk1/2. | ||
| S9190 | Oroxin B | Oroxin B (Hypocretin-2), one of flavonoids isolated from traditional Chinese herbal medicine Oroxylum indicum (L.) Vent, selectively induces tumor-suppressive ER stress in malignant lymphoma cells and has antioxidant activity. Oroxin B significantly inhibits proliferation and induce apoptosis, which may be strongly associated with the inhibiting COX-2/VEGF and PTEN/PI3K/AKT signaling pathway in SMMC-7721 cells, Oroxin B potentially be used as a novel therapeutic agent for liver cancer.COX-2, VEGF, PI3K, and p-AKT expression levels are downregulated, while PTEN is upregulated after Oroxin B treatment. | ||
| S8132 | Deguelin | Deguelin, a natural product isolated from plants in the Mundulea sericea family, is an PI3K/AKT Inhibitor. | ||
| S6847 | ML-9 HCl | ML-9 HCl (ML-9 hydrochloride) is a selective and potent inhibitor of Akt kinase, myosin light chain kinase (MLCK) and stromal interaction molecule 1 (STIM1). ML-9 HCl is also a potent inhibitor of Ca2+-permeable channels. ML-9 HCl is a lysosomotropic agent targeting autophagy and cell death. | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S3296 | Hispidulin | Hispidulin (Dinatin), an active natrual ingredient in a number of traditional Chinese medicinal herbs, exhibits inhibitory activity against the oncogenic protein kinase Pim-1 with IC50 of 2.71 μM. Hispidulin induces apoptosis through mitochondrial dysfunction and inhibition of P13k/Akt signalling pathway in HepG2 cancer cells. Hispidulin exerts anti-osteoporotic and bone resorption attenuating effects via activating the AMPK signaling pathway. | ||
| E2384 | (E)-Akt inhibitor-IV | (E)-Akt inhibitor-IV is a PI3K-Akt inhibitor, with potent cytotoxic. | ||
| S3785 | Notoginsenoside R1 | Notoginsenoside R1 (Sanchinoside R1) is the main ingredient with cardiovascular activity in Panax notoginseng. It inhibits TNF-α-induced PAI-1 overexpression via extracellular signal-related kinases (ERK1/2) and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PKB) signaling pathways. | ||
| S3810 | Scutellarin | Scutellarin (Breviscapine, Breviscapin, Scutellarein-7-glucuronide), the major active principal flavonoids extracted from the Chinese herbal medicines Scutellaria baicalensis and Erigeron breviscapus (Vant.) Hand-Mazz, has many pharmacological effects, such as antioxidant, antitumor, antiviral, and antiinflammatory activities. Scutellarin can down-regulates the STAT3/Girdin/Akt signaling in HCC cells, and inhibits RANKL-mediated MAPK and NF-κB signaling pathway in osteoclasts. | ||
| S3220 | Trigonelline | Trigonelline (Trigenolline) is a plant alkaloid and a major component of coffee and fenugreek with anti-degranulation, anti-diabetic, antioxidant, anti-inflammatory, and neuroprotective effects. Trigonelline inhibits FcεRI-mediated intracellular signaling pathways, such as phosphorylation of PLCγ1, PI3K, and Akt. Trigonelline (Trigenolline) also inhibits the microtubule formation in RBL-2H3 cells. | ||
| F0004 | Akt (pan) Antibody [E16N17] | RAC-beta serine/threonine-protein kinase, Protein kinase Akt-2, PKB beta 1, RAC-PK-beta, RAC-gamma serine/threonine-protein kinase, Akt-3, PKB gamma, RAC-PK-gamma, STK-2, AKT3, PKBG, RAC-alpha serine/threonine-protein kinase, PKB, PKB alpha, Proto-oncogene c-Akt, RAC-PK-alpha, AKT1 | ||
| F1644 | Phospho-AKT1/2/3 (Ser473/474/472) Antibody [B6L18] | |||
| F0699 | Phospho-Akt (Thr450) Antibody [J1B12] | RAC-beta serine/threonine-protein kinase, Protein kinase Akt-2, PKB beta 1, RAC-PK-beta, RAC-gamma serine/threonine-protein kinase, Akt-3, PKB gamma, RAC-PK-gamma, STK-2, AKT3, PKBG, RAC-alpha serine/threonine-protein kinase, PKB, PKB alpha, Proto-oncogene c-Akt, RAC-PK-alpha, AKT1 | ||
| F0217 | Akt1 Antibody [C19L11] | |||
| F0218 | Akt2 Antibody [P3J21] | |||
| F0493 | Phospho-Akt2 (Ser474) Antibody [L16H11] | AKT2 (phospho S474),Phospho-Akt2 (Ser474) | ||
| F0670 | Akt3 Antibody [D4P3] | |||
| F0418 | AS160 Antibody [M24M10] | |||
| S7127 | TIC10 Analogue | TIC10 Analogue is an analogue of TIC10, which inactivates Akt and ERK to induce TRAIL through Foxo3a, possesses superior drug properties: delivery across the blood-brain barrier, superior stability and improved pharmacokinetics. Phase 1/2. | ||
| S3294 | Demethyl-Coclaurine | Demethyl-Coclaurine (Higenamine, Norcoclaurine), the key component of the Chinese herb aconite root, is a beta-2 adrenergic receptor (β2-AR) agonist. Demethyl-Coclaurine stimulates AKT phosphorylation and requires PI3K activation for the anti-apoptotic effect in cardiomyocytes. | ||
| E2947 | Recilisib | Recilisib (ON01210, EX-RAD) is a radioprotectant that activates the activity of AKT and PI3K in cells. It has been studied as prophylactic (use prior to radiation exposure) and therapeutic (after exposure to radiation) drug. | ||
| S4572 | Homosalate | Homosalate (HMS, Homomenthyl salicylate) is an organic ultraviolet filter used in most sunscreens but has been reported to be toxic to marine organisms. Homosalate aggravates the invasion of human trophoblast cells as well as regulates intracellular signaling pathways including PI3K/AKT and MAPK pathways. | ||
| S3289 | Daphnoretin | Daphnoretin (Dephnoretin, Thymelol), a biologically active compound isolated from Wikstroemia indica C.A. Mey., is a protein kinase C (PKC) activator. Daphnoretin inhibits the proliferation, invasion, and migration of tumor cells and promote its apoptosis by regulating the activity of Akt signal pathway. | ||
| S9514 | Rotundic acid | Rotundic acid (Rutundic acid), a natural compound, exhibit cytotoxic activities toward human hepatocellular carcinoma (HepG2), malignant melanoma (A375), SCLC (NCI-H446), breast cancer (MCF-7), and colon cancer (HT-29) cell lines.RA induces cell cycle arrest, DNA damage, and apoptosis by modulating the AKT/mTOR and MAPK pathways. | ||
| S3224 | Cinobufagin | Cinobufagin (Cinobufagine), an active ingredient of Venenum Bufonis, inhibits tumor development. Cinobufagin increases ATM and Chk2 and decreases CDC25C, CDK1, and cyclin B. Cinobufagin inhibits PI3K, AKT and Bcl-2 while increases levels of cleaved caspase-9 and caspase-3. Thus, Cinobufagin induces cell cycle arrest at the G2/M phase and apoptosis. | ||
| S8961 | Alobresib (GS-5829) | Alobresib (GS-5829) is a novel BET inhibitor that represents a highly effective therapeutics agent against recurrent/chemotherapy-resistant USC-overexpressing c-Myc. Alobresib (GS-5829) inhibits CLL cell proliferation and induces leukemia cell apoptosis through deregulation of key signaling pathways, such as BLK, AKT, ERK1/2, and MYC. Alobresib (GS-5829) also inhibits NF-κB signaling. | ||
| S8839 | Borussertib | Borussertib is a covalent-allosteric inhibitor of protein kinase Akt with an IC50 of 0.8 nM and a Ki of 2.2 nM for WT Akt. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| S3243 | Zeaxanthin | Zeaxanthin, the carotenoid alcohol participates in the xanthophyll cycle, activates the extrinsic apoptosis pathway which induces apoptosis on uveal melanoma cells with IC50 value 40.8 µM. | ||
| E1125 | A-443654 | A-443654, a derivative of indazole–pyridine compounds, is a pan Akt (Akt1, 2, & 3 isoforms) inhibitor which binds to the ATP-binding site of Akt. It is an ATP competitive and reversible inhibitor. | ||
| E3656 | Dichroa febrifuga Extract | Dichroa Febrifuga Extract is extracted from Dichroa Febrifuga, which can Suppress PI3K/AKT and MAPK Signaling Pathways. | ||
| S0765 | MAZ51 | MAZ51 is a potent and selective inhibitor of vascular endothelial growth factor receptor (VEGFR)-3 (Flt-4) tyrosine kinase. MAZ51 induces cell rounding and G2/M cell cycle arrest in glioma cells through phosphorylation of Akt/GSK3β and activation of RhoA. MAZ51 inhibits the proliferation and induces the apoptosis of a variety of non-VEGFR-3-expressing tumor cell lines. | ||
| E4864New | Pentamidine | Pentamidine (MP-601205) is an aromatic diamidine drug, an antagonist of TLR4.It also inhibits PI3K/AKT signaling pathway and reduce the expression of MMP-2 and MMP-9. It exhibits anti- protozoal, anti-inflammatory, and anti-tumor activities and is used as an agent for treating African trypanosomiasis, antimony resistant leishmaniasis and Pneumocystis carinii pneumonia. | ||
| S4953 | Usnic acid | Usnic acid (Usniacin) is a furandione found uniquely in lichen that is used widely in cosmetics, deodorants, toothpaste and medicinal creams as well as some herbal products. It exhibits antiviral, antiprotozoal, antiproliferative, anti-inflammatory and analgesic activity. Usnic acid inhibits breast tumor angiogenesis and growth by suppressing VEGFR2-mediated AKT and ERK1/2 signaling pathways. | ||
| E3106 | Dioscoreae Nipponicae Rhizoma Extract | Dioscoreae Nipponicae Rhizoma Extract is extracted from the rhizome of Dioscorea nipponica, of which the main component decreases the phosphorylation in IGF-1R, which in turn inhibits the phosphorylation and activation of PI3K-AKT and Rap1-MEK signaling pathways, promoting cell apoptosis and Graves’ disease remission. | ||
| E1710 | LY2780301 | LY2780301 is a highly selective adenosine triphosphate (ATP)-competitive dual inhibitor of p70S6K and Akt. LY2780301 binds to Akt, inhibiting its activity, which consequently blocks the PI3K/Akt signaling pathway which leads to reduced cell proliferation and the initiation of apoptosis in tumor cells. | ||
| E3241 | Cinnamomi Ramulus Extract | Cinnamomi Ramulus Extract is extracted from Cinnamomi Ramulus, which can inhibit the growth of colon cancer cells via Akt/ERK signaling pathways. | ||
| E0020 | Lupenone | Lupenone (Lup-20(29)-en-3-one, lupeone) is an isolated compound exhibiting anti-oxidative, anti-inflammation, and anti-diabetic activities. Lupenone can protect SH-SY5y cells against METH-induced neuronal apoptosis through the PI3K/Akt pathway. | ||
| E3041 | Alpiniae Katsumadai Extract | Alpiniae Katsumadai Extract is extracted from Alpiniae Katsumadai, which can induce growth inhibition and autophagy‑related apoptosis by regulating the AMPK and Akt/mTOR/p70S6K signaling pathways in cancer cells. | ||
| S9315 | Praeruptorin A | Praeruptorin A, a naturally existing pyranocumarin, is isolated from the dried root of Peucedanum praeruptorum Dunn. Praeruptorin A inhibits p38/Akt-c-Fos-NFATc1 signaling and PLCγ-independent Ca2+ oscillation. Praeruptorin A can significantly upregulates multidrug resistance-associated protein 2 expression via the constitutive androstane receptor-mediated pathway in vitro, and this should be taken as an herb-drug interaction. | ||
| E3365 | Weigela Grandiflora Fortune Extract | Weigela Grandiflora Fortune Extract is extracted from Weigela Grandiflora Fortune, which decreases the infection-mediated expression of inflammatory mediators by inhibiting the AKT/NF-κB and MAPK signaling pathways. | ||
| S6760 | LM22B-10 | LM22B-10 is a small molecule TrkB/TrkC neurotrophin receptor co-activator, LM22B-10 selectively activates TrkB, TrkC, AKT and ERK in vivo and in vitro. | ||
| S3309 | Solasodine | Solasodine (Purapuridine, Solancarpidine, Solasodin, Salasodine, Salasdine) is a poisonous alkaloid chemical compound that occurs in plants of the Solanaceae family. Solasodine reduces the mRNA level of matrix metalloproteinase-2 (MMP-2), MMP-9 and extracellular inducer of matrix metalloproteinase (EMMPRIN), but increases the expression of reversion-inducing cysteine-rich protein with kazal motifs (RECK). Solasodine downregulates oncogenic microRNA-21 (miR-21), which has been known to target RECK. Solasodine also reduces PI3K/Akt signaling pathways and downregulates expression of miR-21. | ||
| E4992New | Afuresertib hydrochloride | Afuresertib hydrochloride (GSK 2110183 hydrochloride, LAE002 hydrochloride) is an orally bioavailable, selective, ATP-competitive and potent inhibitor of pan-Akt kinase with Ki values of 0.08 nM, 2 nM, 2.6 nM for Akt1/Akt2/Akt3 respectively. | ||
| E5820New | AKT Kinase Inhibitor | AKT Kinase Inhibitor is an inhibitor of Akt that reverses the protective effects of Tadalafil (TAD) in LPS-induced RWPE-1 cells by decreasing cell viability, promoting apoptosis, and increasing pro-inflammatory cytokine levels. | ||
| S9054 | Pectolinarin | Pectolinarin is a major compound in Cirsium setidens with anti-inflammatory activity. Pectolinarin inhibits secretion of IL-6 and IL-8, as well as the production of PGE2 and NO. Pectolinarin induces apoptosis via inactivation of the PI3K/Akt pathway. | ||
| E2391 | α-Linolenic acid | α-Linolenic acid, an essential fatty acid isolated from seed oils, affects the process of thrombotic through the modulation of PI3K/Akt signaling, and possesses the anti-arrhythmic properties. | ||
| E0945 | BIA | BIA (TMBIM6 antagonist BIA), a potential TMBIM6 antagonist, is an inhibitor of the interaction between TMBIM6 (Transmembrane B cell lymphoma 2-associated X protein (BAX) inhibitor motif-containing 6) and mTORC2, which ultimately blocks AKT activation and cancer progression. | ||
| S6885 | Ailanthone | Ailanthone (AIL, Δ13-Dehydrochaparrinone), a natural anti-hepatocellular carcinoma (HCC) component in Ailanthus altissima, induces G0/G1-phase cell cycle arrest by decreasing expression of cyclins and CDKs and increases expression of p21 and p27. Ailanthone triggers DNA damage characterized by activation of the ATM/ATR pathway. Ailanthone induces apoptosis which is mitochondrion-mediated and involves the PI3K/AKT signaling pathway in Huh7 cells. Ailanthone is also a potent inhibitor of both full-length Androgen Receptor (AR-FL) and constitutively active truncated AR splice variants (AR-Vs, AR1-651) with IC50 of 69 nM and 309 nM, respectively. | ||
| S2323 | Methyl-Hesperidin | Methyl Hesperidin is a flavanone glycoside (flavonoid) (C28H34O15) found abundantly in citrus fruits. Its aglycone form is called hesperetin. | ||
| E7359New | A-674563 | A-674563 is a selective, orally active inhibitor of Akt1 with a Ki of 11 nM. It also inhibits PKA with Ki of 16 nM and CDK2 with Ki of 46 nM. A-674563 reduces Akt downstream target phosphorylation and inhibits tumor cell proliferation in vitro with an EC50 of 0.4 µM. | ||
| E2682 | RPI-1 | RPI-1 inhibits proliferation of human papillary thyroid carcinoma cell line TPC-1 by inducing accumulation of cells at the G2 cell cycle phase, abolishes Ret/Ptc1 tyrosine phosphorylation along with its binding to Shc and phospholipase Cg, also abolishes the activation of JNK2 and AKT. | ||
| S8500 | BAY1125976 | BAY 1125976 is a selective allosteric AKT1/2 inhibitor,exhibits high efficacy on AKT signaling-dependent tumor growth in mouse models. BAY1125976 inhibits the activity of AKT1 (IC50 = 5.2 nM at 10 µM ATP and 44 nM at 2 mM ATP) and AKT2 (IC50 = 18 nM at 10 µM ATP and 36 nM at 2 mM ATP) very potently.Whereas BAY1125976 is almost inactive on AKT3 (IC50 = 427 nM at 10 µM ATP). | ||
| E2826 | Hematein | Hematein inhibits casein kinase II activity in a selective, dose-dependent and ATP non-competitive manner in vitro, with IC50 of 0.55 μM in the presence of 10 μM ATP. | ||
| S9611 | ABTL-0812 | ABTL0812 (α-Hydroxylinoleic acid, LP-10218, SCLN-0812) inhibits Akt/mTOR axis by inducing the overexpression of TRIB3 and activating autophagy in lung squamous carcinoma cell lines. ABTL0812 also induces AMPK activation and ROS accumulation. | ||
| E2401 | SPP-86 | SPP-86, a potent and selective cell permeable inhibitor of rearranged during transfection (RET) tyrosine kinase with an IC50 of 8 nM, inhibits RET-induced phosphatidylinositide 3-kinases (PI3K)/Akt and MAPK signaling, also inhibits RET-induced estrogen receptorα (ERα) phosphorylation in MCF7 cells. | ||
| S1321 | Urolithin B | Urolithin B inhibits NF-κB activity by reducing the phosphorylation and degradation of IκBα. Urolithin B suppresses the phosphorylation of JNK, ERK, and Akt, and enhances the phosphorylation of AMPK. Urolithin B is also a regulator of skeletal muscle mass. | ||
| S1273 | Amarogentin | Amarogentin (AG), a secoiridoid glycoside mainly extracted from Swertia and Gentiana roots, exhibits anti-oxidative, anti-tumour, and anti-diabetic activities. Amarogentin is an agonist for the bitter taste receptor TAS2R1 and inhibits in LAD-2 cells substance P-induced production of newly synthesized TNF-α. Amarogentin induces apoptosis in human gastric cancer cells (SNU-16) through G2/M cell cycle arrest and PI3K/Akt signalling pathway. Amarogentin (AG) interacts with the α2 subunit of AMP-activated protein kinase (AMPK) and activates the trimeric kinase with EC50 of 277 pM. |
||
| S3241 | Loureirin A | Loureirin A is a flavonoid extracted from the red resin of the herbs of Dracaena cochinchinensis, which is known as Dragon's Blood. Loureirin A inhibits platelet activation by an impairment of PI3K/Akt signaling. Loureirin A inhibits Akt phosphorylation. | ||
| E0785 | YS-49 | YS-49 is a PI3K/Akt (a downstream target of RhoA) activator, to reduce RhoA/PTEN activation in the 3-methylcholanthrene-treated cells, inhibits angiotensin II (Ang II)-stimulated proliferation of VSMCs via induction of heme oxygenase (HO)-1, also is an isoquinoline compound alkaloid, has a strong positive inotropic action through activation of cardiac β-adrenoceptors. | ||
| E1604 | Vevorisertib trihydrochloride | Vevorisertib trihydrochloride(ARQ 751 trihydrochloride) is a selective, allosteric inhibitor of pan-AKT and AKT1-E17K mutant with Kd values of 1.2 nM and 8.6 nM, respectively. It also has IC50 values of 0.55, 0.81, and 1.3 nM for AKT1, AKT2, and AKT3, respectively and can be used for the research of cancer. | ||
| S1078 | MK-2206 2HCl | MK-2206 2HCl is a highly selective inhibitor of Akt1/2/3 with IC50 of 8 nM/12 nM/65 nM in cell-free assays, respectively; no inhibitory activities against 250 other protein kinases observed. MK-2206 2HCl induces autophagy and apoptosis in cancer cells. Phase 2. |

|
|
| S1037 | Perifosine | Perifosine is a novel Akt inhibitor with IC50 of 4.7 μM in MM.1S cells, targets pleckstrin homology domain of Akt. Phase 3. |

|
|
| S8019 | Capivasertib (AZD5363) | Capivasertib (AZD5363) potently inhibits all isoforms of Akt(Akt1/Akt2/Akt3) with IC50 of 3 nM/8 nM/8 nM in cell-free assays, similar to P70S6K/PKA and lower activity towards ROCK1/2. Phase 2. |

|
|
| S1113 | GSK690693 | GSK690693 is a pan-Akt inhibitor targeting Akt1/2/3 with IC50 of 2 nM/13 nM/9 nM in cell-free assays, also sensitive to the AGC kinase family: PKA, PrkX and PKC isozymes. GSK690693 also potently inhibits AMPK and DAPK3 from the CAMK family with IC50 of 50 nM and 81 nM, respectively. GSK690693 affects Unc-51-like autophagy activating kinase 1 (ULK1) activity, robustly inhibits STING-dependent IRF3 activation. Phase 1. |

|
|
| S2808 | Ipatasertib (GDC-0068) | Ipatasertib (GDC-0068, RG7440) is a highly selective pan-Akt inhibitor targeting Akt1/2/3 with IC50 of 5 nM/18 nM/8 nM in cell-free assays, 620-fold selectivity over PKA. Phase 2. |
-S280804W1120150906.gif)
|
|
| S1117 | Triciribine (API-2) | Triciribine (API-2) is a DNA synthesis inhibitor, also inhibits Akt in PC3 cell line and HIV-1 in CEM-SS, H9, H9IIIB, U1 cells with IC50 of 130 nM and 20 nM, respectively; does not inhibit PI3K/PDK1; 5000-fold less active in cells lacking adenosine kinase. Phase 1/2. |

|
|
| S2743 | PF-04691502 | PF-04691502 (PF4691502) is an ATP-competitive PI3K(α/β/δ/γ)/mTOR dual inhibitor with Ki of 1.8 nM/2.1 nM/1.6 nM/1.9 nM and 16 nM in cell-free assays, little activity against either Vps34, AKT, PDK1, p70S6K, MEK, ERK, p38, or JNK. PF-04691502 induces apoptosis. Phase 2. |

|
|
| S1362 | Rigosertib (ON-01910) | Rigosertib (ON-01910) is a non-ATP-competitive inhibitor of PLK1 with IC50 of 9 nM in a cell-free assay. It shows 30-fold greater selectivity against Plk2 and no activity to Plk3. Rigosertib inhibits PI3K/Akt pathway and activates oxidative stress signals. Rigosertib induces apoptosis in various cancer cells. Phase 3. |

|
|
| S2635 | CCT128930 | CCT128930 is a potent, ATP-competitive and selective inhibitor of Akt2 with IC50 of 6 nM in a cell-free assay, 28-fold greater selectivity for Akt2 than the closely related PKA kinase. CCT128930 induces cell cycle arrest, DNA damage, and autophagy independent of Akt inhibition. High dose of CCT128930 triggers cell apoptosis in HepG2 cells. |

|
|
| S2310 | Honokiol | Honokiol is the active principle of magnolia extract that inhibits Akt-phosphorylation and promotes ERK1/2 phosphorylation. Honokiol causes G0/G1 phase arrest, induces apoptosis, and autophagy via the ROS/ERK1/2 signaling pathway. Honokiol inhibits hepatitis C virus (HCV) infection. Phase 3. |

|
|
| S7521 | Afuresertib (GSK2110183) | Afuresertib (GSK2110183) is a potent, orally bioavailable Akt inhibitor with Ki of 0.08 nM, 2 nM, and 2.6 nM for Akt1, Akt2, and Akt3, respectively. Phase 2. |

|
|
| S2670 | A-674563 HCl | A-674563 HCl is an Akt1 inhibitor with Ki of 11 nM in cell-free assays, modest potent to PKA and >30-fold selective for Akt1 over PKC. |

|
|
| S1558 | AT7867 | AT7867 is a potent ATP-competitive inhibitor of Akt1/2/3 and p70S6K/PKA with IC50 of 32 nM/17 nM/47 nM and 85 nM/20 nM in cell-free assays, respectively; little activity outside the AGC kinase family. |

|
|
| S2335 | Oridonin | Oridonin, a diterpenoid purified from Rabdosia rubescens, is a traditional agent with antitumor, anti-bacterial and anti-inflammatory effects. Oridonin inhibits AKT1 and AKT2 kinase activity with IC50 of 8.4 μM and 8.9 μM, respectively. |

|
|
| S7776 | Akti-1/2 | Akti-1/2 (Akt Inhibitor VIII) is a highly selective Akt1/Akt2 inhibitor with IC50 of 58 nM/210 nM, respectively, about 36-fold selectivity for Akt1 over Akt3. Akti-1/2 induces apoptosis. |

|
|
| S1556 | PHT-427 | PHT-427 (CS-0223) is a dual Akt and PDPK1 inhibitor (high affinity binding for the PH domains of Akt and PDPK1) with Ki of 2.7 μM and 5.2 μM, respectively. |

|
|
| S3056 | Miltefosine | Miltefosine (Hexadecylphosphocholine) inhibits PI3K/Akt activity with ED50 of 17.2 μM and 8.1 μM in carcinoma cell lines A431 and HeLa, first oral drug for Visceral leishmaniasis, effective against both promastigotes and amastigotes. |

|
|
| S8339 | Miransertib (ARQ 092) HCl | Miransertib (ARQ 092) HCl is a novel, orally bioavailable and selective AKT pathway inhibitor exhibiting a manageable safety profile among patients with advanced solid tumors. |

|
|
| S7563 | AT13148 | AT13148 is an oral, ATP-competitive, multi-AGC kinase inhibitor with IC50 of 38 nM/402 nM/50 nM, 8 nM, 3 nM, and 6 nM/4 nM for Akt1/2/3, p70S6K, PKA, and ROCKI/II, respectively. Phase 1. |

|
|
| S7492 | Uprosertib (GSK2141795) | Uprosertib (GSK2141795, GSK795) is a selective, ATP-competitive, and orally bioavailable Akt inhibitor with IC50 of 180 nM, 328 nM, and 38 nM for Akt 1, 2 and 3, respectively. Phase 2. |

|
|
| S6811 | Miransertib (ARQ-092) | Miransertib (ARQ-092) is a potent, selective and orally bioavailable allosteric inhibitor of Akt with IC50s of 2.7 nM, 14 nM and 8.1 nM for Akt1, Akt2, Akt3, respectively. | ||
| S5313 | SC66 | SC66 is an allosteric inhibitor which displays a dual-inhibitory function toward AKT activity with IC50 values of 0.77, 2.85 and 0.47 μg/ml in HepG2, Huh7 and Hep3B cells after 72 h treatment, respectively. | ||
| S3901 | Astragaloside IV | Astragaloside IV (AST-IV, AS-IV) is a bioactive saponin first isolated from the dried plant roots of the genus Astragalus, which is used in traditional Chinese medicine. It has various effect on the cardiovascular, immune, digestive, and nervous systems. AS-IV suppresses activation of p-Akt, p-mTOR, p-NF-κB and p-Erk1/2. | ||
| S9190 | Oroxin B | Oroxin B (Hypocretin-2), one of flavonoids isolated from traditional Chinese herbal medicine Oroxylum indicum (L.) Vent, selectively induces tumor-suppressive ER stress in malignant lymphoma cells and has antioxidant activity. Oroxin B significantly inhibits proliferation and induce apoptosis, which may be strongly associated with the inhibiting COX-2/VEGF and PTEN/PI3K/AKT signaling pathway in SMMC-7721 cells, Oroxin B potentially be used as a novel therapeutic agent for liver cancer.COX-2, VEGF, PI3K, and p-AKT expression levels are downregulated, while PTEN is upregulated after Oroxin B treatment. | ||
| S8132 | Deguelin | Deguelin, a natural product isolated from plants in the Mundulea sericea family, is an PI3K/AKT Inhibitor. | ||
| S6847 | ML-9 HCl | ML-9 HCl (ML-9 hydrochloride) is a selective and potent inhibitor of Akt kinase, myosin light chain kinase (MLCK) and stromal interaction molecule 1 (STIM1). ML-9 HCl is also a potent inhibitor of Ca2+-permeable channels. ML-9 HCl is a lysosomotropic agent targeting autophagy and cell death. | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S3296 | Hispidulin | Hispidulin (Dinatin), an active natrual ingredient in a number of traditional Chinese medicinal herbs, exhibits inhibitory activity against the oncogenic protein kinase Pim-1 with IC50 of 2.71 μM. Hispidulin induces apoptosis through mitochondrial dysfunction and inhibition of P13k/Akt signalling pathway in HepG2 cancer cells. Hispidulin exerts anti-osteoporotic and bone resorption attenuating effects via activating the AMPK signaling pathway. | ||
| E2384 | (E)-Akt inhibitor-IV | (E)-Akt inhibitor-IV is a PI3K-Akt inhibitor, with potent cytotoxic. | ||
| S3785 | Notoginsenoside R1 | Notoginsenoside R1 (Sanchinoside R1) is the main ingredient with cardiovascular activity in Panax notoginseng. It inhibits TNF-α-induced PAI-1 overexpression via extracellular signal-related kinases (ERK1/2) and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PKB) signaling pathways. | ||
| S3810 | Scutellarin | Scutellarin (Breviscapine, Breviscapin, Scutellarein-7-glucuronide), the major active principal flavonoids extracted from the Chinese herbal medicines Scutellaria baicalensis and Erigeron breviscapus (Vant.) Hand-Mazz, has many pharmacological effects, such as antioxidant, antitumor, antiviral, and antiinflammatory activities. Scutellarin can down-regulates the STAT3/Girdin/Akt signaling in HCC cells, and inhibits RANKL-mediated MAPK and NF-κB signaling pathway in osteoclasts. | ||
| S3220 | Trigonelline | Trigonelline (Trigenolline) is a plant alkaloid and a major component of coffee and fenugreek with anti-degranulation, anti-diabetic, antioxidant, anti-inflammatory, and neuroprotective effects. Trigonelline inhibits FcεRI-mediated intracellular signaling pathways, such as phosphorylation of PLCγ1, PI3K, and Akt. Trigonelline (Trigenolline) also inhibits the microtubule formation in RBL-2H3 cells. | ||
| S7127 | TIC10 Analogue | TIC10 Analogue is an analogue of TIC10, which inactivates Akt and ERK to induce TRAIL through Foxo3a, possesses superior drug properties: delivery across the blood-brain barrier, superior stability and improved pharmacokinetics. Phase 1/2. | ||
| S3224 | Cinobufagin | Cinobufagin (Cinobufagine), an active ingredient of Venenum Bufonis, inhibits tumor development. Cinobufagin increases ATM and Chk2 and decreases CDC25C, CDK1, and cyclin B. Cinobufagin inhibits PI3K, AKT and Bcl-2 while increases levels of cleaved caspase-9 and caspase-3. Thus, Cinobufagin induces cell cycle arrest at the G2/M phase and apoptosis. | ||
| S8839 | Borussertib | Borussertib is a covalent-allosteric inhibitor of protein kinase Akt with an IC50 of 0.8 nM and a Ki of 2.2 nM for WT Akt. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| S3243 | Zeaxanthin | Zeaxanthin, the carotenoid alcohol participates in the xanthophyll cycle, activates the extrinsic apoptosis pathway which induces apoptosis on uveal melanoma cells with IC50 value 40.8 µM. | ||
| E1125 | A-443654 | A-443654, a derivative of indazole–pyridine compounds, is a pan Akt (Akt1, 2, & 3 isoforms) inhibitor which binds to the ATP-binding site of Akt. It is an ATP competitive and reversible inhibitor. | ||
| E3656 | Dichroa febrifuga Extract | Dichroa Febrifuga Extract is extracted from Dichroa Febrifuga, which can Suppress PI3K/AKT and MAPK Signaling Pathways. | ||
| E4864New | Pentamidine | Pentamidine (MP-601205) is an aromatic diamidine drug, an antagonist of TLR4.It also inhibits PI3K/AKT signaling pathway and reduce the expression of MMP-2 and MMP-9. It exhibits anti- protozoal, anti-inflammatory, and anti-tumor activities and is used as an agent for treating African trypanosomiasis, antimony resistant leishmaniasis and Pneumocystis carinii pneumonia. | ||
| S4953 | Usnic acid | Usnic acid (Usniacin) is a furandione found uniquely in lichen that is used widely in cosmetics, deodorants, toothpaste and medicinal creams as well as some herbal products. It exhibits antiviral, antiprotozoal, antiproliferative, anti-inflammatory and analgesic activity. Usnic acid inhibits breast tumor angiogenesis and growth by suppressing VEGFR2-mediated AKT and ERK1/2 signaling pathways. | ||
| E3106 | Dioscoreae Nipponicae Rhizoma Extract | Dioscoreae Nipponicae Rhizoma Extract is extracted from the rhizome of Dioscorea nipponica, of which the main component decreases the phosphorylation in IGF-1R, which in turn inhibits the phosphorylation and activation of PI3K-AKT and Rap1-MEK signaling pathways, promoting cell apoptosis and Graves’ disease remission. | ||
| E1710 | LY2780301 | LY2780301 is a highly selective adenosine triphosphate (ATP)-competitive dual inhibitor of p70S6K and Akt. LY2780301 binds to Akt, inhibiting its activity, which consequently blocks the PI3K/Akt signaling pathway which leads to reduced cell proliferation and the initiation of apoptosis in tumor cells. | ||
| E3241 | Cinnamomi Ramulus Extract | Cinnamomi Ramulus Extract is extracted from Cinnamomi Ramulus, which can inhibit the growth of colon cancer cells via Akt/ERK signaling pathways. | ||
| E0020 | Lupenone | Lupenone (Lup-20(29)-en-3-one, lupeone) is an isolated compound exhibiting anti-oxidative, anti-inflammation, and anti-diabetic activities. Lupenone can protect SH-SY5y cells against METH-induced neuronal apoptosis through the PI3K/Akt pathway. | ||
| E3041 | Alpiniae Katsumadai Extract | Alpiniae Katsumadai Extract is extracted from Alpiniae Katsumadai, which can induce growth inhibition and autophagy‑related apoptosis by regulating the AMPK and Akt/mTOR/p70S6K signaling pathways in cancer cells. | ||
| S9315 | Praeruptorin A | Praeruptorin A, a naturally existing pyranocumarin, is isolated from the dried root of Peucedanum praeruptorum Dunn. Praeruptorin A inhibits p38/Akt-c-Fos-NFATc1 signaling and PLCγ-independent Ca2+ oscillation. Praeruptorin A can significantly upregulates multidrug resistance-associated protein 2 expression via the constitutive androstane receptor-mediated pathway in vitro, and this should be taken as an herb-drug interaction. | ||
| E3365 | Weigela Grandiflora Fortune Extract | Weigela Grandiflora Fortune Extract is extracted from Weigela Grandiflora Fortune, which decreases the infection-mediated expression of inflammatory mediators by inhibiting the AKT/NF-κB and MAPK signaling pathways. | ||
| S3309 | Solasodine | Solasodine (Purapuridine, Solancarpidine, Solasodin, Salasodine, Salasdine) is a poisonous alkaloid chemical compound that occurs in plants of the Solanaceae family. Solasodine reduces the mRNA level of matrix metalloproteinase-2 (MMP-2), MMP-9 and extracellular inducer of matrix metalloproteinase (EMMPRIN), but increases the expression of reversion-inducing cysteine-rich protein with kazal motifs (RECK). Solasodine downregulates oncogenic microRNA-21 (miR-21), which has been known to target RECK. Solasodine also reduces PI3K/Akt signaling pathways and downregulates expression of miR-21. | ||
| E4992New | Afuresertib hydrochloride | Afuresertib hydrochloride (GSK 2110183 hydrochloride, LAE002 hydrochloride) is an orally bioavailable, selective, ATP-competitive and potent inhibitor of pan-Akt kinase with Ki values of 0.08 nM, 2 nM, 2.6 nM for Akt1/Akt2/Akt3 respectively. | ||
| E5820New | AKT Kinase Inhibitor | AKT Kinase Inhibitor is an inhibitor of Akt that reverses the protective effects of Tadalafil (TAD) in LPS-induced RWPE-1 cells by decreasing cell viability, promoting apoptosis, and increasing pro-inflammatory cytokine levels. | ||
| S9054 | Pectolinarin | Pectolinarin is a major compound in Cirsium setidens with anti-inflammatory activity. Pectolinarin inhibits secretion of IL-6 and IL-8, as well as the production of PGE2 and NO. Pectolinarin induces apoptosis via inactivation of the PI3K/Akt pathway. | ||
| E0945 | BIA | BIA (TMBIM6 antagonist BIA), a potential TMBIM6 antagonist, is an inhibitor of the interaction between TMBIM6 (Transmembrane B cell lymphoma 2-associated X protein (BAX) inhibitor motif-containing 6) and mTORC2, which ultimately blocks AKT activation and cancer progression. | ||
| E7359New | A-674563 | A-674563 is a selective, orally active inhibitor of Akt1 with a Ki of 11 nM. It also inhibits PKA with Ki of 16 nM and CDK2 with Ki of 46 nM. A-674563 reduces Akt downstream target phosphorylation and inhibits tumor cell proliferation in vitro with an EC50 of 0.4 µM. | ||
| E2682 | RPI-1 | RPI-1 inhibits proliferation of human papillary thyroid carcinoma cell line TPC-1 by inducing accumulation of cells at the G2 cell cycle phase, abolishes Ret/Ptc1 tyrosine phosphorylation along with its binding to Shc and phospholipase Cg, also abolishes the activation of JNK2 and AKT. | ||
| S8500 | BAY1125976 | BAY 1125976 is a selective allosteric AKT1/2 inhibitor,exhibits high efficacy on AKT signaling-dependent tumor growth in mouse models. BAY1125976 inhibits the activity of AKT1 (IC50 = 5.2 nM at 10 µM ATP and 44 nM at 2 mM ATP) and AKT2 (IC50 = 18 nM at 10 µM ATP and 36 nM at 2 mM ATP) very potently.Whereas BAY1125976 is almost inactive on AKT3 (IC50 = 427 nM at 10 µM ATP). | ||
| E2826 | Hematein | Hematein inhibits casein kinase II activity in a selective, dose-dependent and ATP non-competitive manner in vitro, with IC50 of 0.55 μM in the presence of 10 μM ATP. | ||
| S9611 | ABTL-0812 | ABTL0812 (α-Hydroxylinoleic acid, LP-10218, SCLN-0812) inhibits Akt/mTOR axis by inducing the overexpression of TRIB3 and activating autophagy in lung squamous carcinoma cell lines. ABTL0812 also induces AMPK activation and ROS accumulation. | ||
| E2401 | SPP-86 | SPP-86, a potent and selective cell permeable inhibitor of rearranged during transfection (RET) tyrosine kinase with an IC50 of 8 nM, inhibits RET-induced phosphatidylinositide 3-kinases (PI3K)/Akt and MAPK signaling, also inhibits RET-induced estrogen receptorα (ERα) phosphorylation in MCF7 cells. | ||
| S1321 | Urolithin B | Urolithin B inhibits NF-κB activity by reducing the phosphorylation and degradation of IκBα. Urolithin B suppresses the phosphorylation of JNK, ERK, and Akt, and enhances the phosphorylation of AMPK. Urolithin B is also a regulator of skeletal muscle mass. | ||
| S1273 | Amarogentin | Amarogentin (AG), a secoiridoid glycoside mainly extracted from Swertia and Gentiana roots, exhibits anti-oxidative, anti-tumour, and anti-diabetic activities. Amarogentin is an agonist for the bitter taste receptor TAS2R1 and inhibits in LAD-2 cells substance P-induced production of newly synthesized TNF-α. Amarogentin induces apoptosis in human gastric cancer cells (SNU-16) through G2/M cell cycle arrest and PI3K/Akt signalling pathway. Amarogentin (AG) interacts with the α2 subunit of AMP-activated protein kinase (AMPK) and activates the trimeric kinase with EC50 of 277 pM. |
||
| S3241 | Loureirin A | Loureirin A is a flavonoid extracted from the red resin of the herbs of Dracaena cochinchinensis, which is known as Dragon's Blood. Loureirin A inhibits platelet activation by an impairment of PI3K/Akt signaling. Loureirin A inhibits Akt phosphorylation. | ||
| E1604 | Vevorisertib trihydrochloride | Vevorisertib trihydrochloride(ARQ 751 trihydrochloride) is a selective, allosteric inhibitor of pan-AKT and AKT1-E17K mutant with Kd values of 1.2 nM and 8.6 nM, respectively. It also has IC50 values of 0.55, 0.81, and 1.3 nM for AKT1, AKT2, and AKT3, respectively and can be used for the research of cancer. | ||
| S7863 | SC79 | SC79 is a brain-penetrable Akt phosphorylation activator and an inhibitor of Akt-PH domain translocation. |

|
|
| S5144 | Neferine | Neferine ((R)-1,2-Dimethoxyaporphine), a natural component of Nelumbo nucifera, has antitumor efficiency. Neferine induces apoptosis in renal cancer cells. Neferine prevents autophagy through activation of Akt/mTOR pathway and Nrf2 in muscle cells. Neferine strongly inhibits NF-κB activation. Neferine possesses a number of therapeutic effects such as anti-diabetic, anti-aging, anti-microbial, anti-thrombotic, anti-arrhythmic, anti-inflammatory and even anti-HIV. | ||
| S3294 | Demethyl-Coclaurine | Demethyl-Coclaurine (Higenamine, Norcoclaurine), the key component of the Chinese herb aconite root, is a beta-2 adrenergic receptor (β2-AR) agonist. Demethyl-Coclaurine stimulates AKT phosphorylation and requires PI3K activation for the anti-apoptotic effect in cardiomyocytes. | ||
| E2947 | Recilisib | Recilisib (ON01210, EX-RAD) is a radioprotectant that activates the activity of AKT and PI3K in cells. It has been studied as prophylactic (use prior to radiation exposure) and therapeutic (after exposure to radiation) drug. | ||
| S0765 | MAZ51 | MAZ51 is a potent and selective inhibitor of vascular endothelial growth factor receptor (VEGFR)-3 (Flt-4) tyrosine kinase. MAZ51 induces cell rounding and G2/M cell cycle arrest in glioma cells through phosphorylation of Akt/GSK3β and activation of RhoA. MAZ51 inhibits the proliferation and induces the apoptosis of a variety of non-VEGFR-3-expressing tumor cell lines. | ||
| S6760 | LM22B-10 | LM22B-10 is a small molecule TrkB/TrkC neurotrophin receptor co-activator, LM22B-10 selectively activates TrkB, TrkC, AKT and ERK in vivo and in vitro. | ||
| E0785 | YS-49 | YS-49 is a PI3K/Akt (a downstream target of RhoA) activator, to reduce RhoA/PTEN activation in the 3-methylcholanthrene-treated cells, inhibits angiotensin II (Ang II)-stimulated proliferation of VSMCs via induction of heme oxygenase (HO)-1, also is an isoquinoline compound alkaloid, has a strong positive inotropic action through activation of cardiac β-adrenoceptors. | ||
| S4572 | Homosalate | Homosalate (HMS, Homomenthyl salicylate) is an organic ultraviolet filter used in most sunscreens but has been reported to be toxic to marine organisms. Homosalate aggravates the invasion of human trophoblast cells as well as regulates intracellular signaling pathways including PI3K/AKT and MAPK pathways. | ||
| S3289 | Daphnoretin | Daphnoretin (Dephnoretin, Thymelol), a biologically active compound isolated from Wikstroemia indica C.A. Mey., is a protein kinase C (PKC) activator. Daphnoretin inhibits the proliferation, invasion, and migration of tumor cells and promote its apoptosis by regulating the activity of Akt signal pathway. | ||
| S9514 | Rotundic acid | Rotundic acid (Rutundic acid), a natural compound, exhibit cytotoxic activities toward human hepatocellular carcinoma (HepG2), malignant melanoma (A375), SCLC (NCI-H446), breast cancer (MCF-7), and colon cancer (HT-29) cell lines.RA induces cell cycle arrest, DNA damage, and apoptosis by modulating the AKT/mTOR and MAPK pathways. | ||
| S8961 | Alobresib (GS-5829) | Alobresib (GS-5829) is a novel BET inhibitor that represents a highly effective therapeutics agent against recurrent/chemotherapy-resistant USC-overexpressing c-Myc. Alobresib (GS-5829) inhibits CLL cell proliferation and induces leukemia cell apoptosis through deregulation of key signaling pathways, such as BLK, AKT, ERK1/2, and MYC. Alobresib (GS-5829) also inhibits NF-κB signaling. | ||
| E2391 | α-Linolenic acid | α-Linolenic acid, an essential fatty acid isolated from seed oils, affects the process of thrombotic through the modulation of PI3K/Akt signaling, and possesses the anti-arrhythmic properties. | ||
| S6885 | Ailanthone | Ailanthone (AIL, Δ13-Dehydrochaparrinone), a natural anti-hepatocellular carcinoma (HCC) component in Ailanthus altissima, induces G0/G1-phase cell cycle arrest by decreasing expression of cyclins and CDKs and increases expression of p21 and p27. Ailanthone triggers DNA damage characterized by activation of the ATM/ATR pathway. Ailanthone induces apoptosis which is mitochondrion-mediated and involves the PI3K/AKT signaling pathway in Huh7 cells. Ailanthone is also a potent inhibitor of both full-length Androgen Receptor (AR-FL) and constitutively active truncated AR splice variants (AR-Vs, AR1-651) with IC50 of 69 nM and 309 nM, respectively. | ||
| E4864New | Pentamidine | Pentamidine (MP-601205) is an aromatic diamidine drug, an antagonist of TLR4.It also inhibits PI3K/AKT signaling pathway and reduce the expression of MMP-2 and MMP-9. It exhibits anti- protozoal, anti-inflammatory, and anti-tumor activities and is used as an agent for treating African trypanosomiasis, antimony resistant leishmaniasis and Pneumocystis carinii pneumonia. | ||
| E4992New | Afuresertib hydrochloride | Afuresertib hydrochloride (GSK 2110183 hydrochloride, LAE002 hydrochloride) is an orally bioavailable, selective, ATP-competitive and potent inhibitor of pan-Akt kinase with Ki values of 0.08 nM, 2 nM, 2.6 nM for Akt1/Akt2/Akt3 respectively. | ||
| E5820New | AKT Kinase Inhibitor | AKT Kinase Inhibitor is an inhibitor of Akt that reverses the protective effects of Tadalafil (TAD) in LPS-induced RWPE-1 cells by decreasing cell viability, promoting apoptosis, and increasing pro-inflammatory cytokine levels. | ||
| E7359New | A-674563 | A-674563 is a selective, orally active inhibitor of Akt1 with a Ki of 11 nM. It also inhibits PKA with Ki of 16 nM and CDK2 with Ki of 46 nM. A-674563 reduces Akt downstream target phosphorylation and inhibits tumor cell proliferation in vitro with an EC50 of 0.4 µM. |