- Bioactive Compounds
- By Signaling Pathways
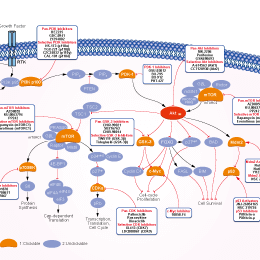
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Afuresertib (GSK2110183)
Afuresertib (GSK2110183) is a potent, orally bioavailable Akt inhibitor with Ki of 0.08 nM, 2 nM, and 2.6 nM for Akt1, Akt2, and Akt3, respectively. Phase 2.
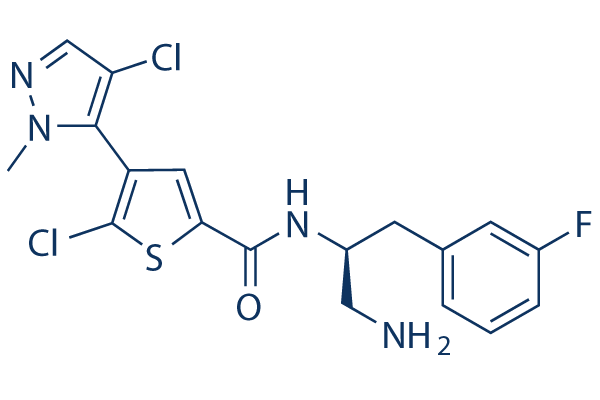
Afuresertib (GSK2110183) Chemical Structure
CAS: 1047644-62-1
Selleck's Afuresertib (GSK2110183) has been cited by 31 publications
Purity & Quality Control
Batch:
Purity:
99.61%
99.61
Afuresertib (GSK2110183) Related Products
| Related Targets | Akt1 Akt2 Akt3 | Click to Expand |
|---|---|---|
| Related Compound Libraries | Kinase Inhibitor Library PI3K/Akt Inhibitor Library Apoptosis Compound Library Cell Cycle compound library NF-κB Signaling Compound Library | Click to Expand |
Signaling Pathway
Choose Selective Akt Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Sf9 | Function assay | 40 mins | Inhibition of full length human AKT1 expressed in Sf9 cells assessed as reduction in substrate phosphorylation using biotin-ahx-ARKRERAYSFGHHA-amide substrate and [gamma-33P]ATP incubated for 40 mins by top count microplate scintillation counting method, IC50 = 0.001 μM. | ChEMBL | ||
| Sf9 | Function assay | 40 mins | Inhibition of full length human AKT3 expressed in Sf9 cells assessed as reduction in substrate phosphorylation using biotin-ahx-ARKRERAYSFGHHA-amide substrate and [gamma-33P]ATP incubated for 40 mins by top count microplate scintillation counting method, IC50 = 0.001585 μM. | ChEMBL | ||
| COLO205 | Growth inhibition assay | 72 hrs | Growth inhibition of human COLO205 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs , IC50 = 0.009 μM. | ChEMBL | ||
| Sf9 | Function assay | 40 mins | Inhibition of full length human AKT2 expressed in Sf9 cells assessed as reduction in substrate phosphorylation using biotin-ahx-ARKRERAYSFGHHA-amide substrate and [gamma-33P]ATP incubated for 40 mins by top count microplate scintillation counting method, IC50 = 0.01 μM. | ChEMBL | ||
| MV522 | Growth inhibition assay | 72 hrs | Growth inhibition of human MV522 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.012 μM. | ChEMBL | ||
| HT-29 | Growth inhibition assay | 72 hrs | Growth inhibition of human HT-29 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.015 μM. | ChEMBL | ||
| NCI-H727 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H727 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.019 μM. | ChEMBL | ||
| SW1417 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW1417 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.03 μM. | ChEMBL | ||
| Calu6 | Growth inhibition assay | 72 hrs | Growth inhibition of human Calu6 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.031 μM. | ChEMBL | ||
| LS1034 | Growth inhibition assay | 72 hrs | Growth inhibition of human LS1034 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.04 μM. | ChEMBL | ||
| SW1463 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW1463 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.041 μM. | ChEMBL | ||
| RKO | Growth inhibition assay | 72 hrs | Growth inhibition of human RKO cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.054 μM. | ChEMBL | ||
| NCI-H508 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H508 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.08 μM. | ChEMBL | ||
| NCI-H1155 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1155 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hr, IC50 = 0.101 μM. | ChEMBL | ||
| A427 | Growth inhibition assay | 72 hrs | Growth inhibition of human A427 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.103 μM. | ChEMBL | ||
| KM12 | Growth inhibition assay | 72 hrs | Growth inhibition of human KM12 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.104 μM. | ChEMBL | ||
| HCT8 | Growth inhibition assay | 72 hrs | Growth inhibition of human HCT8 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.143 μM. | ChEMBL | ||
| MDA-MB-175-VII | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-175-VII cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.155 μM. | ChEMBL | ||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.164 μM. | ChEMBL | ||
| NCI-H23 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H23 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs , IC50 = 0.199 μM. | ChEMBL | ||
| SW837 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW837 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.248 μM. | ChEMBL | ||
| SW480 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW480 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.259 μM. | ChEMBL | ||
| NCI-H1355 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1355 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hr, IC50 = 0.268 μM. | ChEMBL | ||
| EFM19 | Growth inhibition assay | 72 hrs | Growth inhibition of human EFM19 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.3 μM. | ChEMBL | ||
| T84 | Growth inhibition assay | 72 hrs | Growth inhibition of human T84 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.304 μM. | ChEMBL | ||
| COR-L23 | Growth inhibition assay | 72 hrs | Growth inhibition of human COR-L23 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs , IC50 = 0.368 μM. | ChEMBL | ||
| NCI-H1792 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1792 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hr, IC50 = 0.374 μM. | ChEMBL | ||
| SW1573 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW1573 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.382 μM. | ChEMBL | ||
| Calu3 | Growth inhibition assay | 72 hrs | Growth inhibition of human Calu3 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.392 μM. | ChEMBL | ||
| HCC827 | Growth inhibition assay | 72 hrs | Growth inhibition of human HCC827 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.401 μM. | ChEMBL | ||
| EFM19 | Growth inhibition assay | 72 hrs | Growth inhibition of human EFM19 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 0.43 μM. | ChEMBL | ||
| HOP62 | Growth inhibition assay | 72 hrs | Growth inhibition of human HOP62 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.498 μM. | ChEMBL | ||
| NCI-H508 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H508 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 0.522 μM. | ChEMBL | ||
| SW900 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW900 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.719 μM. | ChEMBL | ||
| MDA-MB-175-VII | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-175-VII cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 0.794 μM. | ChEMBL | ||
| NCI-H1155 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1155 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 0.865 μM. | ChEMBL | ||
| DLD1 | Growth inhibition assay | 72 hrs | Growth inhibition of human DLD1 cells at 1:10 molar ratio of compound in presence of N-{3-[3-cyclopropyl-5-(2-fluoro-4-iodo-phenylamino)-6,8-dimethyl- 2,4,7-trioxo-3,4,6,7-tetrahydro-2H-pyrido[4,3-d]pyrimidin-1-yl]phenyl}acetamide incubated for 72 hrs, IC50 = 0.932 μM. | ChEMBL | ||
| HT-29 | Growth inhibition assay | 72 hrs | Growth inhibition of human HT-29 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 1.383 μM. | ChEMBL | ||
| HCT8 | Growth inhibition assay | 72 hrs | Growth inhibition of human HCT8 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 1.541 μM. | ChEMBL | ||
| RKO | Growth inhibition assay | 72 hrs | Growth inhibition of human RKO cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 1.806 μM. | ChEMBL | ||
| Calu6 | Growth inhibition assay | 72 hrs | Growth inhibition of human Calu6 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 2.154 μM. | ChEMBL | ||
| KM12 | Growth inhibition assay | 72 hrs | Growth inhibition of human KM12 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 2.247 μM. | ChEMBL | ||
| COLO205 | Growth inhibition assay | 72 hrs | Growth inhibition of human COLO205 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 2.567 μM. | ChEMBL | ||
| MV522 | Growth inhibition assay | 72 hrs | Growth inhibition of human MV522 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 3.044 μM. | ChEMBL | ||
| LS1034 | Growth inhibition assay | 72 hrs | Growth inhibition of human LS1034 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 4.339 μM. | ChEMBL | ||
| NCI-H727 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H727 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 4.799 μM. | ChEMBL | ||
| COR-L23 | Growth inhibition assay | 72 hrs | Growth inhibition of human COR-L23 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 5.373 μM. | ChEMBL | ||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 5.993 μM. | ChEMBL | ||
| NCI-H1355 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1355 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 6.685 μM. | ChEMBL | ||
| NCI-H23 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H23 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 7.124 μM. | ChEMBL | ||
| NCI-H1792 | Growth inhibition assay | 72 hrs | Growth inhibition of human NCI-H1792 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 7.307 μM. | ChEMBL | ||
| SW1463 | Growth inhibition assay | 72 hrs | Growth inhibition of human SW1463 cells incubated for 72 hrs by Cell-titer-Glo reagent based assay, IC50 = 9.725 μM. | ChEMBL | ||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Afuresertib (GSK2110183) is a potent, orally bioavailable Akt inhibitor with Ki of 0.08 nM, 2 nM, and 2.6 nM for Akt1, Akt2, and Akt3, respectively. Phase 2. | ||||||
|---|---|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | Afuresertib inhibits the kinase activity of the E17K AKT1 mutant protein with EC50 of 0.2 nM. Afuresertib shows a concentration-dependent effect on multiple AKT substrate phosphorylation levels, including GSK3b, PRAS40, FOXO and Caspase 9. Overall 65% of the hematological cell lines are sensitive to afuresertib (EC50 < 1 μM). Among tested solid tumor cell lines, 21% have EC50 < 1 μM in response to afuresertib.[1] | |||
|---|---|---|---|---|
| Kinase Assay | Potency (Ki*) of afuresertib | |||
| The true potency (Ki*) of the inhibitor is initially determined at low enzyme concentrations (0.1 nM AKT1, 0.7 nM AKT2, and 0.2 nM AKT3) using a filter binding assay and then confirmed with progress curve analysis. In the filter binding assay, a pre-mix of enzyme plus inhibitor is incubated for 1 h and then added to a GSKα peptide (Ac-KKGGRARTSS-FAEPG-amide) and [γ33P] ATP. Reactions are terminated after 2 h and the radio labeled AKT peptide product is captured in a phospho-cellulose filter plate. Progress curve analysis utilizes continuous real-time fluorescence detection of product formation using the Sox-AKT-tide substrate (Ac-ARKRERAYSF-d-Pro-Sox-Gly-NH2). | ||||
| Cell Research | Cell lines | Hematological cell lines and solid tumor cell lines | ||
| Concentrations | 30 μM | |||
| Incubation Time | 72 h | |||
| Method | A 3-day proliferation assay using CellTiter-Glo is performed to measure the growth inhibition by the compounds at 0-30 μM. Cell growth is determined relative to untreated (DMSO) controls. EC50’s are calculated from inhibition curves using a 4- or 6-parameter fitting algorithm in the Assay Client application. | |||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | p-GSK3 / GSK3 / p-PRAS40 / PRAS40 / p-FOXO / p-Casp9 / p-AKT / AKT / p-MEK / p-ERK / Erk ATG3 / ATG12 / LC3B |
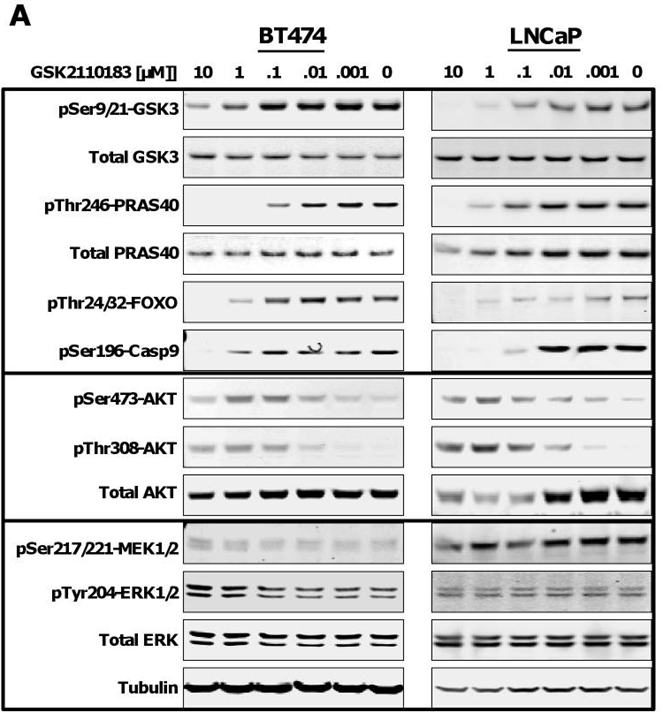
|
24978597 | |
| In Vivo | ||
| In vivo | Mice bearing BT474 breast tumor xenografts are dosed with afuresertib (p.o.) at 10, 30 or 100 mg/kg daily which results in 8, 37 and 61% TGI, respectively. Mice bearing SKOV3 ovarian tumor xenografts are treated with 10, 30 and 100 mg/kg afuresertib which results in 23, 37 and 97% TGI, respectively.[1] | |
|---|---|---|
| Animal Research | Animal Models | Female athymic nude and SCID mice bearing SKOV3 or BT474 tumors |
| Dosages | 100 mg/kg | |
| Administration | p.o. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05489744 | Completed | Healthy Volunteer |
Laekna Limited |
August 6 2022 | Phase 1 |
| NCT04374630 | Active not recruiting | Platinum-resistant Ovarian Cancer |
Laekna Limited |
June 9 2020 | Phase 2 |
| NCT04060394 | Active not recruiting | Metastatic Castration-resistant Prostate Cancer |
Laekna Limited |
September 13 2019 | Phase 1|Phase 2 |
| NCT02235740 | Terminated | Cancer |
Novartis|Amgen |
November 2014 | Phase 1 |
Chemical Information & Solubility
| Molecular Weight | 427.32 | Formula | C18H17Cl2FN4OS |
| CAS No. | 1047644-62-1 | SDF | Download Afuresertib (GSK2110183) SDF |
| Smiles | CN1C(=C(C=N1)Cl)C2=C(SC(=C2)C(=O)NC(CC3=CC(=CC=C3)F)CN)Cl | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 85 mg/mL ( (198.91 mM); Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Ethanol : 85 mg/mL Water : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy Afuresertib (GSK2110183) | Afuresertib (GSK2110183) supplier | purchase Afuresertib (GSK2110183) | Afuresertib (GSK2110183) cost | Afuresertib (GSK2110183) manufacturer | order Afuresertib (GSK2110183) | Afuresertib (GSK2110183) distributor