- Inhibitors
- Antibodies
- Compound Libraries
- New Products
- Contact Us
research use only
Golvatinib (E7050) c-Met inhibitor
Cat.No.S2859
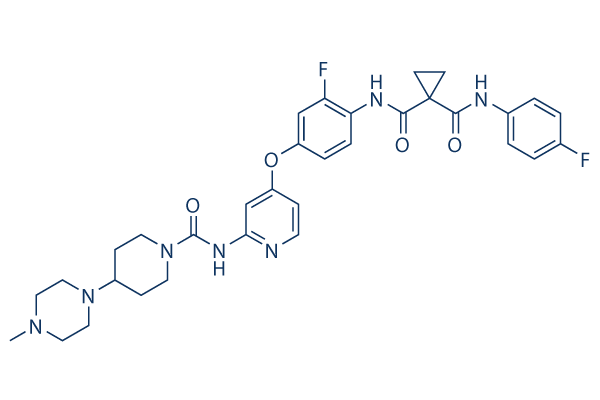
Chemical Structure
Molecular Weight: 633.69
Jump to
Quality Control
| Related Targets | EGFR VEGFR PDGFR FGFR Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other c-Met Products | Tepotinib Dihexa SGX-523 Foretinib PHA-665752 SU11274 BMS-777607 JNJ-38877605 Tivantinib PF-04217903 |
Solubility
|
In vitro |
DMSO
: 20 mg/mL
(31.56 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
Dilution Calculator
Molecular Weight Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
%
DMSO
%
%
Tween 80
%
ddH2O
%
DMSO
+
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 633.69 | Formula | C33H37F2N7O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 928037-13-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1CCN(CC1)C2CCN(CC2)C(=O)NC3=NC=CC(=C3)OC4=CC(=C(C=C4)NC(=O)C5(CC5)C(=O)NC6=CC=C(C=C6)F)F | ||
Mechanism of Action
| Targets/IC50/Ki |
c-Met
14 nM
VEGFR2
16 nM
|
|---|---|
| In vitro |
In vitro studies indicate that Golvatinib (E7050) potently inhibits phosphorylation of both c-Met and VEGFR-2. It also potently represses the growth of both c-met amplified tumor cells and endothelial cells stimulated with either HGF or VEGF. This compound circumvents resistance to all of the reversible, irreversible, and mutant-selective EGFR-TKIs induced by exogenous and/or endogenous HGF in EGFR mutant lung cancer cell lines, by blocking the Met/Gab1/PI3K/Akt pathway in vitro. It also prevents the emergence of gefitinib-resistant HCC827 cells induced by continuous exposure to HGF.
|
| Kinase Assay |
Western blot analysis
|
|
The phosphorylation status of c-Met and VEGFR-2 is detected by Western blot analysis. For c-Met, MKN45 cells are incubated with a serial dilution of Golvatinib (E7050) in complete medium at 37 °C for 2 h. For VEGFR-2, HUVEC are starved with human endothelial serum free medium containing 0.5% FBS for 24 h. Subsequently HUVEC are incubated with a serial dilution of this compound for 1 h and then incubated with 20 ng/mL of human VEGF for 5 min. Cells are lysed by lysis buffer (50 mM HEPES [pH 7.4], 150 mM NaCl, 10% glycerol, 1% Triton X-100, 1.5 mM MgCl2, 1 mM EDTA [pH 8.0], 100 mM NaF, 1 mM phenylmethylsulfonyl fluoride 1 mM sodium orthovanadate, 10 μg/mL aprotinin, 50 μg/mL leupeptin, and 1 μg/mL pepstatin A). The resected tumor samples are homogenized with lysis buffer containing 25 mM β-glycerophosphate and 0.5% (v/v) phosphatase inhibitor cocktail 2 at 4 °C. Cellular debris is removed by centrifugation at 17 860g for 20 min at 4 °C. Aliquots of the supernatants containing 5-20 μg of protein are subjected to SDS-PAGE under reducing conditions. The proteins are then transferred onto PVDF membranes, blocked with TBS containing 0.05% Tween-20 and either 5% skim milk or 5% BSA. The membranes are probed with the following antibodies: anti-c-Met polyclonal antibody (C-28) and anti-VEGFR-2 polyclonal antibody (C-20); mouse anti-phosphotyrosine clone 4G10; and anti-VEGFR-2 polyclonal antibody, anti-phospho-VEGFR-2 (Tyr996) polyclonal antibody, and anti-phospho-c-Met (Tyr1234/1235) polyclonal antibody. Detection is performed using a Super Signal enhanced chemiluminescence kit. Immunoreactive bands are visualized by chemiluminescence with an Image Master-VDS-CL detection system. The intensity of each band is measured by using an image analyzer.
|
|
| In vivo |
Golvatinib (E7050) shows inhibition of the phosphorylation of c-Met and VEGFR-2 in tumors in vivo, with strong suppression of tumor growth and angiogenesis in xenograft models. Treatment of some tumor lines containing c-met amplifications with high doses of this compound (50–200 mg/kg) induces tumor regression and disappearance. In a peritoneal dissemination model, it demonstrates an antitumor effect against peritoneal tumors as well as a significant prolongation of lifespan in treated mice. In another xenograft study, tumors produced by HGF-transfected Ma-1 (Ma-1/HGF) cells are more angiogenic than vector control tumors and show resistance to ZD1839. E7050 alone inhibits angiogenesis and retards growth of Ma-1/HGF tumors, and when combined with ZD1839, it induces marked regression of tumor growth.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01355302 | Terminated | Advanced or Metastatic Solid Tumors|Previously Untreated Gastric Cancer |
Eisai Inc.|Quintiles Inc. |
November 2011 | Phase 1|Phase 2 |
| NCT01433991 | Terminated | Advanced Solid Tumors |
Eisai Inc. |
October 13 2011 | Phase 1|Phase 2 |
| NCT01332266 | Completed | Platinum-Resistant Squamous Cell Carcinoma of the Head and Neck |
Eisai Inc.|PharmaBio Development Inc. |
September 19 2011 | Phase 1|Phase 2 |
| NCT01271504 | Completed | Hepatocellular Carcinoma |
Eisai Inc.|PharmaBio Development Inc. |
July 19 2011 | Phase 1|Phase 2 |
| NCT02533102 | Completed | Healthy Subjects |
Eisai Limited|Eisai Inc. |
November 2010 | Phase 1 |
| NCT00921869 | Completed | Solid Tumors |
Eisai Co. Ltd.|Eisai Inc. |
October 2009 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































