research use only
SU11274 c-Met inhibitor
Cat.No.S1080
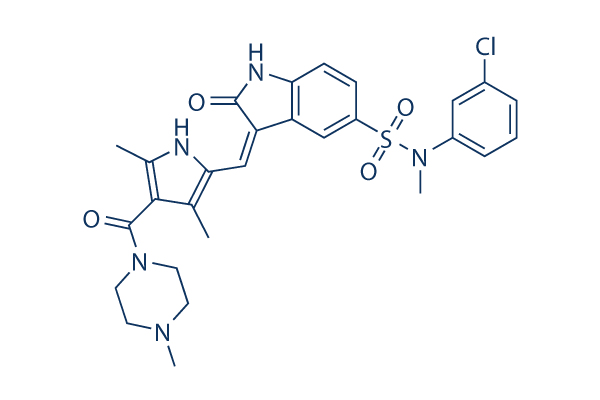
Chemical Structure
Molecular Weight: 568.09
Quality Control
| Related Targets | EGFR VEGFR PDGFR FGFR Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other c-Met Inhibitors | Tepotinib Dihexa SGX-523 Foretinib PHA-665752 BMS-777607 JNJ-38877605 Tivantinib PF-04217903 Savolitinib (AZD6094) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| A549 cells | Function assay | Inhibition of human recombinant c-MET kinase in A549 cells assessed as inhibition of HGF-induced cell growth, IC50=0.01 μM | ||||
| human MDCK cells | Function assay | 24 h | Inhibition of Met-mediated scattering in HGF-stimulated human MDCK cells pre-incubated overnight prior to HGF stimulation for 24 hrs measured after 24 to 48 hrs, IC50=0.152 μM | |||
| mouse BAF3 cells | Proliferation assay | 72 h | Antiproliferative activity against mouse BAF3 cells expressing TPR-Met after 72 hrs in absence of IL-3, IC50=0.53 μM | |||
| human SNU5 cells | Proliferation assay | 72 h | Antiproliferative activity against human SNU5 cells after 72 hrs, IC50=0.8 μM | |||
| human MKN45 cells | Proliferation assay | 72 h | Antiproliferative activity against human MKN45 cells after 72 hrs, IC50=1.3 μM | |||
| human HepG2 cells | Function assay | Inhibition of Met-mediated tumorigenesis in HGF-stimulated human HepG2 cells assessed as impairment in anchorage-independent growth by soft agar growth assay, IC50=1.561 μM | ||||
| mouse NIH/3T3 cells | Proliferation assay | 72 h | Antiproliferative activity against mouse NIH/3T3 cells expressing TPR-Met after 72 hrs, IC50=2 μM | |||
| human MCF7 cells | Proliferation assay | 72 h | Antiproliferative activity against human MCF7 cells after 72 hrs, IC50=6.2 μM | |||
| human SNU1 cells | Proliferation assay | 72 h | Antiproliferative activity against human SNU1 cells after 72 hrs, IC50=7 μM | |||
| human NCI-H1993 cells | Proliferation assay | 72 h | Antiproliferative activity against human NCI-H1993 cells after 72 hrs, IC50=7.3 μM | |||
| human MDA-MB-231 cells | Proliferation assay | 72 h | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs | |||
| human NCI-H441 cells | Proliferation assay | 72 h | Antiproliferative activity against human NCI-H441 cells after 72 hrs | |||
| human BxPC3 cells | Proliferation assay | 72 h | Antiproliferative activity against human BxPC3 cells after 72 hrs | |||
| DLD1 cells | Function assay | 2.5 μM | Inhibition of human p38-alpha phosphorylation in DLD1 cells at 2.5 μM | |||
| DLD1 cells | Function assay | 2.5 μM | 16 h | Inhibition of human MET receptor in DLD1 cells at 2.5 uM after 16 hrs by Western blot | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 92 mg/mL
(161.94 mM)
Ethanol : 2 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 568.09 | Formula | C28H30CIN5O4S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 658084-23-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | PKI-SU11274 | Smiles | CC1=C(NC(=C1C(=O)N2CCN(CC2)C)C)C=C3C4=C(C=CC(=C4)S(=O)(=O)N(C)C5=CC(=CC=C5)Cl)NC3=O | ||
Mechanism of Action
| Targets/IC50/Ki |
Met
(Cell-free assay) 0.01 μM
|
|---|---|
| In vitro |
SU11274 exhibits greater than 50-fold selectivity for Met versus Flk and more than 500 times selectivity versus other tyrosine kinases such as FGFR-1, c-src, PDGFbR, and EGFR. This compound inhibits the phosphorylation of key regulators of the PI3K pathway, including AKT, FKHR, or GSK3β. Treatment with this chemical inhibits the growth of TPR-MET-transformed BaF3 cells in a dose-dependent manner with IC50 of <3 μM in the absence of interleukin 3, without growth inhibition of BaF3 cells transformed by other oncogenic tyrosine kinases, including BCR-ABL, TEL-JAK2, TEL-ABL, and TEL-PDGFβR. In addition to cell growth, this compound treatment significantly inhibits the migration of BaF3. TPR-MET cells by 44.8% and 80% at 1 μM and 5 μM, respectively. It inhibits HGF-dependent phosphorylation of Met as well as HGF-dependent cell proliferation and motility with an IC50 of 1-1.5 μM. In H69 and H345 cells which have functional Met receptor, this inhibitor inhibits the HGF-induced cell growth with IC50 of 3.4 μM and 6.5 μM, respectively. It induces G1 cell cycle arrest with cells in G1 phase increased from 42.4% to 70.6% at 5 μM, and induces caspase-dependent apoptosis by 24% at 1 μM. This compound inhibits cell viability in c-Met-expressing non-small cell lung cancer (NSCLC) cells with IC50 values of 0.8-4.4 μM, and abrogates hepatocyte growth factor-induced phosphorylation of c-Met and its downstream signaling.
|
| Kinase Assay |
In vitro Met kinase assay
|
|
A chimeric protein is constructed containing the cytoplasmic domain of human c-Met fused to Glutathione S-transferase (GST) and expressed in SF9 cells. The c-Met kinase GST-fusion protein is used for an ELISA-based Met biochemical assay using the random copolymer poly(Glu:Tyr) (4:1) immobilized on microtiter plates as a substrate. IC50 value is determined with various concentrations of SU11274 in a buffer containing 5 μM ATP and 10 mM MnCl2, 50 mM HEPES (pH 7.5), 25 mM NaCl, 0.01% BSA, and 0.1 mM Na orthovanadate. The kinase reaction is performed for 5 minutes at room temperature. The extent of substrate phosphorylation is measured using horseradish peroxidase-conjugated anti-pTyr antibodies.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Cell viability |
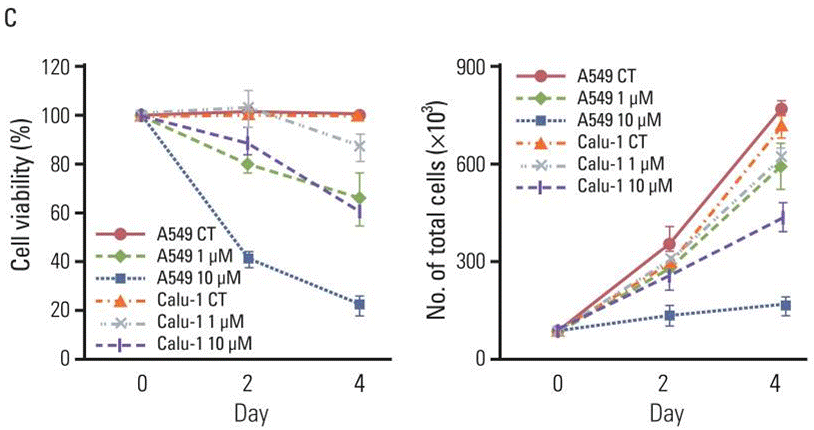
|
23341789 |
| Western blot | p-Met / Met / p-AKT / AKT / p-ERK / ERK PUMA / Bcl-2 / Bax |
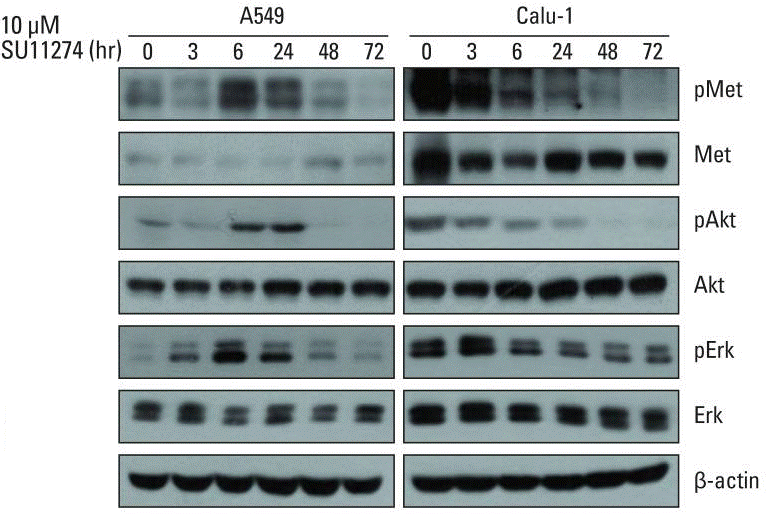
|
23341789 |
| Immunofluorescence | p-SphK1 |
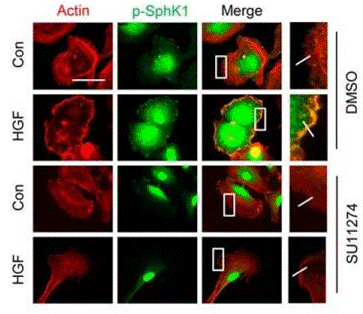
|
27864331 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What is the solubility of this compound in acetone?
Answer:
The solubility of S1080 in acetone is 7 mg/mL.






































