
- Inhibitors
- By product type
- Natural Products
- Inducing Agents
- Peptides
- Antibiotics
- Antibody-drug Conjugates(ADC)
- PROTAC
- Hydrotropic Agents
- Dyes
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
By research - Antibodies
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- Featured Products
- MRTX1133
- Nab-Paclitaxel
- KP-457
- IAG933
- RMC-6236 (Daraxonrasib)
- RMC-7977
- Zoldonrasib (RMC-9805)
- GsMTx4
- Navitoclax (ABT-263)
- TSA (Trichostatin A)
- Y-27632 Dihydrochloride
- SB431542
- SB202190
- MK-2206 Dihydrochloride
- LY294002
- Alisertib (MLN8237)
- XAV-939
- CHIR-99021 (Laduviglusib)
- Bafilomycin A1 (Baf-A1)
- Thiazovivin (TZV)
- CP-673451
- Verteporfin
- DAPT
- Galunisertib (LY2157299)
- MG132
- SBE-β-CD
- Tween 80
- Bavdegalutamide (ARV-110)
- Z-VAD-FMK
- Wnt-C59 (C59)
- IWR-1-endo
- (+)-JQ1
- 3-Deazaneplanocin A (DZNep) Hydrochloride
- RepSox (E-616452)
- Erastin
- Q-VD-Oph
- Puromycin Dihydrochloride
- Cycloheximide
- Telaglenastat (CB-839)
- A-83-01
- Ceralasertib (AZD6738)
- Liproxstatin-1
- Emricasan (IDN-6556)
- PMA (Phorbol 12-myristate 13-acetate)
- Dibutyryl cAMP (Bucladesine) sodium
- Nedisertib (M3814)
- PLX5622
- IKE (Imidazole Ketone Erastin)
- STM2457
- Saruparib (AZD5305)
- New Products
- Contact Us
research use only
Etoposide Topoisomerase II inhibitor
Cat.No.S1225
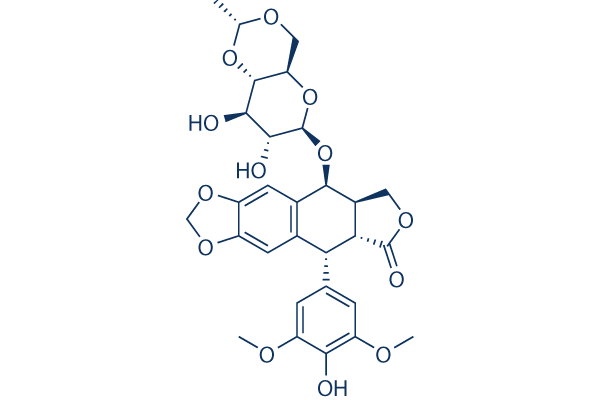
Chemical Structure
Molecular Weight: 588.56
Quality Control
Batch:
Purity:
99.96%
99.96
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Kelly | Growth Inhibition Assay | IC50=0.12 ± 0.01 μM | 25960282 | |||
| KellyCis83 | Growth Inhibition Assay | IC50=0.16 ± 0.02 μM | 25960282 | |||
| SK-N-AS | Growth Inhibition Assay | IC50=0.24 ± 0.03 μM | 25960282 | |||
| SK-N-ASCis24 | Growth Inhibition Assay | IC50=0.57 ± 0.11 μM | 25960282 | |||
| U87 | Growth Inhibition Assay | 0-50 μM | 48 h | decreases cell viability which can be enhanced by silibinin | 25750273 | |
| HCT116 | Growth Inhibition Assay | 0.5-2.5 μM | 48 h | IC50=1.73 ± 0.21 μM | 25746763 | |
| HT-29 | Growth Inhibition Assay | 0.5-2.5 μM | 48 h | IC50=7.2 ± 1.04 μM | 25746763 | |
| Caco2 | Growth Inhibition Assay | 0.5-2.5 μM | 48 h | IC50=7.26 ± 1.68 μM | 25746763 | |
| COLO 205 | Growth Inhibition Assay | 0.5-2.5 μM | 48 h | IC50=1.61 ± 0.02 μM | 25746763 | |
| SW480 | Growth Inhibition Assay | 0.5-2.5 μM | 48 h | IC50=4.92 ± 0.33 μM | 25746763 | |
| HEK293T | Growth Inhibition Assay | 1-5 μM | 48 h | IC50=2.42 ± 0.05 μM | 25746763 | |
| Hep3B | Function Assay | 10 μM | 48 h | reduces the enhancing effect of BMP-6 | 25633564 | |
| Hep3B | Function Assay | 0.1-10 μM | 24 h | suppresses the expression of hepcidin mRNA | 25633564 | |
| HEK293 | Growth Inhibition Assay | IC50=7.14 ± 0.36 μM | 25603122 | |||
| DU145 | Growth Inhibition Assay | IC50=2.28 ± 0.04 μM | 25603122 | |||
| HCT15 | Growth Inhibition Assay | IC50=0.81 ± 0.01 μM | 25603122 | |||
| T47D | Growth Inhibition Assay | IC50=3.18 ± 0.11 μM | 25603122 | |||
| SMMC-7721 | Function Assay | 40 μM | 48 h | DMSO | induces γH2AX foci formation | 25544361 |
| MDA-MB-231 | Growth Inhibition Assay | 72 h | IC50=21.2 ± 4.2 μM | 25486219 | ||
| MCF-7 | Growth Inhibition Assay | 72 h | IC50=10.9 ± 2.1 μM | 25486219 | ||
| Jurkat | Growth Inhibition Assay | 72 h | IC50=1.2 ± 1.5 μM | 25486219 | ||
| HeLa | Growth Inhibition Assay | 72 h | IC50=3.9 ± 2.3 μM | 25486219 | ||
| MCF7 | Growth Inhibition Assay | 5-100 μM | 7 d | inhibits cell growth in a dose-dependent manner | 25472619 | |
| K562 | Growth Inhibition Assay | 72 h | IC50=0.29 μM | 25282653 | ||
| K/VP.5 | Growth Inhibition Assay | 72 h | IC50=4.9 μM | 25282653 | ||
| SH-EP | Function Assay | 20 μg/ml | 24 h | increases the expression of endogenous DEPP | 25261981 | |
| SCC25 | Growth Inhibition Assay | 24 h | IC50=43.3 ± 1.12 μM | 25220729 | ||
| CAL27 | Growth Inhibition Assay | 24 h | IC50=52.1 ± 1.09 μM | 25220729 | ||
| FaDu | Growth Inhibition Assay | 24 h | IC50=25.89 ± 1.13 μM | 25220729 | ||
| SCC25 | Growth Inhibition Assay | 48 h | IC50=20.86 ± 1.07 μM | 25220729 | ||
| CAL27 | Growth Inhibition Assay | 48 h | IC50=18.24 ± 1.15 μM | 25220729 | ||
| FaDu | Growth Inhibition Assay | 48 h | IC50=6.43 ± 1.13 μM | 25220729 | ||
| SCC25 | Growth Inhibition Assay | 72 h | IC50=8.41 ± 1.11 μM | 25220729 | ||
| CAL27 | Growth Inhibition Assay | 72 h | IC50=4.27 ± 1.14 μM | 25220729 | ||
| FaDu | Growth Inhibition Assay | 72 h | IC50=5.02 ± 1.15 μM | 25220729 | ||
| MCF-7 | Growth Inhibition Assay | 48 h | DMSO | IC50=7.2 ± 0.8 μM | 25216378 | |
| T-47D | Growth Inhibition Assay | 48 h | DMSO | IC50=7.7 ± 0.7 μM | 25216378 | |
| MDA-MB-231 | Growth Inhibition Assay | 48 h | DMSO | IC50=12.8 ± 1.0 μM | 25216378 | |
| DU145 | Apoptosis Assay | 10-100 μM | 8 h | DMSO | induces cell death significantly in a very low concentration | 25149681 |
| DU145 stem-like | Apoptosis Assay | 10-100 μM | 8 h | DMSO | induces cell death in a dose-dependent manner | 25149681 |
| DU145 | Function Assay | 10-100 μM | 2 h | DMSO | increases the pCHK1 expression and decreases the pCHK1 expression in a dose-dependent manner | 25149681 |
| DU145 stem-like | Function Assay | 10-100 μM | 2 h | DMSO | increases the pCHK1 expression and decreases the pCHK1 expression in a dose-dependent manner | 25149681 |
| UW228-3 | Growth Inhibition Assay | 0.01-300 μM | 48 h | DMSO | IC50=0.99 μM | 25119185 |
| NSCs | Growth Inhibition Assay | 0.01-300 μM | 48 h | DMSO | IC50=0.3-3 μM | 25119185 |
| MKL-1 | Function Assay | 10-1000 nM | 4 d | induces the induction of MHC-I expression | 25116754 | |
| MCF7 EV | Function Assay | 10-100 μM | 2 h | induces production of γH2AX | 25088203 | |
| MCF 7BMI1 | Function Assay | 10-100 μM | 2 h | induces production of γH2AX | 25088203 | |
| MCF7 EV | Function Assay | 10-100 μM | 2 h | ETOP induces ATM activation | 25088203 | |
| MCF7 BMI1 | Function Assay | 10-100 μM | 2 h | ETOP induces ATM activation | 25088203 | |
| HepG2 | Growth Inhibition Assay | DMSO | IC50=30.16 ± 0.50 μM | 25078311 | ||
| MOLT-3 | Growth Inhibition Assay | DMSO | IC50=0.051 ± 0.002 μM | 25078311 | ||
| HT1080 | Growth Inhibition Assay | 1-100 μM | 4/24/48 h | DMSO | induces cell death significantly in a very low concentration | 25078064 |
| HT1080 | Function Assay | 0.0001-100 μM | 1-24 h | DMSO | induces p-p53(ser15) in both time- and concentration-dependent manner | 25078064 |
| HT1080 | Growth Inhibition Assay | 0.0001-100 μM | 24 h | DMSO | causes an increase in the number of cells in G2/M, while decreasing S and G1 phase cells | 25078064 |
| HD-MY-Z | Growth Inhibition Assay | 24/48/72 h | IC50>100 μM | 25048236 | ||
| DOHH-2 | Growth Inhibition Assay | 24 h | IC50>100 μM | 25048236 | ||
| DOHH-2 | Growth Inhibition Assay | 48 h | IC50=19.9 μM | 25048236 | ||
| DOHH-2 | Growth Inhibition Assay | 72 h | IC50=5 μM | 25048236 | ||
| REH | Growth Inhibition Assay | 24 h | IC50=0.027 μM | 25048236 | ||
| REH | Growth Inhibition Assay | 48 h | IC50=0.014 μM | 25048236 | ||
| REH | Growth Inhibition Assay | 72 h | IC50=0.015 μM | 25048236 | ||
| HH | Growth Inhibition Assay | 24 h | IC50=104.7 μM | 25048236 | ||
| HH | Growth Inhibition Assay | 48 h | IC50=48.6 μM | 25048236 | ||
| HH | Growth Inhibition Assay | 72 h | IC50=14.7 μM | 25048236 | ||
| HuT-78 | Growth Inhibition Assay | 24 h | IC50=9.3 μM | 25048236 | ||
| HuT-78 | Growth Inhibition Assay | 48 h | IC50=4.3 μM | 25048236 | ||
| HuT-78 | Growth Inhibition Assay | 72 h | IC50=4.2 μM | 25048236 | ||
| OPM-2 | Growth Inhibition Assay | 24 h | IC50=24.1 μM | 25048236 | ||
| OPM-2 | Growth Inhibition Assay | 48 h | IC50=4 μM | 25048236 | ||
| OPM-2 | Growth Inhibition Assay | 72 h | IC50=1.3 μM | 25048236 | ||
| RPMI-8226 | Growth Inhibition Assay | 24 h | IC50=106.6 μM | 25048236 | ||
| RPMI-8226 | Growth Inhibition Assay | 48 h | IC50=91.1 μM | 25048236 | ||
| RPMI-8226 | Growth Inhibition Assay | 72 h | IC50=14.9 μM | 25048236 | ||
| U-266 | Growth Inhibition Assay | 24 h | IC50=86.2 μM | 25048236 | ||
| U-266 | Growth Inhibition Assay | 48 h | IC50=68.4 μM | 25048236 | ||
| U-266 | Growth Inhibition Assay | 72 h | IC50=27.4 μM | 25048236 | ||
| Kelly | Growth Inhibition Assay | 0-10 μM | 72 h | IC50=1.518 μM | 25008900 | |
| SH-SY5Y | Growth Inhibition Assay | 0-10 μM | 72 h | IC50=0.754 μM | 25008900 | |
| SK-N-AS | Growth Inhibition Assay | 0-10 μM | 72 h | IC50=1.712 μM | 25008900 | |
| SK-N-DZ | Growth Inhibition Assay | 0-10 μM | 72 h | IC50=5.485 μM | 25008900 | |
| HepG2 | Growth Inhibition Assay | 48 h | DMSO | IC50=13.65 ± 0.92 μM | 24996136 | |
| A549 | Growth Inhibition Assay | 48 h | DMSO | IC50=241.9 ± 31.23 μM | 24996136 | |
| MCF7 | Growth Inhibition Assay | 48 h | DMSO | IC50=81.09 ± 14.21 μM | 24996136 | |
| HL-60 | Growth Inhibition Assay | 72 h | IC50=0.12 μM | 24993014 | ||
| HL-60[R] | Growth Inhibition Assay | 72 h | IC50=3.12 μM | 24993014 | ||
| MIAPACA | Growth Inhibition Assay | GI50=1.3 ± 0.03 μM | 24953821 | |||
| MCF-7 | Growth Inhibition Assay | GI50=0.25 ± 0.1 μM | 24953821 | |||
| HeLa | Growth Inhibition Assay | GI50=0.64 ± 0.4 μM | 24953821 | |||
| MO59K | Growth Inhibition Assay | 7 d | IC50=0.17 μM | 24953561 | ||
| MO59J | Growth Inhibition Assay | 7 d | IC50=0.1 μM | 24953561 | ||
| ME 180 | Growth Inhibition Assay | 48 h | IC50=8.9 ± 0.3 μM | 24953027 | ||
| MCF-7 | Growth Inhibition Assay | 48 h | IC50=23.9 ± 0.3 μM | 24953027 | ||
| HeLa | Growth Inhibition Assay | 48 h | IC50=4.71 ± 1.4 μM | 24953027 | ||
| MDA-MB-453 | Growth Inhibition Assay | 48 h | IC50=12.5 ± 0.85 μM | 24953027 | ||
| MDA-MB-231 | Growth Inhibition Assay | 48 h | IC50=24.22 ± 2.94 μM | 24953027 | ||
| PC-3 | Growth Inhibition Assay | 48 h | IC50=14.4 ± 3.23 μM | 24953027 | ||
| HT-29 | Growth Inhibition Assay | 48 h | IC50=21.45 ± 3.87 μM | 24953027 | ||
| BGC-823 | Growth Inhibition Assay | 48 h | IC50=43.74 ± 5.13 μM | 24793877 | ||
| HeLa | Growth Inhibition Assay | 48 h | IC50=209.90 ± 13.42 μM | 24793877 | ||
| A549 | Growth Inhibition Assay | 48 h | IC50=139.54 ± 7.05 μM | 24793877 | ||
| HK-2 | Growth Inhibition Assay | 48 h | IC50=9.17 ± 1.58 μM | 24793877 | ||
| KB | Cytotoxicity assay | 3 days | Cytotoxicity against human KB cells assessed as reduction in cell number after 3 days, ID50 = 0.2 μM. | 1331331 | ||
| KB | Cytotoxicity assay | 3 days | In vitro cytotoxicity against KB cells after a 3-day incubation., ID50 = 0.2 μM. | 2158562 | ||
| HCT116 | Cytotoxicity assay | Cytotoxicity against HCT116 cells., IC50 = 2.5 μM. | 7473578 | |||
| CCRF CEM | Function assay | Tested for the inhibitory activity against CCRF CEM T- cell leukemia cells, IC50 = 0.002 μM. | 7932571 | |||
| KB ATCC | Function assay | 50% reduction in the number of KB ATCC cells, ID50 = 0.32 μM. | 8120864 | |||
| KB | Cytotoxicity assay | 3 days | Cytotoxicity against KB cells after a 3 day incubation, ID50 = 0.2 μM. | 8120864 | ||
| SCLC | Cytotoxicity assay | Cytotoxicity against human SCLC cells resistant to cisplatin (SCLC/CDDP), IC50 = 1.3 μM. | 8258835 | |||
| P388 | Cytotoxicity assay | 1 hr | In vitro cytotoxicity against P388 murine leukemia cells in culture in a clonogenic assay after 1 hour exposure to drug, IC50 = 0.3 μM. | 8410993 | ||
| HeLa | Function assay | Tested for inhibition of topoisomerase II isolated from HeLa cells by DNA-cleavage assay, EC50 = 0.81 μM. | 8410993 | |||
| Bel7402 | Cytotoxicity assay | 5 days | Cytotoxicity against human Bel7402 cells after 5 days by MTT assay, IC50 = 1.9 μM. | 8778240 | ||
| BGC | Cytotoxicity assay | 5 days | Cytotoxicity against human BGC cells after 5 days by MTT assay, IC50 = 6.7 μM. | 8778240 | ||
| HCT8 | Cytotoxicity assay | 5 days | Cytotoxicity against human HCT8 cells after 5 days by MTT assay, IC50 = 6.4 μM. | 8778240 | ||
| HL60 | Cytotoxicity assay | 5 days | Cytotoxicity against human HL60 cells after 5 days by MTT assay, IC50 = 0.16 μM. | 8778240 | ||
| C6 | Cytotoxicity assay | 3 days | Cytotoxicity against rat C6 cells after 3 days treated 4 hrs before db-cAMP challenge by MTT assay, LC50 = 0.12 μM. | 8988604 | ||
| KB | Growth inhibition assay | Compound concentration required to reduce the exponential growth of KB cells by 50%, CC50 = 0.1 μM. | 9767632 | |||
| MT-4 | Growth inhibition assay | Compound concentration required to reduce the exponential growth of MT-4 cells by 50%, CC50 = 0.08 μM. | 9767632 | |||
| L1210 | Cytotoxicity assay | In vitro cytotoxic activity against murine L1210 leukemia cells., IC50 = 0.2 μM. | 9804687 | |||
| HL-60 | Cytotoxicity assay | Relative cytotoxicity against human promyelocytic leukemia(HL-60) cells, IC50 = 0.367 μM. | 10377219 | |||
| HL-60 | Cytotoxicity assay | Relative cytotoxicity against human promyelocytic leukemia(HL-60) cells, IC90 = 0.73 μM. | 10377219 | |||
| HL-60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.025 μM. | 11170674 | ||
| A549 | Function assay | GI values against A549 cells (lung cancer), GI50 = 6.30957 μM. | 11311065 | |||
| CAKI-1 | Function assay | GI values against CAKI-1 cells(renal carcinoma), GI50 = 6.30957 μM. | 11311065 | |||
| KB | Function assay | GI values against KB cells (nasopharyngeal carcinoma), GI50 = 0.19953 μM. | 11311065 | |||
| HEL | Function assay | Inhibitory activity against human cytomegalovirus (HCMV) in HEL cells, ED50 = 8.64 μM. | 11311065 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by MTT assay, IC50 = 0.5 μM. | 11473438 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 5.4 μM. | 11473438 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by MTT assay, IC50 = 12.3 μM. | 11473438 | |||
| P388 | Cytotoxicity assay | Cytotoxicity against mouse P388 cells by MTT assay, IC50 = 0.1 μM. | 11473438 | |||
| HT-29 | Cytotoxicity assay | Tested for cytotoxicity against Colon carcinoma type HT-29 cell line expressing MDR-1 (-) gene; 35% of cells were affected, IC50 = 10 μM. | 11844671 | |||
| KB | Function assay | Compound concentration required to reduce the cell proliferation of Wild type and Drug- resistant human nasopharyngeal carcinoma KB cells by 50%, IC50 = 0.6 μM. | 12431048 | |||
| P388 | Growth inhibition assay | Growth inhibitory activity against murine P388 leukemia cells, IC50 = 0.15 μM. | 12620081 | |||
| HL60 | Apoptosis assay | In vitro concentration required to induce apoptosis in HL60 cells, AC50 = 0.45 μM. | 12877593 | |||
| HL60R | Apoptosis assay | In vitro concentration required to induce apoptosis in HL60R cells, AC50 = 15 μM. | 12877593 | |||
| HL60 | Function assay | In vitro inhibitory concentration against proliferation of HL60 cells, IC50 = 0.123 μM. | 12877593 | |||
| HL60R | Function assay | In vitro inhibitory concentration against proliferation of HL60R cells, IC50 = 6.5 μM. | 12877593 | |||
| HL60R | Function assay | In vitro inhibitory concentration against proliferation of HL60R cells along with 5 (uM) verapamil, IC50 = 0.5 μM. | 12877593 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 0.2 μM. | 14738377 | |||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by methylene blue assay, IC50 = 2.3 μM. | 14738395 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by methylene blue assay, IC50 = 1.1 μM. | 14738395 | ||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells, ID50 = 0.2 μM. | 14987069 | |||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells, LD50 = 3 μM. | 14987069 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells, GI50 = 4.36516 μM. | 14987069 | |||
| MOLT4 | Cytotoxicity assay | Cytotoxicity against human MOLT4 cells, GI50 = 1.02329 μM. | 14987069 | |||
| HeLa | Function assay | Concentration required to inhibit S phase and G2/M phase in HeLa cells, Concentration = 2 μM. | 15026047 | |||
| HeLa | Growth inhibition assay | Growth inhibitory concentration of compound was determined against HeLa cells by alamar blue assay, GI50 = 40 μM. | 15026047 | |||
| KB | Function assay | 3 days | Dose required for reduction in camptothecin resistant human nasopharyngeal KB cancer cells after 3 days incubation, ED50 = 7 μM. | 15456257 | ||
| KB-7d | Function assay | 3 days | Dose required for reduction in etoposide resistant human nasopharyngeal KB-7d cancer cells after 3 days incubation, ED50 = 17.2 μM. | 15456257 | ||
| A549 | Function assay | 3 days | Dose required for reduction in human lung A549 cancer cells after 3 days incubation, ED50 = 10.6 μM. | 15456257 | ||
| KB | Function assay | 3 days | Dose required to cause reduction in human nasopharyngeal KB cancer cells after 3 days incubation, ED50 = 0.3 μM. | 15456257 | ||
| 1A9 | Function assay | 3 days | Dose required to cause reduction in human ovarian 1A9 cancer cells after 3 days incubation, ED50 = 0.15 μM. | 15456257 | ||
| HSC2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HSC2 cells assessed as cell viability after 24 hrs, IC50 = 41 μM. | 15620262 | ||
| A549 | Function assay | Effective dose on human lung carcinoma (A549) cells, ED50 = 3.39 μM. | 15914002 | |||
| HL60 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HL60 cells after 24 hrs by MTT assay, IC50 = 25 μM. | 15974617 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by SRB assay, IC50 = 0.8775 μM. | 16309315 | ||
| TK10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human TK10 cells after 48 hrs by SRB assay, IC50 = 9.9525 μM. | 16309315 | ||
| UACC62 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human UACC62 cells after 48 hrs by SRB assay, IC50 = 1.13 μM. | 16309315 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, ED50 = 3.76 μM. | 16441087 | ||
| HeLa | Antiproliferative assay | 69 hrs | Antiproliferative effect against human HeLa cells after 69 hrs by MTT assay, IC50 = 3.6 μM. | 16499324 | ||
| HL60 | Antiproliferative assay | 69 hrs | Antiproliferative effect against human HL60 cells after 69 hrs, IC50 = 0.21 μM. | 16499324 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative effect against human MCF7 cells after 72 hrs by MTT assay, IC50 = 0.19 μM. | 16499324 | ||
| K-562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K-562 cells after 72 hrs by MTT assay, IC50 = 1.87 μM. | 16872135 | ||
| LAMA-84 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human LAMA-84 cells after 72 hrs by MTT assay, IC50 = 0.79 μM. | 16872135 | ||
| SKW3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKW3 cells after 72 hrs by MTT assay, IC50 = 0.82 μM. | 16872135 | ||
| HL-60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL-60 cells after 72 hrs by MTT assay, IC50 = 10.5 μM. | 16933865 | ||
| HSC2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HSC2 cells after 24 hrs by MTT assay, IC50 = 24 μM. | 17125230 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs, IC50 = 2 μM. | 17125231 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB assay, IC50 = 1.16 μM. | 17194596 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells by SRB assay, IC50 = 1.87 μM. | 17194596 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by SRB assay, IC50 = 0.37 μM. | 17194596 | |||
| HT1080 | Cytotoxicity assay | Cytotoxicity against human HT1080 cells by SRB assay, IC50 = 0.25 μM. | 17194596 | |||
| SK-MEL-2 | Cytotoxicity assay | Cytotoxicity against human SK-MEL-2 cells by SRB assay, IC50 = 5.72 μM. | 17194596 | |||
| SK-OV-3 | Cytotoxicity assay | Cytotoxicity against human SK-OV-3 cells by SRB assay, IC50 = 4.28 μM. | 17194596 | |||
| SNU638 | Cytotoxicity assay | Cytotoxicity against human SNU638 cells by SRB assay, IC50 = 0.23 μM. | 17194596 | |||
| XF498 | Cytotoxicity assay | Cytotoxicity against human XF498 cells by SRB assay, IC50 = 3.48 μM. | 17194596 | |||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxic activity against human MCF7 cells after 48 hrs by MTT reduction assay, IC50 = 3.2 μM. | 17316916 | ||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells by XTT assay, IC50 = 1.7 μM. | 17368022 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by XTT assay, IC50 = 35.6 μM. | 17368022 | |||
| MDA-MB468 | Cytotoxicity assay | Cytotoxicity against human MDA-MB468 cells by XTT assay, IC50 = 13.6 μM. | 17368022 | |||
| K562 | Growth inhibition assay | 5 days | Growth inhibition of K562 cells by XTT assay after 5 days, IC50 = 0.32 μM. | 17411092 | ||
| mouse astrocyte cells | Antiproliferative assay | Antiproliferative activity against mouse astrocyte cells by MTT assay, EC50 = 0.433 μM. | 17417631 | |||
| mouse medulloblastoma cells | Antiproliferative assay | Antiproliferative activity against mouse medulloblastoma cells harboring heterozygous ptch1 gene by MTT assay, EC50 = 0.208 μM. | 17417631 | |||
| mouse neural precursor cells | Antiproliferative assay | Antiproliferative activity against mouse neural precursor cells by MTT assay, EC50 = 0.34 μM. | 17417631 | |||
| BV173 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BV173 cells after 72 hrs by MTT assay, IC50 = 0.9 μM. | 17459529 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by MTT assay, IC50 = 3.1 μM. | 17459529 | ||
| H460 | Growth inhibition assay | Growth inhibition of human H460 cells by MTT assay, IC50 = 0.2 μM. | 17482821 | |||
| HeLa | Growth inhibition assay | Growth inhibition of human HeLa cells by MTT assay, IC50 = 2.9 μM. | 17482821 | |||
| MiaPaCa2 | Growth inhibition assay | Growth inhibition of human MiaPaCa2 cells by MTT assay, IC50 = 15.4 μM. | 17482821 | |||
| SW620 | Growth inhibition assay | Growth inhibition of human SW620 cells by MTT assay, IC50 = 20 μM. | 17482821 | |||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by SRB method, IC50 = 0.32 μM. | 17493824 | ||
| HCT15 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT15 cells after 48 hrs by SRB method, IC50 = 2.43 μM. | 17493824 | ||
| SK-MEL-2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SK-MEL-2 cells after 48 hrs by SRB method, IC50 = 0.3 μM. | 17493824 | ||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells after 48 hrs by SRB method, IC50 = 1.72 μM. | 17493824 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB microtiter plate assay, IC50 = 1.4 μM. | 17585747 | |||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells by SRB microtiter plate assay, IC50 = 2.4 μM. | 17585747 | |||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells by SRB microtiter plate assay, IC50 = 4.8 μM. | 17585747 | |||
| ZR751 | Cytotoxicity assay | Cytotoxicity against human ZR751cells by SRB microtiter plate assay, IC50 = 3.1 μM. | 17585747 | |||
| HL20 | Growth inhibition assay | 48 hrs | Inhibition of cell growth in human HL20 cells after 48 hrs by MTT assay, IC50 = 0.315 μM. | 17889547 | ||
| HeLa | Growth inhibition assay | 48 hrs | Growth inhibition of HeLa cells after 48 hrs by MTT assay, GI50 = 8 μM. | 17894480 | ||
| HeLa | Antitumor assay | 72 hrs | Antitumor activity against human HeLa cells after 72 hrs by MTT assay, IC50 = 3 μM. | 17935309 | ||
| MiaPaCa2 | Antitumor assay | 72 hrs | Antitumor activity against human MiaPaCa2 cells after 72 hrs by MTT assay, IC50 = 15 μM. | 17935309 | ||
| SW620 | Antitumor assay | 72 hrs | Antitumor activity against human SW620 cells after 72 hrs by MTT assay, IC50 = 20 μM. | 17935309 | ||
| HL60/MX2 | Cytotoxicity assay | Cytotoxicity against DNA topoisomerase-2 deficient human HL60/MX2 cells by MTT assay, GI50 = 5.98 μM. | 17962028 | |||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, GI50 = 0.14 μM. | 17962028 | ||
| 502713 | Cytotoxicity assay | Cytotoxicity against human 502713 cells by sulforhodamine B assay, IC50 = 2.42 μM. | 17988764 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by sulforhodamine B assay, IC50 = 5.62 μM. | 17988764 | |||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells by sulforhodamine B assay, IC50 = 3.85 μM. | 17988764 | |||
| HCT-15 | Cytotoxicity assay | Cytotoxicity against human HCT-15 cells by sulforhodamine B assay, IC50 = 1.48 μM. | 17988764 | |||
| HEP2 | Cytotoxicity assay | Cytotoxicity against human HEP2 cells by sulforhodamine B assay, IC50 = 2.15 μM. | 17988764 | |||
| PC-3 | Cytotoxicity assay | Cytotoxicity against human PC-3 cells by sulforhodamine B assay, IC50 = 17.4 μM. | 17988764 | |||
| SF295 | Cytotoxicity assay | Cytotoxicity against human SF295 cells by sulforhodamine B assay, IC50 = 13.3 μM. | 17988764 | |||
| K562 | Cytotoxicity assay | 5 days | Cytotoxicity against human K562 cells after 5 days by XTT assay, IC50 = 0.32 μM. | 18076140 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by XTT assay, IC50 = 35.6 μM. | 18093835 | |||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells, IC50 = 29.79 μM. | 18178085 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells, IC50 = 15.11 μM. | 18178085 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells, IC50 = 18.24 μM. | 18178085 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells, IC50 = 1.44 μM. | 18178085 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells, IC50 = 16.25 μM. | 18178085 | |||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 2.9 μM. | 18258438 | ||
| AGS | Antitumor assay | 72 hrs | Antitumor against human AGS cells after 72 hrs by MTT assay, IC50 = 0.36 μM. | 18299197 | ||
| HL60 | Antitumor assay | 72 hrs | Antitumor against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.8 μM. | 18299197 | ||
| J82 | Antitumor assay | 72 hrs | Antitumor against human J82 cells after 72 hrs by MTT assay, IC50 = 2.8 μM. | 18299197 | ||
| SK-MES1 | Antitumor assay | 72 hrs | Antitumor against human SK-MES1 cells after 72 hrs by MTT assay, IC50 = 2.5 μM. | 18299197 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells after 72 hrs by MTT assay, IC50 = 3.9 μM. | 18299197 | ||
| H520 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human H520 cells after 72 hrs by Cell Titer 96 Aqueous Non-Radioactive assay, GI50 = 8.7 μM. | 18321710 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by Cell Titer 96 Aqueous Non-Radioactive assay, GI50 = 11.1 μM. | 18321710 | ||
| A549 | Cytotoxicity assay | 3 days | Cytotoxicity against human A549 cells after 3 days by SRB assay, IC50 = 1.16 μM. | 18321715 | ||
| HCT15 | Cytotoxicity assay | 3 days | Cytotoxicity against human HCT15 cells after 3 days by SRB assay, IC50 = 2.8 μM. | 18321715 | ||
| HL60 | Cytotoxicity assay | 3 days | Cytotoxicity against human HL60 cells after 3 days by SRB assay, IC50 = 0.37 μM. | 18321715 | ||
| HT1080 | Cytotoxicity assay | 3 days | Cytotoxicity against human HT1080 cells after 3 days by SRB assay, IC50 = 0.25 μM. | 18321715 | ||
| SK-MEL-2 | Cytotoxicity assay | 3 days | Cytotoxicity against human SK-MEL-2 cells after 3 days by SRB assay, IC50 = 5.72 μM. | 18321715 | ||
| SKOV3 | Cytotoxicity assay | 3 days | Cytotoxicity against human SKOV3 cells after 3 days by SRB assay, IC50 = 4.28 μM. | 18321715 | ||
| SNU638 | Cytotoxicity assay | 3 days | Cytotoxicity against human SNU638 cells after 3 days by SRB assay, IC50 = 0.23 μM. | 18321715 | ||
| XF498 | Cytotoxicity assay | 3 days | Cytotoxicity against human XF498 cells after 3 days by SRB assay, IC50 = 3.48 μM. | 18321715 | ||
| HL60 | Apoptosis assay | 48 hrs | Induction of apoptosis in human HL60 cells after 48 hrs, AC50 = 0.4 μM. | 18339455 | ||
| 5637 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human 5637 cells after 96 hrs by crystal violet assay, IC50 = 0.54 μM. | 18434163 | ||
| A427 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human A427 cells after 96 hrs by crystal violet assay, IC50 = 0.13 μM. | 18434163 | ||
| KYSE70 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human KYSE70 cells after 96 hrs by crystal violet assay, IC50 = 0.94 μM. | 18434163 | ||
| MCF7 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MCF7 cells after 96 hrs by crystal violet assay, IC50 = 0.5 μM. | 18434163 | ||
| NCI-H520 | Growth inhibition assay | Growth inhibition of human NCI-H520 cells, GI50 = 13.5 μM. | 18502124 | |||
| PC3 | Growth inhibition assay | Growth inhibition of human PC3 cells, GI50 = 12.5 μM. | 18502124 | |||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human mitoxantrone-sensitive HL60 cells after 72 hrs, IC50 = 0.486 μM. | 18507368 | ||
| HL60/MX2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mitoxantrone-resistant human HL60/MX2 cells after 72 hrs, IC50 = 48.5 μM. | 18507368 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.2 μM. | 18507472 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.315 μM. | 18513974 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by MTT assay, IC50 = 1.27 μM. | 18514972 | ||
| KB | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KB cells after 48 hrs by MTT assay, IC50 = 0.42 μM. | 18514972 | ||
| L02 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human L02 cells after 48 hrs by MTT assay, IC50 = 13 μM. | 18514972 | ||
| DU145 ATCC HTB 81 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 ATCC HTB 81 cells after 72 hrs by MTT assay, IC50 = 1.5 μM. | 18558490 | ||
| K562 ATCC CCL 243 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 ATCC CCL 243 cells after 72 hrs by MTT assay, IC50 = 4.9 μM. | 18558490 | ||
| MCF7 ATCC HTB 22 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 ATCC HTB 22 cells after 72 hrs by MTT assay, IC50 = 12.2 μM. | 18558490 | ||
| U251 RCB 0641 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U251 RCB 0641 cells after 72 hrs by MTT assay, IC50 = 0.5 μM. | 18558490 | ||
| 3LL CRL 1642 | Cytotoxicity assay | 48 hrs | Cytotoxicity against mouse 3LL CRL 1642 cells after 48 hrs by MTT assay, IC50 = 4 μM. | 18558490 | ||
| L1210 ATCC CCL 219 | Cytotoxicity assay | 48 hrs | Cytotoxicity against mouse L1210 ATCC CCL 219 cells after 48 hrs by MTT assay, IC50 = 0.5 μM. | 18558490 | ||
| SK-Hep1 | Cytotoxicity assay | Cytotoxicity against human SK-Hep1 cells by MTT assay, IC50 = 49.6 μM. | 18637688 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by SRB assay, IC50 = 0.2 μM. | 18651771 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells assessed as reduction of cell growth by SRB method, IC50 = 9.98 μM. | 18782668 | |||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells assessed as reduction of cell growth by SRB method, IC50 = 14.2 μM. | 18782668 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 8.37 μM. | 18844422 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTT assay, IC50 = 1.25 μM. | 18844422 | ||
| AGS | Cytotoxicity assay | 3 days | Cytotoxicity against human AGS cells after 3 days by MTT assay, IC50 = 0.36 μM. | 19013074 | ||
| HL-60 | Cytotoxicity assay | 3 days | Cytotoxicity against human HL-60 cells after 3 days by MTT assay, IC50 = 0.8 μM. | 19013074 | ||
| J82 | Cytotoxicity assay | 3 days | Cytotoxicity against human J82 cells after 3 days by MTT assay, IC50 = 2.8 μM. | 19013074 | ||
| MRC5 | Cytotoxicity assay | 3 days | Cytotoxicity against human MRC5 cells after 3 days by MTT assay, IC50 = 3.9 μM. | 19013074 | ||
| SK-MES-1 | Cytotoxicity assay | 3 days | Cytotoxicity against human SK-MES-1 cells after 3 days by MTT assay, IC50 = 2.5 μM. | 19013074 | ||
| A375 | Cytotoxicity assay | Cytotoxicity against human A375 cells by MTT assay, IC50 = 3.92 μM. | 19036584 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by MTT assay, IC50 = 9.51 μM. | 19036584 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 1.83 μM. | 19036584 | |||
| THP1 | Cytotoxicity assay | Cytotoxicity against human THP1 cells by MTT assay, IC50 = 2.16 μM. | 19036584 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 5.9 μM. | 19053514 | ||
| HL-60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL-60 cells after 72 hrs by MTT assay, IC50 = 0.25 μM. | 19053514 | ||
| U937 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U937 cells after 72 hrs by MTT assay, IC50 = 0.92 μM. | 19053514 | ||
| ACHN | Antiproliferative assay | 96 hrs | Antiproliferative activity against human ACHN cells after 96 hrs by MTT assay, IC50 = 2.15 μM. | 19053767 | ||
| CCRF-CEM | Antiproliferative assay | 96 hrs | Antiproliferative activity against human CCRF-CEM cells after 96 hrs by MTT assay, IC50 = 1.2 μM. | 19053767 | ||
| CCRF-SB | Antiproliferative assay | 96 hrs | Antiproliferative activity against human CCRF-SB cells after 96 hrs by MTT assay, IC50 = 0.1 μM. | 19053767 | ||
| HeLa | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HeLa cells after 96 hrs by MTT assay, IC50 = 3 μM. | 19053767 | ||
| HT-29 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HT-29 cells after 96 hrs by MTT assay, IC50 = 2.5 μM. | 19053767 | ||
| MCF7 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MCF7 cells after 96 hrs by MTT assay, IC50 = 1 μM. | 19053767 | ||
| MRC5 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MRC5 cells after 96 hrs by MTT assay, IC50 = 3.9 μM. | 19053767 | ||
| MT4 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MT4 cells after 96 hrs by MTT assay, IC50 = 1.25 μM. | 19053767 | ||
| SF-268 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human SF-268 cells after 96 hrs by MTT assay, IC50 = 3.25 μM. | 19053767 | ||
| XF498 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human XF498 cells after 96 hrs by MTT assay, IC50 = 3.45 μM. | 19053767 | ||
| KB | Growth inhibition assay | 72 hrs | Growth inhibition of human KB cells after 72 hrs by methylene blue dye assay, IC50 = 1.1 μM. | 19053773 | ||
| KB-TAX50 | Growth inhibition assay | 72 hrs | Growth inhibition of human Pgp170 overexpressing multidrug resistant-KB-TAX50 cells after 72 hrs by methylene blue dye assay, IC50 = 3.5 μM. | 19053773 | ||
| KB-VIN10 | Growth inhibition assay | 72 hrs | Growth inhibition of human Pgp170 overexpressing multidrug resistant-KB-VIN10 cells after 72 hrs by methylene blue dye assay, IC50 = 23 μM. | 19053773 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by WST-8 assay, IC50 = 1.7 μM. | 19072209 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by WST-8 assay, IC50 = 7.6 μM. | 19072209 | ||
| NB4 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human NB4 cells after 48 hrs by WST-8 assay, IC50 = 1.3 μM. | 19072209 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by WST-8 assay, IC50 = 13.6 μM. | 19072209 | ||
| SH-SY5Y | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SH-SY5Y cells after 48 hrs by WST-8 assay, IC50 = 1.7 μM. | 19072209 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs, IC50 = 1.2 μM. | 19115839 | ||
| DLD1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DLD1 cells after 48 hrs, IC50 = 27 μM. | 19115839 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs, IC50 = 0.7 μM. | 19115839 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs, IC50 = 1.7 μM. | 19115839 | ||
| WS1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WS1 cells after 48 hrs, IC50 = 34 μM. | 19115839 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 3.3 μM. | 19251420 | ||
| AGS | Cytotoxicity assay | 72 hrs | Cytotoxicity against human AGS cells after 72 hrs by MTT assay, IC50 = 0.36 μM. | 19269832 | ||
| J82 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human J82 cells after 72 hrs by MTT assay, IC50 = 2.8 μM. | 19269832 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells after 72 hrs by MTT assay, IC50 = 3.9 μM. | 19269832 | ||
| SKMES1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKMES1 cells after 72 hrs by MTT assay, IC50 = 2.5 μM. | 19269832 | ||
| HCT15 | Antiproliferative assay | Antiproliferative activity against human HCT15 cells by SRB assay, GI50 = 1.25 μM. | 19394218 | |||
| MESSA | Antiproliferative assay | Antiproliferative activity against human MESSA cells by SRB assay, GI50 = 0.21 μM. | 19394218 | |||
| HCT15/CLO2 | Antiproliferative assay | Antiproliferative activity against multidrug resistant human HCT15/CLO2 cells by SRB assay, GI50 = 10.02 μM. | 19394218 | |||
| MES-SA/Dx5 | Antiproliferative assay | Antiproliferative activity against multidrug resistant human MES-SA/Dx5 cells by SRB assay, GI50 = 9.72 μM. | 19394218 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 14.3 μM. | 19394829 | ||
| CCRF-CEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human CCRF-CEM cells after 72 hrs by MTT assay, IC50 = 1.1 μM. | 19394829 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 3.9 μM. | 19394829 | ||
| Jurkat | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Jurkat cells after 72 hrs by MTT assay, IC50 = 1.2 μM. | 19394829 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 10.9 μM. | 19394829 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 21.2 μM. | 19394829 | ||
| AGS | Cytotoxicity assay | 3 days | Cytotoxicity against human AGS cells after 3 days by MTT assay, IC50 = 0.36 μM. | 19419803 | ||
| HL60 | Cytotoxicity assay | 3 days | Cytotoxicity against human HL60 cells after 3 days by MTT assay, IC50 = 0.8 μM. | 19419803 | ||
| J82 | Cytotoxicity assay | 3 days | Cytotoxicity against human J82 cells after 3 days by MTT assay, IC50 = 2.5 μM. | 19419803 | ||
| MRC5 | Cytotoxicity assay | 3 days | Cytotoxicity against human MRC5 cells after 3 days by MTT assay, IC50 = 3.93 μM. | 19419803 | ||
| SKMES1 | Cytotoxicity assay | 3 days | Cytotoxicity against human SKMES1 cells after 3 days by MTT assay, IC50 = 2.8 μM. | 19419803 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by SRB method, IC50 = 1.7 μM. | 19425589 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by SRB method, IC50 = 7.6 μM. | 19425589 | ||
| NB4 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human NB4 cells after 48 hrs by SRB method, IC50 = 1.3 μM. | 19425589 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by SRB method, IC50 = 13.6 μM. | 19425589 | ||
| SH-SY5Y | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SH-SY5Y cells after 48 hrs by SRB method, IC50 = 1.7 μM. | 19425589 | ||
| HT-29 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human HT-29 cells after 96 hrs by SRB assay, GI50 = 0.73 μM. | 19572549 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 0.63 μM. | 19685913 | ||
| Bel7402 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Bel7402 cells after 72 hrs by MTT assay, IC50 = 1.03 μM. | 19685913 | ||
| HL60 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HL60 cells after 24 hrs by SRB assay, IC50 = 0.042 μM. | 19685913 | ||
| P388 | Cytotoxicity assay | 24 hrs | Cytotoxicity against mouse P388 cells after 24 hrs by SRB assay, IC50 = 0.05 μM. | 19685913 | ||
| HCT116 | Antiproliferative assay | 3 days | Antiproliferative activity against human HCT116 cells after 3 days by XTT assay, IC50 = 1.7 μM. | 19691293 | ||
| SW948 | Antiproliferative assay | 3 days | Antiproliferative activity against human SW948 cells after 3 days by XTT assay, IC50 = 1.3 μM. | 19691293 | ||
| A549 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human A549 cells after 24 hrs by SRB assay, IC50 = 0.63 μM. | 19769341 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.042 μM. | 19769341 | ||
| A375 | Cytotoxicity assay | Cytotoxicity against human A375 cells by MTT assay, IC50 = 3.92 μM. | 19782567 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by MTT assay, IC50 = 9.51 μM. | 19782567 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 2.16 μM. | 19782567 | |||
| THP1 | Cytotoxicity assay | Cytotoxicity against human THP1 cells by MTT assay, IC50 = 1.83 μM. | 19782567 | |||
| HONE1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HONE1 cells after 72 hrs by methylene blue dye assay, IC50 = 1.2 μM. | 19785430 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by methylene blue dye assay, IC50 = 2.3 μM. | 19785430 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by methylene blue dye assay, IC50 = 1.3 μM. | 19785430 | ||
| H69 | Cytotoxicity assay | Cytotoxicity against human adriamycin resistant H69 cells, IC50 = 46 μM. | 19788270 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells, IC50 = 0.4 μM. | 19788270 | |||
| HuCCa1 | Cytotoxicity assay | Cytotoxicity against human HuCCa1 cells, IC50 = 5.1 μM. | 19788270 | |||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells, IC50 = 0.5 μM. | 19788270 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells, IC50 = 0.3 μM. | 19788270 | |||
| T47D | Cytotoxicity assay | Cytotoxicity against human T47D cells, IC50 = 0.1 μM. | 19788270 | |||
| HCT116 | Cytotoxicity assay | 4 days | Cytotoxicity against human HCT116 cells after 4 days, IC50 = 14.39 μM. | 19800803 | ||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells by MTT assay, IC50 = 5.16 μM. | 19836231 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells by MTT assay, IC50 = 2.15 μM. | 19836231 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 0.93 μM. | 19836231 | |||
| HT-29 | Cytotoxicity assay | Cytotoxicity against human HT-29 cells by MTT assay, IC50 = 6.32 μM. | 19836231 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells by MTT assay, IC50 = 3.5 μM. | 19836231 | |||
| HaCaT | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HaCaT cells after 72 hrs by MTT assay, IC50 = 0.55 μM. | 19919064 | ||
| B16 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse B16 cells after 72 hrs by MTT assay, IC50 = 0.28 μM. | 19919064 | ||
| DU145 | Cytotoxicity assay | 4 days | Cytotoxicity against human DU145 cells after 4 days by ELISA reader assay, IC50 = 1.3 μM. | 19939682 | ||
| HCT15 | Cytotoxicity assay | 4 days | Cytotoxicity against human HCT15 cells after 4 days by ELISA reader assay, IC50 = 1.09 μM. | 19939682 | ||
| HeLa | Cytotoxicity assay | 4 days | Cytotoxicity against human HeLa cells after 4 days by ELISA reader assay, IC50 = 1.29 μM. | 19939682 | ||
| K562 | Cytotoxicity assay | 4 days | Cytotoxicity against human K562 cells after 4 days by ELISA reader assay, IC50 = 2.41 μM. | 19939682 | ||
| MCF7 | Cytotoxicity assay | 4 days | Cytotoxicity against human MCF7 cells after 4 days by ELISA reader assay, IC50 = 1.89 μM. | 19939682 | ||
| DU145 | Cytotoxicity assay | 3 days | Cytotoxicity against human DU145 cells after 3 days, IC50 = 21.07 μM. | 19954977 | ||
| HCT116 | Cytotoxicity assay | 3 days | Cytotoxicity against human HCT116 cells after 3 days, IC50 = 17.92 μM. | 19954977 | ||
| HeLa | Cytotoxicity assay | 3 days | Cytotoxicity against human HeLa cells after 3 days, IC50 = 8.43 μM. | 19954977 | ||
| MCF7 | Cytotoxicity assay | 3 days | Cytotoxicity against human MCF7 cells after 3 days, IC50 = 18.01 μM. | 19954977 | ||
| MDA-MB-231 | Cytotoxicity assay | 3 days | Cytotoxicity against human MDA-MB-231 cells after 3 days, IC50 = 16.85 μM. | 19954977 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by MTS assay, IC50 = 0.42 μM. | 20006518 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs, IC50 = 0.29 μM. | 20053564 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs, IC50 = 0.42 μM. | 20053564 | ||
| RPMI8226 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human RPMI8226 cells after 48 hrs, IC50 = 0.14 μM. | 20053564 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 0.83 μM. | 20071056 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by CCK8 method, IC50 = 0.18 μM. | 20071056 | ||
| RPMI8226 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RPMI8226 cells after 72 hrs by CCK8 method, IC50 = 0.7 μM. | 20071056 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by MTT assay, IC50 = 1.63 μM. | 20093033 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 0.47 μM. | 20093033 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.97 μM. | 20093033 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 1.42 μM. | 20093033 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 2.87 μM. | 20093033 | ||
| KB | Cytotoxicity assay | Cytotoxicity against human KB cells, EC50 = 1.1 μM. | 20102168 | |||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTT assay, ED50 = 0.2 μM. | 20187635 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTT assay, IC50 = 0.6 μM. | 20187635 | ||
| KB1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB1 cells after 72 hrs by MTT assay, IC50 = 34.8 μM. | 20187635 | ||
| KB-7d | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB-7d cells after 72 hrs by MTT assay, ED50 = 25 μM. | 20187635 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human vincristine-resistant KB cells after 72 hrs by MTT assay, IC50 = 28.7 μM. | 20187635 | ||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells in presence of 10% fetal bovine serum, IC50 = 2.45 μM. | 20188578 | |||
| HCT15 | Cytotoxicity assay | Cytotoxicity against human HCT15 cells in presence of 10% fetal bovine serum, IC50 = 1.29 μM. | 20188578 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells in presence of 10% fetal bovine serum, IC50 = 1.57 μM. | 20188578 | |||
| K562 | Cytotoxicity assay | Cytotoxicity against human K562 cells in presence of 10% fetal bovine serum, IC50 = 2.84 μM. | 20188578 | |||
| NCI-H460 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human NCI-H460 cells after 48 hrs by [3H]thymidine incorporation assay, IC50 = 0.09 μM. | 20210346 | ||
| SKHEP1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKHEP1 cells after 48 hrs by [3H]thymidine incorporation assay, IC50 = 0.062 μM. | 20210346 | ||
| U87MG | Cytotoxicity assay | 48 hrs | Cytotoxicity against human U87MG cells after 48 hrs by [3H]thymidine incorporation assay, IC50 = 0.145 μM. | 20210346 | ||
| HepG2 | Growth inhibition assay | 72 hrs | Growth inhibition of human HepG2 cells after 72 hrs by sulforhodamine B assay, IC50 = 4.1 μM. | 20346660 | ||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells by MTT assay, IC50 = 8.93 μM. | 20356735 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells by MTT assay, IC50 = 1.81 μM. | 20356735 | |||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days by automatic ELISA reader system, IC50 = 0.5 μM. | 20392646 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days by automatic ELISA reader system, IC50 = 1.1 μM. | 20392646 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days by automatic ELISA reader system, IC50 = 1.4 μM. | 20392646 | ||
| HL60 | Cytotoxicity assay | 2 days | Cytotoxicity against human HL60 cells after 2 days by automatic ELISA reader system, IC50 = 1 μM. | 20392646 | ||
| MDA-MB-231 | Cytotoxicity assay | 2 days | Cytotoxicity against human MDA-MB-231 cells after 2 days by automatic ELISA reader system, IC50 = 0.7 μM. | 20392646 | ||
| HM-SFME-1 | Antiproliferative assay | 24 hrs | Antiproliferative activity against mouse r/m HM-SFME-1 cells after 24 hrs by MTT assay, IC50 = 9 μM. | 20398972 | ||
| SFME | Antiproliferative assay | 24 hrs | Antiproliferative activity against mouse SFME cells after 24 hrs by MTT assay, IC50 = 0.5 μM. | 20398972 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 30.38 μM. | 20405844 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 23.22 μM. | 20405844 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by resazurin reduction assay, IC50 = 16 μM. | 20413315 | ||
| DLD1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DLD1 cells after 48 hrs by resazurin reduction assay, IC50 = 10 μM. | 20413315 | ||
| WS1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WS1 cells after 48 hrs by resazurin reduction assay, IC50 = 4.3 μM. | 20413315 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by SRB assay, IC50 = 0.63 μM. | 20415462 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.042 μM. | 20415462 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.39 μM. | 20524638 | ||
| HL60/MX2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60/MX2 cells after 72 hrs by MTT assay, IC50 = 11.9 μM. | 20570510 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human topoisomerase 2-sensitive HL60 cells after 72 hrs by MTT assay, IC50 = 1.3 μM. | 20570510 | ||
| A375 | Cytotoxicity assay | 24 hrs | Cytotoxicity activity against human A375 cells after 24 hrs by NRU assay, IC50 = 19 μM. | 20570625 | ||
| A375 | Cytotoxicity assay | 48 hrs | Cytotoxicity activity against human A375 cells after 48 hrs by NRU assay, IC50 = 12 μM. | 20570625 | ||
| B16 | Cytotoxicity assay | 24 hrs | Cytotoxicity activity against mouse B16 cells after 24 hrs by NRU assay, IC50 = 9 μM. | 20570625 | ||
| B16 | Cytotoxicity assay | 48 hrs | Cytotoxicity activity against mouse B16 cells after 48 hrs by NRU assay, IC50 = 0.13 μM. | 20570625 | ||
| DU145 | Cytotoxicity assay | 4 days | Cytotoxicity against human DU145 cells after 4 days by MTT assay, IC50 = 13.8 μM. | 20619511 | ||
| HCT116 | Cytotoxicity assay | 4 days | Cytotoxicity against human HCT116 cells after 4 days by MTT assay, IC50 = 11.9 μM. | 20619511 | ||
| HeLa | Cytotoxicity assay | 4 days | Cytotoxicity against human HeLa cells after 4 days by MTT assay, IC50 = 18.2 μM. | 20619511 | ||
| MCF7 | Cytotoxicity assay | 4 days | Cytotoxicity against human MCF7 cells after 4 days by MTT assay, IC50 = 17.5 μM. | 20619511 | ||
| MDA-MB-231 | Cytotoxicity assay | 4 days | Cytotoxicity against human MDA-MB-231 cells after 4 days by MTT assay, IC50 = 16.3 μM. | 20619511 | ||
| AGS | Cytotoxicity assay | 72 hrs | Cytotoxicity against human AGS cells after 72 hrs by MTT assay, IC50 = 0.36 μM. | 20828890 | ||
| HL-60(TB) | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL-60(TB) cells after 72 hrs by MTT assay, IC50 = 0.8 μM. | 20828890 | ||
| J82 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human J82 cells after 72 hrs by MTT assay, IC50 = 2.8 μM. | 20828890 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells after 72 hrs by MTT assay, IC50 = 3.9 μM. | 20828890 | ||
| SKMES1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKMES1 cells after 72 hrs by MTT assay, IC50 = 2.5 μM. | 20828890 | ||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells, IC50 = 0.49 μM. | 20863598 | |||
| colon cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human colon cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 1.3 μM. | 20884089 | ||
| gastric cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human gastric cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 1.3 μM. | 20884089 | ||
| lung cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human lung cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 1.3 μM. | 20884089 | ||
| HCT15 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT15 cells after 48 hrs by sulforhodamine B assay, IC50 = 1.6 μM. | 20939516 | ||
| MESSA | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MESSA cells after 48 hrs by sulforhodamine B assay, IC50 = 0.3 μM. | 20939516 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs, IC50 = 0.486 μM. | 20961767 | ||
| HL60/MX2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60/MX2 cells after 72 hrs, IC50 = 48.5 μM. | 20961767 | ||
| KB | Cytotoxicity assay | 3 days | Cytotoxicity against human KB cells after 3 days by sulforhodamine B assay, EC50 = 4.5 μM. | 21028898 | ||
| NCI60 | Growth inhibition assay | Growth inhibition of human NCI60 cells by SBR assay, GI50 = 38.9045 μM. | 21035334 | |||
| 502713 | Anticancer assay | 48 hrs | Anticancer activity against human 502713 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 0.1 μM. | 21074444 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 3.08 μM. | 21074444 | ||
| DU145 | Anticancer assay | 48 hrs | Anticancer activity against human DU145 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 3.7 μM. | 21074444 | ||
| HCT15 | Anticancer assay | 48 hrs | Anticancer activity against human HCT15 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 0.9 μM. | 21074444 | ||
| Hep2 | Anticancer assay | 48 hrs | Anticancer activity against human Hep2 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 0.9 μM. | 21074444 | ||
| IMR32 | Anticancer assay | 48 hrs | Anticancer activity against human IMR32 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 6.3 μM. | 21074444 | ||
| PC3 | Anticancer assay | 48 hrs | Anticancer activity against human PC3 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 2.6 μM. | 21074444 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay, GI50 = 31.62 μM. | 21115210 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by MTT assay, GI50 = 12.59 μM. | 21115210 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, GI50 = 19.95 μM. | 21115210 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay, GI50 = 0.63 μM. | 21115210 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay, TGI = 3.98 μM. | 21115210 | ||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells by MTT assay, IC50 = 1.44 μM. | 21115246 | |||
| HCT15 | Cytotoxicity assay | Cytotoxicity against human HCT15 cells by MTT assay, IC50 = 2.03 μM. | 21115246 | |||
| K562 | Cytotoxicity assay | Cytotoxicity against human K562 cells by MTT assay, IC50 = 1.21 μM. | 21115246 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by MTT assay, IC50 = 1.98 μM. | 21115246 | |||
| 95-D | Cytotoxicity assay | 72 hrs | Cytotoxicity against human 95-D cells after 72 hrs by MTT assay, IC50 = 0.0202 μM. | 21145139 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 0.00256 μM. | 21145139 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTT assay, IC50 = 0.00461 μM. | 21145139 | ||
| KB/VCR | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB/VCR cells after 72 hrs by MTT assay, IC50 = 0.0834 μM. | 21145139 | ||
| HepG2 | Cytotoxicity assay | Cytotoxicity against human HepG2 cells by MTT assay, IC50 = 15.8 μM. | 21174408 | |||
| MOLT3 | Cytotoxicity assay | Cytotoxicity against human MOLT3 cells by XTT assay, IC50 = 0.03 μM. | 21174408 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB assay, IC50 = 1.85 μM. | 21182258 | |||
| SK-MEL-2 | Cytotoxicity assay | Cytotoxicity against human SK-MEL-2 cells by SRB assay, IC50 = 1.17 μM. | 21182258 | |||
| SKOV3 | Cytotoxicity assay | Cytotoxicity against human SKOV3 cells by SRB assay, IC50 = 1.81 μM. | 21182258 | |||
| XF498 | Cytotoxicity assay | Cytotoxicity against human XF498 cells by SRB assay, IC50 = 1.72 μM. | 21182258 | |||
| A549 | Cytotoxicity assay | 3 days | Cytotoxicity against human A549 cells assessed as cell growth inhibition after 3 days by MTT assay, IC50 = 21.4 μM. | 21277655 | ||
| HCT116 | Cytotoxicity assay | 3 days | Cytotoxicity against human HCT116 cells assessed as cell growth inhibition after 3 days by MTT assay, IC50 = 13.5 μM. | 21277655 | ||
| HepG2 | Cytotoxicity assay | 3 days | Cytotoxicity against human HepG2 cells assessed as cell growth inhibition after 3 days by MTT assay, IC50 = 4.9 μM. | 21277655 | ||
| HT1080 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HT1080 cells after 96 hrs by MTT assay, IC50 = 0.75 μM. | 21296467 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 11 μM. | 21397370 | ||
| A2780 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A2780 cells after 48 hrs by sulforhodamine B based ELISA, GI50 = 1.3 μM. | 21402478 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by sulforhodamine B based ELISA, GI50 = 3.08 μM. | 21402478 | ||
| COLO205 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human COLO205 cells after 48 hrs by sulforhodamine B based ELISA, GI50 = 0.13 μM. | 21402478 | ||
| KB | Cytotoxicity assay | 48 hrs | Cytotoxicity against human KB cells after 48 hrs by sulforhodamine B based ELISA, GI50 = 0.3 μM. | 21402478 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by sulforhodamine B based ELISA, GI50 = 2.1 μM. | 21402478 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days, IC50 = 2.19 μM. | 21419530 | ||
| HCT116 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT116 cells after 2 days, IC50 = 9.5 μM. | 21419530 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days, IC50 = 2.36 μM. | 21419530 | ||
| HL60 | Cytotoxicity assay | 2 days | Cytotoxicity against human HL60 cells after 2 days, IC50 = 1.28 μM. | 21419530 | ||
| MDA-MB-231 | Cytotoxicity assay | 2 days | Cytotoxicity against human MDA-MB-231 cells after 2 days, IC50 = 1.23 μM. | 21419530 | ||
| A549 | Anticancer assay | Anticancer activity against human A549 cells by SRB assay, IC50 = 1.85 μM. | 21420296 | |||
| SK-MEL-2 | Anticancer assay | Anticancer activity against human SK-MEL-2 cells by SRB assay, IC50 = 1.17 μM. | 21420296 | |||
| SKOV3 | Anticancer assay | Anticancer activity against human SKOV3 cells by SRB assay, IC50 = 1.81 μM. | 21420296 | |||
| XF498 | Anticancer assay | Anticancer activity against human XF498 cells by SRB assay, IC50 = 1.72 μM. | 21420296 | |||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells, IC50 = 1.4 μM. | 21439821 | |||
| HL60/MX2 | Cytotoxicity assay | Cytotoxicity against human HL60/MX2 cells, IC50 = 15.75 μM. | 21439821 | |||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by sulforhodamine B assay, IC50 = 5.62 μM. | 21477899 | ||
| HCT15 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT15 cells after 48 hrs by sulforhodamine B assay, IC50 = 7.15 μM. | 21477899 | ||
| Hep2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human Hep2 cells after 48 hrs by sulforhodamine B assay, IC50 = 2.15 μM. | 21477899 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by sulforhodamine B assay, IC50 = 19 μM. | 21477899 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by sulforhodamine B assay, IC50 = 17.5 μM. | 21477899 | ||
| SF295 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SF295 cells after 48 hrs by sulforhodamine B assay, IC50 = 13.5 μM. | 21477899 | ||
| A2780 | Growth inhibition assay | 48 hrs | Growth inhibition of human A2780 cells after 48 hrs by sulforhodamine B assay, GI50 = 1.3 μM. | 21489802 | ||
| A549 | Growth inhibition assay | 48 hrs | Growth inhibition of human A549 cells after 48 hrs by sulforhodamine B assay, GI50 = 3.08 μM. | 21489802 | ||
| COLO205 | Growth inhibition assay | 48 hrs | Growth inhibition of human COLO205 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.13 μM. | 21489802 | ||
| DWD | Growth inhibition assay | 48 hrs | Growth inhibition of human DWD cells after 48 hrs by sulforhodamine B assay, GI50 = 0.6 μM. | 21489802 | ||
| Gurav | Growth inhibition assay | 48 hrs | Growth inhibition of human Gurav cells after 48 hrs by sulforhodamine B assay, GI50 = 0.5 μM. | 21489802 | ||
| HOP62 | Growth inhibition assay | 48 hrs | Growth inhibition of human HOP62 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.8 μM. | 21489802 | ||
| KB | Growth inhibition assay | 48 hrs | Growth inhibition of human KB cells after 48 hrs by sulforhodamine B assay, GI50 = 0.3 μM. | 21489802 | ||
| MCF7 | Growth inhibition assay | 48 hrs | Growth inhibition of human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50 = 2.1 μM. | 21489802 | ||
| PC3 | Growth inhibition assay | 48 hrs | Growth inhibition of human PC3 cells after 48 hrs by sulforhodamine B assay, GI50 = 2.6 μM. | 21489802 | ||
| SiHa | Growth inhibition assay | 48 hrs | Growth inhibition of human SiHa cells after 48 hrs by sulforhodamine B assay, GI50 = 3.1 μM. | 21489802 | ||
| ZR-75-1 | Growth inhibition assay | 48 hrs | Growth inhibition of human ZR-75-1 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.2 μM. | 21489802 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 4 μM. | 21570846 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by MTT assay, IC50 = 10 μM. | 21570846 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days, IC50 = 0.5 μM. | 21601964 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days, IC50 = 1.1 μM. | 21601964 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days, IC50 = 1.4 μM. | 21601964 | ||
| HL60 | Cytotoxicity assay | 2 days | Cytotoxicity against human HL60 cells after 2 days, IC50 = 1 μM. | 21601964 | ||
| MDA-MB-231 | Cytotoxicity assay | 2 days | Cytotoxicity against human MDA-MB-231 cells after 2 days, INH = 0.7 μM. | 21601964 | ||
| AGS | Anticancer assay | 3 days | Anticancer activity against human AGS cells after 3 days by MTT assay, IC50 = 0.36 μM. | 21621882 | ||
| J82 | Anticancer assay | 3 days | Anticancer activity against human J82 cells after 3 days by MTT assay, IC50 = 0.8 μM. | 21621882 | ||
| MRC5 | Anticancer assay | 3 days | Anticancer activity against human MRC5 cells after 3 days by MTT assay, IC50 = 3.9 μM. | 21621882 | ||
| SKMES1 | Anticancer assay | 3 days | Anticancer activity against human SKMES1 cells after 3 days by MTT assay, IC50 = 2.8 μM. | 21621882 | ||
| Caco2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Caco2 cells after 72 hrs by MTS assay, IC50 = 18.6 μM. | 21724394 | ||
| CCD-18Co | Cytotoxicity assay | 72 hrs | Cytotoxicity against human CCD-18Co cells after 72 hrs by MTS assay, IC50 = 44.4 μM. | 21724394 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells after 72 hrs by MTS assay, IC50 = 25.5 μM. | 21724394 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 14.8 μM. | 21733601 | ||
| SiHa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SiHa cells after 48 hrs by MTT assay, IC50 = 30.7 μM. | 21733601 | ||
| A2780 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A2780 cells after 48 hrs by sulforhodamine B assay, GI50 = 1.3 μM. | 21737288 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by sulforhodamine B assay, GI50 = 3.08 μM. | 21737288 | ||
| COLO205 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human COLO205 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.13 μM. | 21737288 | ||
| DWD | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DWD cells after 48 hrs by sulforhodamine B assay, GI50 = 0.6 μM. | 21737288 | ||
| Gurav | Cytotoxicity assay | 48 hrs | Cytotoxicity against human Gurav cells after 48 hrs by sulforhodamine B assay, GI50 = 0.5 μM. | 21737288 | ||
| HOP62 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HOP62 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.8 μM. | 21737288 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50 = 2.1 μM. | 21737288 | ||
| ZR-75-1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human ZR-75-1 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.2 μM. | 21737288 | ||
| CCRF-CEM | Antiproliferative assay | 96 hrs | Antiproliferative activity against human CCRF-CEM cells after 96 hrs by MTT assay, CC50 = 0.09 μM. | 21741130 | ||
| CCRF-SB | Antiproliferative assay | 96 hrs | Antiproliferative activity against human CCRF-SB cells after 96 hrs by MTT assay, CC50 = 0.1 μM. | 21741130 | ||
| DU145 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human DU145 cells after 96 hrs by MTT assay, CC50 = 0.4 μM. | 21741130 | ||
| HepG2 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HepG2 cells after 96 hrs by MTT assay, CC50 = 0.7 μM. | 21741130 | ||
| MCF7 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MCF7 cells after 96 hrs by MTT assay, CC50 = 1 μM. | 21741130 | ||
| SK-MEL-28 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human SK-MEL-28 cells after 96 hrs by MTT assay, CC50 = 1.2 μM. | 21741130 | ||
| SKMES1 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human SKMES1 cells after 96 hrs by MTT assay, CC50 = 0.3 μM. | 21741130 | ||
| WIL2-NS | Antiproliferative assay | 96 hrs | Antiproliferative activity against human WIL2-NS cells after 96 hrs by MTT assay, CC50 = 0.2 μM. | 21741130 | ||
| MT4 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human MT4 cells infected with HTLV-1 after 96 hrs by MTT assay, CC50 = 0.09 μM. | 21741130 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days by MTT assay, IC50 = 6.27 μM. | 21821321 | ||
| K562 | Cytotoxicity assay | 2 days | Cytotoxicity against human K562 cells after 2 days by MTT assay, IC50 = 2.11 μM. | 21821321 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by sulforhodamine B assay, IC50 = 0.44 μM. | 21936523 | |||
| SK-MEL-2 | Cytotoxicity assay | Cytotoxicity against human SK-MEL-2 cells by sulforhodamine B assay, IC50 = 0.41 μM. | 21936523 | |||
| SKOV3 | Cytotoxicity assay | Cytotoxicity against human SKOV3 cells by sulforhodamine B assay, IC50 = 1.31 μM. | 21936523 | |||
| HeLa | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HeLa cells assessed as inhibition of cell viability after 24 hrs by MTT assay, IC50 = 23.33 μM. | 21983445 | ||
| HBL100 | Growth inhibition assay | Growth inhibition of human HBL100 cells, GI50 = 2.3 μM. | 21986585 | |||
| HeLa | Growth inhibition assay | Growth inhibition of human HeLa cells, GI50 = 3 μM. | 21986585 | |||
| Ishikawa | Growth inhibition assay | Growth inhibition of human Ishikawa cells, GI50 = 11 μM. | 21986585 | |||
| SW1573 | Growth inhibition assay | Growth inhibition of human SW1573 cells, GI50 = 15 μM. | 21986585 | |||
| T47D | Growth inhibition assay | Growth inhibition of human T47D cells, GI50 = 22 μM. | 21986585 | |||
| WiDr | Growth inhibition assay | Growth inhibition of human WiDr cells, GI50 = 23 μM. | 21986585 | |||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 24.9 μM. | 22041063 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as growth inhibition after 48 hrs by CCK-8 assay, IC50 = 2.76 μM. | 22041063 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as growth inhibition after 48 hrs by CCK-8 assay, IC50 = 2.85 μM. | 22041063 | ||
| SiHa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SiHa cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 40.4 μM. | 22041063 | ||
| KB | Growth inhibition assay | 72 hrs | Growth inhibition of human KB cells after 72 hrs by methylene blue assay, IC50 = 1.1 μM. | 22060033 | ||
| KB-S15 | Growth inhibition assay | 72 hrs | Growth inhibition of human KB-S15 cells after 72 hrs by methylene blue assay, IC50 = 3.5 μM. | 22060033 | ||
| KB-VIN10 | Growth inhibition assay | 72 hrs | Growth inhibition of human KB-VIN10 cells after 72 hrs by methylene blue assay, IC50 = 23 μM. | 22060033 | ||
| CCRF-CEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human CCRF-CEM cells after 72 hrs by MTT assay, GI50 = 12.59 μM. | 22104973 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay, GI50 = 31.62 μM. | 22104973 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, GI50 = 19.95 μM. | 22104973 | ||
| THP1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human THP1 cells after 48 hrs by MTT assay, IC50 = 2.16 μM. | 22115593 | ||
| U937 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human U937 cells after 48 hrs by MTT assay, IC50 = 17.94 μM. | 22115593 | ||
| B16F10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against mouse B16F10 cells after 48 hrs by MTT assay, IC50 = 18.69 μM. | 22115593 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells after 72 hrs by sulforhodamine B and MTT assay, IC50 = 0.91 μM. | 22119124 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by sulforhodamine B and MTT assay, IC50 = 5.16 μM. | 22119124 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by sulforhodamine B and MTT assay, IC50 = 0.85 μM. | 22119124 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by sulforhodamine B and MTT assay, IC50 = 1.09 μM. | 22119124 | ||
| A375 | Anticancer assay | 48 hrs | Anticancer activity against human A375 cells after 48 hrs by MTT assay, IC50 = 1.39 μM. | 22136907 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells after 48 hrs by MTT assay, IC50 = 2.34 μM. | 22136907 | ||
| ACHN | Anticancer assay | 48 hrs | Anticancer activity against human ACHN cells after 48 hrs by MTT assay, IC50 = 7.61 μM. | 22136907 | ||
| HT-29 | Anticancer assay | 48 hrs | Anticancer activity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 1.81 μM. | 22136907 | ||
| MCF7 | Anticancer assay | 48 hrs | Anticancer activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 0.68 μM. | 22136907 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 24.9 μM. | 22244588 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by CCK-8 assay, IC50 = 40.4 μM. | 22244588 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by CCK-8 assay, IC50 = 2.85 μM. | 22244588 | ||
| SiHa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SiHa cells after 48 hrs by MTT assay, IC50 = 2.76 μM. | 22244588 | ||
| DU145 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human DU145 cells after 96 hrs, IC50 = 3 μM. | 22276775 | ||
| HepG2 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human HepG2 cells after 96 hrs, IC50 = 4 μM. | 22276775 | ||
| MT4 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human MT4 cells after 96 hrs, IC50 = 0.09 μM. | 22276775 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days, IC50 = 2.94 μM. | 22318164 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days, IC50 = 1.33 μM. | 22318164 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days, IC50 = 3.2 μM. | 22318164 | ||
| K562 | Cytotoxicity assay | 2 days | Cytotoxicity against human K562 cells after 2 days, IC50 = 2.09 μM. | 22318164 | ||
| MCF7 | Cytotoxicity assay | 2 days | Cytotoxicity against human MCF7 cells after 2 days, IC50 = 3.25 μM. | 22318164 | ||
| A549 | Cytotoxicity assay | 2 days | Cytotoxicity against human A549 cells after 2 days by MTT assay, IC50 = 9.51 μM. | 22325897 | ||
| HL60 | Cytotoxicity assay | 2 days | Cytotoxicity against human HL60 cells after 2 days by MTT assay, IC50 = 1.83 μM. | 22325897 | ||
| Jurkat | Cytotoxicity assay | 2 days | Cytotoxicity against human Jurkat cells after 2 days by MTT assay, IC50 = 5.35 μM. | 22325897 | ||
| THP1 | Cytotoxicity assay | 2 days | Cytotoxicity against human THP1 cells after 2 days by MTT assay, IC50 = 2.16 μM. | 22325897 | ||
| U937 | Cytotoxicity assay | 2 days | Cytotoxicity against human U937 cells after 2 days by MTT assay, IC50 = 17.94 μM. | 22325897 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 23.8 μM. | 22342101 | ||
| Hep3B | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Hep3B cells after 72 hrs by MTT assay, IC50 = 6.7 μM. | 22342101 | ||
| U87 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U87 cells after 72 hrs by MTT assay, IC50 = 12.4 μM. | 22342101 | ||
| A2780 | Growth inhibition assay | 48 hrs | Growth inhibition of human A2780 cells after 48 hrs by sulforhodamine B assay, GI50 = 1.31 μM. | 22364746 | ||
| A549 | Growth inhibition assay | 48 hrs | Growth inhibition of human A549 cells after 48 hrs by sulforhodamine B assay, GI50 = 3.08 μM. | 22364746 | ||
| COLO205 | Growth inhibition assay | 48 hrs | Growth inhibition of human COLO205 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.13 μM. | 22364746 | ||
| DWD | Growth inhibition assay | 48 hrs | Growth inhibition of human DWD cells after 48 hrs by sulforhodamine B assay, GI50 = 0.62 μM. | 22364746 | ||
| HOP62 | Growth inhibition assay | 48 hrs | Growth inhibition of human HOP62 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.8 μM. | 22364746 | ||
| KB | Growth inhibition assay | 48 hrs | Growth inhibition of human KB cells after 48 hrs by sulforhodamine B assay, GI50 = 0.31 μM. | 22364746 | ||
| MCF7 | Growth inhibition assay | 48 hrs | Growth inhibition of human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50 = 2.11 μM. | 22364746 | ||
| ZR-75-1 | Growth inhibition assay | 48 hrs | Growth inhibition of human ZR-75-1 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.2 μM. | 22364746 | ||
| Vero | Cytotoxicity assay | 48 hrs | Cytotoxicity against african green monkey Vero cells after 48 hrs by MTT assay, LC50 = 18 μM. | 22381050 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HEK293 cells after 48 hrs by MTT assay, LC50 = 28 μM. | 22381050 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days by cell counting kit-8 analysis, IC50 = 1.21 μM. | 22503656 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days by cell counting kit-8 analysis, IC50 = 1.17 μM. | 22503656 | ||
| HEK293 | Cytotoxicity assay | 2 days | Cytotoxicity against human HEK293 cells after 2 days by cell counting kit-8 analysis, IC50 = 1.21 μM. | 22503656 | ||
| T47D | Cytotoxicity assay | 2 days | Cytotoxicity against human T47D cells after 2 days by cell counting kit-8 analysis, IC50 = 2.39 μM. | 22503656 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 0.48 μM. | 22564383 | ||
| T98G | Cytotoxicity assay | 48 hrs | Cytotoxicity against human T98G cells expressing mutant p53 after 48 hrs by MTT assay, IC50 = 0.46 μM. | 22564383 | ||
| U87 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human U87 cells expressing wild-type p53 after 48 hrs by MTT assay, IC50 = 0.62 μM. | 22564383 | ||
| COLO205 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human COLO205 cells after 48 hrs by MTT assay, IC50 = 0.43 μM. | 22578452 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 4.76 μM. | 22578452 | ||
| THP1 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human THP1 cells after 48 hrs by MTT assay, IC50 = 2.16 μM. | 22578452 | ||
| U937 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human U937 cells after 48 hrs by MTT assay, IC50 = 17.96 μM. | 22578452 | ||
| B16F10 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse B16F10 cells after 48 hrs by MTT assay, IC50 = 18.71 μM. | 22578452 | ||
| CCRF-CEM | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CCRF-CEM cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 0.24 μM. | 22582991 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 0.11 μM. | 22582991 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 2.3 μM. | 22582991 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by SRB assay, GI50 = 3.56 μM. | 22682920 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by SRB assay, TGI = 40.18 μM. | 22682920 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by SRB assay, GI50 = 0.56 μM. | 22682920 | ||
| TK10 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human TK10 cells after 48 hrs by SRB assay, GI50 = 7.19 μM. | 22682920 | ||
| TK10 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human TK10 cells after 48 hrs by SRB assay, TGI = 49.74 μM. | 22682920 | ||
| UACC62 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human UACC62 cells after 48 hrs by SRB assay, GI50 = 0.89 μM. | 22682920 | ||
| Vero | Cytotoxicity assay | 72 hrs | Cytotoxicity against african green monkey Vero cells after 72 hrs by MTT assay, IC50 = 31.14 μM. | 22687745 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 10.12 μM. | 22687745 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HepG2 cells after 72 hrs by MTT assay, IC50 = 5.48 μM. | 22687745 | ||
| SGC7901 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SGC7901 cells after 72 hrs by MTT assay, IC50 = 12.09 μM. | 22687745 | ||
| SH-SY5Y | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SH-SY5Y cells after 72 hrs by MTT assay, IC50 = 5.37 μM. | 22687745 | ||
| L929 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse L929 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 22687745 | ||
| KBCPT100 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human camptothecin-resistant KBCPT100 cells after 72 hrs by ethylene blue dye assay, IC50 = 0.336 μM. | 22698783 | ||
| KB | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KB cells after 72 hrs by ethylene blue dye assay, IC50 = 0.475 μM. | 22698783 | ||
| KBCPT100 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human partially revertant camptothecin-resistant KBCPT100 cells after 72 hrs by ethylene blue dye assay, IC50 = 0.374 μM. | 22698783 | ||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 cells after 72 hrs by MTT assay, IC50 = 1.42 μM. | 22819942 | ||
| HCT15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT15 cells after 72 hrs by MTT assay, IC50 = 0.28 μM. | 22819942 | ||
| HEK293 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HEK293 cells after 72 hrs by MTT assay, IC50 = 0.41 μM. | 22819942 | ||
| T47D | Cytotoxicity assay | 72 hrs | Cytotoxicity against human T47D cells after 72 hrs by MTT assay, IC50 = 1.21 μM. | 22819942 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity at human A549 cells after 72 hrs by MTT assay, IC50 = 0.8 μM. | 22861499 | ||
| DMS53 | Antiproliferative assay | 72 hrs | Antiproliferative activity at human DMS53 cells after 72 hrs by MTT assay, IC50 = 1.5 μM. | 22861499 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.83 μM. | 22889556 | ||
| TK10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human TK10 cells after 48 hrs by sulforhodamine B assay, GI50 = 5.89 μM. | 22889556 | ||
| TK10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human TK10 cells after 48 hrs by sulforhodamine B assay, TGI = 43.33 μM. | 22889556 | ||
| UACC62 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human UACC62 cells after 48 hrs by sulforhodamine B assay, GI50 = 3.41 μM. | 22889556 | ||
| UACC62 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human UACC62 cells after 48 hrs by sulforhodamine B assay, TGI = 4 μM. | 22889556 | ||
| DOGUM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DOGUM cells after 72 hrs by MTT assay, IC50 = 0.075 μM. | 23031590 | ||
| DOGUM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human etoposide-resistant DOGUM cells after 72 hrs by MTT assay, IC50 = 0.28 μM. | 23031590 | ||
| GUMBUS | Cytotoxicity assay | 72 hrs | Cytotoxicity against human etoposide-resistant GUMBUS cells after 72 hrs by MTT assay, IC50 = 0.57 μM. | 23031590 | ||
| GUMBUS | Cytotoxicity assay | 72 hrs | Cytotoxicity against human GUMBUS cells after 72 hrs by MTT assay, IC50 = 0.042 μM. | 23031590 | ||
| Bel7402 | Cytotoxicity assay | Cytotoxicity against human Bel7402 cells, IC50 = 0.08 μM. | 23092389 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells, IC50 = 0.08 μM. | 23092389 | |||
| SGC7901 | Cytotoxicity assay | Cytotoxicity against human SGC7901 cells, IC50 = 0.08 μM. | 23092389 | |||
| P388 | Cytotoxicity assay | Cytotoxicity against mouse P388 cells, IC50 = 0.08 μM. | 23092389 | |||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells after 72 hrs by SRB assay, GI50 = 2.2 μM. | 23153810 | ||
| HT-29 | Growth inhibition assay | 72 hrs | Growth inhibition of human HT-29 cells after 72 hrs by SRB assay, GI50 = 9.4 μM. | 23153810 | ||
| MDA-MB-231 | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-231 cells after 72 hrs by SRB assay, GI50 = 7.2 μM. | 23153810 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 2.8 μM. | 23182091 | ||
| ACHN | Cytotoxicity assay | 72 hrs | Cytotoxicity against human ACHN cells after 72 hrs by MTT assay, IC50 = 7.61 μM. | 23182091 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 1.52 μM. | 23182091 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay, IC50 = 1.81 μM. | 23182091 | ||
| B16 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse B16 cells after 72 hrs by MTT assay, IC50 = 1.39 μM. | 23182091 | ||
| HeLa | Antitumor assay | 48 hrs | Antitumor activity against human HeLa cells assessed as growth inhibition measured after 48 hrs by MTT assay, IC50 = 8.32 μM. | 23313635 | ||
| HepG2 | Antitumor assay | 48 hrs | Antitumor activity against human HepG2 cells assessed as growth inhibition measured after 48 hrs by MTT assay, IC50 = 9.14 μM. | 23313635 | ||
| Vero | Cytotoxicity assay | 48 hrs | Cytotoxicity against african green monkey Vero cells after 48 hrs by MTT assay, LC50 = 29 μM. | 23321564 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by MTT assay, LC50 = 17 μM. | 23321564 | ||
| BGC832 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human BGC832 cells after 24 hrs by MTT assay, IC50 = 26.85 μM. | 23369687 | ||
| HeLa | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HeLa cells after 24 hrs by MTT assay, IC50 = 17.68 μM. | 23369687 | ||
| K562 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human K562 cells after 24 hrs by MTT assay, IC50 = 2.85 μM. | 23369687 | ||
| AGS | Cytotoxicity assay | 72 hrs | Cytotoxicity against human AGS cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 0.36 μM. | 23454511 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 1.8 μM. | 23454511 | ||
| J82 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human J82 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 2.8 μM. | 23454511 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 3.9 μM. | 23454511 | ||
| SKMES1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKMES1 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 2.5 μM. | 23454511 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by SRB assay, IC50 = 2.58 μM. | 23490151 | ||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 cells after 72 hrs by SRB assay, IC50 = 2.03 μM. | 23490151 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by SRB assay, IC50 = 3.88 μM. | 23490151 | ||
| SNU638 | Cytotoxicity assay | Cytotoxicity against human SNU638 cells by MTT assay, IC50 = 2.57 μM. | 23608763 | |||
| T47D | Cytotoxicity assay | Cytotoxicity against human T47D cells by MTT assay, IC50 = 4.98 μM. | 23608763 | |||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 6.41 μM. | 23668986 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 3.2 μM. | 23711769 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 7.06 μM. | 23711769 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 2.91 μM. | 23711769 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as growth inhibition after 48 hrs by CCK-8 assay, IC50 = 1.15 μM. | 23711769 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay, GI50 = 31.62 μM. | 23831811 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, GI50 = 19.95 μM. | 23831811 | ||
| A2780 | Growth inhibition assay | 48 hrs | Growth inhibition of human A2780 cells after 48 hrs by sulforhodamine B assay, GI50 = 1.31 μM. | 23849207 | ||
| A549 | Growth inhibition assay | 48 hrs | Growth inhibition of human A549 cells after 48 hrs by sulforhodamine B assay, GI50 = 3.08 μM. | 23849207 | ||
| COLO205 | Growth inhibition assay | 48 hrs | Growth inhibition of human COLO205 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.13 μM. | 23849207 | ||
| DWD | Growth inhibition assay | 48 hrs | Growth inhibition of human DWD cells after 48 hrs by sulforhodamine B assay, GI50 = 0.62 μM. | 23849207 | ||
| Gurav | Growth inhibition assay | 48 hrs | Growth inhibition of human Gurav cells after 48 hrs by sulforhodamine B assay, GI50 = 0.51 μM. | 23849207 | ||
| HOP62 | Growth inhibition assay | 48 hrs | Growth inhibition of human HOP62 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.8 μM. | 23849207 | ||
| KB | Growth inhibition assay | 48 hrs | Growth inhibition of human KB cells after 48 hrs by sulforhodamine B assay, GI50 = 0.31 μM. | 23849207 | ||
| MCF7 | Growth inhibition assay | 48 hrs | Growth inhibition of human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50 = 2.11 μM. | 23849207 | ||
| SiHa | Growth inhibition assay | 48 hrs | Growth inhibition of human SiHa cells after 48 hrs by sulforhodamine B assay, GI50 = 3.11 μM. | 23849207 | ||
| ZR-75-1 | Growth inhibition assay | 48 hrs | Growth inhibition of human ZR-75-1 cells after 48 hrs by sulforhodamine B assay, GI50 = 0.2 μM. | 23849207 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.5 μM. | 23865778 | ||
| U937 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U937 cells after 72 hrs by MTT assay, IC50 = 1.4 μM. | 23865778 | ||
| H460 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human H460 cells after 48 hrs by MTT assay, IC50 = 1 μM. | 23867385 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 5.48 μM. | 23867385 | ||
| H460 | Cytotoxicity assay | Cytotoxicity against human H460 cells assessed as growth inhibition by MTT assay, IC50 = 30 μM. | 23920485 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells assessed as growth inhibition by MTT assay, IC50 = 20.9 μM. | 23920485 | |||
| PC3 | Cytotoxicity assay | Cytotoxicity against human PC3 cells assessed as growth inhibition by MTT assay, IC50 = 18.2 μM. | 23920485 | |||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 32.05 μM. | 23968711 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.33 μM. | 23968711 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells after 48 hrs by MTT assay, IC50 = 8.45 μM. | 23968711 | ||
| MCF10A | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF10A cells after 72 hrs by MTT assay, IC50 = 36.26 μM. | 23999041 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by SRB assay, GI50 = 0.87 μM. | 23999041 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 1.2 μM. | 23999041 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by SRB assay, GI50 = 1.67 μM. | 23999041 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 1.5 μM. | 23999041 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 14.3 μM. | 24012378 | ||
| CCRF-CEM | Antiproliferative assay | 72 hrs | Antiproliferative activity against human CCRF-CEM cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 1.1 μM. | 24012378 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 3.9 μM. | 24012378 | ||
| Jurkat | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Jurkat cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 1.2 μM. | 24012378 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 10.9 μM. | 24012378 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 21.2 μM. | 24012378 | ||
| HEK293 | Cytotoxicity assay | 2 days | Cytotoxicity against HEK293 cells after 2 days by CCK-8 assay, IC50 = 0.72 μM. | 24013413 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days by CCK-8 assay, IC50 = 9.29 μM. | 24013413 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days by CCK-8 assay, IC50 = 5.12 μM. | 24013413 | ||
| T47D | Cytotoxicity assay | 2 days | Cytotoxicity against human T47D cells after 2 days by CCK-8 assay, IC50 = 3.42 μM. | 24013413 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 1.71 μM. | 24095018 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells assessed as cell viability after 72 hrs by MTT assay, IC50 = 0.87 μM. | 24095018 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 14.9 μM. | 24095019 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells after 48 hrs by MTT assay, IC50 = 6.54 μM. | 24095019 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 4.73 μM. | 24095019 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 11.52 μM. | 24095019 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells assessed as growth inhibition after 72 hrs by methylene blue staining-based assay, IC50 = 1.1 μM. | 24106982 | ||
| KB-S15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB-S15 cells overexpressing P-gp170/MDR assessed as growth inhibition after 72 hrs by methylene blue staining-based assay, IC50 = 3.5 μM. | 24106982 | ||
| KB-VIN10 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB-VIN10 cells overexpressing P-gp170/MDR assessed as growth inhibition after 72 hrs by methylene blue staining-based assay, IC50 = 23 μM. | 24106982 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 3.2 μM. | 24140948 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells after 48 hrs by MTT assay, IC50 = 7.06 μM. | 24140948 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 2.91 μM. | 24140948 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by cell counting kit-8 assay, IC50 = 1.15 μM. | 24140948 | ||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells after 72 hrs by MTT assay, IC50 = 2 μM. | 24180210 | ||
| Hs683 | Growth inhibition assay | 72 hrs | Growth inhibition of human Hs683 cells after 72 hrs by MTT assay, IC50 = 2 μM. | 24180210 | ||
| MCF7 | Growth inhibition assay | 72 hrs | Growth inhibition of human MCF7 cells after 72 hrs by MTT assay, IC50 = 4 μM. | 24180210 | ||
| SK-MEL-28 | Growth inhibition assay | 72 hrs | Growth inhibition of human SK-MEL-28 cells after 72 hrs by MTT assay, IC50 = 3 μM. | 24180210 | ||
| U373 | Growth inhibition assay | 72 hrs | Growth inhibition of human U373 cells after 72 hrs by MTT assay, IC50 = 25 μM. | 24180210 | ||
| HBL100 | Growth inhibition assay | 48 hrs | Growth inhibition of human HBL100 cells after 48 hrs by SRB assay, GI50 = 1.4 μM. | 24211632 | ||
| HeLa | Growth inhibition assay | 48 hrs | Growth inhibition of human HeLa cells after 48 hrs by SRB assay, GI50 = 3.3 μM. | 24211632 | ||
| SW1573 | Growth inhibition assay | 48 hrs | Growth inhibition of human SW1573 cells after 48 hrs by SRB assay, GI50 = 15 μM. | 24211632 | ||
| T47D | Growth inhibition assay | 48 hrs | Growth inhibition of human T47D cells after 48 hrs by SRB assay, GI50 = 22 μM. | 24211632 | ||
| WiDr | Growth inhibition assay | 48 hrs | Growth inhibition of human WiDr cells after 48 hrs by SRB assay, GI50 = 23 μM. | 24211632 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells assessed as cell viability after 72 hrs by MTS assay, IC50 = 1 μM. | 24328269 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by sulforhodamine B assay, IC50 = 2.58 μM. | 24332656 | ||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 cells after 72 hrs by sulforhodamine B assay, IC50 = 2.03 μM. | 24332656 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by sulforhodamine B assay, IC50 = 3.88 μM. | 24332656 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay relative to untreated control, GI50 = 4.46 μM. | 24389510 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay relative to untreated control, GI50 = 31.62 μM. | 24389510 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay relative to untreated control, GI50 = 19.95 μM. | 24389510 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay relative to untreated control, GI50 = 0.63 μM. | 24389510 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay relative to untreated control, TGI = 3.98 μM. | 24389510 | ||
| RWPE1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RWPE1 cells after 72 hrs by MTT assay relative to untreated control, GI50 = 0.9 μM. | 24389510 | ||
| RWPE1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RWPE1 cells after 72 hrs by MTT assay relative to untreated control, TGI = 4.8 μM. | 24389510 | ||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells by MTT assay, IC50 = 0.0057 μM. | 24422592 | |||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 4.71 μM. | 24487188 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 21.45 μM. | 24487188 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 23.9 μM. | 24487188 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 24.22 μM. | 24487188 | ||
| ME180 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human ME180 cells after 48 hrs by MTT assay, IC50 = 8.9 μM. | 24487188 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by MTT assay, IC50 = 14.4 μM. | 24487188 | ||
| DLD1 | Growth inhibition assay | 72 hrs | Growth inhibition of human DLD1 cells assessed as inhibition of cell viability after 72 hrs by MTT assay, IC50 = 20 μM. | 24527835 | ||
| HeLa | Growth inhibition assay | 72 hrs | Growth inhibition of human HeLa cells assessed as inhibition of cell viability after 72 hrs by MTT assay, IC50 = 16 μM. | 24527835 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by SRB assay, IC50 = 2.58 μM. | 24553146 | ||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 cells after 72 hrs by SRB assay, IC50 = 2.03 μM. | 24553146 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by SRB assay, IC50 = 3.88 μM. | 24553146 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by methylene blue assay, IC50 = 1.5 μM. | 24657567 | ||
| KB-S15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human vincristine/paclitaxel-resistant KB-S15 cells overexpressing P-gp170 after 72 hrs by methylene blue assay, IC50 = 3.5 μM. | 24657567 | ||
| KB-VIN10 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human vincristine/paclitaxel-resistant KB-VIN10 cells overexpressing P-gp170 after 72 hrs by methylene blue assay, IC50 = 26.4 μM. | 24657567 | ||
| KB-7d | Cytotoxicity assay | 72 hrs | Cytotoxicity against human VP16-resistant KB-7d cells overexpressing MRP after 72 hrs by methylene blue assay, IC50 = 46.1 μM. | 24657567 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells after 48 hrs by MTT assay, IC50 = 2.45 μM. | 24721832 | ||
| ACHN | Anticancer assay | 48 hrs | Anticancer activity against human ACHN cells after 48 hrs by MTT assay, IC50 = 2.04 μM. | 24721832 | ||
| DU145 | Anticancer assay | 48 hrs | Anticancer activity against human DU145 cells after 48 hrs by MTT assay, IC50 = 3.17 μM. | 24721832 | ||
| HeLa | Anticancer assay | 48 hrs | Anticancer activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 2.75 μM. | 24721832 | ||
| HT-29 | Anticancer assay | 48 hrs | Anticancer activity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 1.47 μM. | 24721832 | ||
| BGC823 | Cytotoxicity assay | Cytotoxicity against human BGC823 cells, IC50 = 25.72 μM. | 24775914 | |||
| BGC823 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BGC823 cells after 48 hrs by MTT assay, IC50 = 30.74 μM. | 24775914 | ||
| HepG2 | Cytotoxicity assay | Cytotoxicity against human HepG2 cells, IC50 = 1.15 μM. | 24775914 | |||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 15.32 μM. | 24775914 | ||
| HL-7702 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL-7702 cells after 48 hrs by MTT assay, IC50 = 24.61 μM. | 24775914 | ||
| H460 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human H460 cells after 72 hrs by MTT assay, IC50 = 0.1 μM. | 24780599 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by MTT assay, IC50 = 5 μM. | 24780599 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 24780599 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 30.16 μM. | 24836071 | ||
| MOLT3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MOLT3 cells after 48 hrs by MTT assay, IC50 = 0.051 μM. | 24836071 | ||
| EPG85-257P | Antiproliferative assay | 5 days | Antiproliferative activity against human EPG85-257P cells after 5 days by sulforhodamine B assay, IC50 = 0.105 μM. | 24864039 | ||
| EPG85-257RD | Antiproliferative assay | 5 days | Antiproliferative activity against human EPG85-257RD cells after 5 days by sulforhodamine B assay, IC50 = 6.2 μM. | 24864039 | ||
| EPG85-257RN | Antiproliferative assay | 5 days | Antiproliferative activity against human EPG85-257RN cells after 5 days by sulforhodamine B assay, IC50 = 1.55 μM. | 24864039 | ||
| EPP85-181P | Antiproliferative assay | 5 days | Antiproliferative activity against human EPP85-181P cells after 5 days by sulforhodamine B assay, IC50 = 0.58 μM. | 24864039 | ||
| EPP85-181RN | Antiproliferative assay | 5 days | Antiproliferative activity against human EPP85-181RN cells after 5 days by sulforhodamine B assay, IC50 = 4.5 μM. | 24864039 | ||
| HT-29 | Antiproliferative assay | 5 days | Antiproliferative activity against human HT-29 cells after 5 days by sulforhodamine B assay, IC50 = 2.3 μM. | 24864039 | ||
| HT-29RD | Antiproliferative assay | 5 days | Antiproliferative activity against human HT-29RD cells after 5 days by sulforhodamine B assay, IC50 = 26 μM. | 24864039 | ||
| HT-29RN | Antiproliferative assay | 5 days | Antiproliferative activity against human HT-29RN cells after 5 days by sulforhodamine B assay, IC50 = 35 μM. | 24864039 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days, IC50 = 13.85 μM. | 24904965 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days, IC50 = 9.45 μM. | 24904965 | ||
| MCF10A | Cytotoxicity assay | 2 days | Cytotoxicity against human MCF10A cells after 2 days, IC50 = 12.23 μM. | 24904965 | ||
| T47D | Cytotoxicity assay | 2 days | Cytotoxicity against human T47D cells after 2 days, IC50 = 2.57 μM. | 24904965 | ||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells after 72 hrs by MTT assay, IC50 = 0.1 μM. | 24950030 | ||
| Hs683 | Growth inhibition assay | 72 hrs | Growth inhibition of human Hs683 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 24950030 | ||
| MCF7 | Growth inhibition assay | 72 hrs | Growth inhibition of human MCF7 cells after 72 hrs by MTT assay, IC50 = 17 μM. | 24950030 | ||
| PC3 | Growth inhibition assay | 72 hrs | Growth inhibition of human PC3 cells after 72 hrs by MTT assay, IC50 = 2 μM. | 24950030 | ||
| SK-MEL-28 | Growth inhibition assay | 72 hrs | Growth inhibition of human SK-MEL-28 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 24950030 | ||
| U373 | Growth inhibition assay | 72 hrs | Growth inhibition of human U373 cells after 72 hrs by MTT assay, IC50 = 7 μM. | 24950030 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50 = 0.6 μM. | 25014747 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 20.389 μM. | 25019480 | ||
| MOLT3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MOLT3 cells after 48 hrs by MTT assay, IC50 = 0.051 μM. | 25019480 | ||
| KB | Antiproliferative assay | Antiproliferative activity against human KB cells, IC50 = 1.1 μM. | 25059503 | |||
| KB-S15 | Antiproliferative assay | Antiproliferative activity against human KB-S15 cells, IC50 = 3.5 μM. | 25059503 | |||
| KB-VIN10 | Antiproliferative assay | Antiproliferative activity against human KB-VIN10 cells, IC50 = 23 μM. | 25059503 | |||
| HEK293 | Cytotoxicity assay | 4 days | Cytotoxicity against HEK293 cells after 4 days by CCK-8 assay, IC50 = 1.04 μM. | 25062006 | ||
| DU145 | Cytotoxicity assay | 4 days | Cytotoxicity against human DU145 cells after 4 days by CCK-8 assay, IC50 = 0.001 μM. | 25062006 | ||
| HCT15 | Cytotoxicity assay | 4 days | Cytotoxicity against human HCT15 cells after 4 days by CCK-8 assay, IC50 = 7.74 μM. | 25062006 | ||
| T47D | Cytotoxicity assay | 4 days | Cytotoxicity against human T47D cells after 4 days by CCK-8 assay, IC50 = 8.74 μM. | 25062006 | ||
| Vero | Cytotoxicity assay | 48 hrs | Cytotoxicity against african green monkey Vero cells after 48 hrs by MTT assay, LC50 = 28 μM. | 25078313 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by MTT assay, LC50 = 18 μM. | 25078313 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 6.27 μM. | 25113878 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells after 48 hrs by MTT assay, IC50 = 3.39 μM. | 25113878 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 0.62 μM. | 25131956 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 0.25 μM. | 25131956 | ||
| MIAPaCa2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MIAPaCa2 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 1.1 μM. | 25131956 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by sulforhodamine B assay, IC50 = 13.78 μM. | 25240702 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by sulforhodamine B assay, IC50 = 4.36 μM. | 25240702 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by sulforhodamine B assay, IC50 = 3.67 μM. | 25240702 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50 = 9.3 μM. | 25402632 | ||
| HCT15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT15 cells after 72 hrs by MTT assay, IC50 = 4.6 μM. | 25402632 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 3.4 μM. | 25402632 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HepG2 cells after 72 hrs by MTT assay, IC50 = 3.6 μM. | 25402632 | ||
| U373 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U373 cells after 72 hrs by MTT assay, IC50 = 8.5 μM. | 25402632 | ||
| ZR-7530 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human ZR-7530 cells after 72 hrs by MTT assay, IC50 = 2.5 μM. | 25402632 | ||
| SKMES1 | Cytotoxicity assay | Cytotoxicity against human SKMES1 cells, IC50 = 1.83 μM. | 25440884 | |||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 4.71 μM. | 25453799 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 23.9 μM. | 25453799 | ||
| ME180 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human ME180 cells after 72 hrs by MTT assay, IC50 = 8.9 μM. | 25453799 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay, IC50 = 14.4 μM. | 25453799 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 10.12 μM. | 25462233 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 5.48 μM. | 25462233 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by Alamar Blue assay, IC50 = 2.87 μM. | 25466187 | ||
| L6 | Cytotoxicity assay | 71.5 hrs | Cytotoxicity against rat L6 cells after 71.5 hrs by Presto Blue assay, IC50 = 18.99 μM. | 25466187 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days by CCK-8 assay, IC50 = 0.5 μM. | 25481396 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days by CCK-8 assay, IC50 = 1.1 μM. | 25481396 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells after 2 days by CCK-8 assay, IC50 = 1.26 μM. | 25481396 | ||
| MCF7 | Cytotoxicity assay | 2 days | Cytotoxicity against human MCF7 cells after 2 days by CCK-8 assay, IC50 = 0.73 μM. | 25481396 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.8 μM. | 25495422 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells after 72 hrs by sulforhodamine B assay, IC50 = 1.9 μM. | 25495422 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by sulforhodamine B assay, IC50 = 3.1 μM. | 25495422 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by sulforhodamine B assay, IC50 = 10.6 μM. | 25495422 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells after 72 hrs by sulforhodamine B assay, IC50 = 1.1 μM. | 25495422 | ||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.5 μM. | 25495422 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 8.91 μM. | 25599951 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 0.54 μM. | 25599951 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.06 μM. | 25599951 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells after 48 hrs by MTT assay, IC50 = 1.64 μM. | 25599951 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells after 48 hrs by MTT assay, IC50 = 3.01 μM. | 25615796 | ||
| DU145 | Anticancer assay | 48 hrs | Anticancer activity against human DU145 cells after 48 hrs by MTT assay, IC50 = 1.98 μM. | 25615796 | ||
| HeLa | Anticancer assay | 48 hrs | Anticancer activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 3.23 μM. | 25615796 | ||
| HepG2 | Anticancer assay | 48 hrs | Anticancer activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 2.45 μM. | 25615796 | ||
| MCF7 | Anticancer assay | 48 hrs | Anticancer activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 2.69 μM. | 25615796 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HepG2 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 1.4 μM. | 25644672 | ||
| MDA-MB-468 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-468 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 6.69 μM. | 25644672 | ||
| THP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human THP1 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 1.82 μM. | 25644672 | ||
| U937 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U937 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 1.1 μM. | 25644672 | ||
| EL4 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse EL4 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 4.9 μM. | 25644672 | ||
| N9 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse N9 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 1.95 μM. | 25644672 | ||
| RAW264.7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse RAW264.7 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 0.82 μM. | 25644672 | ||
| S17 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse S17 cells assessed as reduction in cell viability after 72 hrs by MTT colorimetric assay, IC50 = 10.4 μM. | 25644672 | ||
| A549 | Antiproliferative assay | 3 days | Antiproliferative activity against human A549 cells after 3 days by sulforhodamine B assay, IC50 = 0.8 μM. | 25700232 | ||
| HCT116 | Antiproliferative assay | 3 days | Antiproliferative activity against human HCT116 cells after 3 days by sulforhodamine B assay, IC50 = 1.9 μM. | 25700232 | ||
| K562 | Antiproliferative assay | 3 days | Antiproliferative activity against human K562 cells after 3 days by sulforhodamine B assay, IC50 = 3.1 μM. | 25700232 | ||
| MDA-MB-231 | Antiproliferative assay | 3 days | Antiproliferative activity against human MDA-MB-231 cells after 3 days by sulforhodamine B assay, IC50 = 11 μM. | 25700232 | ||
| SKHEP1 | Antiproliferative assay | 3 days | Antiproliferative activity against human SKHEP1 cells after 3 days by sulforhodamine B assay, IC50 = 1.1 μM. | 25700232 | ||
| SNU638 | Antiproliferative assay | 3 days | Antiproliferative activity against human SNU638 cells after 3 days by sulforhodamine B assay, IC50 = 0.5 μM. | 25700232 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells after 48 hrs by MTT assay, IC50 = 11.92 μM. | 25744190 | ||
| HL60 | Anticancer assay | 48 hrs | Anticancer activity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.31 μM. | 25744190 | ||
| MCF7 | Anticancer assay | 48 hrs | Anticancer activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 32.82 μM. | 25744190 | ||
| SMMC7721 | Anticancer assay | 48 hrs | Anticancer activity against human SMMC7721 cells after 48 hrs by MTT assay, IC50 = 8.12 μM. | 25744190 | ||
| SW480 | Anticancer assay | 48 hrs | Anticancer activity against human SW480 cells after 48 hrs by MTT assay, IC50 = 17.11 μM. | 25744190 | ||
| HepG2 | Cytotoxicity assay | Cytotoxicity against human HepG2 cells by MTT assay, IC50 = 40.47 μM. | 25747499 | |||
| MOLT3 | Cytotoxicity assay | Cytotoxicity against human MOLT3 cells by XTT assay, IC50 = 0.11 μM. | 25747499 | |||
| CCRF-CEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human CCRF-CEM cells after 72 hrs by MTT assay, GI50 = 1.6 μM. | 25792142 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by MTT assay, GI50 = 31.6 μM. | 25792142 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay, GI50 = 19.9 μM. | 25792142 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay, GI50 = 0.6 μM. | 25792142 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells after 72 hrs by MTT assay, TGI = 4 μM. | 25792142 | ||
| DU145 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human DU145 cells assessed as cell viability incubated for 72 hrs by MTT assay, IC50 = 0.4 μM. | 25799376 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells assessed as cell viability incubated for 72 hrs by MTT assay, IC50 = 4.33 μM. | 25799376 | ||
| U937 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human U937 cells after 24 hrs by MTT assay, IC50 = 1.5 μM. | 25800433 | ||
| MDA-MB-435 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-435 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 2.81 μM. | 25800514 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by MTT assay, IC50 = 7.6 μM. | 25847767 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells by MTT assay, IC50 = 10.3 μM. | 25847767 | |||
| T47D | Cytotoxicity assay | Cytotoxicity against human T47D cells by MTT assay, IC50 = 8.9 μM. | 25847767 | |||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as growth inhibition after 48 hrs by trypan blue staining-based hemocytometric analysis, GI50 = 0.4 μM. | 25874335 | ||
| HBL100 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HBL100 cells after 48 hrs incubation by SRB assay, GI50 = 1.9 μM. | 25899335 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs incubation by SRB assay, GI50 = 2 μM. | 25899335 | ||
| SW1573 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human SW1573 cells after 48 hrs incubation by SRB assay, GI50 = 3 μM. | 25899335 | ||
| T47D | Antiproliferative assay | 48 hrs | Antiproliferative activity against human T47D cells after 48 hrs incubation by SRB assay, GI50 = 15 μM. | 25899335 | ||
| WiDr | Antiproliferative assay | 48 hrs | Antiproliferative activity against human WiDr cells after 48 hrs incubation by SRB assay, GI50 = 26 μM. | 25899335 | ||
| K/VP.5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against etoposide-resistant human K/VP.5 cells assessed as cell growth inhibition after 72 hrs by MTS assay, IC50 = 2.5 μM. | 25922181 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells assessed as cell growth inhibition after 72 hrs by MTS assay, IC50 = 0.163 μM. | 25922181 | ||
| DU145 | Cytotoxicity assay | 4 days | Cytotoxicity against human DU145 cells measured on day 4 by CCK8 assay, IC50 = 2.09 μM. | 25936262 | ||
| HCT15 | Cytotoxicity assay | 4 days | Cytotoxicity against human HCT15 cells measured on day 4 by CCK8 assay, IC50 = 18.87 μM. | 25936262 | ||
| HeLa | Cytotoxicity assay | 4 days | Cytotoxicity against human HeLa cells measured on day 4 by CCK8 assay, IC50 = 7.32 μM. | 25936262 | ||
| T47D | Cytotoxicity assay | 4 days | Cytotoxicity against human T47D cells measured on day 4 by CCK8 assay, IC50 = 13.7 μM. | 25936262 | ||
| NALM6 | Cytotoxicity assay | 5 days | Cytotoxicity against human NALM6 cells expressing 50% TOP2A assessed as growth inhibition after 5 days by XTT assay, IC50 = 0.234 μM. | 25945730 | ||
| NALM6 | Cytotoxicity assay | 5 days | Cytotoxicity against TOP2B double knockout human NALM6 cells assessed as growth inhibition after 5 days by XTT assay, IC50 = 0.182 μM. | 25945730 | ||
| NALM6 | Cytotoxicity assay | 5 days | Cytotoxicity against wild type human NALM6 cells assessed as growth inhibition after 5 days by XTT assay, IC50 = 0.134 μM. | 25945730 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 0.4 μM. | 26010585 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 1.9 μM. | 26010585 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 7.7 μM. | 26010585 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 2.8 μM. | 26010585 | ||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 0.3 μM. | 26010585 | ||
| DU145 | Cytotoxicity assay | 2 to 4 days | Cytotoxicity against human DU145 cells incubated from day 2 to day 4 by CCK8 assay, IC50 = 0.5 μM. | 26022080 | ||
| HCT15 | Cytotoxicity assay | 2 to 4 days | Cytotoxicity against human HCT15 cells incubated from day 2 to day 4 by CCK8 assay, IC50 = 1.1 μM. | 26022080 | ||
| HeLa | Cytotoxicity assay | 2 to 4 days | Cytotoxicity against human HeLa cells incubated from day 2 to day 4 by CCK8 assay, IC50 = 1.41 μM. | 26022080 | ||
| T47D | Cytotoxicity assay | 2 to 4 days | Cytotoxicity against human T47D cells incubated from day 2 to day 4 by CCK8 assay, IC50 = 0.7 μM. | 26022080 | ||
| KB | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KB cells after 72 hrs by methylene blue assay, IC50 = 1.5 μM. | 26160020 | ||
| KB-7d | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KB-7d cells expressing MRP after 72 hrs by methylene blue assay, IC50 = 46.1 μM. | 26160020 | ||
| KB-S15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KB-S15 cells expressing P-gp170/MDR after 72 hrs by methylene blue assay, IC50 = 3.5 μM. | 26160020 | ||
| KB-VIN10 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human KB-VIN10 cells expressing P-gp170/MDR after 72 hrs by methylene blue assay, IC50 = 26.4 μM. | 26160020 | ||
| A549 | Antiproliferative assay | Antiproliferative activity against human A549 cells by MTT assay, IC50 = 18.2 μM. | 26216018 | |||
| HepG2 | Antiproliferative assay | Antiproliferative activity against human HepG2 cells by MTT assay, IC50 = 20.9 μM. | 26216018 | |||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as inhibition of cell growth incubated for 48 hrs by Hoechst assay, IC50 = 0.48 μM. | 26225905 | ||
| DLD1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DLD1 cells assessed as inhibition of cell growth incubated for 48 hrs by Hoechst assay, IC50 = 3.3 μM. | 26225905 | ||
| HepG2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HepG2 cells after 72 hrs by SRB assay, IC50 = 30 μM. | 26291037 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by SRB assay, IC50 = 3.6 μM. | 26291037 | ||
| MDA-MB-435 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-435 cells after 72 hrs by SRB assay, IC50 = 1.7 μM. | 26291037 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells after 72 hrs by SRB assay, IC50 = 1 μM. | 26291037 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 38.5 μM. | 26292628 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by MTT assay, IC50 = 5.21 μM. | 26292628 | ||
| HGC | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HGC cells after 48 hrs by MTT assay, IC50 = 10.9 μM. | 26292628 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 49.35 μM. | 26292628 | ||
| RWPE1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human RWPE1 cells after 48 hrs by MTT assay, IC50 = 27.64 μM. | 26292628 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells measured after 2 days by MTT assay, IC50 = 5.54 μM. | 26334499 | ||
| HeLa | Cytotoxicity assay | 2 days | Cytotoxicity against human HeLa cells measured after 2 days by MTT assay, IC50 = 7.6 μM. | 26334499 | ||
| T47D | Cytotoxicity assay | 2 days | Cytotoxicity against human T47D cells measured after 2 days by MTT assay, IC50 = 9.28 μM. | 26334499 | ||
| GBM2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GBM2 cells assessed as reduction in cell viability incubated for 72 hrs by WST-1 method, IC50 = 1.1 μM. | 26355532 | ||
| DU145 | Cytotoxicity assay | 2 days | Cytotoxicity against human DU145 cells after 2 days by CCK8 assay, IC50 = 13.1 μM. | 26361737 | ||
| HCT15 | Cytotoxicity assay | 2 days | Cytotoxicity against human HCT15 cells after 2 days by CCK8 assay, IC50 = 2.6 μM. | 26361737 | ||
| MCF10A | Cytotoxicity assay | 2 days | Cytotoxicity against human MCF10A cells after 2 days by CCK8 assay, IC50 = 10.14 μM. | 26361737 | ||
| T47D | Cytotoxicity assay | 2 days | Cytotoxicity against human T47D cells after 2 days by CCK8 assay, IC50 = 0.94 μM. | 26361737 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 33.98 μM. | 26397393 | ||
| MOLT3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MOLT3 cells after 48 hrs by XTT assay, IC50 = 0.041 μM. | 26397393 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 48 hrs by resazurin assay, IC50 = 1.7 μM. | 26592897 | ||
| DLD1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DLD1 cells assessed as growth inhibition after 48 hrs by resazurin assay, IC50 = 2 μM. | 26592897 | ||
| A549 | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human A549 cells after 24 to 72 hrs by SRB assay, IC50 = 0.8 μM. | 26595875 | ||
| CAKI-1 | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human CAKI-1 cells after 24 to 72 hrs by SRB assay, IC50 = 1.1 μM. | 26595875 | ||
| HCT116 | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human HCT116 cells after 24 to 72 hrs by SRB assay, IC50 = 3.3 μM. | 26595875 | ||
| HCT116 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT116 cells assessed as cell viability after 48 hrs by SRB assay, IC50 = 2.3 μM. | 26595875 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 0.3 μM. | 26595875 | ||
| K562 | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human K562 cells after 24 to 72 hrs by SRB assay, IC50 = 6.7 μM. | 26595875 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against p53-expressing human HCT116 cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 1.1 μM. | 26595875 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against p53-null human HCT116 cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 3.1 μM. | 26595875 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells assessed as growth inhibition after 72 hrs by CCK-8 assay, IC50 = 2.675 μM. | 26615886 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells assessed as growth inhibition after 72 hrs by CCK-8 assay, IC50 = 1.605 μM. | 26615886 | ||
| HepG2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HepG2 cells assessed as growth inhibition after 72 hrs by CCK-8 assay, IC50 = 1.748 μM. | 26615886 | ||
| K562 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562 cells assessed as growth inhibition after 72 hrs by CCK-8 assay, IC50 = 0.413 μM. | 26615886 | ||
| K562/ADR | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562/ADR cells assessed as growth inhibition after 72 hrs by CCK-8 assay, IC50 = 2.025 μM. | 26615886 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 26.1 μM. | 26618211 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 3.2 μM. | 26618211 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells assessed as cell viability after 72 hrs by SRB assay, IC50 = 4.2 μM. | 26618211 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by MTT assay, IC50 = 6.74 μM. | 26649766 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 32.45 μM. | 26649766 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.16 μM. | 26649766 | ||
| HL60/MX2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60/MX2 cells after 48 hrs by MTT assay, IC50 = 9.02 μM. | 26649766 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by MTT assay, IC50 = 0.51 μM. | 26649766 | ||
| MDA-MB-201 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-201 cells after 48 hrs by MTT assay, IC50 = 31.33 μM. | 26649766 | ||
| Raji | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Raji cells after 48 hrs by MTT assay, IC50 = 1.68 μM. | 26649766 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by MTT assay, IC50 = 0.69 μM. | 26649766 | ||
| GM07492A | Cytotoxicity assay | 24 hrs | Cytotoxicity against human GM07492A cells assessed as cell growth inhibition after 24 hrs by XTT assay, IC50 = 1.2 μM. | 26649907 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by SRB assay, IC50 = 1.74 μM. | 26706168 | ||
| HCT15 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT15 cells after 48 hrs by SRB assay, IC50 = 2.95 μM. | 26706168 | ||
| SK-MEL-2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SK-MEL-2 cells after 48 hrs by SRB assay, IC50 = 1.33 μM. | 26706168 | ||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells after 48 hrs by SRB assay, IC50 = 1.96 μM. | 26706168 | ||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells assessed as cellular DNA content after 72 hrs by CyQUANT NF fluorescence assay, IC50 = 2.2 μM. | 26731300 | ||
| MCF7 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MCF7 cells assessed as cellular DNA content after 96 hrs by CyQUANT NF fluorescence assay, IC50 = 0.8 μM. | 26731300 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells assessed as cellular DNA content after 72 hrs by CyQUANT NF fluorescence assay, IC50 = 1.1 μM. | 26731300 | ||
| T84 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human T84 cells assessed as cellular DNA content after 96 hrs by CyQUANT NF fluorescence assay, IC50 = 3 μM. | 26731300 | ||
| A375 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A375 cells assessed as growth inhibition after 72 hrs by ATPlite assay, IC50 = 0.24 μM. | 26793989 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 72 hrs by ATPlite assay, IC50 = 0.59 μM. | 26793989 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as growth inhibition after 72 hrs by ATPlite assay, IC50 = 0.86 μM. | 26793989 | ||
| HCT116 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT116 cells after 48 hrs by MTT assay, IC50 = 1.7 μM. | 26804229 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 5.62 μM. | 26804229 | ||
| A549 | Cytotoxicity assay | Cytotoxicity in human A549 cells by SRB assay, IC50 = 1.4 μM. | 26812172 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity in human HCT116 cells by SRB assay, IC50 = 1 μM. | 26812172 | |||
| SK-MEL-2 | Cytotoxicity assay | Cytotoxicity in human SK-MEL-2 cells by SRB assay, IC50 = 1.5 μM. | 26812172 | |||
| SKOV3 | Cytotoxicity assay | Cytotoxicity in human SKOV3 cells by SRB assay, IC50 = 2.6 μM. | 26812172 | |||
| 22Rv1 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human 22Rv1 cells after 96 hrs by propidium iodide-based monolayer assay, IC50 = 0.364 μM. | 26854430 | ||
| MCF7 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MCF7 cells after 96 hrs by propidium iodide-based monolayer assay, IC50 = 1.264 μM. | 26854430 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 23.94 μM. | 26873416 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 10.89 μM. | 26873416 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 12.12 μM. | 26873416 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 27.07 μM. | 26873416 | ||
| WI38 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WI38 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 35.8 μM. | 26873416 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 12.4 μM. | 26890115 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 15.7 μM. | 26890115 | ||
| T47D | Cytotoxicity assay | 48 hrs | Cytotoxicity against human T47D cells after 48 hrs by MTT assay, IC50 = 11.8 μM. | 26890115 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells assessed as reduction in viable cells after 48 hrs by MTT assay, IC50 = 4.39 μM. | 26916436 | ||
| HONE1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HONE1 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 1.2 μM. | 26920801 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 2 μM. | 26920801 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 2.2 μM. | 26920801 | ||
| P388 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse P388 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 1.3 μM. | 26920801 | ||
| HCT15 | Cytotoxicity assay | 3 days | Cytotoxicity against human HCT15 cells after 3 days by CCK8 assay, IC50 = 2.82 μM. | 26927425 | ||
| HeLa | Cytotoxicity assay | 3 days | Cytotoxicity against human HeLa cells after 3 days by CCK8 assay, IC50 = 0.18 μM. | 26927425 | ||
| T47D | Cytotoxicity assay | 3 days | Cytotoxicity against human T47D cells after 3 days by CCK8 assay, IC50 = 1.84 μM. | 26927425 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by CCK8 assay, IC50 = 2.09 μM. | 26945111 | ||
| HCT15 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT15 cells after 48 hrs by CCK8 assay, IC50 = 18.87 μM. | 26945111 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by CCK8 assay, IC50 = 7.32 μM. | 26945111 | ||
| T47D | Cytotoxicity assay | 48 hrs | Cytotoxicity against human T47D cells after 48 hrs by CCK8 assay, IC50 = 13.7 μM. | 26945111 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB assay, IC50 = 1.85 μM. | 26988298 | |||
| SK-MEL-2 | Cytotoxicity assay | Cytotoxicity against human SK-MEL-2 cells by SRB assay, IC50 = 1.17 μM. | 26988298 | |||
| SKOV3 | Cytotoxicity assay | Cytotoxicity against human SKOV3 cells by SRB assay, IC50 = 0.84 μM. | 26988298 | |||
| XF498 | Cytotoxicity assay | Cytotoxicity against human XF498 cells by SRB assay, IC50 = 1.72 μM. | 26988298 | |||
| BT474 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human BT474 cells after 72 hrs by CCK8 assay, IC50 = 6.6 μM. | 26988802 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK8 assay, IC50 = 6.9 μM. | 26988802 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK8 assay, IC50 = 6.4 μM. | 26988802 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by MTT assay, IC50 = 3.82 μM. | 27060757 | ||
| CCRF-CEM | Antiproliferative assay | 48 hrs | Antiproliferative activity against human CCRF-CEM cells after 48 hrs by MTT assay, IC50 = 0.12 μM. | 27060757 | ||
| HCT116 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT116 cells after 48 hrs by MTT assay, IC50 = 6.03 μM. | 27060757 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 41.21 μM. | 27060757 | ||
| A375 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A375 cells assessed as reduction in cell viability after 72 hrs by ATPlite assay, IC50 = 0.24 μM. | 27096049 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells assessed as reduction in cell viability after 72 hrs by ATPlite assay, IC50 = 0.59 μM. | 27096049 | ||
| HL60 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HL60 cells assessed as reduction in cell viability after 72 hrs by ATPlite assay, IC50 = 0.86 μM. | 27096049 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 0.5 μM. | 27145162 | ||
| MOLT3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MOLT3 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 0.7 μM. | 27145162 | ||
| HL60 | Cytotoxicity assay | Cytotoxicity against human HL60 cells assessed as reduction in cell survival, IC50 = 0.025 μM. | 27234147 | |||
| HEK293T | Cytotoxicity assay | 36 hrs | Cytotoxicity against HEK293T cells after 36 hrs by MTT assay, IC50 = 11.47 μM. | 27262599 | ||
| A549 | Cytotoxicity assay | 36 hrs | Cytotoxicity against human A549 cells after 36 hrs by MTT assay, IC50 = 8.01 μM. | 27262599 | ||
| HeLa | Cytotoxicity assay | 36 hrs | Cytotoxicity against human HeLa cells after 36 hrs by MTT assay, IC50 = 4.39 μM. | 27262599 | ||
| MCF7 | Cytotoxicity assay | 36 hrs | Cytotoxicity against human MCF7 cells after 36 hrs by MTT assay, IC50 = 1.74 μM. | 27262599 | ||
| HEK293 | Anticlonogenic assay | 48 hrs | Anticlonogenic activity against HEK293 cells assessed as inhibition of colony formation incubated for 48 hrs measured after 6 days by crystal violet staining based assay, LC50 = 20 μM. | 27343852 | ||
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells assessed as reduction in cell viability after 48 hrs by MTT assay, LC50 = 22 μM. | 27343852 | ||
| H460 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human H460 cells after 72 hrs by MTT assay, IC50 = 0.1 μM. | 27448912 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by MTT assay, IC50 = 5 μM. | 27448912 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 27448912 | ||
| shGFP-fused HMLE | Antiproliferative assay | 72 hrs | Antiproliferative activity against human shGFP-fused HMLE cells after 72 hrs by MTT assay, IC50 = 0.4 μM. | 27448912 | ||
| HCT15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT15 cells assessed as reduction in cell growth after 72 hrs by CCK8 assay, IC50 = 1.06 μM. | 27484510 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells assessed as reduction in cell growth after 72 hrs by CCK8 assay, IC50 = 10.03 μM. | 27484510 | ||
| NCI-N87 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NCI-N87 cells assessed as reduction in cell growth after 72 hrs by CCK8 assay, IC50 = 1.29 μM. | 27484510 | ||
| T47D | Cytotoxicity assay | 72 hrs | Cytotoxicity against human T47D cells assessed as reduction in cell growth after 72 hrs by CCK8 assay, IC50 = 2.8 μM. | 27484510 | ||
| K562 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562 cells measured after 72 hrs by CCK8 assay, IC50 = 0.345 μM. | 27484511 | ||
| K562/ADR | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562/ADR cells measured after 72 hrs by CCK8 assay, IC50 = 2.211 μM. | 27484511 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells measured after 48 hrs by MTT assay, IC50 = 29.7 μM. | 27517805 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells measured after 48 hrs by MTT assay, IC50 = 10 μM. | 27517805 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells measured after 48 hrs by MTT assay, IC50 = 13.5 μM. | 27517805 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells measured after 48 hrs by MTT assay, IC50 = 5.21 μM. | 27517805 | ||
| WI38 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WI38 cells measured after 48 hrs by MTT assay, IC50 = 17.6 μM. | 27517805 | ||
| HEK293 | Cytotoxicity assay | 24 hrs | Cytotoxicity against HEK293 cells assessed as reduction in cell viability after 24 hrs by MTT assay, IC50 = 0.6 μM. | 27533261 | ||
| H1975-1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human H1975-1 cells assessed as reduction in cell viability measured after 72 hrs by MTT assay, IC50 = 9.5 μM. | 27560280 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells assessed as reduction in cell viability measured after 72 hrs by MTT assay, IC50 = 8.2 μM. | 27560280 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as reduction in cell viability measured after 72 hrs by MTT assay, IC50 = 2.1 μM. | 27560280 | ||
| Vero | Cytotoxicity assay | 72 hrs | Cytotoxicity against human Vero cells assessed as reduction in cell viability measured after 72 hrs by MTT assay, IC50 = 1.5 μM. | 27560280 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against resistant human HCT116 cells assessed as reduction in cell viability measured after 72 hrs by MTT assay, IC50 = 2.8 μM. | 27560280 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 0.3 μM. | 27598828 | ||
| HEK293 | Antiproliferative assay | 72 hrs | Antiproliferative activity against HEK293 cells after 72 hrs by CCK8 assay, IC50 = 1.74 μM. | 27643560 | ||
| DU145 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human DU145 cells after 72 hrs by CCK8 assay, IC50 = 0.02 μM. | 27643560 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK8 assay, IC50 = 1.1 μM. | 27643560 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by CCK8 assay, IC50 = 0.18 μM. | 27643560 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK8 assay, IC50 = 1.84 μM. | 27643560 | ||
| DU145 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human DU145 cells measured after 72 hrs by CCK8 assay, IC50 = 2.19 μM. | 27654394 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells measured after 72 hrs by CCK8 assay, IC50 = 1.27 μM. | 27654394 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells measured after 72 hrs by CCK8 assay, IC50 = 35.55 μM. | 27654394 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells measured after 72 hrs by CCK8 assay, IC50 = 4.85 μM. | 27654394 | ||
| HepG2 | Cytotoxicity assay | Cytotoxicity against human HepG2 cells by MTT assay, IC50 = 15.8 μM. | 27759375 | |||
| HBL100 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HBL100 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 1.4 μM. | 27765506 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 3.3 μM. | 27765506 | ||
| SW1573 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SW1573 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 15 μM. | 27765506 | ||
| WiDr | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WiDr cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 23 μM. | 27765506 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 27.41 μM. | 27863370 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 13.35 μM. | 27863370 | ||
| T47D | Cytotoxicity assay | 72 hrs | Cytotoxicity against human T47D cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 9.8 μM. | 27863370 | ||
| HEK293T | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293T cells assessed as cells growth inhibition after 48 hrs by MTT assay, IC50 = 22 μM. | 27994737 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as cell growth inhibition after 48 hrs by SRB assay, GI50 = 0.56 μM. | 28041801 | ||
| TK10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human TK10 cells assessed as cell growth inhibition after 48 hrs by SRB assay, GI50 = 7.19 μM. | 28041801 | ||
| TK10 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human TK10 cells assessed as cell growth inhibition after 48 hrs by SRB assay, TGI = 49.74 μM. | 28041801 | ||
| UACC62 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human UACC62 cells assessed as cell growth inhibition after 48 hrs by SRB assay, GI50 = 0.89 μM. | 28041801 | ||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells assessed as growth inhibition by MTS assay, IC50 = 0.49 μM. | 28055208 | |||
| THP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human THP1 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 1.82 μM. | 28062194 | ||
| U937 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U937 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 1.1 μM. | 28062194 | ||
| EL4 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse EL4 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 4.9 μM. | 28062194 | ||
| S17 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse S17 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 10.4 μM. | 28062194 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 5.87 μM. | 28068602 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 3.1 μM. | 28068602 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 20.3 μM. | 28068602 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 13.9 μM. | 28068602 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK8 assay, IC50 = 5.4 μM. | 28068603 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by CCK8 assay, IC50 = 9.1 μM. | 28068603 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK8 assay, IC50 = 7 μM. | 28068603 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells assessed as growth inhibition after 48 hrs by Cell Titer-Glo assay, EC50 = 15.9 μM. | 28080063 | ||
| HEK293 | Antiproliferative assay | 48 hrs | Antiproliferative activity against HEK293 cells after 48 hrs by SRB assay, IC50 = 7.51 μM. | 28094224 | ||
| DLD1 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human DLD1 cells after 48 hrs by SRB assay, IC50 = 8.11 μM. | 28094224 | ||
| DU145 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human DU145 cells after 48 hrs by SRB assay, IC50 = 10.64 μM. | 28094224 | ||
| FADU | Antiproliferative assay | 48 hrs | Antiproliferative activity against human FADU cells after 48 hrs by SRB assay, IC50 = 16.01 μM. | 28094224 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by SRB assay, IC50 = 0.92 μM. | 28165740 | ||
| BT549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BT549 cells after 48 hrs by SRB assay, IC50 = 1.24 μM. | 28165740 | ||
| SK-MEL-2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SK-MEL-2 cells after 48 hrs by SRB assay, IC50 = 1.1 μM. | 28165740 | ||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells after 48 hrs by SRB assay, IC50 = 1.95 μM. | 28165740 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 0.83 μM. | 28169166 | ||
| LO2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human LO2 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 3.78 μM. | 28169166 | ||
| SMMC7721 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SMMC7721 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 0.15 μM. | 28169166 | ||
| HCT15 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human HCT15 cells after 24 hrs by WST-1 assay, IC50 = 19.7 μM. | 28181805 | ||
| HeLaS3 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human HeLaS3 cells after 24 hrs by WST-1 assay, IC50 = 43.1 μM. | 28181805 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK8 assay, IC50 = 1.33 μM. | 28384547 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by CCK8 assay, IC50 = 3.2 μM. | 28384547 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK8 assay, IC50 = 3.25 μM. | 28384547 | ||
| Vero | Cytotoxicity assay | 72 hrs | Cytotoxicity against African green monkey Vero cells assessed as decrease in cell viability after 72 hrs by MTT assay, IC50 = 16 μM. | 28385594 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as decrease in cell viability after 72 hrs by MTT assay, IC50 = 3 μM. | 28385594 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells assessed as decrease in cell viability after 72 hrs by MTT assay, IC50 = 8 μM. | 28385594 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells assessed as decrease in cell viability after 72 hrs by MTT assay, IC50 = 20 μM. | 28385594 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells assessed as decrease in cell viability after 72 hrs by MTT assay, IC50 = 12 μM. | 28385594 | ||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells after 72 hrs by MTT assay, GI50 = 1.7 μM. | 28406636 | ||
| Hs683 | Growth inhibition assay | 72 hrs | Growth inhibition of human Hs683 cells after 72 hrs by MTT assay, GI50 = 1.5 μM. | 28406636 | ||
| MCF7 | Growth inhibition assay | 72 hrs | Growth inhibition of human MCF7 cells after 72 hrs by MTT assay, GI50 = 4 μM. | 28406636 | ||
| SK-MEL-28 | Growth inhibition assay | 72 hrs | Growth inhibition of human SK-MEL-28 cells after 72 hrs by MTT assay, GI50 = 3.2 μM. | 28406636 | ||
| U373 | Growth inhibition assay | 72 hrs | Growth inhibition of human U373 cells after 72 hrs by MTT assay, GI50 = 24.9 μM. | 28406636 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, IC50 = 7.89 μM. | 28431878 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 29.27 μM. | 28431878 | ||
| SMMC7721 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SMMC7721 cells after 72 hrs by MTT assay, IC50 = 40.44 μM. | 28431878 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by MTT assay, IC50 = 19.83 μM. | 28462840 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 15.93 μM. | 28462840 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 24.01 μM. | 28462840 | ||
| MGC803 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MGC803 cells after 48 hrs by MTT assay, IC50 = 20.91 μM. | 28462840 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 14.7 μM. | 28512026 | ||
| HCT8 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT8 cells after 48 hrs by MTT assay, IC50 = 26.3 μM. | 28512026 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 24.9 μM. | 28512026 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 13.4 μM. | 28512026 | ||
| WI38 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WI38 cells after 48 hrs by MTT assay, IC50 = 46.9 μM. | 28512026 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells assessed as reduction in cell viability by MTT assay, IC50 = 16.6 μM. | 28571311 | |||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells assessed as reduction in cell viability by MTT assay, IC50 = 15 μM. | 28571311 | |||
| HepG2 | Cytotoxicity assay | Cytotoxicity against human HepG2 cells assessed as reduction in cell viability by MTT assay, IC50 = 16.1 μM. | 28571311 | |||
| A549 | Growth inhibition assay | 72 hrs | Growth inhibition of human A549 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 0.3 μM. | 28625715 | ||
| HCT116 | Growth inhibition assay | 72 hrs | Growth inhibition of human HCT116 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 1.06 μM. | 28625715 | ||
| K562 | Growth inhibition assay | 72 hrs | Growth inhibition of human K562 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 0.76 μM. | 28625715 | ||
| MDA-MB-231 | Growth inhibition assay | 72 hrs | Growth inhibition of human MDA-MB-231 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 1.53 μM. | 28625715 | ||
| MRC5 | Growth inhibition assay | 72 hrs | Growth inhibition of human MRC5 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 11.73 μM. | 28625715 | ||
| SKHEP1 | Growth inhibition assay | 72 hrs | Growth inhibition of human SKHEP1 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 0.63 μM. | 28625715 | ||
| SNU638 | Growth inhibition assay | 72 hrs | Growth inhibition of human SNU638 cells incubated for 72 hrs by sulforhodamine B assay, IC50 = 1.05 μM. | 28625715 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK8 assay, IC50 = 9.27 μM. | 28633898 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by CCK8 assay, IC50 = 4.12 μM. | 28633898 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK8 assay, IC50 = 8.81 μM. | 28633898 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxic activity against human HeLa cells after 72 hrs by MTT assay, IC50 = 7.21 μM. | 28733083 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxic activity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 16.27 μM. | 28733083 | ||
| SMMC7721 | Cytotoxicity assay | 72 hrs | Cytotoxic activity against human SMMC7721 cells after 72 hrs by MTT assay, IC50 = 21.03 μM. | 28733083 | ||
| A549 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human A549 cells after 24 hrs by MTT assay, IC50 = 6.21 μM. | 28757065 | ||
| HeLa | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HeLa cells after 24 hrs by MTT assay, IC50 = 1.58 μM. | 28757065 | ||
| HepG2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HepG2 cells after 24 hrs by MTT assay, IC50 = 5.52 μM. | 28757065 | ||
| L02 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human L02 cells after 24 hrs by MTT assay, IC50 = 7.75 μM. | 28757065 | ||
| MCF7 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MCF7 cells after 24 hrs by MTT assay, IC50 = 2.08 μM. | 28757065 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by SRB assay, IC50 = 1.1 μM. | 28841320 | ||
| BT549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BT549 cells after 48 hrs by SRB assay, IC50 = 1 μM. | 28841320 | ||
| SK-MEL-2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SK-MEL-2 cells after 48 hrs by SRB assay, IC50 = 0.2 μM. | 28841320 | ||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells after 48 hrs by SRB assay, IC50 = 2.9 μM. | 28841320 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by MTT assay, IC50 = 12.4 μM. | 28886509 | |||
| SK-MEL-1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SK-MEL-1 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IC50 = 8 μM. | 29032930 | ||
| HeLaS3 | Antiproliferative assay | Antiproliferative activity against human HeLaS3 cells by MTT assay, IC50 = 0.5 μM. | 29072457 | |||
| HT-29 | Antiproliferative assay | Antiproliferative activity against human HT-29 cells by MTT assay, IC50 = 7.6 μM. | 29072457 | |||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by MTT assay, IC50 = 1.3 μM. | 29077404 | ||
| DU145 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human DU145 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 29077404 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by MTT assay, IC50 = 2.4 μM. | 29077404 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 10.5 μM. | 29077404 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by SRB assay, GI50 = 1.5 μM. | 29129514 | ||
| HBL100 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HBL100 cells after 48 hrs by SRB assay, GI50 = 2.3 μM. | 29129514 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by SRB assay, GI50 = 3 μM. | 29129514 | ||
| SW1573 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human SW1573 cells after 48 hrs by SRB assay, GI50 = 15 μM. | 29129514 | ||
| T47D | Antiproliferative assay | 48 hrs | Antiproliferative activity against human T47D cells after 48 hrs by SRB assay, GI50 = 22 μM. | 29129514 | ||
| WiDr | Antiproliferative assay | 48 hrs | Antiproliferative activity against human WiDr cells after 48 hrs by SRB assay, GI50 = 23 μM. | 29129514 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as reduction in inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 9.1 μM. | 29248296 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as reduction in inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 11.2 μM. | 29248296 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells assessed as reduction in inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 9.2 μM. | 29248296 | ||
| LoVo | Cytotoxicity assay | 48 hrs | Cytotoxicity against human LoVo cells assessed as reduction in inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 18.7 μM. | 29248296 | ||
| WI38 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human WI38 cells assessed as reduction in inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 25.7 μM. | 29248296 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 2.03 μM. | 29269253 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by MTT assay, IC50 = 2.03 μM. | 29269253 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 2.03 μM. | 29269253 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 2.03 μM. | 29269253 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 2.27 μM. | 29289881 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by MTT assay, IC50 = 1.97 μM. | 29289881 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 1.93 μM. | 29289881 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 1.64 μM. | 29289881 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 1.91 μM. | 29289881 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by MTT assay, IC50 = 1.97 μM. | 29289884 | ||
| DU145 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human DU145 cells after 48 hrs by MTT assay, IC50 = 2.35 μM. | 29289884 | ||
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 2.71 μM. | 29289884 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 2.84 μM. | 29289884 | ||
| HT-29 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HT-29 cells after 48 hrs by MTT assay, IC50 = 1.87 μM. | 29289884 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 1.62 μM. | 29289884 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 32.61 μM. | 29335211 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 22.35 μM. | 29335211 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 0.19 μM. | 29335211 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells assessed as growth inhibition after 48 hrs by MTT assay, IC50 = 5.73 μM. | 29335211 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells incubated for 72 hrs by CCK8 assay, IC50 = 2.94 μM. | 29402741 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells incubated for 72 hrs by CCK8 assay, IC50 = 9.21 μM. | 29402741 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells incubated for 72 hrs by CCK8 assay, IC50 = 0.4 μM. | 29402741 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by CCK-8 assay, IC50 = 2.45 μM. | 29510948 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by CCK-8 assay, IC50 = 10.03 μM. | 29510948 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by CCK-8 assay, IC50 = 0.32 μM. | 29510948 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 3.25 μM. | 29525336 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 2.13 μM. | 29525336 | ||
| HCT116 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT116 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 2.6 μM. | 29525336 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 1.8 μM. | 29525336 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 2.03 μM. | 29525336 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 5.4 μM. | 29525336 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 13.71 μM. | 29545100 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 32.05 μM. | 29545100 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.33 μM. | 29545100 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells after 48 hrs by MTT assay, IC50 = 8.45 μM. | 29545100 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human etoposide-sensitive K562 cells after 72 hrs by CellTiter96 AQueous one solution cell proliferation assay, IC50 = 1.22 μM. | 29656990 | ||
| HT-29 | Cytotoxicity assay | 24 to 72 hrs | Cytotoxicity against human HT-29 cells measured after 24 to 72 hrs by SRB assay, IC50 = 15 μM. | 29656990 | ||
| K/VP.5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K/VP.5 cells after 72 hrs by CellTiter96 AQueous one solution cell proliferation assay, IC50 = 35.36 μM. | 29656990 | ||
| OVCAR3 | Cytotoxicity assay | 24 to 72 hrs | Cytotoxicity against human OVCAR3 cells measured after 24 to 72 hrs by SRB assay, IC50 = 2.9 μM. | 29656990 | ||
| CCRF-CEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human CCRF-CEM cells assessed as growth inhibition after 72 hrs by MTT assay, GI50 = 1.26 μM. | 29670691 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as growth inhibition after 72 hrs by MTT assay, GI50 = 31.62 μM. | 29670691 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells assessed as growth inhibition after 72 hrs by MTT assay, GI50 = 12.59 μM. | 29670691 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 72 hrs by MTT assay, GI50 = 19.95 μM. | 29670691 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells assessed as growth inhibition after 72 hrs by MTT assay, GI50 = 0.63 μM. | 29670691 | ||
| K562 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562 cells after 72 hrs by CCK8 assay, IC50 = 0.764 μM. | 29678463 | ||
| K562/VCR | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562/VCR cells after 72 hrs by CCK8 assay, IC50 = 11.84 μM. | 29678463 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HepG2 cells assessed as reduction in cell proliferation after 72 hrs by MTS assay, IC50 = 20.1 μM. | 29778894 | ||
| K562 | Antiproliferative assay | Antiproliferative activity against human K562 cells, IC50 = 0.3 μM. | 29871847 | |||
| K/VP.5 | Function assay | Inhibition of human topoisomerase-2 in etoposide-resistant K/VP.5 cells assessed as growth inhibition, IC50 = 5.1 μM. | 29871847 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 9.4695 μM. | 29886322 | ||
| L02 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human L02 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 3.4805 μM. | 29886322 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 4.7165 μM. | 29886322 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.72 μM. | 29893558 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.68 μM. | 29893558 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells after 72 hrs by sulforhodamine B assay, IC50 = 1.3 μM. | 29893558 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells after 72 hrs by sulforhodamine B assay, IC50 = 4.4 μM. | 29893558 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.93 μM. | 29893558 | ||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.43 μM. | 29893558 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.3 μM. | 30041947 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 1.06 μM. | 30041947 | ||
| K562 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human K562 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.76 μM. | 30041947 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 1.53 μM. | 30041947 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 11.73 μM. | 30041947 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.63 μM. | 30041947 | ||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 1.05 μM. | 30041947 | ||
| CCRF-CEM | Antiproliferative assay | 72 hrs | Antiproliferative activity against human CCRF-CEM cells after 72 hrs by MTT assay, GI50 = 1.6 μM. | 30071406 | ||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells after 72 hrs by MTT assay, GI50 = 31.6 μM. | 30071406 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, GI50 = 19.9 μM. | 30071406 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells after 72 hrs by MTT assay, GI50 = 0.6 μM. | 30071406 | ||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells after 72 hrs by MTT assay, TGI = 4 μM. | 30071406 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by EZ-CYTOX reagent based assay, IC50 = 2.94 μM. | 30262132 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by EZ-CYTOX reagent based assay, IC50 = 9.21 μM. | 30262132 | ||
| T47D | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T47D cells after 72 hrs by EZ-CYTOX reagent based assay, IC50 = 0.4 μM. | 30262132 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 22.5 μM. | 30265994 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells after 48 hrs by MTT assay, IC50 = 21.4 μM. | 30265994 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 20 μM. | 30265994 | ||
| HL7702 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL7702 cells after 48 hrs by MTT assay, IC50 = 24.6 μM. | 30265994 | ||
| MRC5 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MRC5 cells after 48 hrs by MTT assay, IC50 = 33.5 μM. | 30265994 | ||
| RH-1 | Growth inhibition assay | IC50 = 0.01778 μM | SANGER | |||
| A253 | Growth inhibition assay | IC50 = 0.01843 μM | SANGER | |||
| HT-144 | Growth inhibition assay | IC50 = 0.02009 μM | SANGER | |||
| EW-16 | Growth inhibition assay | IC50 = 0.02148 μM | SANGER | |||
| NTERA-S-cl-D1 | Growth inhibition assay | IC50 = 0.02502 μM | SANGER | |||
| KE-37 | Growth inhibition assay | IC50 = 0.04072 μM | SANGER | |||
| SK-UT-1 | Growth inhibition assay | IC50 = 0.04237 μM | SANGER | |||
| OCUB-M | Growth inhibition assay | IC50 = 0.04455 μM | SANGER | |||
| 8-MG-BA | Growth inhibition assay | IC50 = 0.04687 μM | SANGER | |||
| TE-9 | Growth inhibition assay | IC50 = 0.05593 μM | SANGER | |||
| CGTH-W-1 | Growth inhibition assay | IC50 = 0.05741 μM | SANGER | |||
| BB30-HNC | Growth inhibition assay | IC50 = 0.06418 μM | SANGER | |||
| ACN | Growth inhibition assay | IC50 = 0.07896 μM | SANGER | |||
| EW-13 | Growth inhibition assay | IC50 = 0.08885 μM | SANGER | |||
| DU-4475 | Growth inhibition assay | IC50 = 0.09615 μM | SANGER | |||
| HGC-27 | Growth inhibition assay | IC50 = 0.10212 μM | SANGER | |||
| A427 | Growth inhibition assay | IC50 = 0.10338 μM | SANGER | |||
| HT-1080 | Growth inhibition assay | IC50 = 0.10853 μM | SANGER | |||
| MC-IXC | Growth inhibition assay | IC50 = 0.11512 μM | SANGER | |||
| MRK-nu-1 | Growth inhibition assay | IC50 = 0.1162 μM | SANGER | |||
| EW-7 | Growth inhibition assay | IC50 = 0.12506 μM | SANGER | |||
| SW982 | Growth inhibition assay | IC50 = 0.13069 μM | SANGER | |||
| SIG-M5 | Growth inhibition assay | IC50 = 0.13082 μM | SANGER | |||
| PA-1 | Growth inhibition assay | IC50 = 0.13219 μM | SANGER | |||
| ES8 | Growth inhibition assay | IC50 = 0.14221 μM | SANGER | |||
| MV-4-11 | Growth inhibition assay | IC50 = 0.15121 μM | SANGER | |||
| QIMR-WIL | Growth inhibition assay | IC50 = 0.15458 μM | SANGER | |||
| COLO-679 | Growth inhibition assay | IC50 = 0.16303 μM | SANGER | |||
| CCRF-CEM | Growth inhibition assay | IC50 = 0.17043 μM | SANGER | |||
| IGROV-1 | Growth inhibition assay | IC50 = 0.17072 μM | SANGER | |||
| LOUCY | Growth inhibition assay | IC50 = 0.1766 μM | SANGER | |||
| ML-2 | Growth inhibition assay | IC50 = 0.17847 μM | SANGER | |||
| RS4-11 | Growth inhibition assay | IC50 = 0.19612 μM | SANGER | |||
| DJM-1 | Growth inhibition assay | IC50 = 0.20294 μM | SANGER | |||
| ATN-1 | Growth inhibition assay | IC50 = 0.20386 μM | SANGER | |||
| NALM-6 | Growth inhibition assay | IC50 = 0.20584 μM | SANGER | |||
| HUTU-80 | Growth inhibition assay | IC50 = 0.21676 μM | SANGER | |||
| REH | Growth inhibition assay | IC50 = 0.22424 μM | SANGER | |||
| NCI-H2030 | Growth inhibition assay | IC50 = 0.22992 μM | SANGER | |||
| A204 | Growth inhibition assay | IC50 = 0.23248 μM | SANGER | |||
| NCI-H460 | Growth inhibition assay | IC50 = 0.23571 μM | SANGER | |||
| 697 | Growth inhibition assay | IC50 = 0.2432 μM | SANGER | |||
| KYSE-270 | Growth inhibition assay | IC50 = 0.25188 μM | SANGER | |||
| ES1 | Growth inhibition assay | IC50 = 0.25434 μM | SANGER | |||
| COLO-205 | Growth inhibition assay | IC50 = 0.26497 μM | SANGER | |||
| SK-HEP-1 | Growth inhibition assay | IC50 = 0.26712 μM | SANGER | |||
| RPMI-8402 | Growth inhibition assay | IC50 = 0.27109 μM | SANGER | |||
| H4 | Growth inhibition assay | IC50 = 0.29654 μM | SANGER | |||
| LU-134-A | Growth inhibition assay | IC50 = 0.29919 μM | SANGER | |||
| EoL-1-cell | Growth inhibition assay | IC50 = 0.30172 μM | SANGER | |||
| SBC-5 | Growth inhibition assay | IC50 = 0.30262 μM | SANGER | |||
| DEL | Growth inhibition assay | IC50 = 0.31149 μM | SANGER | |||
| P12-ICHIKAWA | Growth inhibition assay | IC50 = 0.31679 μM | SANGER | |||
| AGS | Growth inhibition assay | IC50 = 0.31823 μM | SANGER | |||
| KS-1 | Growth inhibition assay | IC50 = 0.33444 μM | SANGER | |||
| SK-PN-DW | Growth inhibition assay | IC50 = 0.34228 μM | SANGER | |||
| DOHH-2 | Growth inhibition assay | IC50 = 0.34733 μM | SANGER | |||
| KGN | Growth inhibition assay | IC50 = 0.35189 μM | SANGER | |||
| A3-KAW | Growth inhibition assay | IC50 = 0.35826 μM | SANGER | |||
| M059J | Growth inhibition assay | IC50 = 0.36401 μM | SANGER | |||
| Daoy | Growth inhibition assay | IC50 = 0.36773 μM | SANGER | |||
| MKN1 | Growth inhibition assay | IC50 = 0.38169 μM | SANGER | |||
| CESS | Growth inhibition assay | IC50 = 0.38386 μM | SANGER | |||
| SW620 | Growth inhibition assay | IC50 = 0.38448 μM | SANGER | |||
| NCI-H1355 | Growth inhibition assay | IC50 = 0.38972 μM | SANGER | |||
| EB2 | Growth inhibition assay | IC50 = 0.39146 μM | SANGER | |||
| ABC-1 | Growth inhibition assay | IC50 = 0.40453 μM | SANGER | |||
| GT3TKB | Growth inhibition assay | IC50 = 0.40644 μM | SANGER | |||
| CTV-1 | Growth inhibition assay | IC50 = 0.40969 μM | SANGER | |||
| VA-ES-BJ | Growth inhibition assay | IC50 = 0.41481 μM | SANGER | |||
| KP-4 | Growth inhibition assay | IC50 = 0.41615 μM | SANGER | |||
| MOLT-4 | Growth inhibition assay | IC50 = 0.41754 μM | SANGER | |||
| NKM-1 | Growth inhibition assay | IC50 = 0.42516 μM | SANGER | |||
| A2058 | Growth inhibition assay | IC50 = 0.42537 μM | SANGER | |||
| BFTC-905 | Growth inhibition assay | IC50 = 0.42621 μM | SANGER | |||
| SR | Growth inhibition assay | IC50 = 0.44265 μM | SANGER | |||
| NCI-H526 | Growth inhibition assay | IC50 = 0.45612 μM | SANGER | |||
| MES-SA | Growth inhibition assay | IC50 = 0.45821 μM | SANGER | |||
| MHH-ES-1 | Growth inhibition assay | IC50 = 0.46271 μM | SANGER | |||
| IMR-5 | Growth inhibition assay | IC50 = 0.46935 μM | SANGER | |||
| RKO | Growth inhibition assay | IC50 = 0.47075 μM | SANGER | |||
| HOS | Growth inhibition assay | IC50 = 0.47376 μM | SANGER | |||
| 647-V | Growth inhibition assay | IC50 = 0.48734 μM | SANGER | |||
| 786-0 | Growth inhibition assay | IC50 = 0.4922 μM | SANGER | |||
| Calu-3 | Growth inhibition assay | IC50 = 0.49556 μM | SANGER | |||
| U251 | Growth inhibition assay | IC50 = 0.50083 μM | SANGER | |||
| 23132-87 | Growth inhibition assay | IC50 = 0.50139 μM | SANGER | |||
| BE-13 | Growth inhibition assay | IC50 = 0.50368 μM | SANGER | |||
| SW1710 | Growth inhibition assay | IC50 = 0.51838 μM | SANGER | |||
| ES4 | Growth inhibition assay | IC50 = 0.52145 μM | SANGER | |||
| HOP-62 | Growth inhibition assay | IC50 = 0.52437 μM | SANGER | |||
| MHH-NB-11 | Growth inhibition assay | IC50 = 0.52443 μM | SANGER | |||
| KYSE-150 | Growth inhibition assay | IC50 = 0.53039 μM | SANGER | |||
| ESS-1 | Growth inhibition assay | IC50 = 0.53078 μM | SANGER | |||
| HLE | Growth inhibition assay | IC50 = 0.53357 μM | SANGER | |||
| NEC8 | Growth inhibition assay | IC50 = 0.53895 μM | SANGER | |||
| HC-1 | Growth inhibition assay | IC50 = 0.54032 μM | SANGER | |||
| BC-1 | Growth inhibition assay | IC50 = 0.54533 μM | SANGER | |||
| SK-LMS-1 | Growth inhibition assay | IC50 = 0.5528 μM | SANGER | |||
| NCI-H2342 | Growth inhibition assay | IC50 = 0.55508 μM | SANGER | |||
| OCI-AML2 | Growth inhibition assay | IC50 = 0.55935 μM | SANGER | |||
| KYSE-510 | Growth inhibition assay | IC50 = 0.56639 μM | SANGER | |||
| HMV-II | Growth inhibition assay | IC50 = 0.56879 μM | SANGER | |||
| RPMI-2650 | Growth inhibition assay | IC50 = 0.57196 μM | SANGER | |||
| DMS-273 | Growth inhibition assay | IC50 = 0.57348 μM | SANGER | |||
| MC-CAR | Growth inhibition assay | IC50 = 0.57962 μM | SANGER | |||
| DU-145 | Growth inhibition assay | IC50 = 0.59125 μM | SANGER | |||
| HCC1599 | Growth inhibition assay | IC50 = 0.61627 μM | SANGER | |||
| RT-112 | Growth inhibition assay | IC50 = 0.62053 μM | SANGER | |||
| CAL-148 | Growth inhibition assay | IC50 = 0.62544 μM | SANGER | |||
| ES3 | Growth inhibition assay | IC50 = 0.62849 μM | SANGER | |||
| A375 | Growth inhibition assay | IC50 = 0.63084 μM | SANGER | |||
| SW872 | Growth inhibition assay | IC50 = 0.6404 μM | SANGER | |||
| LCLC-103H | Growth inhibition assay | IC50 = 0.6511 μM | SANGER | |||
| MEL-JUSO | Growth inhibition assay | IC50 = 0.6571 μM | SANGER | |||
| NH-12 | Growth inhibition assay | IC50 = 0.67198 μM | SANGER | |||
| NCI-H187 | Growth inhibition assay | IC50 = 0.67362 μM | SANGER | |||
| CAL-33 | Growth inhibition assay | IC50 = 0.6858 μM | SANGER | |||
| ALL-PO | Growth inhibition assay | IC50 = 0.68979 μM | SANGER | |||
| 8505C | Growth inhibition assay | IC50 = 0.69275 μM | SANGER | |||
| NCI-H2009 | Growth inhibition assay | IC50 = 0.69655 μM | SANGER | |||
| KURAMOCHI | Growth inhibition assay | IC50 = 0.6986 μM | SANGER | |||
| HSC-2 | Growth inhibition assay | IC50 = 0.70578 μM | SANGER | |||
| SUP-T1 | Growth inhibition assay | IC50 = 0.70586 μM | SANGER | |||
| GI-1 | Growth inhibition assay | IC50 = 0.71317 μM | SANGER | |||
| TUR | Growth inhibition assay | IC50 = 0.72224 μM | SANGER | |||
| PF-382 | Growth inhibition assay | IC50 = 0.72388 μM | SANGER | |||
| MEL-HO | Growth inhibition assay | IC50 = 0.72874 μM | SANGER | |||
| BCPAP | Growth inhibition assay | IC50 = 0.73695 μM | SANGER | |||
| MFH-ino | Growth inhibition assay | IC50 = 0.7513 μM | SANGER | |||
| ONS-76 | Growth inhibition assay | IC50 = 0.75429 μM | SANGER | |||
| MFE-296 | Growth inhibition assay | IC50 = 0.76297 μM | SANGER | |||
| NCI-H2087 | Growth inhibition assay | IC50 = 0.76306 μM | SANGER | |||
| LCLC-97TM1 | Growth inhibition assay | IC50 = 0.76624 μM | SANGER | |||
| ES7 | Growth inhibition assay | IC50 = 0.76713 μM | SANGER | |||
| A431 | Growth inhibition assay | IC50 = 0.79754 μM | SANGER | |||
| HCE-4 | Growth inhibition assay | IC50 = 0.80135 μM | SANGER | |||
| SK-MES-1 | Growth inhibition assay | IC50 = 0.80842 μM | SANGER | |||
| MZ1-PC | Growth inhibition assay | IC50 = 0.8111 μM | SANGER | |||
| ME-180 | Growth inhibition assay | IC50 = 0.8238 μM | SANGER | |||
| MCF7 | Growth inhibition assay | IC50 = 0.8305 μM | SANGER | |||
| G-361 | Growth inhibition assay | IC50 = 0.83387 μM | SANGER | |||
| ACHN | Growth inhibition assay | IC50 = 0.83874 μM | SANGER | |||
| A4-Fuk | Growth inhibition assay | IC50 = 0.84103 μM | SANGER | |||
| SCC-9 | Growth inhibition assay | IC50 = 0.84957 μM | SANGER | |||
| IST-SL2 | Growth inhibition assay | IC50 = 0.85003 μM | SANGER | |||
| CAL-62 | Growth inhibition assay | IC50 = 0.85117 μM | SANGER | |||
| C-33-A | Growth inhibition assay | IC50 = 0.86169 μM | SANGER | |||
| 5637 | Growth inhibition assay | IC50 = 0.86693 μM | SANGER | |||
| EFM-19 | Growth inhibition assay | IC50 = 0.86752 μM | SANGER | |||
| SW48 | Growth inhibition assay | IC50 = 0.86895 μM | SANGER | |||
| NCI-H748 | Growth inhibition assay | IC50 = 0.87069 μM | SANGER | |||
| CHL-1 | Growth inhibition assay | IC50 = 0.8829 μM | SANGER | |||
| G-402 | Growth inhibition assay | IC50 = 0.89819 μM | SANGER | |||
| LS-411N | Growth inhibition assay | IC50 = 0.92538 μM | SANGER | |||
| GCIY | Growth inhibition assay | IC50 = 0.92549 μM | SANGER | |||
| HuO9 | Growth inhibition assay | IC50 = 0.92578 μM | SANGER | |||
| BPH-1 | Growth inhibition assay | IC50 = 0.94282 μM | SANGER | |||
| ECC12 | Growth inhibition assay | IC50 = 0.95438 μM | SANGER | |||
| SU-DHL-1 | Growth inhibition assay | IC50 = 0.95915 μM | SANGER | |||
| TE-15 | Growth inhibition assay | IC50 = 0.95964 μM | SANGER | |||
| BB65-RCC | Growth inhibition assay | IC50 = 0.97887 μM | SANGER | |||
| NUGC-3 | Growth inhibition assay | IC50 = 0.98081 μM | SANGER | |||
| HCT-116 | Growth inhibition assay | IC50 = 1.00749 μM | SANGER | |||
| 639-V | Growth inhibition assay | IC50 = 1.00797 μM | SANGER | |||
| RPMI-6666 | Growth inhibition assay | IC50 = 1.03221 μM | SANGER | |||
| DoTc2-4510 | Growth inhibition assay | IC50 = 1.04831 μM | SANGER | |||
| L-540 | Growth inhibition assay | IC50 = 1.05791 μM | SANGER | |||
| NB12 | Growth inhibition assay | IC50 = 1.08357 μM | SANGER | |||
| CAL-51 | Growth inhibition assay | IC50 = 1.08397 μM | SANGER | |||
| NB69 | Growth inhibition assay | IC50 = 1.0875 μM | SANGER | |||
| OVCAR-8 | Growth inhibition assay | IC50 = 1.09145 μM | SANGER | |||
| A2780 | Growth inhibition assay | IC50 = 1.10527 μM | SANGER | |||
| RL95-2 | Growth inhibition assay | IC50 = 1.11034 μM | SANGER | |||
| HT-3 | Growth inhibition assay | IC50 = 1.12847 μM | SANGER | |||
| UM-UC-3 | Growth inhibition assay | IC50 = 1.14554 μM | SANGER | |||
| BFTC-909 | Growth inhibition assay | IC50 = 1.16148 μM | SANGER | |||
| SNG-M | Growth inhibition assay | IC50 = 1.16368 μM | SANGER | |||
| MG-63 | Growth inhibition assay | IC50 = 1.17999 μM | SANGER | |||
| TE-5 | Growth inhibition assay | IC50 = 1.18327 μM | SANGER | |||
| A101D | Growth inhibition assay | IC50 = 1.19812 μM | SANGER | |||
| NCI-H209 | Growth inhibition assay | IC50 = 1.20699 μM | SANGER | |||
| SCC-3 | Growth inhibition assay | IC50 = 1.21017 μM | SANGER | |||
| P30-OHK | Growth inhibition assay | IC50 = 1.21124 μM | SANGER | |||
| YKG-1 | Growth inhibition assay | IC50 = 1.22754 μM | SANGER | |||
| CAPAN-1 | Growth inhibition assay | IC50 = 1.24645 μM | SANGER | |||
| HCC2998 | Growth inhibition assay | IC50 = 1.25705 μM | SANGER | |||
| HSC-4 | Growth inhibition assay | IC50 = 1.26252 μM | SANGER | |||
| SAS | Growth inhibition assay | IC50 = 1.2672 μM | SANGER | |||
| COR-L279 | Growth inhibition assay | IC50 = 1.26744 μM | SANGER | |||
| FADU | Growth inhibition assay | IC50 = 1.28154 μM | SANGER | |||
| SK-LU-1 | Growth inhibition assay | IC50 = 1.28962 μM | SANGER | |||
| NCI-H1703 | Growth inhibition assay | IC50 = 1.29939 μM | SANGER | |||
| KASUMI-1 | Growth inhibition assay | IC50 = 1.32239 μM | SANGER | |||
| NCI-H1694 | Growth inhibition assay | IC50 = 1.37183 μM | SANGER | |||
| LK-2 | Growth inhibition assay | IC50 = 1.40757 μM | SANGER | |||
| CHP-212 | Growth inhibition assay | IC50 = 1.41096 μM | SANGER | |||
| HSC-3 | Growth inhibition assay | IC50 = 1.43589 μM | SANGER | |||
| G-401 | Growth inhibition assay | IC50 = 1.43693 μM | SANGER | |||
| SF126 | Growth inhibition assay | IC50 = 1.44086 μM | SANGER | |||
| HuP-T4 | Growth inhibition assay | IC50 = 1.45259 μM | SANGER | |||
| EW-1 | Growth inhibition assay | IC50 = 1.45847 μM | SANGER | |||
| MDA-MB-468 | Growth inhibition assay | IC50 = 1.46554 μM | SANGER | |||
| NCI-H1734 | Growth inhibition assay | IC50 = 1.50503 μM | SANGER | |||
| 22RV1 | Growth inhibition assay | IC50 = 1.53054 μM | SANGER | |||
| BHY | Growth inhibition assay | IC50 = 1.53169 μM | SANGER | |||
| OVCAR-5 | Growth inhibition assay | IC50 = 1.53748 μM | SANGER | |||
| EM-2 | Growth inhibition assay | IC50 = 1.57782 μM | SANGER | |||
| NCI-H810 | Growth inhibition assay | IC50 = 1.58283 μM | SANGER | |||
| IGR-1 | Growth inhibition assay | IC50 = 1.59112 μM | SANGER | |||
| J-RT3-T3-5 | Growth inhibition assay | IC50 = 1.59438 μM | SANGER | |||
| SF539 | Growth inhibition assay | IC50 = 1.60689 μM | SANGER | |||
| GB-1 | Growth inhibition assay | IC50 = 1.60936 μM | SANGER | |||
| SK-CO-1 | Growth inhibition assay | IC50 = 1.62804 μM | SANGER | |||
| BOKU | Growth inhibition assay | IC50 = 1.63222 μM | SANGER | |||
| PANC-03-27 | Growth inhibition assay | IC50 = 1.6369 μM | SANGER | |||
| SF295 | Growth inhibition assay | IC50 = 1.65606 μM | SANGER | |||
| COLO-668 | Growth inhibition assay | IC50 = 1.66431 μM | SANGER | |||
| KM-H2 | Growth inhibition assay | IC50 = 1.66942 μM | SANGER | |||
| C8166 | Growth inhibition assay | IC50 = 1.6773 μM | SANGER | |||
| CTB-1 | Growth inhibition assay | IC50 = 1.68766 μM | SANGER | |||
| NCI-H650 | Growth inhibition assay | IC50 = 1.70971 μM | SANGER | |||
| C-4-II | Growth inhibition assay | IC50 = 1.71356 μM | SANGER | |||
| NCI-H1650 | Growth inhibition assay | IC50 = 1.73565 μM | SANGER | |||
| NCI-SNU-1 | Growth inhibition assay | IC50 = 1.75327 μM | SANGER | |||
| SNU-423 | Growth inhibition assay | IC50 = 1.75342 μM | SANGER | |||
| COLO-829 | Growth inhibition assay | IC50 = 1.76857 μM | SANGER | |||
| SW13 | Growth inhibition assay | IC50 = 1.80203 μM | SANGER | |||
| NCI-H2170 | Growth inhibition assay | IC50 = 1.85285 μM | SANGER | |||
| MKN45 | Growth inhibition assay | IC50 = 1.87359 μM | SANGER | |||
| KM12 | Growth inhibition assay | IC50 = 1.87938 μM | SANGER | |||
| SN12C | Growth inhibition assay | IC50 = 1.88379 μM | SANGER | |||
| SK-NEP-1 | Growth inhibition assay | IC50 = 1.90148 μM | SANGER | |||
| HuCCT1 | Growth inhibition assay | IC50 = 1.91064 μM | SANGER | |||
| SW954 | Growth inhibition assay | IC50 = 1.96311 μM | SANGER | |||
| VMRC-RCZ | Growth inhibition assay | IC50 = 1.96786 μM | SANGER | |||
| HT-29 | Growth inhibition assay | IC50 = 1.97537 μM | SANGER | |||
| LB647-SCLC | Growth inhibition assay | IC50 = 2.00119 μM | SANGER | |||
| KU812 | Growth inhibition assay | IC50 = 2.03197 μM | SANGER | |||
| LoVo | Growth inhibition assay | IC50 = 2.06081 μM | SANGER | |||
| LC-2-ad | Growth inhibition assay | IC50 = 2.06407 μM | SANGER | |||
| KYSE-180 | Growth inhibition assay | IC50 = 2.06953 μM | SANGER | |||
| NCI-H1299 | Growth inhibition assay | IC50 = 2.0703 μM | SANGER | |||
| LXF-289 | Growth inhibition assay | IC50 = 2.07522 μM | SANGER | |||
| BL-41 | Growth inhibition assay | IC50 = 2.08098 μM | SANGER | |||
| COLO-800 | Growth inhibition assay | IC50 = 2.09855 μM | SANGER | |||
| LB1047-RCC | Growth inhibition assay | IC50 = 2.13516 μM | SANGER | |||
| OAW-28 | Growth inhibition assay | IC50 = 2.15234 μM | SANGER | |||
| CAL-85-1 | Growth inhibition assay | IC50 = 2.15944 μM | SANGER | |||
| A673 | Growth inhibition assay | IC50 = 2.16359 μM | SANGER | |||
| MZ2-MEL | Growth inhibition assay | IC50 = 2.17939 μM | SANGER | |||
| NOMO-1 | Growth inhibition assay | IC50 = 2.18798 μM | SANGER | |||
| Ca9-22 | Growth inhibition assay | IC50 = 2.18807 μM | SANGER | |||
| ARH-77 | Growth inhibition assay | IC50 = 2.18911 μM | SANGER | |||
| D-283MED | Growth inhibition assay | IC50 = 2.19537 μM | SANGER | |||
| AN3-CA | Growth inhibition assay | IC50 = 2.20226 μM | SANGER | |||
| ST486 | Growth inhibition assay | IC50 = 2.24988 μM | SANGER | |||
| SW1573 | Growth inhibition assay | IC50 = 2.25339 μM | SANGER | |||
| 769-P | Growth inhibition assay | IC50 = 2.25653 μM | SANGER | |||
| HCC38 | Growth inhibition assay | IC50 = 2.27106 μM | SANGER | |||
| RPMI-7951 | Growth inhibition assay | IC50 = 2.28584 μM | SANGER | |||
| HL-60 | Growth inhibition assay | IC50 = 2.29181 μM | SANGER | |||
| NCI-H1304 | Growth inhibition assay | IC50 = 2.30406 μM | SANGER | |||
| 8305C | Growth inhibition assay | IC50 = 2.32067 μM | SANGER | |||
| Ca-Ski | Growth inhibition assay | IC50 = 2.32829 μM | SANGER | |||
| DSH1 | Growth inhibition assay | IC50 = 2.36138 μM | SANGER | |||
| HEC-1 | Growth inhibition assay | IC50 = 2.3745 μM | SANGER | |||
| HO-1-N-1 | Growth inhibition assay | IC50 = 2.37868 μM | SANGER | |||
| TE-10 | Growth inhibition assay | IC50 = 2.38102 μM | SANGER | |||
| TYK-nu | Growth inhibition assay | IC50 = 2.41237 μM | SANGER | |||
| LU-99A | Growth inhibition assay | IC50 = 2.42772 μM | SANGER | |||
| NCI-H2405 | Growth inhibition assay | IC50 = 2.43564 μM | SANGER | |||
| BC-3 | Growth inhibition assay | IC50 = 2.47545 μM | SANGER | |||
| RERF-LC-MS | Growth inhibition assay | IC50 = 2.49413 μM | SANGER | |||
| JiyoyeP-2003 | Growth inhibition assay | IC50 = 2.5428 μM | SANGER | |||
| MKN28 | Growth inhibition assay | IC50 = 2.54669 μM | SANGER | |||
| SF268 | Growth inhibition assay | IC50 = 2.54745 μM | SANGER | |||
| HuH-7 | Growth inhibition assay | IC50 = 2.56377 μM | SANGER | |||
| VM-CUB-1 | Growth inhibition assay | IC50 = 2.57875 μM | SANGER | |||
| AU565 | Growth inhibition assay | IC50 = 2.61126 μM | SANGER | |||
| GAMG | Growth inhibition assay | IC50 = 2.61355 μM | SANGER | |||
| DBTRG-05MG | Growth inhibition assay | IC50 = 2.62832 μM | SANGER | |||
| NCI-H2228 | Growth inhibition assay | IC50 = 2.65509 μM | SANGER | |||
| MIA-PaCa-2 | Growth inhibition assay | IC50 = 2.6613 μM | SANGER | |||
| NCI-H1882 | Growth inhibition assay | IC50 = 2.67868 μM | SANGER | |||
| CMK | Growth inhibition assay | IC50 = 2.68446 μM | SANGER | |||
| ETK-1 | Growth inhibition assay | IC50 = 2.74528 μM | SANGER | |||
| NB13 | Growth inhibition assay | IC50 = 2.76358 μM | SANGER | |||
| PANC-10-05 | Growth inhibition assay | IC50 = 2.88779 μM | SANGER | |||
| UACC-62 | Growth inhibition assay | IC50 = 2.91997 μM | SANGER | |||
| SW756 | Growth inhibition assay | IC50 = 2.98046 μM | SANGER | |||
| SJSA-1 | Growth inhibition assay | IC50 = 2.98416 μM | SANGER | |||
| L-428 | Growth inhibition assay | IC50 = 2.98894 μM | SANGER | |||
| IST-MEL1 | Growth inhibition assay | IC50 = 2.99943 μM | SANGER | |||
| JEG-3 | Growth inhibition assay | IC50 = 3.04067 μM | SANGER | |||
| SW1783 | Growth inhibition assay | IC50 = 3.05871 μM | SANGER | |||
| BHT-101 | Growth inhibition assay | IC50 = 3.10736 μM | SANGER | |||
| SH-4 | Growth inhibition assay | IC50 = 3.1346 μM | SANGER | |||
| NCI-H358 | Growth inhibition assay | IC50 = 3.15154 μM | SANGER | |||
| LC4-1 | Growth inhibition assay | IC50 = 3.22759 μM | SANGER | |||
| GCT | Growth inhibition assay | IC50 = 3.26576 μM | SANGER | |||
| EGI-1 | Growth inhibition assay | IC50 = 3.27013 μM | SANGER | |||
| NMC-G1 | Growth inhibition assay | IC50 = 3.32432 μM | SANGER | |||
| A388 | Growth inhibition assay | IC50 = 3.32957 μM | SANGER | |||
| J82 | Growth inhibition assay | IC50 = 3.33987 μM | SANGER | |||
| NCI-H522 | Growth inhibition assay | IC50 = 3.35766 μM | SANGER | |||
| EFO-27 | Growth inhibition assay | IC50 = 3.35777 μM | SANGER | |||
| NCI-H1437 | Growth inhibition assay | IC50 = 3.38115 μM | SANGER | |||
| MLMA | Growth inhibition assay | IC50 = 3.38191 μM | SANGER | |||
| GDM-1 | Growth inhibition assay | IC50 = 3.40206 μM | SANGER | |||
| IA-LM | Growth inhibition assay | IC50 = 3.43569 μM | SANGER | |||
| no-10 | Growth inhibition assay | IC50 = 3.48873 μM | SANGER | |||
| NOS-1 | Growth inhibition assay | IC50 = 3.58057 μM | SANGER | |||
| HCC1569 | Growth inhibition assay | IC50 = 3.58372 μM | SANGER | |||
| FTC-133 | Growth inhibition assay | IC50 = 3.58908 μM | SANGER | |||
| AM-38 | Growth inhibition assay | IC50 = 3.66706 μM | SANGER | |||
| KYSE-450 | Growth inhibition assay | IC50 = 3.68871 μM | SANGER | |||
| ChaGo-K-1 | Growth inhibition assay | IC50 = 3.79409 μM | SANGER | |||
| HAL-01 | Growth inhibition assay | IC50 = 3.80443 μM | SANGER | |||
| NCI-H1770 | Growth inhibition assay | IC50 = 3.86539 μM | SANGER | |||
| HEL | Growth inhibition assay | IC50 = 3.89764 μM | SANGER | |||
| NCI-H64 | Growth inhibition assay | IC50 = 3.99787 μM | SANGER | |||
| LU-139 | Growth inhibition assay | IC50 = 4.00867 μM | SANGER | |||
| LS-123 | Growth inhibition assay | IC50 = 4.02035 μM | SANGER | |||
| Calu-6 | Growth inhibition assay | IC50 = 4.05854 μM | SANGER | |||
| KP-N-YS | Growth inhibition assay | IC50 = 4.23582 μM | SANGER | |||
| MZ7-mel | Growth inhibition assay | IC50 = 4.29859 μM | SANGER | |||
| KARPAS-422 | Growth inhibition assay | IC50 = 4.3292 μM | SANGER | |||
| IPC-298 | Growth inhibition assay | IC50 = 4.33485 μM | SANGER | |||
| NCI-H661 | Growth inhibition assay | IC50 = 4.34179 μM | SANGER | |||
| WSU-NHL | Growth inhibition assay | IC50 = 4.36044 μM | SANGER | |||
| HH | Growth inhibition assay | IC50 = 4.44423 μM | SANGER | |||
| NY | Growth inhibition assay | IC50 = 4.48988 μM | SANGER | |||
| BT-20 | Growth inhibition assay | IC50 = 4.5794 μM | SANGER | |||
| NCI-H1417 | Growth inhibition assay | IC50 = 4.60594 μM | SANGER | |||
| CAL-39 | Growth inhibition assay | IC50 = 4.63617 μM | SANGER | |||
| RPMI-8866 | Growth inhibition assay | IC50 = 4.64882 μM | SANGER | |||
| NCI-H292 | Growth inhibition assay | IC50 = 4.7095 μM | SANGER | |||
| SJRH30 | Growth inhibition assay | IC50 = 4.72366 μM | SANGER | |||
| HCC1806 | Growth inhibition assay | IC50 = 4.73101 μM | SANGER | |||
| MEG-01 | Growth inhibition assay | IC50 = 4.74917 μM | SANGER | |||
| EW-11 | Growth inhibition assay | IC50 = 4.76565 μM | SANGER | |||
| RD | Growth inhibition assay | IC50 = 4.80409 μM | SANGER | |||
| KYSE-140 | Growth inhibition assay | IC50 = 4.91746 μM | SANGER | |||
| NCI-H2081 | Growth inhibition assay | IC50 = 4.91765 μM | SANGER | |||
| HOP-92 | Growth inhibition assay | IC50 = 5.02277 μM | SANGER | |||
| BV-173 | Growth inhibition assay | IC50 = 5.05329 μM | SANGER | |||
| OE33 | Growth inhibition assay | IC50 = 5.12216 μM | SANGER | |||
| NCI-H1793 | Growth inhibition assay | IC50 = 5.30922 μM | SANGER | |||
| Daudi | Growth inhibition assay | IC50 = 5.36115 μM | SANGER | |||
| NCI-H2122 | Growth inhibition assay | IC50 = 5.39424 μM | SANGER | |||
| SW1990 | Growth inhibition assay | IC50 = 5.639 μM | SANGER | |||
| MS-1 | Growth inhibition assay | IC50 = 5.64297 μM | SANGER | |||
| HT-1376 | Growth inhibition assay | IC50 = 5.81886 μM | SANGER | |||
| GP5d | Growth inhibition assay | IC50 = 5.8311 μM | SANGER | |||
| OAW-42 | Growth inhibition assay | IC50 = 6.05896 μM | SANGER | |||
| LB831-BLC | Growth inhibition assay | IC50 = 6.15047 μM | SANGER | |||
| COR-L23 | Growth inhibition assay | IC50 = 6.18279 μM | SANGER | |||
| CP66-MEL | Growth inhibition assay | IC50 = 6.25378 μM | SANGER | |||
| CHP-126 | Growth inhibition assay | IC50 = 6.27397 μM | SANGER | |||
| GOTO | Growth inhibition assay | IC50 = 6.37509 μM | SANGER | |||
| DB | Growth inhibition assay | IC50 = 6.5298 μM | SANGER | |||
| L-363 | Growth inhibition assay | IC50 = 6.58998 μM | SANGER | |||
| SCC-25 | Growth inhibition assay | IC50 = 6.63406 μM | SANGER | |||
| CAKI-1 | Growth inhibition assay | IC50 = 6.65287 μM | SANGER | |||
| GR-ST | Growth inhibition assay | IC50 = 6.68733 μM | SANGER | |||
| Mewo | Growth inhibition assay | IC50 = 6.79226 μM | SANGER | |||
| MMAC-SF | Growth inhibition assay | IC50 = 6.87553 μM | SANGER | |||
| CCF-STTG1 | Growth inhibition assay | IC50 = 6.91149 μM | SANGER | |||
| NCI-H1563 | Growth inhibition assay | IC50 = 7.06829 μM | SANGER | |||
| BL-70 | Growth inhibition assay | IC50 = 7.07066 μM | SANGER | |||
| SCC-4 | Growth inhibition assay | IC50 = 7.20234 μM | SANGER | |||
| TE-11 | Growth inhibition assay | IC50 = 7.45503 μM | SANGER | |||
| TK10 | Growth inhibition assay | IC50 = 7.49225 μM | SANGER | |||
| NCI-H2196 | Growth inhibition assay | IC50 = 7.49486 μM | SANGER | |||
| no-11 | Growth inhibition assay | IC50 = 7.55794 μM | SANGER | |||
| HuO-3N1 | Growth inhibition assay | IC50 = 7.71346 μM | SANGER | |||
| HCT-15 | Growth inhibition assay | IC50 = 7.87059 μM | SANGER | |||
| Calu-1 | Growth inhibition assay | IC50 = 7.89022 μM | SANGER | |||
| NCI-SNU-5 | Growth inhibition assay | IC50 = 7.91903 μM | SANGER | |||
| C3A | Growth inhibition assay | IC50 = 7.93051 μM | SANGER | |||
| M14 | Growth inhibition assay | IC50 = 7.94538 μM | SANGER | |||
| KMOE-2 | Growth inhibition assay | IC50 = 8.03697 μM | SANGER | |||
| NCI-H520 | Growth inhibition assay | IC50 = 8.03907 μM | SANGER | |||
| NCI-H69 | Growth inhibition assay | IC50 = 8.18234 μM | SANGER | |||
| SK-MEL-30 | Growth inhibition assay | IC50 = 8.24405 μM | SANGER | |||
| COLO-680N | Growth inhibition assay | IC50 = 8.25621 μM | SANGER | |||
| Raji | Growth inhibition assay | IC50 = 8.31941 μM | SANGER | |||
| NCI-H2141 | Growth inhibition assay | IC50 = 8.52227 μM | SANGER | |||
| HD-MY-Z | Growth inhibition assay | IC50 = 8.53777 μM | SANGER | |||
| CaR-1 | Growth inhibition assay | IC50 = 8.60091 μM | SANGER | |||
| A172 | Growth inhibition assay | IC50 = 8.64964 μM | SANGER | |||
| NCI-H510A | Growth inhibition assay | IC50 = 8.65064 μM | SANGER | |||
| TE-8 | Growth inhibition assay | IC50 = 8.75118 μM | SANGER | |||
| T98G | Growth inhibition assay | IC50 = 8.81581 μM | SANGER | |||
| SIMA | Growth inhibition assay | IC50 = 8.85231 μM | SANGER | |||
| SW626 | Growth inhibition assay | IC50 = 8.90372 μM | SANGER | |||
| CAL-54 | Growth inhibition assay | IC50 = 8.92125 μM | SANGER | |||
| MDA-MB-231 | Growth inhibition assay | IC50 = 9.08645 μM | SANGER | |||
| NCI-H630 | Growth inhibition assay | IC50 = 9.12079 μM | SANGER | |||
| LAMA-84 | Growth inhibition assay | IC50 = 9.20893 μM | SANGER | |||
| COR-L105 | Growth inhibition assay | IC50 = 9.56953 μM | SANGER | |||
| NCI-H2347 | Growth inhibition assay | IC50 = 9.66391 μM | SANGER | |||
| SK-MM-2 | Growth inhibition assay | IC50 = 9.68223 μM | SANGER | |||
| PC-14 | Growth inhibition assay | IC50 = 9.72234 μM | SANGER | |||
| KG-1 | Growth inhibition assay | IC50 = 9.72465 μM | SANGER | |||
| COLO-320-HSR | Growth inhibition assay | IC50 = 9.73489 μM | SANGER | |||
| GI-ME-N | Growth inhibition assay | IC50 = 9.82616 μM | SANGER | |||
| KMS-12-PE | Growth inhibition assay | IC50 = 9.91106 μM | SANGER | |||
| Ramos-2G6-4C10 | Growth inhibition assay | IC50 = 9.93808 μM | SANGER | |||
| HCC1954 | Growth inhibition assay | IC50 = 10.0429 μM | SANGER | |||
| SK-MEL-3 | Growth inhibition assay | IC50 = 10.1322 μM | SANGER | |||
| NB1 | Growth inhibition assay | IC50 = 10.2077 μM | SANGER | |||
| KLE | Growth inhibition assay | IC50 = 10.2925 μM | SANGER | |||
| KALS-1 | Growth inhibition assay | IC50 = 10.3029 μM | SANGER | |||
| NCI-H2126 | Growth inhibition assay | IC50 = 10.3993 μM | SANGER | |||
| CAL-27 | Growth inhibition assay | IC50 = 10.4443 μM | SANGER | |||
| SK-OV-3 | Growth inhibition assay | IC50 = 10.8274 μM | SANGER | |||
| SNB75 | Growth inhibition assay | IC50 = 10.9411 μM | SANGER | |||
| NCI-H1155 | Growth inhibition assay | IC50 = 11.02 μM | SANGER | |||
| Detroit562 | Growth inhibition assay | IC50 = 11.0755 μM | SANGER | |||
| CAL-12T | Growth inhibition assay | IC50 = 11.136 μM | SANGER | |||
| NCI-H226 | Growth inhibition assay | IC50 = 11.1518 μM | SANGER | |||
| NCI-H441 | Growth inhibition assay | IC50 = 11.166 μM | SANGER | |||
| OS-RC-2 | Growth inhibition assay | IC50 = 11.2175 μM | SANGER | |||
| U031 | Growth inhibition assay | IC50 = 11.2885 μM | SANGER | |||
| NCI-H2052 | Growth inhibition assay | IC50 = 11.4502 μM | SANGER | |||
| TCCSUP | Growth inhibition assay | IC50 = 11.6187 μM | SANGER | |||
| T47D | Growth inhibition assay | IC50 = 11.6637 μM | SANGER | |||
| Becker | Growth inhibition assay | IC50 = 11.8156 μM | SANGER | |||
| EFO-21 | Growth inhibition assay | IC50 = 11.8503 μM | SANGER | |||
| NCI-H1963 | Growth inhibition assay | IC50 = 12.0942 μM | SANGER | |||
| MONO-MAC-6 | Growth inhibition assay | IC50 = 12.1309 μM | SANGER | |||
| MOLT-16 | Growth inhibition assay | IC50 = 12.4619 μM | SANGER | |||
| U-698-M | Growth inhibition assay | IC50 = 12.4627 μM | SANGER | |||
| MDA-MB-157 | Growth inhibition assay | IC50 = 12.6094 μM | SANGER | |||
| KYSE-410 | Growth inhibition assay | IC50 = 12.8456 μM | SANGER | |||
| CAMA-1 | Growth inhibition assay | IC50 = 13.0046 μM | SANGER | |||
| SCC-15 | Growth inhibition assay | IC50 = 13.1497 μM | SANGER | |||
| NCI-H23 | Growth inhibition assay | IC50 = 13.1861 μM | SANGER | |||
| KNS-42 | Growth inhibition assay | IC50 = 13.6511 μM | SANGER | |||
| HDLM-2 | Growth inhibition assay | IC50 = 13.7938 μM | SANGER | |||
| NCI-H1623 | Growth inhibition assay | IC50 = 13.9776 μM | SANGER | |||
| RPMI-8226 | Growth inhibition assay | IC50 = 14.3243 μM | SANGER | |||
| EW-3 | Growth inhibition assay | IC50 = 14.3337 μM | SANGER | |||
| HT | Growth inhibition assay | IC50 = 15.2889 μM | SANGER | |||
| CAL-120 | Growth inhibition assay | IC50 = 15.6326 μM | SANGER | |||
| HCC1937 | Growth inhibition assay | IC50 = 15.7725 μM | SANGER | |||
| NCI-N87 | Growth inhibition assay | IC50 = 15.8106 μM | SANGER | |||
| NB10 | Growth inhibition assay | IC50 = 15.9987 μM | SANGER | |||
| UACC-812 | Growth inhibition assay | IC50 = 16.4249 μM | SANGER | |||
| MHH-PREB-1 | Growth inhibition assay | IC50 = 16.4817 μM | SANGER | |||
| SNU-449 | Growth inhibition assay | IC50 = 16.6657 μM | SANGER | |||
| COLO-684 | Growth inhibition assay | IC50 = 16.7953 μM | SANGER | |||
| BxPC-3 | Growth inhibition assay | IC50 = 16.8611 μM | SANGER | |||
| DMS-153 | Growth inhibition assay | IC50 = 16.9246 μM | SANGER | |||
| SW962 | Growth inhibition assay | IC50 = 16.9729 μM | SANGER | |||
| OVCAR-4 | Growth inhibition assay | IC50 = 17.3428 μM | SANGER | |||
| CAS-1 | Growth inhibition assay | IC50 = 17.5833 μM | SANGER | |||
| HPAF-II | Growth inhibition assay | IC50 = 17.6767 μM | SANGER | |||
| NCI-H838 | Growth inhibition assay | IC50 = 17.9844 μM | SANGER | |||
| D-247MG | Growth inhibition assay | IC50 = 18.052 μM | SANGER | |||
| T-24 | Growth inhibition assay | IC50 = 18.0868 μM | SANGER | |||
| YT | Growth inhibition assay | IC50 = 18.2712 μM | SANGER | |||
| NCI-H596 | Growth inhibition assay | IC50 = 18.2999 μM | SANGER | |||
| CPC-N | Growth inhibition assay | IC50 = 18.426 μM | SANGER | |||
| HN | Growth inhibition assay | IC50 = 18.5533 μM | SANGER | |||
| KARPAS-299 | Growth inhibition assay | IC50 = 19.2708 μM | SANGER | |||
| ES6 | Growth inhibition assay | IC50 = 19.6334 μM | SANGER | |||
| IST-SL1 | Growth inhibition assay | IC50 = 19.8912 μM | SANGER | |||
| LOXIMVI | Growth inhibition assay | IC50 = 19.9253 μM | SANGER | |||
| U-266 | Growth inhibition assay | IC50 = 19.9794 μM | SANGER | |||
| HCC2157 | Growth inhibition assay | IC50 = 20.3328 μM | SANGER | |||
| MPP-89 | Growth inhibition assay | IC50 = 20.7595 μM | SANGER | |||
| MDA-MB-361 | Growth inhibition assay | IC50 = 20.8051 μM | SANGER | |||
| RCM-1 | Growth inhibition assay | IC50 = 20.9952 μM | SANGER | |||
| SW780 | Growth inhibition assay | IC50 = 21.109 μM | SANGER | |||
| SK-MEL-2 | Growth inhibition assay | IC50 = 21.2698 μM | SANGER | |||
| TE-1 | Growth inhibition assay | IC50 = 21.4838 μM | SANGER | |||
| NB5 | Growth inhibition assay | IC50 = 21.8618 μM | SANGER | |||
| OVCAR-3 | Growth inhibition assay | IC50 = 22.2138 μM | SANGER | |||
| LNCaP-Clone-FGC | Growth inhibition assay | IC50 = 23.5156 μM | SANGER | |||
| KYSE-520 | Growth inhibition assay | IC50 = 24.0956 μM | SANGER | |||
| UMC-11 | Growth inhibition assay | IC50 = 24.3195 μM | SANGER | |||
| NCCIT | Growth inhibition assay | IC50 = 24.4842 μM | SANGER | |||
| OPM-2 | Growth inhibition assay | IC50 = 24.6118 μM | SANGER | |||
| GAK | Growth inhibition assay | IC50 = 25.1015 μM | SANGER | |||
| PLC-PRF-5 | Growth inhibition assay | IC50 = 25.4198 μM | SANGER | |||
| K5 | Growth inhibition assay | IC50 = 25.5192 μM | SANGER | |||
| HT55 | Growth inhibition assay | IC50 = 25.82 μM | SANGER | |||
| HT-1197 | Growth inhibition assay | IC50 = 25.8682 μM | SANGER | |||
| Saos-2 | Growth inhibition assay | IC50 = 26.0963 μM | SANGER | |||
| NCI-H1648 | Growth inhibition assay | IC50 = 26.3623 μM | SANGER | |||
| NCI-H747 | Growth inhibition assay | IC50 = 27.2478 μM | SANGER | |||
| U-87-MG | Growth inhibition assay | IC50 = 27.5565 μM | SANGER | |||
| LU-165 | Growth inhibition assay | IC50 = 28.8005 μM | SANGER | |||
| Caov-3 | Growth inhibition assay | IC50 = 29.2228 μM | SANGER | |||
| MN-60 | Growth inhibition assay | IC50 = 29.5512 μM | SANGER | |||
| TE-6 | Growth inhibition assay | IC50 = 29.723 μM | SANGER | |||
| EHEB | Growth inhibition assay | IC50 = 30.3605 μM | SANGER | |||
| SNU-C2B | Growth inhibition assay | IC50 = 30.4898 μM | SANGER | |||
| YAPC | Growth inhibition assay | IC50 = 30.5644 μM | SANGER | |||
| LU-65 | Growth inhibition assay | IC50 = 30.8083 μM | SANGER | |||
| HTC-C3 | Growth inhibition assay | IC50 = 30.8175 μM | SANGER | |||
| HuP-T3 | Growth inhibition assay | IC50 = 30.9157 μM | SANGER | |||
| LB2241-RCC | Growth inhibition assay | IC50 = 31.1032 μM | SANGER | |||
| SK-N-DZ | Growth inhibition assay | IC50 = 31.4228 μM | SANGER | |||
| KYSE-70 | Growth inhibition assay | IC50 = 32.3637 μM | SANGER | |||
| EW-18 | Growth inhibition assay | IC50 = 32.7339 μM | SANGER | |||
| NCI-H1651 | Growth inhibition assay | IC50 = 32.7543 μM | SANGER | |||
| LS-1034 | Growth inhibition assay | IC50 = 33.1173 μM | SANGER | |||
| RXF393 | Growth inhibition assay | IC50 = 33.7866 μM | SANGER | |||
| OMC-1 | Growth inhibition assay | IC50 = 36.8562 μM | SANGER | |||
| OE19 | Growth inhibition assay | IC50 = 36.9759 μM | SANGER | |||
| MHH-CALL-2 | Growth inhibition assay | IC50 = 37.3808 μM | SANGER | |||
| BB49-HNC | Growth inhibition assay | IC50 = 37.3849 μM | SANGER | |||
| A498 | Growth inhibition assay | IC50 = 37.4282 μM | SANGER | |||
| NCI-H1581 | Growth inhibition assay | IC50 = 37.647 μM | SANGER | |||
| LB996-RCC | Growth inhibition assay | IC50 = 38.6567 μM | SANGER | |||
| SKG-IIIa | Growth inhibition assay | IC50 = 38.976 μM | SANGER | |||
| SBC-1 | Growth inhibition assay | IC50 = 39.1276 μM | SANGER | |||
| TE-441-T | Growth inhibition assay | IC50 = 39.1447 μM | SANGER | |||
| KINGS-1 | Growth inhibition assay | IC50 = 39.2733 μM | SANGER | |||
| NCI-H28 | Growth inhibition assay | IC50 = 39.4002 μM | SANGER | |||
| NCI-H1618 | Growth inhibition assay | IC50 = 40.1041 μM | SANGER | |||
| MFE-280 | Growth inhibition assay | IC50 = 40.5005 μM | SANGER | |||
| EB-3 | Growth inhibition assay | IC50 = 40.6243 μM | SANGER | |||
| IM-9 | Growth inhibition assay | IC50 = 41.6341 μM | SANGER | |||
| DV-90 | Growth inhibition assay | IC50 = 41.9741 μM | SANGER | |||
| NCI-H1792 | Growth inhibition assay | IC50 = 42.6858 μM | SANGER | |||
| SNU-387 | Growth inhibition assay | IC50 = 43.2181 μM | SANGER | |||
| HCC1395 | Growth inhibition assay | IC50 = 44.7953 μM | SANGER | |||
| TGW | Growth inhibition assay | IC50 = 46.3289 μM | SANGER | |||
| KY821 | Growth inhibition assay | IC50 = 47.0932 μM | SANGER | |||
| ECC4 | Growth inhibition assay | IC50 = 48.0182 μM | SANGER | |||
| KARPAS-45 | Growth inhibition assay | IC50 = 48.2359 μM | SANGER | |||
| LB2518-MEL | Growth inhibition assay | IC50 = 48.5056 μM | SANGER | |||
| COR-L88 | Growth inhibition assay | IC50 = 48.735 μM | SANGER | |||
| COLO-741 | Growth inhibition assay | IC50 = 48.8966 μM | SANGER | |||
| HCE-T | Growth inhibition assay | IC50 = 49.301 μM | SANGER | |||
| NCI-H1755 | Growth inhibition assay | IC50 = 49.3468 μM | SANGER | |||
| Click to View More Cell Line Experimental Data | ||||||
Chemical Information, Storage & Stability
| Molecular Weight | 588.56 | Formula | C29H32O13 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 33419-42-0 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | VP-16213,VP-16 | Smiles | CC1OCC2C(O1)C(C(C(O2)OC3C4COC(=O)C4C(C5=CC6=C(C=C35)OCO6)C7=CC(=C(C(=C7)OC)O)OC)O)O | ||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(169.9 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
%
DMSO
%
%
Tween 80
%
ddH2O
%
DMSO
+
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Mechanism of Action
| Targets/IC50/Ki |
Topo II [2]
(Cell-free assay) |
|---|---|
| In vitro |
Etoposide inhibits DNA synthesis by forming a complex with topoisomerase II and DNA, which induces breaks in double stranded DNA and prevents repair by topoisomerase II binding. Accumulated breaks in DNA prevent entry into the mitotic phase of cell division, and lead to cell death. This compound acts primarily in the G2 and S phases of the cell cycle. [1] It inhibits the growth of murine angiosarcoma cell line (ISOS-1) in a 5 days-period with IC50 of 0.25 μg/mL. Cell growth of normal murine microvascular endothelial cells (mECs) is less sensitive to this chemical with IC50 of 10 μg/mL). [2] This agent treated for 6 hr inhibits colonies of tetraploid variant of the human leukemic lymphoblast line CCRF-CEM with IC50 of 0.6 μM. [3] It treated for 2 hr inhibits growth of human pancreatic cancer cell line Y1, Y3, Y5, Y19, YM. YS, and YT with IC50s of 300 μg/mL, 300 μg/mL, 300 μg/mL, 91 μg/mL, 0.68 μg/mL, 300 μg/mL, 300 μg/mL, and 260 μg/mL, respectively. [4] This compound exposed for 1 hr inhibits growth of human glioma cell lines CL5, G142, G152, G111, and G5 with IC50 of 8, 9, 9.8, 10, and 15.8 μg/mL respectively for 12 days. Under same condition, the IC90 value is attained in cell lines CL5, G152, G142, and G111 at 26, 27, 32, and 33 μg/mL. Its inhibition of topoisomerase II is homogeneous for each cell. The average inhibition rates are 15%, 21.8%, 31.8%, 41.5%, and 49.5% for 1, 2, 4, 8, and 16 μg of this agent, respectively. [5] |
| Kinase Assay |
Topoisomerase II activity assay
|
|
Nuclear extracts are prepared, and nuclei are isolated. The activity of topoisomerase II is calculated from the percentage of decatenation obtained. Tritiated kinoplast DNA (KDNA 0.22 μg) is used as a substrate. Etoposide and topoisomerase II are incubated for 30 min at 37 ℃ and are stopped with 1% sodium dodecyl sulfate (SDS) and proteinase K (100 μg/mL). The percentages of decatenation and inhibition of topoisomerase II by this compound are obtained.
|
|
| In vivo |
Etoposide administrated as a single agent is found to been ineffective in many xenografts growth, such as Heterotransplanted Hepatoblastoma NMHB1, and NMHB 2, [6] human neuroblastoma xenograft, [7] and human gastrointestinal cancer xenograft, [8] while the dose of 10 mg/kg i.p. of this compound inhibits murine angiosarcoma cell ISOS-1 tumors in 36% of controls. [2] This chemical induces tumor immunity in Lewis lung cancer. A single administration of 50 mg/kg of this agent i.p., induces a 60% survival of C57B1/6 mice injected with Lewis lung cancer cell (3LL) over 60 days. About 40% of these surviving mice reject a subsequent challenge with 3LL, while none of control mice survive beyond 30 days. 3LL cells which have survived an 90% lethal concentration of this compound in vitro kill 75% of recipient mice, but 60% surviving mice reject challenge with 3LL. Splenocytes harvested from tumor rejecting mice protect naive mice injected with 3LL. [9] |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p53 / p-p53 / Acetyl-p53 / TF-IIB p53 / p21 / c-Myc / PARP |
|
23800173 |
| Immunofluorescence | YAP p53 pDNA-PKcs MDC1 γH2AX / pATM COX IV |
|
27515249 |
| Growth inhibition assay | Cell viability |
|
22615609 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06393816 | Not yet recruiting | Large Cell Neuroendocrine Carcinoma of the Lung |
Centre Leon Berard|Groupe Français de Pneumo-Cancérologie |
May 2024 | Phase 2 |
| NCT06406465 | Not yet recruiting | Carcinoma Neuroendocrine|Tumor Neuroendocrine|Tumors Neuroendocrine|Neuroendocrine; Carcinoma|Small Cell; Receptors |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 2 |
| NCT06086288 | Not yet recruiting | Merkel Cell Carcinoma |
Fondazione IRCCS Istituto Nazionale dei Tumori Milano |
November 2023 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Regarding the S1225, do you have any data of the inhibition specificity of this product? It will inhibit other enzymes other than TOP2A?
Answer:
According to the available published data, its inhibition is specific to TOP2A. But there're also two papers showing that it could inhibit the p34cdc2 Kinase Activity: 1. http://cancerres.aacrjournals.org/content/52/7/1817.short ; 2. http://cancerres.aacrjournals.org/content/50/12/3761.short.






































