research use only
Clotrimazole Antineoplastic and Immunosuppressive Antibiotics inhibitor
Cat.No.S1606
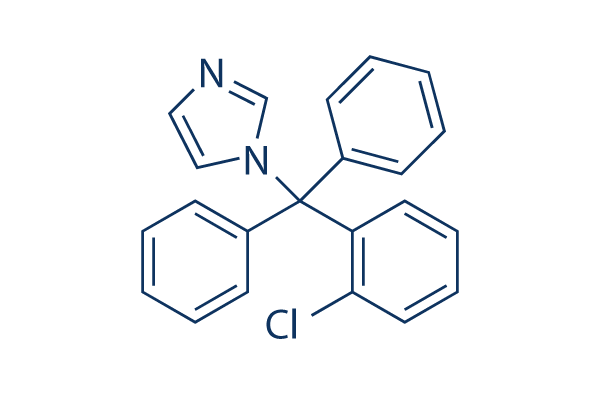
Chemical Structure
Molecular Weight: 344.84
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV |
|---|---|
| Other Antineoplastic and Immunosuppressive Antibiotics Inhibitors | Staurosporine (STS) Cyclosporin A Oligomycin A (MCH 32) Puromycin Dihydrochloride Nigericin sodium salt Geldanamycin (NSC 122750) Honokiol Streptozotocin (STZ) Sodium Monensin (NSC 343257) Cephalomannine |
Solubility
|
In vitro |
Ethanol : 69 mg/mL
DMSO
: 11 mg/mL
(31.89 mM)
Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 344.84 | Formula | C22H17ClN2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 23593-75-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | BAY b 5097, FB 5097 | Smiles | C1=CC=C(C=C1)C(C2=CC=CC=C2)(C3=CC=CC=C3Cl)N4C=CN=C4 | ||
Mechanism of Action
| In vitro |
Clotrimazole causes a sustained depletion of intracellular Ca2+ stores, which results in activation of PKR, phosphorylation of eIF2alpha, and thereby in inhibition of protein synthesis at the level of translation initiation. This compound preferentially decreases the expression of the growth promoting proteins cyclin A, E and D1, resulting in inhibition of cyclin-dependent kinase activity and blockage of cell cycle in G1. It inhibits ADP-ribose-activated currents in HEK-293 cells expressing recombinant human TRPM2(hTRPM2). This chemical (3 mM to 30 mM) produces an essentially complete inhibition of theTRPM2-mediated current. It antagonizes ADP-ribose-activated whole-cell and single-channel currents in the rat insulinoma cell-line CRI-G1. This compound (2 mM) causes a sharp decline in parasitemia, complete inhibition of parasite replication, and destruction of parasites and host cells within a single intraerythrocytic asexual cycle (approximately 48 hours). It effectively and rapidly inhibits parasite growth in five different strains of P. falciparum, in vitro, irrespective of their Chloroquine sensitivity.
|
|---|---|
| In vivo |
Clotrimazole stimulates a subset of capsaicin-sensitive and mustard oil-sensitive trigeminal neurons, and evoked nocifensive behavior and thermal hypersensitivity with intraplantar injection in mice. This compound-induced pain behavior is suppressed by the TRPV1-antagonist BCTC [(N-(-4-tertiarybutylphenyl)-4-(3-cholorpyridin-2-yl)tetrahydropyrazine-1(2H)-carboxamide)] and absent in TRPV1-deficient mice. It inhibits the cold and menthol receptor TRPM8, and blocks menthol-induced responses in capsaicin- and mustard oil-insensitive trigeminal neurons.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































