- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
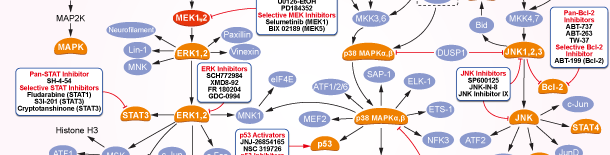
MEK
MEK Products
- All (30)
- MEK Inhibitors (29)
- MEK Antagonist (1)
- New MEK Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
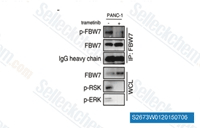
| S2673 | Trametinib (GSK1120212) | Trametinib (GSK1120212, JTP-74057) is a highly specific and potent MEK1/2 inhibitor with IC50 of 0.92 nM/1.8 nM in cell-free assays, no inhibition of the kinase activities of c-Raf, B-Raf, ERK1/2. Trametinib activates autophagy and induces apoptosis. |
|
|
| S1036 | Mirdametinib (PD0325901) | Mirdametinib (PD0325901) is a selective and non ATP-competitive MEK inhibitor with IC50 of 0.33 nM in cell-free assays, roughly 500-fold more potent than CI-1040 on phosphorylation of ERK1 and ERK2. Phase 2. |

|
|
| S1102 | U0126-EtOH | U0126-EtOH is a highly selective inhibitor of MEK1/2 with IC50 of 0.07 μM/0.06 μM in cell-free assays, 100-fold higher affinity for ΔN3-S218E/S222D MEK than PD98059. U0126 inhibits autophagy and mitophagy with antiviral activity. |

|
|
| S1008 | Selumetinib (AZD6244) | Selumetinib (AZD6244, ARRY-142886) is a potent, highly selective MEK inhibitor with IC50 of 14 nM for MEK1 and Kd value of 530 nM for MEK2. It also inhibits ERK1/2 phosphorylation with IC50 of 10 nM, no inhibition to p38α, MKK6, EGFR, ErbB2, ERK2, B-Raf, etc. Selumetinib suppresses cell proliferation, migration and trigger apoptosis. Phase 3. |

|
|
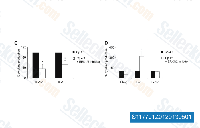
| S1177 | PD98059 | PD98059 is a non-ATP competitive MEK inhibitor with IC50 of 2 μM in a cell-free assay, specifically inhibits MEK-1-mediated activation of MAPK; does not directly inhibit ERK1 or ERK2. PD98059 is a ligand for the aryl hydrocarbon receptor (AHR) and functions as an AHR antagonist. |
|
|
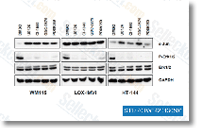
| S1020 | PD184352 (CI-1040) | PD184352 (CI-1040) is an ATP non-competitive MEK1/2 inhibitor with IC50 of 17 nM in cell-based assays, 100-fold more selective for MEK1/2 than MEK5. PD184352 (CI-1040) selectively induces apoptosis. |
|
|
| S8041 | Cobimetinib (GDC-0973) | Cobimetinib (GDC-0973, RG7420) is a potent and highly selective MEK1 inhibitor with IC50 of 4.2 nM, and showing no significant inhibition when tested against a panel of more than 100 of serine-threonine and tyrosine kinases. Cobimetinib induces apoptosis. Phase 3. |
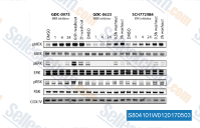
|
|
| S7007 | Binimetinib (MEK162) | Binimetinib (MEK162, ARRY-162, ARRY-438162) is a potent inhibitor of MEK1/2 with IC50 of 12 nM in a cell-free assay. Binimetinib induces G1 cell cycle arrest and apoptosis in human NSCLC cell lines and induces autophagy. Phase 3. |

|
|
| S1531 | BIX 02189 | BIX02189 is a selective inhibitor of MEK5 with IC50 of 1.5 nM, also inhibits ERK5 catalytic activity with IC50 of 59 nM in cell-free assays, and does not inhibit closely related kinases MEK1, MEK2, ERK2, and JNK2. |

|
|
| S1475 | Pimasertib (AS-703026) | Pimasertib (AS-703026, MSC1936369B, SAR 245509) is a highly selective, potent, ATP non-competitive allosteric inhibitor of MEK1/2 with IC50 of 5 nM-2 μM in MM cell lines. Phase 2. |
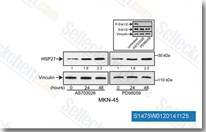
|
|
| S2310 | Honokiol | Honokiol is the active principle of magnolia extract that inhibits Akt-phosphorylation and promotes ERK1/2 phosphorylation. Honokiol causes G0/G1 phase arrest, induces apoptosis, and autophagy via the ROS/ERK1/2 signaling pathway. Honokiol inhibits hepatitis C virus (HCV) infection. Phase 3. |

|
|
| S1089 | Refametinib (RDEA119) | Refametinib (RDEA119, Bay 86-9766) is a potent, ATP non-competitive and highly selective inhibitor of MEK1 and MEK2 with IC50 of 19 nM and 47 nM, respectively. |

|
|
| S2617 | TAK-733 | TAK-733 is a potent and selective MEK allosteric site inhibitor for MEK1 with IC50 of 3.2 nM, inactive to Abl1, AKT3, c-RAF, CamK1, CDK2, c-Met, etc. Phase 1. |

|
|
| S2134 | AZD8330 | AZD8330 (ARRY704) is a novel, selective, non-ATP competitive MEK 1/2 inhibitor with IC50 of 7 nM. Phase 1. |
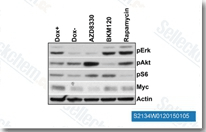
|
|
| S7170 | Avutometinib | Avutometinib(RO5126766,CH5126766,VS 6766, CKI-27, R-7304, RG-7304) is a dual RAF/MEK inhibitor with IC50 of 8.2 nM,19 nM, 56 nM, and 160 nM for BRAF V600E, BRAF, CRAF, and MEK1, respectively. Phase 1. | ||
| S1530 | BIX 02188 | BIX02188 is a selective inhibitor of MEK5 with IC50 of 4.3 nM, also inhibits ERK5 catalytic activity with IC50 of 810 nM, and does not inhibit closely related kinases MEK1, MEK2, ERK2, and JNK2. |
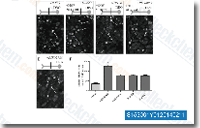
|
|
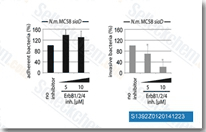
| S1392 | Pelitinib (EKB-569) | Pelitinib (EKB-569) is a potent irreversible EGFR inhibitor with IC50 of 38.5 nM. Pelitinib (EKB-569) also slightly inhibits Src, MEK/ERK and ErbB2 with IC50s of 282 nM, 800 nM and 1255 nM, respectively. Phase2. |
|
|
| S7553 | GDC-0623 | GDC-0623 (G-868) is a potent and ATP-uncompetitive MEK1 inhibitor with Ki of 0.13 nM. Phase 1. |

|
|
| S1066 | SL-327 | SL327 is a selective inhibitor for MEK1/2 with IC50 of 0.18 μM/ 0.22 μM, no activity towards Erk1, MKK3, MKK4, c-JUN, PKC, PKA, or CamKII;capable of transport through the blood-brain barrier. | ||
| S2326 | Myricetin | Myricetin, a natural flavonoid with antioxidant and anti tumor properties, is a novel inhibitor of MEK1 activity and transformation of JB6 P+ mouse epidermal cells. It also inhibits PI3Kγ with Kd of 0.17 μM. | ||
| S7843 | BI-847325 | BI-847325 is an orally bioavailable, and selective dual MEK/Aurora kinase inhibitor with IC50 of 3 nM, 25 nM, 15 nM, 25 nM, and 4 nM for Xenopus laevis Aurora B, human Aurora A and Aurora C, as well as human MEK1 and MEK2, respectively. Phase 1. | ||
| S1568 | PD318088 | PD318088 is a non-ATP competitive allosteric MEK1/2 inhibitor, binds simultaneously with ATP in a region of the MEK1 active site that is adjacent to the ATP-binding site. |

|
|
| S4484 | Trametinib DMSO solvate | Trametinib DMSO solvate is a highly specific and potent MEK1/2 inhibitor with IC50 of 0.92 nM/1.8 nM in cell-free assay. Trametinib activates autophagy and induces apoptosis. | ||
| S8355 | APS-2-79 HCl | APS-2-79 is a MAPK antagonist that modulating KSR-dependent MAPK signalling by antagonizing RAF heterodimerization as well as the conformational changes required for phosphorylation and activation of KSR-bound MEK. IC50=120 ± 23 nM. |

|
|
| S0739 | PD184161 | PD184161, an orally active MEK inhibitor with IC50 of 10-100 nM in a time- and concentration-dependent manner, inhibits cell proliferation and induces apoptosis, and produces depressive-like behavior. | ||
| E3106 | Dioscoreae Nipponicae Rhizoma Extract | Dioscoreae Nipponicae Rhizoma Extract is extracted from the rhizome of Dioscorea nipponica, of which the main component decreases the phosphorylation in IGF-1R, which in turn inhibits the phosphorylation and activation of PI3K-AKT and Rap1-MEK signaling pathways, promoting cell apoptosis and Graves’ disease remission. | ||
| E2821 | RGB-286638 free base | RGB-286638 free base is a Cyclin-dependent kinase (CDK) inhibitor that inhibits the kinase activity of cyclin T1-CDK9, cyclin B1-CDK1, cyclin E-CDK2, cyclin D1-CDK4, cyclin E-CDK3, and p35-CDK5 with IC50s of 1, 2, 3, 4, 5 and 5 nM, respectively; also inhibits GSK-3β, TAK1, Jak2 and MEK1, with IC50s of 3, 5, 50, and 54 nM. | ||
| S6641 | GW284543 (UNC10225170) | GW284543 (UNC10225170), a MEK5-selective inhibitor, inhibits MEK5 in a dose-dependently way as determined by reductions in pERK5, and decreases endogenous MYC protein. | ||
| S0372New | RO4987655 | RO4987655(RG 7167,CH4987655) is an allosteric MEK inhibitor with IC50 of 5 nM. RO4987655 exhibits a significant anti-proliferative and antitumor activity. | ||
| E0404 | Zapnometinib (PD0184264) | Zapnometinib (PD0184264, ATR-002), an major active metabolite of CI-1040, is a novel MEK inhibitor with an IC50 of 5.7 nM. | ||
| S2673 | Trametinib (GSK1120212) | Trametinib (GSK1120212, JTP-74057) is a highly specific and potent MEK1/2 inhibitor with IC50 of 0.92 nM/1.8 nM in cell-free assays, no inhibition of the kinase activities of c-Raf, B-Raf, ERK1/2. Trametinib activates autophagy and induces apoptosis. |

|
|
| S1036 | Mirdametinib (PD0325901) | Mirdametinib (PD0325901) is a selective and non ATP-competitive MEK inhibitor with IC50 of 0.33 nM in cell-free assays, roughly 500-fold more potent than CI-1040 on phosphorylation of ERK1 and ERK2. Phase 2. |

|
|
| S1102 | U0126-EtOH | U0126-EtOH is a highly selective inhibitor of MEK1/2 with IC50 of 0.07 μM/0.06 μM in cell-free assays, 100-fold higher affinity for ΔN3-S218E/S222D MEK than PD98059. U0126 inhibits autophagy and mitophagy with antiviral activity. |

|
|
| S1008 | Selumetinib (AZD6244) | Selumetinib (AZD6244, ARRY-142886) is a potent, highly selective MEK inhibitor with IC50 of 14 nM for MEK1 and Kd value of 530 nM for MEK2. It also inhibits ERK1/2 phosphorylation with IC50 of 10 nM, no inhibition to p38α, MKK6, EGFR, ErbB2, ERK2, B-Raf, etc. Selumetinib suppresses cell proliferation, migration and trigger apoptosis. Phase 3. |

|
|
| S1177 | PD98059 | PD98059 is a non-ATP competitive MEK inhibitor with IC50 of 2 μM in a cell-free assay, specifically inhibits MEK-1-mediated activation of MAPK; does not directly inhibit ERK1 or ERK2. PD98059 is a ligand for the aryl hydrocarbon receptor (AHR) and functions as an AHR antagonist. |

|
|
| S1020 | PD184352 (CI-1040) | PD184352 (CI-1040) is an ATP non-competitive MEK1/2 inhibitor with IC50 of 17 nM in cell-based assays, 100-fold more selective for MEK1/2 than MEK5. PD184352 (CI-1040) selectively induces apoptosis. |

|
|
| S8041 | Cobimetinib (GDC-0973) | Cobimetinib (GDC-0973, RG7420) is a potent and highly selective MEK1 inhibitor with IC50 of 4.2 nM, and showing no significant inhibition when tested against a panel of more than 100 of serine-threonine and tyrosine kinases. Cobimetinib induces apoptosis. Phase 3. |

|
|
| S7007 | Binimetinib (MEK162) | Binimetinib (MEK162, ARRY-162, ARRY-438162) is a potent inhibitor of MEK1/2 with IC50 of 12 nM in a cell-free assay. Binimetinib induces G1 cell cycle arrest and apoptosis in human NSCLC cell lines and induces autophagy. Phase 3. |

|
|
| S1531 | BIX 02189 | BIX02189 is a selective inhibitor of MEK5 with IC50 of 1.5 nM, also inhibits ERK5 catalytic activity with IC50 of 59 nM in cell-free assays, and does not inhibit closely related kinases MEK1, MEK2, ERK2, and JNK2. |

|
|
| S1475 | Pimasertib (AS-703026) | Pimasertib (AS-703026, MSC1936369B, SAR 245509) is a highly selective, potent, ATP non-competitive allosteric inhibitor of MEK1/2 with IC50 of 5 nM-2 μM in MM cell lines. Phase 2. |

|
|
| S2310 | Honokiol | Honokiol is the active principle of magnolia extract that inhibits Akt-phosphorylation and promotes ERK1/2 phosphorylation. Honokiol causes G0/G1 phase arrest, induces apoptosis, and autophagy via the ROS/ERK1/2 signaling pathway. Honokiol inhibits hepatitis C virus (HCV) infection. Phase 3. |

|
|
| S1089 | Refametinib (RDEA119) | Refametinib (RDEA119, Bay 86-9766) is a potent, ATP non-competitive and highly selective inhibitor of MEK1 and MEK2 with IC50 of 19 nM and 47 nM, respectively. |

|
|
| S2617 | TAK-733 | TAK-733 is a potent and selective MEK allosteric site inhibitor for MEK1 with IC50 of 3.2 nM, inactive to Abl1, AKT3, c-RAF, CamK1, CDK2, c-Met, etc. Phase 1. |

|
|
| S2134 | AZD8330 | AZD8330 (ARRY704) is a novel, selective, non-ATP competitive MEK 1/2 inhibitor with IC50 of 7 nM. Phase 1. |

|
|
| S7170 | Avutometinib | Avutometinib(RO5126766,CH5126766,VS 6766, CKI-27, R-7304, RG-7304) is a dual RAF/MEK inhibitor with IC50 of 8.2 nM,19 nM, 56 nM, and 160 nM for BRAF V600E, BRAF, CRAF, and MEK1, respectively. Phase 1. | ||
| S1530 | BIX 02188 | BIX02188 is a selective inhibitor of MEK5 with IC50 of 4.3 nM, also inhibits ERK5 catalytic activity with IC50 of 810 nM, and does not inhibit closely related kinases MEK1, MEK2, ERK2, and JNK2. |

|
|
| S1392 | Pelitinib (EKB-569) | Pelitinib (EKB-569) is a potent irreversible EGFR inhibitor with IC50 of 38.5 nM. Pelitinib (EKB-569) also slightly inhibits Src, MEK/ERK and ErbB2 with IC50s of 282 nM, 800 nM and 1255 nM, respectively. Phase2. |

|
|
| S7553 | GDC-0623 | GDC-0623 (G-868) is a potent and ATP-uncompetitive MEK1 inhibitor with Ki of 0.13 nM. Phase 1. |

|
|
| S1066 | SL-327 | SL327 is a selective inhibitor for MEK1/2 with IC50 of 0.18 μM/ 0.22 μM, no activity towards Erk1, MKK3, MKK4, c-JUN, PKC, PKA, or CamKII;capable of transport through the blood-brain barrier. | ||
| S2326 | Myricetin | Myricetin, a natural flavonoid with antioxidant and anti tumor properties, is a novel inhibitor of MEK1 activity and transformation of JB6 P+ mouse epidermal cells. It also inhibits PI3Kγ with Kd of 0.17 μM. | ||
| S7843 | BI-847325 | BI-847325 is an orally bioavailable, and selective dual MEK/Aurora kinase inhibitor with IC50 of 3 nM, 25 nM, 15 nM, 25 nM, and 4 nM for Xenopus laevis Aurora B, human Aurora A and Aurora C, as well as human MEK1 and MEK2, respectively. Phase 1. | ||
| S1568 | PD318088 | PD318088 is a non-ATP competitive allosteric MEK1/2 inhibitor, binds simultaneously with ATP in a region of the MEK1 active site that is adjacent to the ATP-binding site. |

|
|
| S4484 | Trametinib DMSO solvate | Trametinib DMSO solvate is a highly specific and potent MEK1/2 inhibitor with IC50 of 0.92 nM/1.8 nM in cell-free assay. Trametinib activates autophagy and induces apoptosis. | ||
| S0739 | PD184161 | PD184161, an orally active MEK inhibitor with IC50 of 10-100 nM in a time- and concentration-dependent manner, inhibits cell proliferation and induces apoptosis, and produces depressive-like behavior. | ||
| E3106 | Dioscoreae Nipponicae Rhizoma Extract | Dioscoreae Nipponicae Rhizoma Extract is extracted from the rhizome of Dioscorea nipponica, of which the main component decreases the phosphorylation in IGF-1R, which in turn inhibits the phosphorylation and activation of PI3K-AKT and Rap1-MEK signaling pathways, promoting cell apoptosis and Graves’ disease remission. | ||
| E2821 | RGB-286638 free base | RGB-286638 free base is a Cyclin-dependent kinase (CDK) inhibitor that inhibits the kinase activity of cyclin T1-CDK9, cyclin B1-CDK1, cyclin E-CDK2, cyclin D1-CDK4, cyclin E-CDK3, and p35-CDK5 with IC50s of 1, 2, 3, 4, 5 and 5 nM, respectively; also inhibits GSK-3β, TAK1, Jak2 and MEK1, with IC50s of 3, 5, 50, and 54 nM. | ||
| S6641 | GW284543 (UNC10225170) | GW284543 (UNC10225170), a MEK5-selective inhibitor, inhibits MEK5 in a dose-dependently way as determined by reductions in pERK5, and decreases endogenous MYC protein. | ||
| S0372New | RO4987655 | RO4987655(RG 7167,CH4987655) is an allosteric MEK inhibitor with IC50 of 5 nM. RO4987655 exhibits a significant anti-proliferative and antitumor activity. | ||
| E0404 | Zapnometinib (PD0184264) | Zapnometinib (PD0184264, ATR-002), an major active metabolite of CI-1040, is a novel MEK inhibitor with an IC50 of 5.7 nM. | ||
| S8355 | APS-2-79 HCl | APS-2-79 is a MAPK antagonist that modulating KSR-dependent MAPK signalling by antagonizing RAF heterodimerization as well as the conformational changes required for phosphorylation and activation of KSR-bound MEK. IC50=120 ± 23 nM. |

|
|
| S0372New | RO4987655 | RO4987655(RG 7167,CH4987655) is an allosteric MEK inhibitor with IC50 of 5 nM. RO4987655 exhibits a significant anti-proliferative and antitumor activity. |
Choose Selective MEK Inhibitors
Tags: MEK inhibition | MEK pathway | MEK activation | MEK inhibitor cancer | MEK phosphorylation | MEK activity | MEK inhibitor clinical trial | MEK inhibitor side effects | MEK inhibitor drugs | MEK inhibitor review