
- Inhibitors
- By product type
- Natural Products
- Inducing Agents
- Peptides
- Antibiotics
- Antibody-drug Conjugates(ADC)
- PROTAC
- Hydrotropic Agents
- Dyes
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
By research - Antibodies
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- Featured Products
- MRTX1133
- Nab-Paclitaxel
- KP-457
- IAG933
- RMC-6236 (Daraxonrasib)
- RMC-7977
- Zoldonrasib (RMC-9805)
- GsMTx4
- Navitoclax (ABT-263)
- TSA (Trichostatin A)
- Y-27632 Dihydrochloride
- SB431542
- SB202190
- MK-2206 Dihydrochloride
- LY294002
- Alisertib (MLN8237)
- XAV-939
- CHIR-99021 (Laduviglusib)
- Bafilomycin A1 (Baf-A1)
- Thiazovivin (TZV)
- CP-673451
- Verteporfin
- DAPT
- Galunisertib (LY2157299)
- MG132
- SBE-β-CD
- Tween 80
- Bavdegalutamide (ARV-110)
- Z-VAD-FMK
- Wnt-C59 (C59)
- IWR-1-endo
- (+)-JQ1
- 3-Deazaneplanocin A (DZNep) Hydrochloride
- RepSox (E-616452)
- Erastin
- Q-VD-Oph
- Puromycin Dihydrochloride
- Cycloheximide
- Telaglenastat (CB-839)
- A-83-01
- Ceralasertib (AZD6738)
- Liproxstatin-1
- Emricasan (IDN-6556)
- PMA (Phorbol 12-myristate 13-acetate)
- Dibutyryl cAMP (Bucladesine) sodium
- Nedisertib (M3814)
- PLX5622
- IKE (Imidazole Ketone Erastin)
- STM2457
- Saruparib (AZD5305)
- New Products
- Contact Us
research use only
PD0325901 (Mirdametinib) MEK inhibitor
Cat.No.S1036
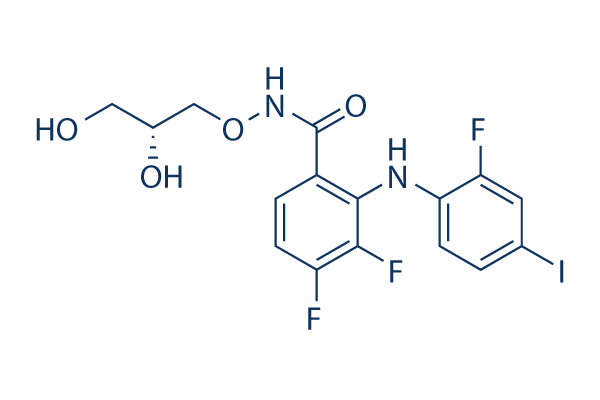
Chemical Structure
Molecular Weight: 482.19
Quality Control
Batch:
Purity:
99.99%
99.99
Products Often Used Together with PD0325901 (Mirdametinib)
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| A549 | Inhibition of MEK1 in human | 10 uM | 1 hr | Inhibition of MEK1 in human A549 cells assessed as reduction in pErk1/2 level at 10 uM after 1 hr by Western blotting analysis | 25766633 | |
| PANC1 | Inhibition of MEK1 in human | 10 uM | 1 hr | Inhibition of MEK1 in human PANC1 cells assessed as reduction in pErk1/2 level at 10 uM after 1 hr by Western blotting analysis | 25766633 | |
| A549 | Inhibition of MEk1/2 in human | 75 mins | Inhibition of MEk1/2 in human A549 cells assessed as decrease in ERK phosphorylation after 75 mins by Western blotting analysis | 23398453 | ||
| CCD-1070sk | Inhibition of MEk1/2 in human | 75 mins | Inhibition of MEk1/2 in human CCD-1070sk cells assessed as decrease in ERK phosphorylation after 75 mins by Western blotting analysis | 23398453 | ||
| A375 | Inhibition of MEk1/2 in human | 75 mins | Inhibition of MEk1/2 in human A375 cells assessed as decrease in ERK phosphorylation after 75 mins by Western blotting analysis | 23398453 | ||
| MCF7 | Inhibition of MEk1/2 in human | 75 mins | Inhibition of MEk1/2 in human MCF7 cells assessed as decrease in ERK phosphorylation after 75 mins by Western blotting analysis | 23398453 | ||
| NIH/3T3 | Inhibition of MEK1/MEK2 in mouse | 75 mins | Inhibition of MEK1/MEK2 in mouse NIH/3T3 cells assessed as decrease in ERK phosphorylation after 75 mins by Western blotting analysis | 23398453 | ||
| HT-29 | Inhibition of MEK in human | Inhibition of MEK in human HT-29 cells xenografted in BALB/c nu/nu mouse tumor assessed as phosphorylated ERK level at maximum tolerable dose administered as po | 21316218 | |||
| HT-29 | Inhibition of MEK in human | Inhibition of MEK in human HT-29 cells xenografted in BALB/c nu/nu mouse brain assessed as phosphorylated ERK level at maximum tolerable dose administered as po | 21316218 | |||
| HeLa | Concentration-dependent inhibition of ERK1, ERK2 phosphorylation in EGF-stimulated | Concentration-dependent inhibition of ERK1, ERK2 phosphorylation in EGF-stimulated HeLa cells | 17850214 | |||
| HeLa | Concentration-dependent inhibition of ERK5 phosphorylation shift in EGF-stimulated | Concentration-dependent inhibition of ERK5 phosphorylation shift in EGF-stimulated HeLa cells | 17850214 | |||
| rhabdomyosarcoma | Induction of MEK1/2 phosphorylation in human | 0.4 to 10 uM | 1 hr | Induction of MEK1/2 phosphorylation in human rhabdomyosarcoma cells infected with EV71 BC08 GU475127 at 0.4 to 10 uM pre-treated for 1 hr followed by viral infection measured after 24 hrs by western blotting analysis | 27288186 | |
| rhabdomyosarcoma | Inhibition of MEK1 phosphorylation in human | 0.4 to 10 uM | 1 hr | Inhibition of MEK1 phosphorylation in human rhabdomyosarcoma cells infected with EV71 BC08 GU475127 assessed as inhibition of ERK1/2 phosphorylation at 0.4 to 10 uM pre-treated for 1 hr followed by viral infection measured after 24 hrs by western blotting | 27288186 | |
| D54 | Cytotoxic activity against human | 48 hrs | Cytotoxic activity against human D54 cells assessed as reduction in cell viability incubated for 48 hrs in presence of ZSTK474 by alamar blue assay | 26943489 | ||
| colon 26 | Inhibition of MEK1 in mouse | 1 hr | Inhibition of MEK1 in mouse colon 26 cells assessed as reduction in ERK1/2 phosphorylation after 1 hr by Western blot analysis, IC50 = 0.00033 μM. | 28835793 | ||
| Sf9 | Inhibition of recombinant human | 30 mins | Inhibition of recombinant human GST-tagged MEK1 expressed in baculovirus infected Sf9 insect cells using inactive ERK2 as substrate preincubated for 30 mins followed by substrate addition measured after 2 hrs by kinase-glo reagent based bioluminescence as, IC50 = 0.015 μM. | 28835793 | ||
| A431 | Inhibition of MEK/ERK signaling pathway in EGF-stimulated human | 60 mins | Inhibition of MEK/ERK signaling pathway in EGF-stimulated human A431 cells transfected with COT siRNA assessed as reduction in ERK1/2 phosphorylation incubated for 60 mins followed by stimulation with EGF for 10 mins by Western blot analysis, IC50 = 0.038 μM. | 27502541 | ||
| A431 | Inhibition of MEK/ERK signaling pathway in TNFalpha-stimulated human | 60 mins | Inhibition of MEK/ERK signaling pathway in TNFalpha-stimulated human A431 cells transfected with COT siRNA assessed as reduction in ERK1/2 phosphorylation incubated for 60 mins followed by stimulation with TNFalpha for 10 mins by Western blot analysis, IC50 = 0.014 μM. | 27502541 | ||
| MCF7 | Antiproliferative activity against human | Antiproliferative activity against human MCF7 cells, IC50 = 9.55 μM. | 26611920 | |||
| HCT116 | Antiproliferative activity against human | Antiproliferative activity against human HCT116 cells harboring K-Ras G13D mutation, IC50 = 0.185 μM. | 26611920 | |||
| A549 | Antiproliferative activity against human | Antiproliferative activity against human A549 cells harboring K-Ras mutation, IC50 = 0.166 μM. | 26611920 | |||
| HepG2 | Antiproliferative activity against human | Antiproliferative activity against human HepG2 cells harboring N-Ras mutation, IC50 = 0.005 μM. | 26611920 | |||
| COLO205 | Antiproliferative activity against human | Antiproliferative activity against human COLO205 cells harboring BRAF mutation, IC50 = 0.005 μM. | 26611920 | |||
| C26 | Inhibition of MEK-stimulated ERK phosphorylation in PDGF-stimulated mouse | Inhibition of MEK-stimulated ERK phosphorylation in PDGF-stimulated mouse C26 cells treated as single oral dose before PDGF challenge by whole cell assay, IC50 = 0.00033 μM. | 18952427 | |||
| A549 | Antitumor activity against human | 3 mg/kg | Antitumor activity against human A549 cells xenografted in nude mouse assessed as tumor growth inhibition at 3 mg/kg, po qd | 23474388 | ||
| A549 | Antiproliferative activity against human | Antiproliferative activity against human A549 cells, IC50 = 0.166 μM. | 23474388 | |||
| HCT116 | Antiproliferative activity against human | Antiproliferative activity against human HCT116 cells expressing KRAS G13D mutant, IC50 = 0.194 μM. | 23474388 | |||
| LoVo | Antiproliferative activity against human | Antiproliferative activity against human LoVo cells, IC50 = 0.128 μM. | 23474388 | |||
| MIAPaCa2 | Proliferation assay | Antiproliferative activity against human MIAPaCa2 cells, IC50=17 nM. | 23474388 | |||
| A375 | Proliferation assay | 72 h | Antiproliferative activity against human A375 cells expressing BRAF V600E mutant after 72 hrs by Cell titer-glo assay, IC50=13 nM. | 23474388 | ||
| CHP-212 | Growth inhibition assay | Inhibition of human CHP-212 cell growth in a cell viability assay, IC50 = 0.0005273 μM. | SANGER | |||
| M14 | Growth inhibition assay | Inhibition of human M14 cell growth in a cell viability assay, IC50 = 0.001448 μM. | SANGER | |||
| SK-MEL-28 | Growth inhibition assay | Inhibition of human SK-MEL-28 cell growth in a cell viability assay, IC50 = 0.001519 μM. | SANGER | |||
| NOMO-1 | Growth inhibition assay | Inhibition of human NOMO-1 cell growth in a cell viability assay, IC50 = 0.002072 μM. | SANGER | |||
| A375 | Growth inhibition assay | Inhibition of human A375 cell growth in a cell viability assay, IC50 = 0.002687 μM. | SANGER | |||
| DU-4475 | Growth inhibition assay | Inhibition of human DU-4475 cell growth in a cell viability assay, IC50 = 0.003566 μM. | SANGER | |||
| C32 | Growth inhibition assay | Inhibition of human C32 cell growth in a cell viability assay, IC50 = 0.003668 μM. | SANGER | |||
| BPH-1 | Growth inhibition assay | Inhibition of human BPH-1 cell growth in a cell viability assay, IC50 = 0.00395 μM. | SANGER | |||
| CP50-MEL-B | Growth inhibition assay | Inhibition of human CP50-MEL-B cell growth in a cell viability assay, IC50 = 0.004575 μM. | SANGER | |||
| H9 | Growth inhibition assay | Inhibition of human H9 cell growth in a cell viability assay, IC50 = 0.005768 μM. | SANGER | |||
| HTC-C3 | Growth inhibition assay | Inhibition of human HTC-C3 cell growth in a cell viability assay, IC50 = 0.005894 μM. | SANGER | |||
| BHT-101 | Growth inhibition assay | Inhibition of human BHT-101 cell growth in a cell viability assay, IC50 = 0.006704 μM. | SANGER | |||
| COLO-741 | Growth inhibition assay | Inhibition of human COLO-741 cell growth in a cell viability assay, IC50 = 0.0071 μM. | SANGER | |||
| OVCAR-5 | Growth inhibition assay | Inhibition of human OVCAR-5 cell growth in a cell viability assay, IC50 = 0.007823 μM. | SANGER | |||
| A549 | Growth inhibition assay | Inhibition of human A549 cell growth in a cell viability assay, IC50 = 0.007893 μM. | SANGER | |||
| SH-4 | Growth inhibition assay | Inhibition of human SH-4 cell growth in a cell viability assay, IC50 = 0.00826 μM. | SANGER | |||
| SK-N-AS | Growth inhibition assay | Inhibition of human SK-N-AS cell growth in a cell viability assay, IC50 = 0.008438 μM. | SANGER | |||
| HT-144 | Growth inhibition assay | Inhibition of human HT-144 cell growth in a cell viability assay, IC50 = 0.008974 μM. | SANGER | |||
| MEL-HO | Growth inhibition assay | Inhibition of human MEL-HO cell growth in a cell viability assay, IC50 = 0.009492 μM. | SANGER | |||
| COLO-679 | Growth inhibition assay | Inhibition of human COLO-679 cell growth in a cell viability assay, IC50 = 0.01001 μM. | SANGER | |||
| HuP-T4 | Growth inhibition assay | Inhibition of human HuP-T4 cell growth in a cell viability assay, IC50 = 0.01098 μM. | SANGER | |||
| H-EMC-SS | Growth inhibition assay | Inhibition of human H-EMC-SS cell growth in a cell viability assay, IC50 = 0.01102 μM. | SANGER | |||
| LB2518-MEL | Growth inhibition assay | Inhibition of human LB2518-MEL cell growth in a cell viability assay, IC50 = 0.01113 μM. | SANGER | |||
| HL-60 | Growth inhibition assay | Inhibition of human HL-60 cell growth in a cell viability assay, IC50 = 0.01115 μM. | SANGER | |||
| NCI-H1666 | Growth inhibition assay | Inhibition of human NCI-H1666 cell growth in a cell viability assay, IC50 = 0.01117 μM. | SANGER | |||
| A101D | Growth inhibition assay | Inhibition of human A101D cell growth in a cell viability assay, IC50 = 0.01145 μM. | SANGER | |||
| RVH-421 | Growth inhibition assay | Inhibition of human RVH-421 cell growth in a cell viability assay, IC50 = 0.01264 μM. | SANGER | |||
| Hs-578-T | Growth inhibition assay | Inhibition of human Hs-578-T cell growth in a cell viability assay, IC50 = 0.01279 μM. | SANGER | |||
| DOK | Growth inhibition assay | Inhibition of human DOK cell growth in a cell viability assay, IC50 = 0.0134 μM. | SANGER | |||
| Mewo | Growth inhibition assay | Inhibition of human Mewo cell growth in a cell viability assay, IC50 = 0.01445 μM. | SANGER | |||
| ONS-76 | Growth inhibition assay | Inhibition of human ONS-76 cell growth in a cell viability assay, IC50 = 0.01451 μM. | SANGER | |||
| UACC-257 | Growth inhibition assay | Inhibition of human UACC-257 cell growth in a cell viability assay, IC50 = 0.01462 μM. | SANGER | |||
| SW626 | Growth inhibition assay | Inhibition of human SW626 cell growth in a cell viability assay, IC50 = 0.01491 μM. | SANGER | |||
| SW620 | Growth inhibition assay | Inhibition of human SW620 cell growth in a cell viability assay, IC50 = 0.01495 μM. | SANGER | |||
| TYK-nu | Growth inhibition assay | Inhibition of human TYK-nu cell growth in a cell viability assay, IC50 = 0.01513 μM. | SANGER | |||
| ACN | Growth inhibition assay | Inhibition of human ACN cell growth in a cell viability assay, IC50 = 0.01576 μM. | SANGER | |||
| T-24 | Growth inhibition assay | Inhibition of human T-24 cell growth in a cell viability assay, IC50 = 0.01971 μM. | SANGER | |||
| AGS | Growth inhibition assay | Inhibition of human AGS cell growth in a cell viability assay, IC50 = 0.02041 μM. | SANGER | |||
| SW872 | Growth inhibition assay | Inhibition of human SW872 cell growth in a cell viability assay, IC50 = 0.02799 μM. | SANGER | |||
| C2BBe1 | Growth inhibition assay | Inhibition of human C2BBe1 cell growth in a cell viability assay, IC50 = 0.02854 μM. | SANGER | |||
| MZ7-mel | Growth inhibition assay | Inhibition of human MZ7-mel cell growth in a cell viability assay, IC50 = 0.02943 μM. | SANGER | |||
| HCC2998 | Growth inhibition assay | Inhibition of human HCC2998 cell growth in a cell viability assay, IC50 = 0.03366 μM. | SANGER | |||
| HO-1-N-1 | Growth inhibition assay | Inhibition of human HO-1-N-1 cell growth in a cell viability assay, IC50 = 0.03443 μM. | SANGER | |||
| SW756 | Growth inhibition assay | Inhibition of human SW756 cell growth in a cell viability assay, IC50 = 0.03445 μM. | SANGER | |||
| NCI-H1437 | Growth inhibition assay | Inhibition of human NCI-H1437 cell growth in a cell viability assay, IC50 = 0.03449 μM. | SANGER | |||
| NCI-H747 | Growth inhibition assay | Inhibition of human NCI-H747 cell growth in a cell viability assay, IC50 = 0.03498 μM. | SANGER | |||
| SK-MEL-2 | Growth inhibition assay | Inhibition of human SK-MEL-2 cell growth in a cell viability assay, IC50 = 0.0352 μM. | SANGER | |||
| MZ2-MEL | Growth inhibition assay | Inhibition of human MZ2-MEL cell growth in a cell viability assay, IC50 = 0.03565 μM. | SANGER | |||
| PSN1 | Growth inhibition assay | Inhibition of human PSN1 cell growth in a cell viability assay, IC50 = 0.03845 μM. | SANGER | |||
| CAL-39 | Growth inhibition assay | Inhibition of human CAL-39 cell growth in a cell viability assay, IC50 = 0.03904 μM. | SANGER | |||
| LOXIMVI | Growth inhibition assay | Inhibition of human LOXIMVI cell growth in a cell viability assay, IC50 = 0.03939 μM. | SANGER | |||
| COLO-792 | Growth inhibition assay | Inhibition of human COLO-792 cell growth in a cell viability assay, IC50 = 0.04315 μM. | SANGER | |||
| CAL-27 | Growth inhibition assay | Inhibition of human CAL-27 cell growth in a cell viability assay, IC50 = 0.04491 μM. | SANGER | |||
| AsPC-1 | Growth inhibition assay | Inhibition of human AsPC-1 cell growth in a cell viability assay, IC50 = 0.04528 μM. | SANGER | |||
| NCI-H2291 | Growth inhibition assay | Inhibition of human NCI-H2291 cell growth in a cell viability assay, IC50 = 0.04646 μM. | SANGER | |||
| RCM-1 | Growth inhibition assay | Inhibition of human RCM-1 cell growth in a cell viability assay, IC50 = 0.04685 μM. | SANGER | |||
| NCI-H292 | Growth inhibition assay | Inhibition of human NCI-H292 cell growth in a cell viability assay, IC50 = 0.04739 μM. | SANGER | |||
| WM-115 | Growth inhibition assay | Inhibition of human WM-115 cell growth in a cell viability assay, IC50 = 0.0485 μM. | SANGER | |||
| RT-112 | Growth inhibition assay | Inhibition of human RT-112 cell growth in a cell viability assay, IC50 = 0.04882 μM. | SANGER | |||
| HT-29 | Growth inhibition assay | Inhibition of human HT-29 cell growth in a cell viability assay, IC50 = 0.05049 μM. | SANGER | |||
| RKO | Growth inhibition assay | Inhibition of human RKO cell growth in a cell viability assay, IC50 = 0.05202 μM. | SANGER | |||
| KY821 | Growth inhibition assay | Inhibition of human KY821 cell growth in a cell viability assay, IC50 = 0.0533 μM. | SANGER | |||
| LB1047-RCC | Growth inhibition assay | Inhibition of human LB1047-RCC cell growth in a cell viability assay, IC50 = 0.05965 μM. | SANGER | |||
| SW1116 | Growth inhibition assay | Inhibition of human SW1116 cell growth in a cell viability assay, IC50 = 0.06049 μM. | SANGER | |||
| P12-ICHIKAWA | Growth inhibition assay | Inhibition of human P12-ICHIKAWA cell growth in a cell viability assay, IC50 = 0.06227 μM. | SANGER | |||
| HCC70 | Growth inhibition assay | Inhibition of human HCC70 cell growth in a cell viability assay, IC50 = 0.06301 μM. | SANGER | |||
| MIA-PaCa-2 | Growth inhibition assay | Inhibition of human MIA-PaCa-2 cell growth in a cell viability assay, IC50 = 0.06353 μM. | SANGER | |||
| LoVo | Growth inhibition assay | Inhibition of human LoVo cell growth in a cell viability assay, IC50 = 0.06529 μM. | SANGER | |||
| LB2241-RCC | Growth inhibition assay | Inhibition of human LB2241-RCC cell growth in a cell viability assay, IC50 = 0.06552 μM. | SANGER | |||
| GAK | Growth inhibition assay | Inhibition of human GAK cell growth in a cell viability assay, IC50 = 0.06687 μM. | SANGER | |||
| RD | Growth inhibition assay | Inhibition of human RD cell growth in a cell viability assay, IC50 = 0.0671 μM. | SANGER | |||
| KNS-62 | Growth inhibition assay | Inhibition of human KNS-62 cell growth in a cell viability assay, IC50 = 0.06999 μM. | SANGER | |||
| HD-MY-Z | Growth inhibition assay | Inhibition of human HD-MY-Z cell growth in a cell viability assay, IC50 = 0.07112 μM. | SANGER | |||
| COR-L105 | Growth inhibition assay | Inhibition of human COR-L105 cell growth in a cell viability assay, IC50 = 0.07171 μM. | SANGER | |||
| IA-LM | Growth inhibition assay | Inhibition of human IA-LM cell growth in a cell viability assay, IC50 = 0.07328 μM. | SANGER | |||
| EM-2 | Growth inhibition assay | Inhibition of human EM-2 cell growth in a cell viability assay, IC50 = 0.0747 μM. | SANGER | |||
| NB69 | Growth inhibition assay | Inhibition of human NB69 cell growth in a cell viability assay, IC50 = 0.078 μM. | SANGER | |||
| HuP-T3 | Growth inhibition assay | Inhibition of human HuP-T3 cell growth in a cell viability assay, IC50 = 0.08393 μM. | SANGER | |||
| BB30-HNC | Growth inhibition assay | Inhibition of human BB30-HNC cell growth in a cell viability assay, IC50 = 0.08551 μM. | SANGER | |||
| HT-1080 | Growth inhibition assay | Inhibition of human HT-1080 cell growth in a cell viability assay, IC50 = 0.08588 μM. | SANGER | |||
| RMG-I | Growth inhibition assay | Inhibition of human RMG-I cell growth in a cell viability assay, IC50 = 0.08734 μM. | SANGER | |||
| HCC1419 | Growth inhibition assay | Inhibition of human HCC1419 cell growth in a cell viability assay, IC50 = 0.09138 μM. | SANGER | |||
| SW780 | Growth inhibition assay | Inhibition of human SW780 cell growth in a cell viability assay, IC50 = 0.09232 μM. | SANGER | |||
| SNU-387 | Growth inhibition assay | Inhibition of human SNU-387 cell growth in a cell viability assay, IC50 = 0.09336 μM. | SANGER | |||
| LAMA-84 | Growth inhibition assay | Inhibition of human LAMA-84 cell growth in a cell viability assay, IC50 = 0.09468 μM. | SANGER | |||
| MV-4-11 | Growth inhibition assay | Inhibition of human MV-4-11 cell growth in a cell viability assay, IC50 = 0.09473 μM. | SANGER | |||
| EGI-1 | Growth inhibition assay | Inhibition of human EGI-1 cell growth in a cell viability assay, IC50 = 0.09581 μM. | SANGER | |||
| NCI-SNU-1 | Growth inhibition assay | Inhibition of human NCI-SNU-1 cell growth in a cell viability assay, IC50 = 0.09673 μM. | SANGER | |||
| MEG-01 | Growth inhibition assay | Inhibition of human MEG-01 cell growth in a cell viability assay, IC50 = 0.09777 μM. | SANGER | |||
| OMC-1 | Growth inhibition assay | Inhibition of human OMC-1 cell growth in a cell viability assay, IC50 = 0.10023 μM. | SANGER | |||
| NB10 | Growth inhibition assay | Inhibition of human NB10 cell growth in a cell viability assay, IC50 = 0.10042 μM. | SANGER | |||
| CAL-62 | Growth inhibition assay | Inhibition of human CAL-62 cell growth in a cell viability assay, IC50 = 0.10078 μM. | SANGER | |||
| NCI-H2087 | Growth inhibition assay | Inhibition of human NCI-H2087 cell growth in a cell viability assay, IC50 = 0.10114 μM. | SANGER | |||
| MDA-MB-175-VII | Growth inhibition assay | Inhibition of human MDA-MB-175-VII cell growth in a cell viability assay, IC50 = 0.10926 μM. | SANGER | |||
| LS-513 | Growth inhibition assay | Inhibition of human LS-513 cell growth in a cell viability assay, IC50 = 0.11483 μM. | SANGER | |||
| HN | Growth inhibition assay | Inhibition of human HN cell growth in a cell viability assay, IC50 = 0.12259 μM. | SANGER | |||
| ABC-1 | Growth inhibition assay | Inhibition of human ABC-1 cell growth in a cell viability assay, IC50 = 0.12302 μM. | SANGER | |||
| SJSA-1 | Growth inhibition assay | Inhibition of human SJSA-1 cell growth in a cell viability assay, IC50 = 0.12319 μM. | SANGER | |||
| LAN-6 | Growth inhibition assay | Inhibition of human LAN-6 cell growth in a cell viability assay, IC50 = 0.12324 μM. | SANGER | |||
| LCLC-97TM1 | Growth inhibition assay | Inhibition of human LCLC-97TM1 cell growth in a cell viability assay, IC50 = 0.12416 μM. | SANGER | |||
| NCI-H1355 | Growth inhibition assay | Inhibition of human NCI-H1355 cell growth in a cell viability assay, IC50 = 0.12438 μM. | SANGER | |||
| HPAF-II | Growth inhibition assay | Inhibition of human HPAF-II cell growth in a cell viability assay, IC50 = 0.12508 μM. | SANGER | |||
| BFTC-905 | Growth inhibition assay | Inhibition of human BFTC-905 cell growth in a cell viability assay, IC50 = 0.12916 μM. | SANGER | |||
| HSC-3 | Growth inhibition assay | Inhibition of human HSC-3 cell growth in a cell viability assay, IC50 = 0.13461 μM. | SANGER | |||
| BCPAP | Growth inhibition assay | Inhibition of human BCPAP cell growth in a cell viability assay, IC50 = 0.13525 μM. | SANGER | |||
| OS-RC-2 | Growth inhibition assay | Inhibition of human OS-RC-2 cell growth in a cell viability assay, IC50 = 0.13664 μM. | SANGER | |||
| NCI-H460 | Growth inhibition assay | Inhibition of human NCI-H460 cell growth in a cell viability assay, IC50 = 0.14466 μM. | SANGER | |||
| ML-2 | Growth inhibition assay | Inhibition of human ML-2 cell growth in a cell viability assay, IC50 = 0.14612 μM. | SANGER | |||
| D-263MG | Growth inhibition assay | Inhibition of human D-263MG cell growth in a cell viability assay, IC50 = 0.15625 μM. | SANGER | |||
| EoL-1-cell | Growth inhibition assay | Inhibition of human EoL-1-cell cell growth in a cell viability assay, IC50 = 0.15662 μM. | SANGER | |||
| CAL-33 | Growth inhibition assay | Inhibition of human CAL-33 cell growth in a cell viability assay, IC50 = 0.15725 μM. | SANGER | |||
| MDA-MB-231 | Growth inhibition assay | Inhibition of human MDA-MB-231 cell growth in a cell viability assay, IC50 = 0.16152 μM. | SANGER | |||
| HT55 | Growth inhibition assay | Inhibition of human HT55 cell growth in a cell viability assay, IC50 = 0.16725 μM. | SANGER | |||
| COLO-678 | Growth inhibition assay | Inhibition of human COLO-678 cell growth in a cell viability assay, IC50 = 0.17331 μM. | SANGER | |||
| DK-MG | Growth inhibition assay | Inhibition of human DK-MG cell growth in a cell viability assay, IC50 = 0.17976 μM. | SANGER | |||
| EFO-27 | Growth inhibition assay | Inhibition of human EFO-27 cell growth in a cell viability assay, IC50 = 0.18084 μM. | SANGER | |||
| CHL-1 | Growth inhibition assay | Inhibition of human CHL-1 cell growth in a cell viability assay, IC50 = 0.18151 μM. | SANGER | |||
| SK-HEP-1 | Growth inhibition assay | Inhibition of human SK-HEP-1 cell growth in a cell viability assay, IC50 = 0.18198 μM. | SANGER | |||
| NB5 | Growth inhibition assay | Inhibition of human NB5 cell growth in a cell viability assay, IC50 = 0.18486 μM. | SANGER | |||
| NCI-H1299 | Growth inhibition assay | Inhibition of human NCI-H1299 cell growth in a cell viability assay, IC50 = 0.18606 μM. | SANGER | |||
| SKG-IIIa | Growth inhibition assay | Inhibition of human SKG-IIIa cell growth in a cell viability assay, IC50 = 0.18838 μM. | SANGER | |||
| Ca9-22 | Growth inhibition assay | Inhibition of human Ca9-22 cell growth in a cell viability assay, IC50 = 0.19035 μM. | SANGER | |||
| LS-1034 | Growth inhibition assay | Inhibition of human LS-1034 cell growth in a cell viability assay, IC50 = 0.19276 μM. | SANGER | |||
| 8305C | Growth inhibition assay | Inhibition of human 8305C cell growth in a cell viability assay, IC50 = 0.19623 μM. | SANGER | |||
| COLO-829 | Growth inhibition assay | Inhibition of human COLO-829 cell growth in a cell viability assay, IC50 = 0.19677 μM. | SANGER | |||
| GP5d | Growth inhibition assay | Inhibition of human GP5d cell growth in a cell viability assay, IC50 = 0.19981 μM. | SANGER | |||
| NKM-1 | Growth inhibition assay | Inhibition of human NKM-1 cell growth in a cell viability assay, IC50 = 0.19989 μM. | SANGER | |||
| PANC-03-27 | Growth inhibition assay | Inhibition of human PANC-03-27 cell growth in a cell viability assay, IC50 = 0.20085 μM. | SANGER | |||
| KU812 | Growth inhibition assay | Inhibition of human KU812 cell growth in a cell viability assay, IC50 = 0.20296 μM. | SANGER | |||
| KM12 | Growth inhibition assay | Inhibition of human KM12 cell growth in a cell viability assay, IC50 = 0.20431 μM. | SANGER | |||
| KYSE-450 | Growth inhibition assay | Inhibition of human KYSE-450 cell growth in a cell viability assay, IC50 = 0.2089 μM. | SANGER | |||
| NCI-H23 | Growth inhibition assay | Inhibition of human NCI-H23 cell growth in a cell viability assay, IC50 = 0.216 μM. | SANGER | |||
| NCI-H1563 | Growth inhibition assay | Inhibition of human NCI-H1563 cell growth in a cell viability assay, IC50 = 0.21961 μM. | SANGER | |||
| A498 | Growth inhibition assay | Inhibition of human A498 cell growth in a cell viability assay, IC50 = 0.22186 μM. | SANGER | |||
| 8505C | Growth inhibition assay | Inhibition of human 8505C cell growth in a cell viability assay, IC50 = 0.22439 μM. | SANGER | |||
| SCC-25 | Growth inhibition assay | Inhibition of human SCC-25 cell growth in a cell viability assay, IC50 = 0.23219 μM. | SANGER | |||
| KYSE-520 | Growth inhibition assay | Inhibition of human KYSE-520 cell growth in a cell viability assay, IC50 = 0.23635 μM. | SANGER | |||
| U251 | Growth inhibition assay | Inhibition of human U251 cell growth in a cell viability assay, IC50 = 0.23846 μM. | SANGER | |||
| D-502MG | Growth inhibition assay | Inhibition of human D-502MG cell growth in a cell viability assay, IC50 = 0.24174 μM. | SANGER | |||
| CAL-85-1 | Growth inhibition assay | Inhibition of human CAL-85-1 cell growth in a cell viability assay, IC50 = 0.24705 μM. | SANGER | |||
| RXF393 | Growth inhibition assay | Inhibition of human RXF393 cell growth in a cell viability assay, IC50 = 0.25469 μM. | SANGER | |||
| TE-8 | Growth inhibition assay | Inhibition of human TE-8 cell growth in a cell viability assay, IC50 = 0.25575 μM. | SANGER | |||
| NCI-H2347 | Growth inhibition assay | Inhibition of human NCI-H2347 cell growth in a cell viability assay, IC50 = 0.25746 μM. | SANGER | |||
| HuCCT1 | Growth inhibition assay | Inhibition of human HuCCT1 cell growth in a cell viability assay, IC50 = 0.26168 μM. | SANGER | |||
| SW1463 | Growth inhibition assay | Inhibition of human SW1463 cell growth in a cell viability assay, IC50 = 0.26181 μM. | SANGER | |||
| KU-19-19 | Growth inhibition assay | Inhibition of human KU-19-19 cell growth in a cell viability assay, IC50 = 0.26516 μM. | SANGER | |||
| Capan-2 | Growth inhibition assay | Inhibition of human Capan-2 cell growth in a cell viability assay, IC50 = 0.27126 μM. | SANGER | |||
| KYSE-510 | Growth inhibition assay | Inhibition of human KYSE-510 cell growth in a cell viability assay, IC50 = 0.27505 μM. | SANGER | |||
| JVM-3 | Growth inhibition assay | Inhibition of human JVM-3 cell growth in a cell viability assay, IC50 = 0.28592 μM. | SANGER | |||
| SK-LU-1 | Growth inhibition assay | Inhibition of human SK-LU-1 cell growth in a cell viability assay, IC50 = 0.28757 μM. | SANGER | |||
| HCT-116 | Growth inhibition assay | Inhibition of human HCT-116 cell growth in a cell viability assay, IC50 = 0.29071 μM. | SANGER | |||
| BHY | Growth inhibition assay | Inhibition of human BHY cell growth in a cell viability assay, IC50 = 0.29713 μM. | SANGER | |||
| SK-N-FI | Growth inhibition assay | Inhibition of human SK-N-FI cell growth in a cell viability assay, IC50 = 0.30401 μM. | SANGER | |||
| RPMI-7951 | Growth inhibition assay | Inhibition of human RPMI-7951 cell growth in a cell viability assay, IC50 = 0.30987 μM. | SANGER | |||
| NCI-H358 | Growth inhibition assay | Inhibition of human NCI-H358 cell growth in a cell viability assay, IC50 = 0.31197 μM. | SANGER | |||
| SK-MES-1 | Growth inhibition assay | Inhibition of human SK-MES-1 cell growth in a cell viability assay, IC50 = 0.32084 μM. | SANGER | |||
| RPMI-8226 | Growth inhibition assay | Inhibition of human RPMI-8226 cell growth in a cell viability assay, IC50 = 0.32811 μM. | SANGER | |||
| TGBC11TKB | Growth inhibition assay | Inhibition of human TGBC11TKB cell growth in a cell viability assay, IC50 = 0.32969 μM. | SANGER | |||
| MG-63 | Growth inhibition assay | Inhibition of human MG-63 cell growth in a cell viability assay, IC50 = 0.33049 μM. | SANGER | |||
| LS-411N | Growth inhibition assay | Inhibition of human LS-411N cell growth in a cell viability assay, IC50 = 0.33898 μM. | SANGER | |||
| IGR-1 | Growth inhibition assay | Inhibition of human IGR-1 cell growth in a cell viability assay, IC50 = 0.34507 μM. | SANGER | |||
| NUGC-3 | Growth inhibition assay | Inhibition of human NUGC-3 cell growth in a cell viability assay, IC50 = 0.35165 μM. | SANGER | |||
| TGW | Growth inhibition assay | Inhibition of human TGW cell growth in a cell viability assay, IC50 = 0.35413 μM. | SANGER | |||
| EPLC-272H | Growth inhibition assay | Inhibition of human EPLC-272H cell growth in a cell viability assay, IC50 = 0.35599 μM. | SANGER | |||
| KS-1 | Growth inhibition assay | Inhibition of human KS-1 cell growth in a cell viability assay, IC50 = 0.36742 μM. | SANGER | |||
| MZ1-PC | Growth inhibition assay | Inhibition of human MZ1-PC cell growth in a cell viability assay, IC50 = 0.36881 μM. | SANGER | |||
| HCE-T | Growth inhibition assay | Inhibition of human HCE-T cell growth in a cell viability assay, IC50 = 0.37029 μM. | SANGER | |||
| BxPC-3 | Growth inhibition assay | Inhibition of human BxPC-3 cell growth in a cell viability assay, IC50 = 0.39116 μM. | SANGER | |||
| SN12C | Growth inhibition assay | Inhibition of human SN12C cell growth in a cell viability assay, IC50 = 0.39248 μM. | SANGER | |||
| SW948 | Growth inhibition assay | Inhibition of human SW948 cell growth in a cell viability assay, IC50 = 0.39388 μM. | SANGER | |||
| KOSC-2 | Growth inhibition assay | Inhibition of human KOSC-2 cell growth in a cell viability assay, IC50 = 0.4001 μM. | SANGER | |||
| 786-0 | Growth inhibition assay | Inhibition of human 786-0 cell growth in a cell viability assay, IC50 = 0.4174 μM. | SANGER | |||
| KG-1 | Growth inhibition assay | Inhibition of human KG-1 cell growth in a cell viability assay, IC50 = 0.41767 μM. | SANGER | |||
| QIMR-WIL | Growth inhibition assay | Inhibition of human QIMR-WIL cell growth in a cell viability assay, IC50 = 0.43049 μM. | SANGER | |||
| SCC-4 | Growth inhibition assay | Inhibition of human SCC-4 cell growth in a cell viability assay, IC50 = 0.43806 μM. | SANGER | |||
| BB65-RCC | Growth inhibition assay | Inhibition of human BB65-RCC cell growth in a cell viability assay, IC50 = 0.44466 μM. | SANGER | |||
| VM-CUB-1 | Growth inhibition assay | Inhibition of human VM-CUB-1 cell growth in a cell viability assay, IC50 = 0.45126 μM. | SANGER | |||
| CFPAC-1 | Growth inhibition assay | Inhibition of human CFPAC-1 cell growth in a cell viability assay, IC50 = 0.45303 μM. | SANGER | |||
| SW1417 | Growth inhibition assay | Inhibition of human SW1417 cell growth in a cell viability assay, IC50 = 0.45409 μM. | SANGER | |||
| HAL-01 | Growth inhibition assay | Inhibition of human HAL-01 cell growth in a cell viability assay, IC50 = 0.45412 μM. | SANGER | |||
| GI-ME-N | Growth inhibition assay | Inhibition of human GI-ME-N cell growth in a cell viability assay, IC50 = 0.45431 μM. | SANGER | |||
| RERF-LC-MS | Growth inhibition assay | Inhibition of human RERF-LC-MS cell growth in a cell viability assay, IC50 = 0.45432 μM. | SANGER | |||
| 647-V | Growth inhibition assay | Inhibition of human 647-V cell growth in a cell viability assay, IC50 = 0.45541 μM. | SANGER | |||
| YKG-1 | Growth inhibition assay | Inhibition of human YKG-1 cell growth in a cell viability assay, IC50 = 0.45662 μM. | SANGER | |||
| SW954 | Growth inhibition assay | Inhibition of human SW954 cell growth in a cell viability assay, IC50 = 0.45696 μM. | SANGER | |||
| A2058 | Growth inhibition assay | Inhibition of human A2058 cell growth in a cell viability assay, IC50 = 0.46197 μM. | SANGER | |||
| NB7 | Growth inhibition assay | Inhibition of human NB7 cell growth in a cell viability assay, IC50 = 0.47746 μM. | SANGER | |||
| GCIY | Growth inhibition assay | Inhibition of human GCIY cell growth in a cell viability assay, IC50 = 0.48453 μM. | SANGER | |||
| SNU-C2B | Growth inhibition assay | Inhibition of human SNU-C2B cell growth in a cell viability assay, IC50 = 0.49237 μM. | SANGER | |||
| GAMG | Growth inhibition assay | Inhibition of human GAMG cell growth in a cell viability assay, IC50 = 0.49757 μM. | SANGER | |||
| GMS-10 | Growth inhibition assay | Inhibition of human GMS-10 cell growth in a cell viability assay, IC50 = 0.49872 μM. | SANGER | |||
| A3-KAW | Growth inhibition assay | Inhibition of human A3-KAW cell growth in a cell viability assay, IC50 = 0.50127 μM. | SANGER | |||
| HT-3 | Growth inhibition assay | Inhibition of human HT-3 cell growth in a cell viability assay, IC50 = 0.5075 μM. | SANGER | |||
| HCC1954 | Growth inhibition assay | Inhibition of human HCC1954 cell growth in a cell viability assay, IC50 = 0.51208 μM. | SANGER | |||
| SNU-423 | Growth inhibition assay | Inhibition of human SNU-423 cell growth in a cell viability assay, IC50 = 0.51706 μM. | SANGER | |||
| TE-12 | Growth inhibition assay | Inhibition of human TE-12 cell growth in a cell viability assay, IC50 = 0.52162 μM. | SANGER | |||
| VA-ES-BJ | Growth inhibition assay | Inhibition of human VA-ES-BJ cell growth in a cell viability assay, IC50 = 0.53045 μM. | SANGER | |||
| PANC-10-05 | Growth inhibition assay | Inhibition of human PANC-10-05 cell growth in a cell viability assay, IC50 = 0.54176 μM. | SANGER | |||
| LS-123 | Growth inhibition assay | Inhibition of human LS-123 cell growth in a cell viability assay, IC50 = 0.5565 μM. | SANGER | |||
| TK10 | Growth inhibition assay | Inhibition of human TK10 cell growth in a cell viability assay, IC50 = 0.55826 μM. | SANGER | |||
| ES8 | Growth inhibition assay | Inhibition of human ES8 cell growth in a cell viability assay, IC50 = 0.56044 μM. | SANGER | |||
| A427 | Growth inhibition assay | Inhibition of human A427 cell growth in a cell viability assay, IC50 = 0.56656 μM. | SANGER | |||
| IGROV-1 | Growth inhibition assay | Inhibition of human IGROV-1 cell growth in a cell viability assay, IC50 = 0.5672 μM. | SANGER | |||
| U-118-MG | Growth inhibition assay | Inhibition of human U-118-MG cell growth in a cell viability assay, IC50 = 0.57081 μM. | SANGER | |||
| SNU-449 | Growth inhibition assay | Inhibition of human SNU-449 cell growth in a cell viability assay, IC50 = 0.57519 μM. | SANGER | |||
| HOP-92 | Growth inhibition assay | Inhibition of human HOP-92 cell growth in a cell viability assay, IC50 = 0.57549 μM. | SANGER | |||
| U-266 | Growth inhibition assay | Inhibition of human U-266 cell growth in a cell viability assay, IC50 = 0.58161 μM. | SANGER | |||
| KALS-1 | Growth inhibition assay | Inhibition of human KALS-1 cell growth in a cell viability assay, IC50 = 0.58424 μM. | SANGER | |||
| SK-MEL-3 | Growth inhibition assay | Inhibition of human SK-MEL-3 cell growth in a cell viability assay, IC50 = 0.60472 μM. | SANGER | |||
| NCI-H2122 | Growth inhibition assay | Inhibition of human NCI-H2122 cell growth in a cell viability assay, IC50 = 0.60641 μM. | SANGER | |||
| HCC1395 | Growth inhibition assay | Inhibition of human HCC1395 cell growth in a cell viability assay, IC50 = 0.62956 μM. | SANGER | |||
| BB49-HNC | Growth inhibition assay | Inhibition of human BB49-HNC cell growth in a cell viability assay, IC50 = 0.63678 μM. | SANGER | |||
| NB17 | Growth inhibition assay | Inhibition of human NB17 cell growth in a cell viability assay, IC50 = 0.64094 μM. | SANGER | |||
| A431 | Growth inhibition assay | Inhibition of human A431 cell growth in a cell viability assay, IC50 = 0.64803 μM. | SANGER | |||
| ME-180 | Growth inhibition assay | Inhibition of human ME-180 cell growth in a cell viability assay, IC50 = 0.66369 μM. | SANGER | |||
| ChaGo-K-1 | Growth inhibition assay | Inhibition of human ChaGo-K-1 cell growth in a cell viability assay, IC50 = 0.66457 μM. | SANGER | |||
| SF539 | Growth inhibition assay | Inhibition of human SF539 cell growth in a cell viability assay, IC50 = 0.67262 μM. | SANGER | |||
| CAPAN-1 | Growth inhibition assay | Inhibition of human CAPAN-1 cell growth in a cell viability assay, IC50 = 0.68975 μM. | SANGER | |||
| IST-MEL1 | Growth inhibition assay | Inhibition of human IST-MEL1 cell growth in a cell viability assay, IC50 = 0.69388 μM. | SANGER | |||
| MHH-NB-11 | Growth inhibition assay | Inhibition of human MHH-NB-11 cell growth in a cell viability assay, IC50 = 0.70144 μM. | SANGER | |||
| HSC-2 | Growth inhibition assay | Inhibition of human HSC-2 cell growth in a cell viability assay, IC50 = 0.70376 μM. | SANGER | |||
| NCI-H2126 | Growth inhibition assay | Inhibition of human NCI-H2126 cell growth in a cell viability assay, IC50 = 0.70549 μM. | SANGER | |||
| SiHa | Growth inhibition assay | Inhibition of human SiHa cell growth in a cell viability assay, IC50 = 0.70939 μM. | SANGER | |||
| COR-L23 | Growth inhibition assay | Inhibition of human COR-L23 cell growth in a cell viability assay, IC50 = 0.71358 μM. | SANGER | |||
| HuH-7 | Growth inhibition assay | Inhibition of human HuH-7 cell growth in a cell viability assay, IC50 = 0.72041 μM. | SANGER | |||
| HOS | Growth inhibition assay | Inhibition of human HOS cell growth in a cell viability assay, IC50 = 0.72579 μM. | SANGER | |||
| KYSE-150 | Growth inhibition assay | Inhibition of human KYSE-150 cell growth in a cell viability assay, IC50 = 0.72671 μM. | SANGER | |||
| 639-V | Growth inhibition assay | Inhibition of human 639-V cell growth in a cell viability assay, IC50 = 0.72735 μM. | SANGER | |||
| SW1710 | Growth inhibition assay | Inhibition of human SW1710 cell growth in a cell viability assay, IC50 = 0.73416 μM. | SANGER | |||
| SW837 | Growth inhibition assay | Inhibition of human SW837 cell growth in a cell viability assay, IC50 = 0.74158 μM. | SANGER | |||
| NCI-H1792 | Growth inhibition assay | Inhibition of human NCI-H1792 cell growth in a cell viability assay, IC50 = 0.76481 μM. | SANGER | |||
| NCI-H1623 | Growth inhibition assay | Inhibition of human NCI-H1623 cell growth in a cell viability assay, IC50 = 0.78037 μM. | SANGER | |||
| TE-11 | Growth inhibition assay | Inhibition of human TE-11 cell growth in a cell viability assay, IC50 = 0.78161 μM. | SANGER | |||
| HLE | Growth inhibition assay | Inhibition of human HLE cell growth in a cell viability assay, IC50 = 0.78726 μM. | SANGER | |||
| NH-12 | Growth inhibition assay | Inhibition of human NH-12 cell growth in a cell viability assay, IC50 = 0.80005 μM. | SANGER | |||
| DoTc2-4510 | Growth inhibition assay | Inhibition of human DoTc2-4510 cell growth in a cell viability assay, IC50 = 0.80141 μM. | SANGER | |||
| OCUB-M | Growth inhibition assay | Inhibition of human OCUB-M cell growth in a cell viability assay, IC50 = 0.80564 μM. | SANGER | |||
| TE-1 | Growth inhibition assay | Inhibition of human TE-1 cell growth in a cell viability assay, IC50 = 0.80826 μM. | SANGER | |||
| MOLT-4 | Growth inhibition assay | Inhibition of human MOLT-4 cell growth in a cell viability assay, IC50 = 0.81854 μM. | SANGER | |||
| MKN7 | Growth inhibition assay | Inhibition of human MKN7 cell growth in a cell viability assay, IC50 = 0.82115 μM. | SANGER | |||
| OC-314 | Growth inhibition assay | Inhibition of human OC-314 cell growth in a cell viability assay, IC50 = 0.85486 μM. | SANGER | |||
| HCC1806 | Growth inhibition assay | Inhibition of human HCC1806 cell growth in a cell viability assay, IC50 = 0.85806 μM. | SANGER | |||
| FTC-133 | Growth inhibition assay | Inhibition of human FTC-133 cell growth in a cell viability assay, IC50 = 0.86254 μM. | SANGER | |||
| FADU | Growth inhibition assay | Inhibition of human FADU cell growth in a cell viability assay, IC50 = 0.86449 μM. | SANGER | |||
| NCI-H1581 | Growth inhibition assay | Inhibition of human NCI-H1581 cell growth in a cell viability assay, IC50 = 0.87337 μM. | SANGER | |||
| Calu-6 | Growth inhibition assay | Inhibition of human Calu-6 cell growth in a cell viability assay, IC50 = 0.91715 μM. | SANGER | |||
| MKN28 | Growth inhibition assay | Inhibition of human MKN28 cell growth in a cell viability assay, IC50 = 0.91737 μM. | SANGER | |||
| SK-PN-DW | Growth inhibition assay | Inhibition of human SK-PN-DW cell growth in a cell viability assay, IC50 = 0.93142 μM. | SANGER | |||
| NEC8 | Growth inhibition assay | Inhibition of human NEC8 cell growth in a cell viability assay, IC50 = 0.93577 μM. | SANGER | |||
| ACHN | Growth inhibition assay | Inhibition of human ACHN cell growth in a cell viability assay, IC50 = 0.93993 μM. | SANGER | |||
| KINGS-1 | Growth inhibition assay | Inhibition of human KINGS-1 cell growth in a cell viability assay, IC50 = 0.94028 μM. | SANGER | |||
| G-401 | Growth inhibition assay | Inhibition of human G-401 cell growth in a cell viability assay, IC50 = 0.94291 μM. | SANGER | |||
| OAW-28 | Growth inhibition assay | Inhibition of human OAW-28 cell growth in a cell viability assay, IC50 = 0.9594 μM. | SANGER | |||
| SW962 | Growth inhibition assay | Inhibition of human SW962 cell growth in a cell viability assay, IC50 = 0.96911 μM. | SANGER | |||
| SW1990 | Growth inhibition assay | Inhibition of human SW1990 cell growth in a cell viability assay, IC50 = 0.97181 μM. | SANGER | |||
| KGN | Growth inhibition assay | Inhibition of human KGN cell growth in a cell viability assay, IC50 = 0.97828 μM. | SANGER | |||
| Saos-2 | Growth inhibition assay | Inhibition of human Saos-2 cell growth in a cell viability assay, IC50 = 0.97892 μM. | SANGER | |||
| NCI-H2405 | Growth inhibition assay | Inhibition of human NCI-H2405 cell growth in a cell viability assay, IC50 = 0.98083 μM. | SANGER | |||
| SK-MEL-24 | Growth inhibition assay | Inhibition of human SK-MEL-24 cell growth in a cell viability assay, IC50 = 0.98568 μM. | SANGER | |||
| NCI-H2228 | Growth inhibition assay | Inhibition of human NCI-H2228 cell growth in a cell viability assay, IC50 = 0.99589 μM. | SANGER | |||
| NCI-H630 | Growth inhibition assay | Inhibition of human NCI-H630 cell growth in a cell viability assay, IC50 = 1.00781 μM. | SANGER | |||
| OE19 | Growth inhibition assay | Inhibition of human OE19 cell growth in a cell viability assay, IC50 = 1.01075 μM. | SANGER | |||
| C3A | Growth inhibition assay | Inhibition of human C3A cell growth in a cell viability assay, IC50 = 1.01654 μM. | SANGER | |||
| TE-5 | Growth inhibition assay | Inhibition of human TE-5 cell growth in a cell viability assay, IC50 = 1.02565 μM. | SANGER | |||
| TGBC24TKB | Growth inhibition assay | Inhibition of human TGBC24TKB cell growth in a cell viability assay, IC50 = 1.02745 μM. | SANGER | |||
| SW1088 | Growth inhibition assay | Inhibition of human SW1088 cell growth in a cell viability assay, IC50 = 1.03055 μM. | SANGER | |||
| EFO-21 | Growth inhibition assay | Inhibition of human EFO-21 cell growth in a cell viability assay, IC50 = 1.03927 μM. | SANGER | |||
| BV-173 | Growth inhibition assay | Inhibition of human BV-173 cell growth in a cell viability assay, IC50 = 1.04423 μM. | SANGER | |||
| NCI-H1395 | Growth inhibition assay | Inhibition of human NCI-H1395 cell growth in a cell viability assay, IC50 = 1.04616 μM. | SANGER | |||
| CAL-12T | Growth inhibition assay | Inhibition of human CAL-12T cell growth in a cell viability assay, IC50 = 1.07163 μM. | SANGER | |||
| SK-N-DZ | Growth inhibition assay | Inhibition of human SK-N-DZ cell growth in a cell viability assay, IC50 = 1.10354 μM. | SANGER | |||
| SHP-77 | Growth inhibition assay | Inhibition of human SHP-77 cell growth in a cell viability assay, IC50 = 1.13357 μM. | SANGER | |||
| EW-16 | Growth inhibition assay | Inhibition of human EW-16 cell growth in a cell viability assay, IC50 = 1.14517 μM. | SANGER | |||
| NB6 | Growth inhibition assay | Inhibition of human NB6 cell growth in a cell viability assay, IC50 = 1.18022 μM. | SANGER | |||
| 769-P | Growth inhibition assay | Inhibition of human 769-P cell growth in a cell viability assay, IC50 = 1.19576 μM. | SANGER | |||
| SK-MEL-30 | Growth inhibition assay | Inhibition of human SK-MEL-30 cell growth in a cell viability assay, IC50 = 1.19699 μM. | SANGER | |||
| MDA-MB-415 | Growth inhibition assay | Inhibition of human MDA-MB-415 cell growth in a cell viability assay, IC50 = 1.20664 μM. | SANGER | |||
| H4 | Growth inhibition assay | Inhibition of human H4 cell growth in a cell viability assay, IC50 = 1.20837 μM. | SANGER | |||
| YH-13 | Growth inhibition assay | Inhibition of human YH-13 cell growth in a cell viability assay, IC50 = 1.24608 μM. | SANGER | |||
| HT-1376 | Growth inhibition assay | Inhibition of human HT-1376 cell growth in a cell viability assay, IC50 = 1.25039 μM. | SANGER | |||
| D-566MG | Growth inhibition assay | Inhibition of human D-566MG cell growth in a cell viability assay, IC50 = 1.25289 μM. | SANGER | |||
| A253 | Growth inhibition assay | Inhibition of human A253 cell growth in a cell viability assay, IC50 = 1.2597 μM. | SANGER | |||
| no-10 | Growth inhibition assay | Inhibition of human no-10 cell growth in a cell viability assay, IC50 = 1.26433 μM. | SANGER | |||
| SW48 | Growth inhibition assay | Inhibition of human SW48 cell growth in a cell viability assay, IC50 = 1.27289 μM. | SANGER | |||
| NCI-H1793 | Growth inhibition assay | Inhibition of human NCI-H1793 cell growth in a cell viability assay, IC50 = 1.28912 μM. | SANGER | |||
| G-402 | Growth inhibition assay | Inhibition of human G-402 cell growth in a cell viability assay, IC50 = 1.31335 μM. | SANGER | |||
| BT-20 | Growth inhibition assay | Inhibition of human BT-20 cell growth in a cell viability assay, IC50 = 1.32858 μM. | SANGER | |||
| PANC-08-13 | Growth inhibition assay | Inhibition of human PANC-08-13 cell growth in a cell viability assay, IC50 = 1.34586 μM. | SANGER | |||
| CAKI-1 | Growth inhibition assay | Inhibition of human CAKI-1 cell growth in a cell viability assay, IC50 = 1.35324 μM. | SANGER | |||
| NCI-H596 | Growth inhibition assay | Inhibition of human NCI-H596 cell growth in a cell viability assay, IC50 = 1.37493 μM. | SANGER | |||
| OAW-42 | Growth inhibition assay | Inhibition of human OAW-42 cell growth in a cell viability assay, IC50 = 1.39905 μM. | SANGER | |||
| AM-38 | Growth inhibition assay | Inhibition of human AM-38 cell growth in a cell viability assay, IC50 = 1.40448 μM. | SANGER | |||
| IST-MES1 | Growth inhibition assay | Inhibition of human IST-MES1 cell growth in a cell viability assay, IC50 = 1.41761 μM. | SANGER | |||
| NCI-H441 | Growth inhibition assay | Inhibition of human NCI-H441 cell growth in a cell viability assay, IC50 = 1.45035 μM. | SANGER | |||
| CAL-54 | Growth inhibition assay | Inhibition of human CAL-54 cell growth in a cell viability assay, IC50 = 1.45489 μM. | SANGER | |||
| MDA-MB-361 | Growth inhibition assay | Inhibition of human MDA-MB-361 cell growth in a cell viability assay, IC50 = 1.49536 μM. | SANGER | |||
| C-33-A | Growth inhibition assay | Inhibition of human C-33-A cell growth in a cell viability assay, IC50 = 1.522 μM. | SANGER | |||
| KYSE-410 | Growth inhibition assay | Inhibition of human KYSE-410 cell growth in a cell viability assay, IC50 = 1.5665 μM. | SANGER | |||
| D-392MG | Growth inhibition assay | Inhibition of human D-392MG cell growth in a cell viability assay, IC50 = 1.59113 μM. | SANGER | |||
| JAR | Growth inhibition assay | Inhibition of human JAR cell growth in a cell viability assay, IC50 = 1.59307 μM. | SANGER | |||
| U-2-OS | Growth inhibition assay | Inhibition of human U-2-OS cell growth in a cell viability assay, IC50 = 1.60021 μM. | SANGER | |||
| OVCAR-8 | Growth inhibition assay | Inhibition of human OVCAR-8 cell growth in a cell viability assay, IC50 = 1.62046 μM. | SANGER | |||
| NCI-H1573 | Growth inhibition assay | Inhibition of human NCI-H1573 cell growth in a cell viability assay, IC50 = 1.63636 μM. | SANGER | |||
| DJM-1 | Growth inhibition assay | Inhibition of human DJM-1 cell growth in a cell viability assay, IC50 = 1.63857 μM. | SANGER | |||
| DBTRG-05MG | Growth inhibition assay | Inhibition of human DBTRG-05MG cell growth in a cell viability assay, IC50 = 1.65068 μM. | SANGER | |||
| KP-4 | Growth inhibition assay | Inhibition of human KP-4 cell growth in a cell viability assay, IC50 = 1.65304 μM. | SANGER | |||
| NOS-1 | Growth inhibition assay | Inhibition of human NOS-1 cell growth in a cell viability assay, IC50 = 1.66033 μM. | SANGER | |||
| NCI-H727 | Growth inhibition assay | Inhibition of human NCI-H727 cell growth in a cell viability assay, IC50 = 1.66682 μM. | SANGER | |||
| AN3-CA | Growth inhibition assay | Inhibition of human AN3-CA cell growth in a cell viability assay, IC50 = 1.68565 μM. | SANGER | |||
| OE33 | Growth inhibition assay | Inhibition of human OE33 cell growth in a cell viability assay, IC50 = 1.71458 μM. | SANGER | |||
| MSTO-211H | Growth inhibition assay | Inhibition of human MSTO-211H cell growth in a cell viability assay, IC50 = 1.77036 μM. | SANGER | |||
| OVCAR-3 | Growth inhibition assay | Inhibition of human OVCAR-3 cell growth in a cell viability assay, IC50 = 1.77078 μM. | SANGER | |||
| Becker | Growth inhibition assay | Inhibition of human Becker cell growth in a cell viability assay, IC50 = 1.80387 μM. | SANGER | |||
| NCI-SNU-5 | Growth inhibition assay | Inhibition of human NCI-SNU-5 cell growth in a cell viability assay, IC50 = 1.82515 μM. | SANGER | |||
| SNB75 | Growth inhibition assay | Inhibition of human SNB75 cell growth in a cell viability assay, IC50 = 1.84345 μM. | SANGER | |||
| DEL | Growth inhibition assay | Inhibition of human DEL cell growth in a cell viability assay, IC50 = 1.87238 μM. | SANGER | |||
| SW982 | Growth inhibition assay | Inhibition of human SW982 cell growth in a cell viability assay, IC50 = 1.91727 μM. | SANGER | |||
| NB14 | Growth inhibition assay | Inhibition of human NB14 cell growth in a cell viability assay, IC50 = 1.95882 μM. | SANGER | |||
| ESS-1 | Growth inhibition assay | Inhibition of human ESS-1 cell growth in a cell viability assay, IC50 = 1.96064 μM. | SANGER | |||
| NMC-G1 | Growth inhibition assay | Inhibition of human NMC-G1 cell growth in a cell viability assay, IC50 = 2.14133 μM. | SANGER | |||
| TE-6 | Growth inhibition assay | Inhibition of human TE-6 cell growth in a cell viability assay, IC50 = 2.14472 μM. | SANGER | |||
| NCI-H661 | Growth inhibition assay | Inhibition of human NCI-H661 cell growth in a cell viability assay, IC50 = 2.15295 μM. | SANGER | |||
| LB996-RCC | Growth inhibition assay | Inhibition of human LB996-RCC cell growth in a cell viability assay, IC50 = 2.1951 μM. | SANGER | |||
| A4-Fuk | Growth inhibition assay | Inhibition of human A4-Fuk cell growth in a cell viability assay, IC50 = 2.21558 μM. | SANGER | |||
| DMS-114 | Growth inhibition assay | Inhibition of human DMS-114 cell growth in a cell viability assay, IC50 = 2.29075 μM. | SANGER | |||
| LXF-289 | Growth inhibition assay | Inhibition of human LXF-289 cell growth in a cell viability assay, IC50 = 2.35474 μM. | SANGER | |||
| ES1 | Growth inhibition assay | Inhibition of human ES1 cell growth in a cell viability assay, IC50 = 2.39127 μM. | SANGER | |||
| U-87-MG | Growth inhibition assay | Inhibition of human U-87-MG cell growth in a cell viability assay, IC50 = 2.4688 μM. | SANGER | |||
| A204 | Growth inhibition assay | Inhibition of human A204 cell growth in a cell viability assay, IC50 = 2.48396 μM. | SANGER | |||
| HCC1569 | Growth inhibition assay | Inhibition of human HCC1569 cell growth in a cell viability assay, IC50 = 2.51747 μM. | SANGER | |||
| RO82-W-1 | Growth inhibition assay | Inhibition of human RO82-W-1 cell growth in a cell viability assay, IC50 = 2.52385 μM. | SANGER | |||
| OVCAR-4 | Growth inhibition assay | Inhibition of human OVCAR-4 cell growth in a cell viability assay, IC50 = 2.52792 μM. | SANGER | |||
| 8-MG-BA | Growth inhibition assay | Inhibition of human 8-MG-BA cell growth in a cell viability assay, IC50 = 2.53238 μM. | SANGER | |||
| LC-2-ad | Growth inhibition assay | Inhibition of human LC-2-ad cell growth in a cell viability assay, IC50 = 2.54292 μM. | SANGER | |||
| KNS-81-FD | Growth inhibition assay | Inhibition of human KNS-81-FD cell growth in a cell viability assay, IC50 = 2.56819 μM. | SANGER | |||
| NCI-N87 | Growth inhibition assay | Inhibition of human NCI-N87 cell growth in a cell viability assay, IC50 = 2.59379 μM. | SANGER | |||
| SCC-15 | Growth inhibition assay | Inhibition of human SCC-15 cell growth in a cell viability assay, IC50 = 2.60118 μM. | SANGER | |||
| NCI-H1693 | Growth inhibition assay | Inhibition of human NCI-H1693 cell growth in a cell viability assay, IC50 = 2.62345 μM. | SANGER | |||
| GOTO | Growth inhibition assay | Inhibition of human GOTO cell growth in a cell viability assay, IC50 = 2.65348 μM. | SANGER | |||
| NCI-H2452 | Growth inhibition assay | Inhibition of human NCI-H2452 cell growth in a cell viability assay, IC50 = 2.65915 μM. | SANGER | |||
| EKVX | Growth inhibition assay | Inhibition of human EKVX cell growth in a cell viability assay, IC50 = 2.68857 μM. | SANGER | |||
| CGTH-W-1 | Growth inhibition assay | Inhibition of human CGTH-W-1 cell growth in a cell viability assay, IC50 = 2.69191 μM. | SANGER | |||
| MLMA | Growth inhibition assay | Inhibition of human MLMA cell growth in a cell viability assay, IC50 = 2.70122 μM. | SANGER | |||
| D-423MG | Growth inhibition assay | Inhibition of human D-423MG cell growth in a cell viability assay, IC50 = 2.80291 μM. | SANGER | |||
| NCI-H1650 | Growth inhibition assay | Inhibition of human NCI-H1650 cell growth in a cell viability assay, IC50 = 2.82476 μM. | SANGER | |||
| CaR-1 | Growth inhibition assay | Inhibition of human CaR-1 cell growth in a cell viability assay, IC50 = 2.85893 μM. | SANGER | |||
| KYSE-140 | Growth inhibition assay | Inhibition of human KYSE-140 cell growth in a cell viability assay, IC50 = 2.87157 μM. | SANGER | |||
| SW13 | Growth inhibition assay | Inhibition of human SW13 cell growth in a cell viability assay, IC50 = 2.87373 μM. | SANGER | |||
| NCI-H520 | Growth inhibition assay | Inhibition of human NCI-H520 cell growth in a cell viability assay, IC50 = 2.93664 μM. | SANGER | |||
| A388 | Growth inhibition assay | Inhibition of human A388 cell growth in a cell viability assay, IC50 = 2.94309 μM. | SANGER | |||
| NCI-H1048 | Growth inhibition assay | Inhibition of human NCI-H1048 cell growth in a cell viability assay, IC50 = 2.9524 μM. | SANGER | |||
| ETK-1 | Growth inhibition assay | Inhibition of human ETK-1 cell growth in a cell viability assay, IC50 = 2.99687 μM. | SANGER | |||
| GI-1 | Growth inhibition assay | Inhibition of human GI-1 cell growth in a cell viability assay, IC50 = 3.01145 μM. | SANGER | |||
| KLE | Growth inhibition assay | Inhibition of human KLE cell growth in a cell viability assay, IC50 = 3.06759 μM. | SANGER | |||
| KYSE-70 | Growth inhibition assay | Inhibition of human KYSE-70 cell growth in a cell viability assay, IC50 = 3.11193 μM. | SANGER | |||
| NCI-H2030 | Growth inhibition assay | Inhibition of human NCI-H2030 cell growth in a cell viability assay, IC50 = 3.15086 μM. | SANGER | |||
| CW-2 | Growth inhibition assay | Inhibition of human CW-2 cell growth in a cell viability assay, IC50 = 3.20269 μM. | SANGER | |||
| DMS-273 | Growth inhibition assay | Inhibition of human DMS-273 cell growth in a cell viability assay, IC50 = 3.25967 μM. | SANGER | |||
| COLO-668 | Growth inhibition assay | Inhibition of human COLO-668 cell growth in a cell viability assay, IC50 = 3.29772 μM. | SANGER | |||
| BEN | Growth inhibition assay | Inhibition of human BEN cell growth in a cell viability assay, IC50 = 3.33742 μM. | SANGER | |||
| UACC-62 | Growth inhibition assay | Inhibition of human UACC-62 cell growth in a cell viability assay, IC50 = 3.34938 μM. | SANGER | |||
| TGBC1TKB | Growth inhibition assay | Inhibition of human TGBC1TKB cell growth in a cell viability assay, IC50 = 3.39718 μM. | SANGER | |||
| SF295 | Growth inhibition assay | Inhibition of human SF295 cell growth in a cell viability assay, IC50 = 3.43272 μM. | SANGER | |||
| D-247MG | Growth inhibition assay | Inhibition of human D-247MG cell growth in a cell viability assay, IC50 = 3.47503 μM. | SANGER | |||
| MN-60 | Growth inhibition assay | Inhibition of human MN-60 cell growth in a cell viability assay, IC50 = 3.60496 μM. | SANGER | |||
| EW-1 | Growth inhibition assay | Inhibition of human EW-1 cell growth in a cell viability assay, IC50 = 3.67598 μM. | SANGER | |||
| NCI-H28 | Growth inhibition assay | Inhibition of human NCI-H28 cell growth in a cell viability assay, IC50 = 3.71695 μM. | SANGER | |||
| SW900 | Growth inhibition assay | Inhibition of human SW900 cell growth in a cell viability assay, IC50 = 3.72151 μM. | SANGER | |||
| GB-1 | Growth inhibition assay | Inhibition of human GB-1 cell growth in a cell viability assay, IC50 = 3.79554 μM. | SANGER | |||
| PA-1 | Growth inhibition assay | Inhibition of human PA-1 cell growth in a cell viability assay, IC50 = 3.79782 μM. | SANGER | |||
| HT-1197 | Growth inhibition assay | Inhibition of human HT-1197 cell growth in a cell viability assay, IC50 = 3.84358 μM. | SANGER | |||
| A704 | Growth inhibition assay | Inhibition of human A704 cell growth in a cell viability assay, IC50 = 3.85783 μM. | SANGER | |||
| MCF7 | Growth inhibition assay | Inhibition of human MCF7 cell growth in a cell viability assay, IC50 = 3.85795 μM. | SANGER | |||
| DSH1 | Growth inhibition assay | Inhibition of human DSH1 cell growth in a cell viability assay, IC50 = 3.86997 μM. | SANGER | |||
| HCC1937 | Growth inhibition assay | Inhibition of human HCC1937 cell growth in a cell viability assay, IC50 = 4.02607 μM. | SANGER | |||
| SK-OV-3 | Growth inhibition assay | Inhibition of human SK-OV-3 cell growth in a cell viability assay, IC50 = 4.06226 μM. | SANGER | |||
| NTERA-S-cl-D1 | Growth inhibition assay | Inhibition of human NTERA-S-cl-D1 cell growth in a cell viability assay, IC50 = 4.15724 μM. | SANGER | |||
| RCC10RGB | Growth inhibition assay | Inhibition of human RCC10RGB cell growth in a cell viability assay, IC50 = 4.1742 μM. | SANGER | |||
| HuO9 | Growth inhibition assay | Inhibition of human HuO9 cell growth in a cell viability assay, IC50 = 4.17433 μM. | SANGER | |||
| NCI-H2342 | Growth inhibition assay | Inhibition of human NCI-H2342 cell growth in a cell viability assay, IC50 = 4.18262 μM. | SANGER | |||
| NCI-H522 | Growth inhibition assay | Inhibition of human NCI-H522 cell growth in a cell viability assay, IC50 = 4.22634 μM. | SANGER | |||
| TE-10 | Growth inhibition assay | Inhibition of human TE-10 cell growth in a cell viability assay, IC50 = 4.24796 μM. | SANGER | |||
| SF126 | Growth inhibition assay | Inhibition of human SF126 cell growth in a cell viability assay, IC50 = 4.30385 μM. | SANGER | |||
| KP-N-YS | Growth inhibition assay | Inhibition of human KP-N-YS cell growth in a cell viability assay, IC50 = 4.33466 μM. | SANGER | |||
| COLO-800 | Growth inhibition assay | Inhibition of human COLO-800 cell growth in a cell viability assay, IC50 = 4.42427 μM. | SANGER | |||
| COLO-680N | Growth inhibition assay | Inhibition of human COLO-680N cell growth in a cell viability assay, IC50 = 4.44325 μM. | SANGER | |||
| Detroit562 | Growth inhibition assay | Inhibition of human Detroit562 cell growth in a cell viability assay, IC50 = 4.45258 μM. | SANGER | |||
| SF268 | Growth inhibition assay | Inhibition of human SF268 cell growth in a cell viability assay, IC50 = 4.68825 μM. | SANGER | |||
| 22RV1 | Growth inhibition assay | Inhibition of human 22RV1 cell growth in a cell viability assay, IC50 = 4.70727 μM. | SANGER | |||
| Ca-Ski | Growth inhibition assay | Inhibition of human Ca-Ski cell growth in a cell viability assay, IC50 = 4.80097 μM. | SANGER | |||
| MHH-ES-1 | Growth inhibition assay | Inhibition of human MHH-ES-1 cell growth in a cell viability assay, IC50 = 4.81086 μM. | SANGER | |||
| SK-NEP-1 | Growth inhibition assay | Inhibition of human SK-NEP-1 cell growth in a cell viability assay, IC50 = 4.84277 μM. | SANGER | |||
| A673 | Growth inhibition assay | Inhibition of human A673 cell growth in a cell viability assay, IC50 = 4.93448 μM. | SANGER | |||
| SJRH30 | Growth inhibition assay | Inhibition of human SJRH30 cell growth in a cell viability assay, IC50 = 4.94997 μM. | SANGER | |||
| KP-N-YN | Growth inhibition assay | Inhibition of human KP-N-YN cell growth in a cell viability assay, IC50 = 4.98905 μM. | SANGER | |||
| VMRC-RCZ | Growth inhibition assay | Inhibition of human VMRC-RCZ cell growth in a cell viability assay, IC50 = 5.00475 μM. | SANGER | |||
| KARPAS-299 | Growth inhibition assay | Inhibition of human KARPAS-299 cell growth in a cell viability assay, IC50 = 5.0724 μM. | SANGER | |||
| HCC1187 | Growth inhibition assay | Inhibition of human HCC1187 cell growth in a cell viability assay, IC50 = 5.09638 μM. | SANGER | |||
| NCI-H1155 | Growth inhibition assay | Inhibition of human NCI-H1155 cell growth in a cell viability assay, IC50 = 5.14672 μM. | SANGER | |||
| LU-99A | Growth inhibition assay | Inhibition of human LU-99A cell growth in a cell viability assay, IC50 = 5.21153 μM. | SANGER | |||
| D-283MED | Growth inhibition assay | Inhibition of human D-283MED cell growth in a cell viability assay, IC50 = 5.31099 μM. | SANGER | |||
| S-117 | Growth inhibition assay | Inhibition of human S-117 cell growth in a cell viability assay, IC50 = 5.3125 μM. | SANGER | |||
| RH-18 | Growth inhibition assay | Inhibition of human RH-18 cell growth in a cell viability assay, IC50 = 5.37589 μM. | SANGER | |||
| TE-9 | Growth inhibition assay | Inhibition of human TE-9 cell growth in a cell viability assay, IC50 = 5.3849 μM. | SANGER | |||
| NCI-H226 | Growth inhibition assay | Inhibition of human NCI-H226 cell growth in a cell viability assay, IC50 = 5.45133 μM. | SANGER | |||
| Daoy | Growth inhibition assay | Inhibition of human Daoy cell growth in a cell viability assay, IC50 = 5.72642 μM. | SANGER | |||
| MPP-89 | Growth inhibition assay | Inhibition of human MPP-89 cell growth in a cell viability assay, IC50 = 5.73747 μM. | SANGER | |||
| SAS | Growth inhibition assay | Inhibition of human SAS cell growth in a cell viability assay, IC50 = 5.8738 μM. | SANGER | |||
| CAS-1 | Growth inhibition assay | Inhibition of human CAS-1 cell growth in a cell viability assay, IC50 = 5.91441 μM. | SANGER | |||
| AU565 | Growth inhibition assay | Inhibition of human AU565 cell growth in a cell viability assay, IC50 = 5.99149 μM. | SANGER | |||
| GT3TKB | Growth inhibition assay | Inhibition of human GT3TKB cell growth in a cell viability assay, IC50 = 6.20685 μM. | SANGER | |||
| L-428 | Growth inhibition assay | Inhibition of human L-428 cell growth in a cell viability assay, IC50 = 6.30127 μM. | SANGER | |||
| DB | Growth inhibition assay | Inhibition of human DB cell growth in a cell viability assay, IC50 = 6.35319 μM. | SANGER | |||
| BT-549 | Growth inhibition assay | Inhibition of human BT-549 cell growth in a cell viability assay, IC50 = 6.38058 μM. | SANGER | |||
| ES6 | Growth inhibition assay | Inhibition of human ES6 cell growth in a cell viability assay, IC50 = 6.39694 μM. | SANGER | |||
| SW684 | Growth inhibition assay | Inhibition of human SW684 cell growth in a cell viability assay, IC50 = 6.51032 μM. | SANGER | |||
| OCI-AML2 | Growth inhibition assay | Inhibition of human OCI-AML2 cell growth in a cell viability assay, IC50 = 6.55132 μM. | SANGER | |||
| Ramos-2G6-4C10 | Growth inhibition assay | Inhibition of human Ramos-2G6-4C10 cell growth in a cell viability assay, IC50 = 6.57105 μM. | SANGER | |||
| RH-1 | Growth inhibition assay | Inhibition of human RH-1 cell growth in a cell viability assay, IC50 = 6.63743 μM. | SANGER | |||
| no-11 | Growth inhibition assay | Inhibition of human no-11 cell growth in a cell viability assay, IC50 = 6.73215 μM. | SANGER | |||
| UM-UC-3 | Growth inhibition assay | Inhibition of human UM-UC-3 cell growth in a cell viability assay, IC50 = 6.80995 μM. | SANGER | |||
| ECC10 | Growth inhibition assay | Inhibition of human ECC10 cell growth in a cell viability assay, IC50 = 7.06506 μM. | SANGER | |||
| CAL-120 | Growth inhibition assay | Inhibition of human CAL-120 cell growth in a cell viability assay, IC50 = 7.1739 μM. | SANGER | |||
| HCC2218 | Growth inhibition assay | Inhibition of human HCC2218 cell growth in a cell viability assay, IC50 = 7.20089 μM. | SANGER | |||
| T98G | Growth inhibition assay | Inhibition of human T98G cell growth in a cell viability assay, IC50 = 7.30092 μM. | SANGER | |||
| LU-134-A | Growth inhibition assay | Inhibition of human LU-134-A cell growth in a cell viability assay, IC50 = 7.36234 μM. | SANGER | |||
| BT-474 | Growth inhibition assay | Inhibition of human BT-474 cell growth in a cell viability assay, IC50 = 7.44003 μM. | SANGER | |||
| NBsusSR | Growth inhibition assay | Inhibition of human NBsusSR cell growth in a cell viability assay, IC50 = 7.91153 μM. | SANGER | |||
| HCE-4 | Growth inhibition assay | Inhibition of human HCE-4 cell growth in a cell viability assay, IC50 = 8.07171 μM. | SANGER | |||
| NCI-H1651 | Growth inhibition assay | Inhibition of human NCI-H1651 cell growth in a cell viability assay, IC50 = 8.22561 μM. | SANGER | |||
| EW-13 | Growth inhibition assay | Inhibition of human EW-13 cell growth in a cell viability assay, IC50 = 8.2479 μM. | SANGER | |||
| CAMA-1 | Growth inhibition assay | Inhibition of human CAMA-1 cell growth in a cell viability assay, IC50 = 8.52162 μM. | SANGER | |||
| KURAMOCHI | Growth inhibition assay | Inhibition of human KURAMOCHI cell growth in a cell viability assay, IC50 = 8.58994 μM. | SANGER | |||
| GR-ST | Growth inhibition assay | Inhibition of human GR-ST cell growth in a cell viability assay, IC50 = 8.59763 μM. | SANGER | |||
| TI-73 | Growth inhibition assay | Inhibition of human TI-73 cell growth in a cell viability assay, IC50 = 8.60681 μM. | SANGER | |||
| A2780 | Growth inhibition assay | Inhibition of human A2780 cell growth in a cell viability assay, IC50 = 8.76448 μM. | SANGER | |||
| LB831-BLC | Growth inhibition assay | Inhibition of human LB831-BLC cell growth in a cell viability assay, IC50 = 8.78041 μM. | SANGER | |||
| KNS-42 | Growth inhibition assay | Inhibition of human KNS-42 cell growth in a cell viability assay, IC50 = 8.93018 μM. | SANGER | |||
| EW-3 | Growth inhibition assay | Inhibition of human EW-3 cell growth in a cell viability assay, IC50 = 8.93635 μM. | SANGER | |||
| HH | Growth inhibition assay | Inhibition of human HH cell growth in a cell viability assay, IC50 = 9.04475 μM. | SANGER | |||
| PC-3 | Growth inhibition assay | Inhibition of human PC-3 cell growth in a cell viability assay, IC50 = 9.0686 μM. | SANGER | |||
| UACC-893 | Growth inhibition assay | Inhibition of human UACC-893 cell growth in a cell viability assay, IC50 = 9.36349 μM. | SANGER | |||
| TCCSUP | Growth inhibition assay | Inhibition of human TCCSUP cell growth in a cell viability assay, IC50 = 9.4848 μM. | SANGER | |||
| MC-IXC | Growth inhibition assay | Inhibition of human MC-IXC cell growth in a cell viability assay, IC50 = 9.53129 μM. | SANGER | |||
| BE-13 | Growth inhibition assay | Inhibition of human BE-13 cell growth in a cell viability assay, IC50 = 9.6499 μM. | SANGER | |||
| J-RT3-T3-5 | Growth inhibition assay | Inhibition of human J-RT3-T3-5 cell growth in a cell viability assay, IC50 = 9.72422 μM. | SANGER | |||
| KARPAS-45 | Growth inhibition assay | Inhibition of human KARPAS-45 cell growth in a cell viability assay, IC50 = 9.82596 μM. | SANGER | |||
| RPMI-2650 | Growth inhibition assay | Inhibition of human RPMI-2650 cell growth in a cell viability assay, IC50 = 9.86434 μM. | SANGER | |||
| L-363 | Growth inhibition assay | Inhibition of human L-363 cell growth in a cell viability assay, IC50 = 9.8843 μM. | SANGER | |||
| HEC-1 | Growth inhibition assay | Inhibition of human HEC-1 cell growth in a cell viability assay, IC50 = 9.91889 μM. | SANGER | |||
| HT | Growth inhibition assay | Inhibition of human HT cell growth in a cell viability assay, IC50 = 9.94018 μM. | SANGER | |||
| Calu-3 | Growth inhibition assay | Inhibition of human Calu-3 cell growth in a cell viability assay, IC50 = 10.37 μM. | SANGER | |||
| ES5 | Growth inhibition assay | Inhibition of human ES5 cell growth in a cell viability assay, IC50 = 10.4087 μM. | SANGER | |||
| NCI-H1092 | Growth inhibition assay | Inhibition of human NCI-H1092 cell growth in a cell viability assay, IC50 = 10.5235 μM. | SANGER | |||
| MDA-MB-157 | Growth inhibition assay | Inhibition of human MDA-MB-157 cell growth in a cell viability assay, IC50 = 10.5769 μM. | SANGER | |||
| ES4 | Growth inhibition assay | Inhibition of human ES4 cell growth in a cell viability assay, IC50 = 10.604 μM. | SANGER | |||
| IST-SL1 | Growth inhibition assay | Inhibition of human IST-SL1 cell growth in a cell viability assay, IC50 = 10.6383 μM. | SANGER | |||
| DOHH-2 | Growth inhibition assay | Inhibition of human DOHH-2 cell growth in a cell viability assay, IC50 = 10.64 μM. | SANGER | |||
| CAL-72 | Growth inhibition assay | Inhibition of human CAL-72 cell growth in a cell viability assay, IC50 = 11.036 μM. | SANGER | |||
| KYSE-270 | Growth inhibition assay | Inhibition of human KYSE-270 cell growth in a cell viability assay, IC50 = 11.2253 μM. | SANGER | |||
| LN-405 | Growth inhibition assay | Inhibition of human LN-405 cell growth in a cell viability assay, IC50 = 11.3201 μM. | SANGER | |||
| LCLC-103H | Growth inhibition assay | Inhibition of human LCLC-103H cell growth in a cell viability assay, IC50 = 11.3543 μM. | SANGER | |||
| SBC-5 | Growth inhibition assay | Inhibition of human SBC-5 cell growth in a cell viability assay, IC50 = 11.4071 μM. | SANGER | |||
| MOLT-16 | Growth inhibition assay | Inhibition of human MOLT-16 cell growth in a cell viability assay, IC50 = 11.4348 μM. | SANGER | |||
| MFE-280 | Growth inhibition assay | Inhibition of human MFE-280 cell growth in a cell viability assay, IC50 = 11.633 μM. | SANGER | |||
| SK-MEL-1 | Growth inhibition assay | Inhibition of human SK-MEL-1 cell growth in a cell viability assay, IC50 = 11.7648 μM. | SANGER | |||
| ALL-PO | Growth inhibition assay | Inhibition of human ALL-PO cell growth in a cell viability assay, IC50 = 11.9757 μM. | SANGER | |||
| CHP-134 | Growth inhibition assay | Inhibition of human CHP-134 cell growth in a cell viability assay, IC50 = 11.9962 μM. | SANGER | |||
| SBC-1 | Growth inhibition assay | Inhibition of human SBC-1 cell growth in a cell viability assay, IC50 = 12.216 μM. | SANGER | |||
| GCT | Growth inhibition assay | Inhibition of human GCT cell growth in a cell viability assay, IC50 = 12.2728 μM. | SANGER | |||
| EW-11 | Growth inhibition assay | Inhibition of human EW-11 cell growth in a cell viability assay, IC50 = 12.6095 μM. | SANGER | |||
| SW1573 | Growth inhibition assay | Inhibition of human SW1573 cell growth in a cell viability assay, IC50 = 12.9986 μM. | SANGER | |||
| 697 | Growth inhibition assay | Inhibition of human 697 cell growth in a cell viability assay, IC50 = 13.0067 μM. | SANGER | |||
| NCI-H720 | Growth inhibition assay | Inhibition of human NCI-H720 cell growth in a cell viability assay, IC50 = 13.0914 μM. | SANGER | |||
| CTV-1 | Growth inhibition assay | Inhibition of human CTV-1 cell growth in a cell viability assay, IC50 = 13.1166 μM. | SANGER | |||
| LU-139 | Growth inhibition assay | Inhibition of human LU-139 cell growth in a cell viability assay, IC50 = 13.2313 μM. | SANGER | |||
| SCC-9 | Growth inhibition assay | Inhibition of human SCC-9 cell growth in a cell viability assay, IC50 = 13.284 μM. | SANGER | |||
| NCI-H2052 | Growth inhibition assay | Inhibition of human NCI-H2052 cell growth in a cell viability assay, IC50 = 13.4706 μM. | SANGER | |||
| NCI-H526 | Growth inhibition assay | Inhibition of human NCI-H526 cell growth in a cell viability assay, IC50 = 13.5368 μM. | SANGER | |||
| KM-H2 | Growth inhibition assay | Inhibition of human KM-H2 cell growth in a cell viability assay, IC50 = 13.6672 μM. | SANGER | |||
| J82 | Growth inhibition assay | Inhibition of human J82 cell growth in a cell viability assay, IC50 = 14.0329 μM. | SANGER | |||
| CAL-51 | Growth inhibition assay | Inhibition of human CAL-51 cell growth in a cell viability assay, IC50 = 14.1108 μM. | SANGER | |||
| NCI-H1838 | Growth inhibition assay | Inhibition of human NCI-H1838 cell growth in a cell viability assay, IC50 = 14.201 μM. | SANGER | |||
| RS4-11 | Growth inhibition assay | Inhibition of human RS4-11 cell growth in a cell viability assay, IC50 = 14.2158 μM. | SANGER | |||
| PC-14 | Growth inhibition assay | Inhibition of human PC-14 cell growth in a cell viability assay, IC50 = 14.2546 μM. | SANGER | |||
| HGC-27 | Growth inhibition assay | Inhibition of human HGC-27 cell growth in a cell viability assay, IC50 = 14.3385 μM. | SANGER | |||
| BFTC-909 | Growth inhibition assay | Inhibition of human BFTC-909 cell growth in a cell viability assay, IC50 = 14.4163 μM. | SANGER | |||
| HEL | Growth inhibition assay | Inhibition of human HEL cell growth in a cell viability assay, IC50 = 14.5225 μM. | SANGER | |||
| BALL-1 | Growth inhibition assay | Inhibition of human BALL-1 cell growth in a cell viability assay, IC50 = 14.5964 μM. | SANGER | |||
| NCI-H209 | Growth inhibition assay | Inhibition of human NCI-H209 cell growth in a cell viability assay, IC50 = 14.7764 μM. | SANGER | |||
| NCI-H1770 | Growth inhibition assay | Inhibition of human NCI-H1770 cell growth in a cell viability assay, IC50 = 14.8218 μM. | SANGER | |||
| 5637 | Growth inhibition assay | Inhibition of human 5637 cell growth in a cell viability assay, IC50 = 15.0297 μM. | SANGER | |||
| NCI-H650 | Growth inhibition assay | Inhibition of human NCI-H650 cell growth in a cell viability assay, IC50 = 15.1644 μM. | SANGER | |||
| COR-L88 | Growth inhibition assay | Inhibition of human COR-L88 cell growth in a cell viability assay, IC50 = 15.2851 μM. | SANGER | |||
| EW-18 | Growth inhibition assay | Inhibition of human EW-18 cell growth in a cell viability assay, IC50 = 15.3363 μM. | SANGER | |||
| M059J | Growth inhibition assay | Inhibition of human M059J cell growth in a cell viability assay, IC50 = 15.61 μM. | SANGER | |||
| NCI-H1648 | Growth inhibition assay | Inhibition of human NCI-H1648 cell growth in a cell viability assay, IC50 = 15.7745 μM. | SANGER | |||
| HuO-3N1 | Growth inhibition assay | Inhibition of human HuO-3N1 cell growth in a cell viability assay, IC50 = 15.9391 μM. | SANGER | |||
| CTB-1 | Growth inhibition assay | Inhibition of human CTB-1 cell growth in a cell viability assay, IC50 = 16.0164 μM. | SANGER | |||
| NCI-H82 | Growth inhibition assay | Inhibition of human NCI-H82 cell growth in a cell viability assay, IC50 = 16.1215 μM. | SANGER | |||
| JVM-2 | Growth inhibition assay | Inhibition of human JVM-2 cell growth in a cell viability assay, IC50 = 16.3872 μM. | SANGER | |||
| ES7 | Growth inhibition assay | Inhibition of human ES7 cell growth in a cell viability assay, IC50 = 16.4652 μM. | SANGER | |||
| EFM-19 | Growth inhibition assay | Inhibition of human EFM-19 cell growth in a cell viability assay, IC50 = 16.5392 μM. | SANGER | |||
| NB13 | Growth inhibition assay | Inhibition of human NB13 cell growth in a cell viability assay, IC50 = 16.5477 μM. | SANGER | |||
| NCI-H2170 | Growth inhibition assay | Inhibition of human NCI-H2170 cell growth in a cell viability assay, IC50 = 16.587 μM. | SANGER | |||
| MOLT-13 | Growth inhibition assay | Inhibition of human MOLT-13 cell growth in a cell viability assay, IC50 = 16.9188 μM. | SANGER | |||
| U031 | Growth inhibition assay | Inhibition of human U031 cell growth in a cell viability assay, IC50 = 16.9663 μM. | SANGER | |||
| NCI-H2029 | Growth inhibition assay | Inhibition of human NCI-H2029 cell growth in a cell viability assay, IC50 = 17.1856 μM. | SANGER | |||
| ES3 | Growth inhibition assay | Inhibition of human ES3 cell growth in a cell viability assay, IC50 = 17.7386 μM. | SANGER | |||
| C8166 | Growth inhibition assay | Inhibition of human C8166 cell growth in a cell viability assay, IC50 = 17.7393 μM. | SANGER | |||
| MS-1 | Growth inhibition assay | Inhibition of human MS-1 cell growth in a cell viability assay, IC50 = 18.0461 μM. | SANGER | |||
| LK-2 | Growth inhibition assay | Inhibition of human LK-2 cell growth in a cell viability assay, IC50 = 18.1229 μM. | SANGER | |||
| NCI-H1755 | Growth inhibition assay | Inhibition of human NCI-H1755 cell growth in a cell viability assay, IC50 = 18.127 μM. | SANGER | |||
| NCI-H810 | Growth inhibition assay | Inhibition of human NCI-H810 cell growth in a cell viability assay, IC50 = 18.2045 μM. | SANGER | |||
| T84 | Growth inhibition assay | Inhibition of human T84 cell growth in a cell viability assay, IC50 = 18.2297 μM. | SANGER | |||
| KYSE-180 | Growth inhibition assay | Inhibition of human KYSE-180 cell growth in a cell viability assay, IC50 = 18.2843 μM. | SANGER | |||
| COLO-320-HSR | Growth inhibition assay | Inhibition of human COLO-320-HSR cell growth in a cell viability assay, IC50 = 18.4454 μM. | SANGER | |||
| MFM-223 | Growth inhibition assay | Inhibition of human MFM-223 cell growth in a cell viability assay, IC50 = 18.4863 μM. | SANGER | |||
| HCC2157 | Growth inhibition assay | Inhibition of human HCC2157 cell growth in a cell viability assay, IC50 = 19.3323 μM. | SANGER | |||
| LU-135 | Growth inhibition assay | Inhibition of human LU-135 cell growth in a cell viability assay, IC50 = 19.3385 μM. | SANGER | |||
| NY | Growth inhibition assay | Inhibition of human NY cell growth in a cell viability assay, IC50 = 19.5526 μM. | SANGER | |||
| SW1783 | Growth inhibition assay | Inhibition of human SW1783 cell growth in a cell viability assay, IC50 = 19.5775 μM. | SANGER | |||
| NCI-H1993 | Growth inhibition assay | Inhibition of human NCI-H1993 cell growth in a cell viability assay, IC50 = 20.0664 μM. | SANGER | |||
| HDLM-2 | Growth inhibition assay | Inhibition of human HDLM-2 cell growth in a cell viability assay, IC50 = 20.2067 μM. | SANGER | |||
| DU-145 | Growth inhibition assay | Inhibition of human DU-145 cell growth in a cell viability assay, IC50 = 20.2796 μM. | SANGER | |||
| RPMI-8866 | Growth inhibition assay | Inhibition of human RPMI-8866 cell growth in a cell viability assay, IC50 = 20.4216 μM. | SANGER | |||
| MHH-PREB-1 | Growth inhibition assay | Inhibition of human MHH-PREB-1 cell growth in a cell viability assay, IC50 = 20.5493 μM. | SANGER | |||
| LU-65 | Growth inhibition assay | Inhibition of human LU-65 cell growth in a cell viability assay, IC50 = 20.7551 μM. | SANGER | |||
| T47D | Growth inhibition assay | Inhibition of human T47D cell growth in a cell viability assay, IC50 = 20.9271 μM. | SANGER | |||
| NCI-H1304 | Growth inhibition assay | Inhibition of human NCI-H1304 cell growth in a cell viability assay, IC50 = 21.076 μM. | SANGER | |||
| COLO-684 | Growth inhibition assay | Inhibition of human COLO-684 cell growth in a cell viability assay, IC50 = 21.442 μM. | SANGER | |||
| KE-37 | Growth inhibition assay | Inhibition of human KE-37 cell growth in a cell viability assay, IC50 = 21.8047 μM. | SANGER | |||
| EW-22 | Growth inhibition assay | Inhibition of human EW-22 cell growth in a cell viability assay, IC50 = 22.3169 μM. | SANGER | |||
| MKN1 | Growth inhibition assay | Inhibition of human MKN1 cell growth in a cell viability assay, IC50 = 22.496 μM. | SANGER | |||
| PFSK-1 | Growth inhibition assay | Inhibition of human PFSK-1 cell growth in a cell viability assay, IC50 = 22.5608 μM. | SANGER | |||
| JEG-3 | Growth inhibition assay | Inhibition of human JEG-3 cell growth in a cell viability assay, IC50 = 22.589 μM. | SANGER | |||
| CCRF-CEM | Growth inhibition assay | Inhibition of human CCRF-CEM cell growth in a cell viability assay, IC50 = 22.6479 μM. | SANGER | |||
| P30-OHK | Growth inhibition assay | Inhibition of human P30-OHK cell growth in a cell viability assay, IC50 = 22.7802 μM. | SANGER | |||
| HCC38 | Growth inhibition assay | Inhibition of human HCC38 cell growth in a cell viability assay, IC50 = 23.0073 μM. | SANGER | |||
| MDA-MB-453 | Growth inhibition assay | Inhibition of human MDA-MB-453 cell growth in a cell viability assay, IC50 = 23.9976 μM. | SANGER | |||
| LNCaP-Clone-FGC | Growth inhibition assay | Inhibition of human LNCaP-Clone-FGC cell growth in a cell viability assay, IC50 = 24.4266 μM. | SANGER | |||
| COLO-824 | Growth inhibition assay | Inhibition of human COLO-824 cell growth in a cell viability assay, IC50 = 24.7279 μM. | SANGER | |||
| NB12 | Growth inhibition assay | Inhibition of human NB12 cell growth in a cell viability assay, IC50 = 25.3007 μM. | SANGER | |||
| NCI-H446 | Growth inhibition assay | Inhibition of human NCI-H446 cell growth in a cell viability assay, IC50 = 25.5814 μM. | SANGER | |||
| YAPC | Growth inhibition assay | Inhibition of human YAPC cell growth in a cell viability assay, IC50 = 26.8375 μM. | SANGER | |||
| K-562 | Growth inhibition assay | Inhibition of human K-562 cell growth in a cell viability assay, IC50 = 27.349 μM. | SANGER | |||
| ATN-1 | Growth inhibition assay | Inhibition of human ATN-1 cell growth in a cell viability assay, IC50 = 28.176 μM. | SANGER | |||
| SCH | Growth inhibition assay | Inhibition of human SCH cell growth in a cell viability assay, IC50 = 28.6137 μM. | SANGER | |||
| MC116 | Growth inhibition assay | Inhibition of human MC116 cell growth in a cell viability assay, IC50 = 29.3019 μM. | SANGER | |||
| ZR-75-30 | Growth inhibition assay | Inhibition of human ZR-75-30 cell growth in a cell viability assay, IC50 = 30.0889 μM. | SANGER | |||
| NCI-H69 | Growth inhibition assay | Inhibition of human NCI-H69 cell growth in a cell viability assay, IC50 = 31.1626 μM. | SANGER | |||
| LB771-HNC | Growth inhibition assay | Inhibition of human LB771-HNC cell growth in a cell viability assay, IC50 = 38.4743 μM. | SANGER | |||
| UMC-11 | Growth inhibition assay | Inhibition of human UMC-11 cell growth in a cell viability assay, IC50 = 52.3124 μM. | SANGER | |||
| Click to View More Cell Line Experimental Data | ||||||
Chemical Information, Storage & Stability
| Molecular Weight | 482.19 | Formula | C16H14F3IN2O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 391210-10-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1=CC(=C(C=C1I)F)NC2=C(C=CC(=C2F)F)C(=O)NOCC(CO)O | ||
Solubility
|
In vitro |
DMSO
: 96 mg/mL
(199.09 mM)
Ethanol : 96 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
%
DMSO
%
%
Tween 80
%
ddH2O
%
DMSO
+
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Mechanism of Action
| Targets/IC50/Ki |
MEK [1]
(Cell-free assay) 0.33 nM
|
|---|---|
| In vitro |
Mirdametinib (PD0325901) shows higher permeability than CI-1040, another MEK inhibitor, and should be able to achieve higher systemic exposures. [1] It is exquisitely specific and highly potent against purified MEK, revealing a Kiapp of 1 nM against activated MEK1 and MEK2. [2] This compound is roughly 500-fold more potent than CI-1040 with respect to its cellular effects on phosphorylation of ERK1 and ERK2, displaying subnanomolar activity. [2] It prevents the growth of melanoma cell lines and inhibits the growth of TPC-1 cells and K2 cells with GI50 of 11 nM and 6.3 nM, respectively. [3] It significantly prevents the growth of PTC cells harboring a BRAF mutation at very low concentration (10 nM) and only moderately increases the growth of the PTC cells carrying the RET/PTC1 rearrangement at the same concentration. It effectively inhibits the phosphorylation of ERK1/2 in multiple PTC cell lines. [3]
|
| Kinase Assay |
In vitro cascade assay
|
|
Incorporation of 32P into myelin basic protein (MBP) is assayed in the presence of a glutathione S-transferase fusion protein containing p44MAP kinase (GST-MAPK) and a glutathione S-transferase protein containing p45MEK (GST-MEK). The assay solution contained 20 mM HEPES, pH 7.4, 10 mM MgCl2, 1 mM MnCl2, 1 mM EGTA, 50 mM [gamma-32P]ATP, 10 mg GST-MEK, 0.5 mg GST-MAPK and 40 mg MBP in a final volume of 100 mL. Reactions are stopped after 20 minutes by addition of trichloroacetic acid and filtered through a GF/C filter mat. 32P retained on the filter mat is determined using a 1205 Betaplate. Mirdametinib (PD0325901) is assessed at various dose ranges in order to determine dose response curves.
|
|
| In vivo |
The improved potency of PD0325901 relative to CI-1040 is evident. A single oral dose of this compound (25 mg/kg) inhibits phosphorylation of ERK by more than 50% at 24 hours post-dosing. In contrast, CI-1040 at a much higher dose (150 mg/kg) only inhibit pERK levels for roughly 8 hours, returning to control levels by 24 hours after treatment. [2] Therefore, the dose required to produce a 70% incidence of complete tumor responses (C26 model) is 25 mg/kg/day versus 900 mg/kg/day for Mirdametinib (PD0325901) and CI-1040, respectively. Anticancer activity of it has been demonstrated for a broad spectrum of human tumor xenografts. [2] After 1 week of oral administration of the compound (20–25 mg/kg/day) in mice, no tumor growth is detected in mice inoculated with PTC cells bearing a BRAF mutation. [3] For PTC with the RET/PTC1 rearrangement, the average tumor volume of the orthotopic tumor is decreased by 58% as compared with controls. In conclusion, PTC cells carrying a BRAF mutation are more sensitive to it than are PTC cells carrying the RET/PTC1 rearrangement. [3]
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-ERK / p-AKT / Cyclin D1 / p27 p-p70S6K(T389) / p4EBP1(S65) / pS6(S235) cleaved PARP DR5 |
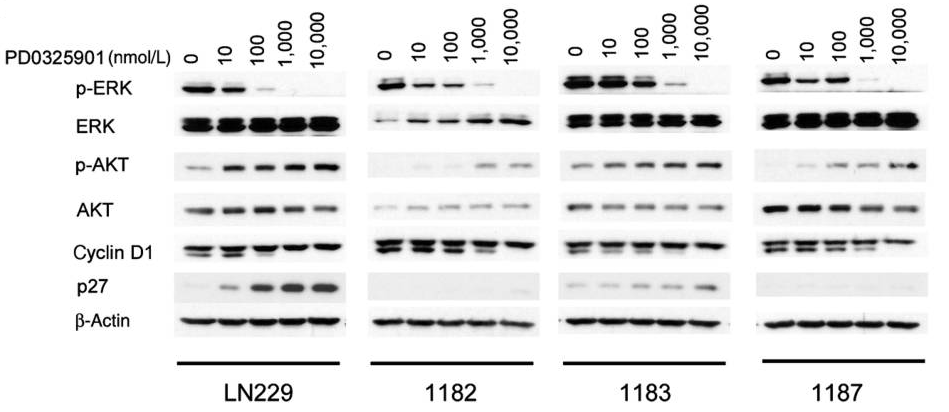
|
22573716 |
| Immunofluorescence | Fibronectin / Actin / pFAK ZO-1 |
|
28187762 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02510001 | Completed | Solid Tumor|Colorectal Cancer |
University of Oxford|Queen''s University Belfast|Oxford University Hospitals NHS Trust|Velindre NHS Trust|University Hospital Antwerp|Hospital Vall d''Hebron|Saint Antoine University Hospital|European Georges Pompidou Hospital|Pfizer|University of Turin Italy|Belfast Health and Social Care Trust|Beaumont Hospital|European Commission|Array BioPharma|University of Paris 5 - Rene Descartes |
November 2014 | Phase 1 |
| NCT02096471 | Completed | Neurofibromatosis Type 1 and Growing or Symptomatic Inoperable PN |
University of Alabama at Birmingham |
June 2014 | Phase 2 |
| NCT02022982 | Completed | KRAS Mutant Non-Small Cell Lung Cancer|Solid Tumors |
Dana-Farber Cancer Institute |
January 2014 | Phase 1 |
| NCT01347866 | Terminated | Advanced Cancer |
Pfizer |
October 2011 | Phase 1 |
| NCT00174369 | Terminated | Carcinoma Non-Small-Cell Lung |
Pfizer |
November 2005 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Whether it interacts with other targets other than MEK?
Answer:
It has very high selectivity to MEK. In addition, this compound can also inhibits VEGF activity according to the reference: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2713590/






































