research use only
U0126-EtOH MEK1/2 inhibitor
Cat.No.S1102
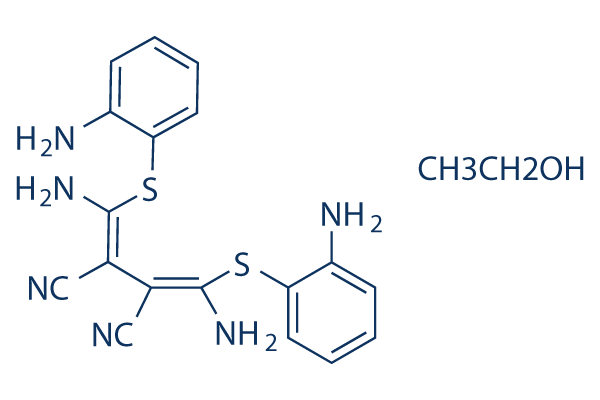
Chemical Structure
Molecular Weight: 426.56
Quality Control
Products Often Used Together with U0126-EtOH
| Related Targets | ERK p38 MAPK Raf JNK Ras KRas S6 Kinase MAP4K TAK1 Mixed Lineage Kinase |
|---|---|
| Other MEK Inhibitors | PD0325901 (Mirdametinib) PD 98059 CI-1040 (PD184352) BIX 02189 Pimasertib (AS-703026) Refametinib (RDEA119) TAK-733 AZD8330 GDC-0623 BIX 02188 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| rat PC12 cells | Function assay | 10 μM | 1 h | Activation of Nrf2/ARE in rat PC12 cells assessed as HO-1 protein induction at 10 uM after 5 hrs pretreated with JNK inhibitor SP600125 for 1 hr before compound addition by Western blot analysis | 21345685 | |
| mouse RAS-3T3 cells | Function assay | 10-40 μM | Inhibition of MEK-mediated ERK1/2 phosphorylation in mouse RAS-3T3 cells at 10 to 40 uM by ELISA. | 24507826 | ||
| HCT116 cells | Function assay | Ability of compound to inhibit anchorage independent colony formation (soft agar growth assay) in HCT116 cells, IC50=19.4 μM. | 15225706 | |||
| COS-7 cells | Function assay | Inhibitory concentration against AP-1 transcription in COS-7 cells, IC50=1 μM. | 15006386 | |||
| HeLa cells | Function assay | Inhibition of EGF-stimulated Elk1-luciferase reporter assay in HeLa cells, IC50=0.29 μM. | 15225706 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 85 mg/mL
(199.26 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 426.56 | Formula | C18H16N6S2.C2H6O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1173097-76-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CCO.C1=CC=C(C(=C1)N)SC(=C(C#N)C(=C(N)SC2=CC=CC=C2N)C#N)N | ||
Mechanism of Action
| Features |
A chemically synthesized and highly selective inhibitor of both MEK1 and MEK2.
|
|---|---|
| Targets/IC50/Ki |
MEK2
(Cell-free assay) 0.06 μM
MEK1
(Cell-free assay) 0.07 μM
|
| In vitro |
U0126-EtOH functionally antagonizes AP- 1 transcriptional activity and blocks the production of a variety of cytokines and metalloproteinases involved in the inflammatory response. This compound inhibits T cell proliferation in response to antigenic stimulation or cross-linked anti-CD3 plus anti-CD28 Abs without effect on IL-2-induced proliferation by down-regulating IL-2 mRNA levels. A recent study shows that this chemical antagonizes resveratrol-induced apoptosis in castration-resistant human prostate cancer C4-2 cells, inhibits mitochondrial function and shifts cells to aerobic glycolysis independently of MEK. |
| Kinase Assay |
In Vitro Kinase Assays
|
|
The amount of immunoprecipitated wild type MEK used in these assays is adjusted to give a similar amount of activity units as obtained with 10 nM recombinant MEK. Reaction velocities are measured using a 96-well nitrocellulose filter apparatus as described below. Unless otherwise noted, reactions are carried out at an enzyme concentration of 10 nM, in 20 mM Hepes, 10 mM MgCl2, 5 mM β-mercaptoethanol, 0.1 mg/mL BSA, pH 7.4, at room temperature. Reactions are initiated by the addition of [γ-33P]ATP into the premixed MEK/ERK/this compound reaction mixture, and an aliquot of 100 μL is taken every 6 minutes and transferred to the 96-well nitrocellulose membrane plate which has 50 mM EDTA to stop the reaction. The membrane plate is drawn and washed 4 times with buffer under vacuum. Wells are then filled with 30 μL of Microscint-20 scintillation fluid, and the radioactivity of 33P-phosphorylated ERK is counted with a Top Count scintillation counter. Velocities are obtained from the slopes of radioactivity versus time plots. Concentrations of ERK and ATP are 400 nM and 40 μM, respectively, unless otherwise indicated. For all of the in vitro enzyme assays, the percent inhibition is calculated 100 (1 −Vi/Vo) where Vi and Vo are the initial reaction velocities in the presence and absence of this chemical, respectively. The data are then plotted as percent inhibition as a function of this compound concentration and fit, by nonlinear least squares regression, to the standard equation for a Langmuir isotherm to determine the IC50. As reported, enzyme concentrations are based upon molecular weights and mass of protein used in the final assay volume and not on active site titration. Thus, the actual enzyme active site concentration may differ from that reported.
|
|
| In vivo |
U0126-EtOH, as the inhibitor of intracellular Raf/MEK/ERK signaling pathway, demonstrates antiviral activity by suppressing propagation of the 2009 pandemic IV H1N1 variant and highly pathogenic avian influenza viruses (HPAIV) in vivo in the mouse lung by inhibiting. This compound shows the potential neuroprotective effect and improving spatial learning in Morris water maze (MWM) by activating peroxisome proliferator-activated receptor gamma coactivator-1a, nuclear respiratory factor 1, and mitochondrial transcription factor A in Aβ-injected rats. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
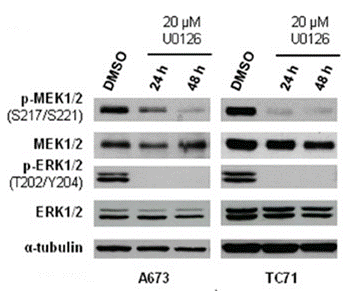
| Western blot | p-MEK / MEK / p-ERK / ERK E-cadherin / Vimentin / Twist-2 |
|
27487136 |
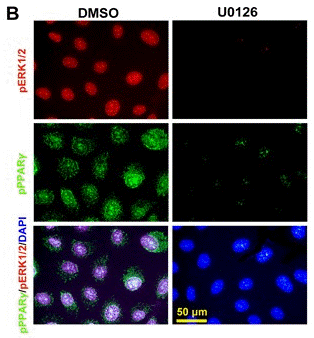
| Immunofluorescence | pERK / pPPARγ E-cadherin / Vimentin CD40 |
|
27145370 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
I want to know whether it is light-sensitive?
Answer:
It is not stable. It should be stored as powder at -20°C, and prepared the solution just before use.






































