- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
LDN-193189
Synonyms: DM3189
LDN-193189 (DM3189) is a selective BMP signaling inhibitor, inhibits the ALK1, ALK2, ALK3 and ALK6 with IC50s of 0.8 nM, 0.8 nM, 5.3 nM and 16.7 nM in the kinase assay, respectively. LDN-193189 inhibits the transcriptional activity of the BMP type I receptors ALK2 and ALK3 with IC50s of 5 nM and 30 nM in C2C12 cells, respectively, exhibits 200-fold selectivity for BMP versus TGF-β. For cell testing, the water-soluble S7507 LDN-193189 2HCl is recommended.This product has poor solubility, animal experiments are available, cell experiments please choose carefully!
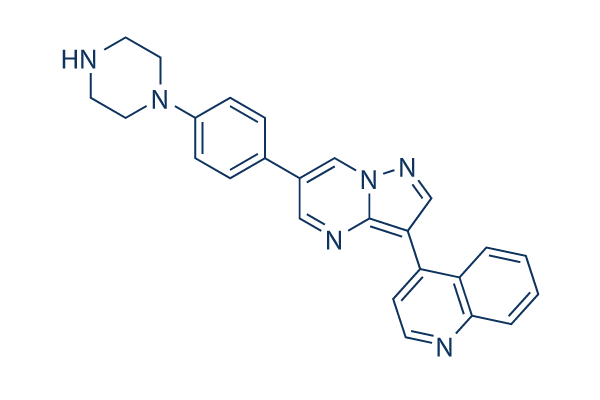
LDN-193189 Chemical Structure
CAS No. 1062368-24-4
Purity & Quality Control
Batch:
Purity:
99.43%
99.43
LDN-193189 Related Products
| Related Targets | ALK1 ALK2 ALK3 ALK4 TGFβRI/ALK5 ALK6 TGFβRII TGF-β Smad3 Smad4 Smad5 ALK7 | Click to Expand |
|---|---|---|
| Related Products | SB431542 Galunisertib (LY2157299) LY2109761 SB525334 LDN-193189 2HCl SIS3 HCl RepSox (E-616452) LY364947 DMH1 SB505124 GW788388 A-83-01 K02288 SD-208 Vactosertib (TEW-7197) SIS3 Kartogenin ITD-1 LDN-212854 ML347 | Click to Expand |
| Related Compound Libraries | Kinase Inhibitor Library PI3K/Akt Inhibitor Library MAPK Inhibitor Library Cell Cycle compound library TGF-beta/Smad compound library | Click to Expand |
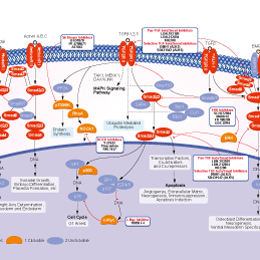
Signaling Pathway
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| C2C12 | Kinase Assay | inhibits the kinase activity of ALK4 and ActRIIA with IC50 values of 101 and 210 nm, respectively | 25368322 | |||
| EOC216 | Growth Inhibition Assay | 0.1-10 μM | 2-10 d | DMSO | induce cell death in a dose-dependent manner | 25227893 |
| OVAC429 | Growth Inhibition Assay | 2/5 μM | 48 h | DMSO | decreases the percentage of cells in S phas | 25227893 |
| SKOV3 | Growth Inhibition Assay | 2/5 μM | 48 h | DMSO | decreases the percentage of cells in S phas | 25227893 |
| OVCA429 | Growth Inhibition Assay | 2/5 μM | 48 h | DMSO | decreases the percentage of cells in S phas | 25227893 |
| EOC219 | Growth Inhibition Assay | 2/5 μM | 48 h | DMSO | decreases the percentage of cells in S phas | 25227893 |
| A549 | Growth Inhibition Assay | 0.5-16 μM | DMSO | inhibits cell growth in a dose-dependent manner | 24778011 | |
| BEAS-2B | Growth Inhibition Assay | 0.5-16 μM | DMSO | inhibits cell growth in a dose-dependent manner | 24778011 | |
| hBMSCs | Function Assay | 0.2 μM | 7 d | abolishes the silibinin-promoted ALP activity partly | 24076187 | |
| PANC-1 | Growth Inhibition Assay | 5-500 nM | 48 h | inhibits cell growth in a dose-dependent manner | 23969729 | |
| MIA PaCa-2 | Growth Inhibition Assay | 5-500 nM | 48 h | inhibits cell growth in a dose-dependent manner | 23969729 | |
| Bx-PC3 | Growth Inhibition Assay | 5-500 nM | 48 h | inhibits cell growth in a dose-dependent manner | 23969729 | |
| PC3 | Function Assay | 500 nM | 2 h | represses activation of Smad1/5/8 and P-Smad3 levels | 22452883 | |
| LNCaP | Function Assay | 0–500 nM | 2 h | reverses rapamycin-induced cell death | 22452883 | |
| EOC | Function Assay | 10-1000 nM | 72 h | reduces the phosphorylated BMP R-Smad1/5/8 | 22249415 | |
| HaCaT | Function Assay | 0.001-10 μM | 2 h | inhibits BMP2-induced phosphorylation of Smad1/5/8 with an IC50 of ~0.005 μM | 21740966 | |
| HaCaT | Function Assay | 0.001-10 μM | 2 h | inhibits the ability of ALK2 to phosphorylate GST-Smad1 with an IC50 of 45 nM | 21740966 | |
| HaCaT | Function Assay | 0.001-10 μM | 2 h | inhibits the ability of ALK3 to phosphorylate Smad1 with an IC50 of 100 nM | 21740966 | |
| HaCaT | Function Assay | 0.001-10 μM | 2 h | inhibits ALK4 and ALK5 with IC50 values of 0.3 μM and 0.5 μM respectively | 21740966 | |
| C2C12 | Function assay | 30 mins | Inhibition of BMP6-induced ALK2 transcriptional activity in mouse C2C12 cells expressing BRE-Luc after 30 mins by luciferase reporter gene assay, EC50 = 0.014 μM. | 30227946 | ||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| fibroblast cell | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for control Hh wild type fibroblast cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| KB-3-1 | Function assay | P-glycoprotein substrates identified in KB-3-1 adenocarcinoma cell line, qHTS therapeutic library screen | 31515284 | |||
| C2C12 | Function assay | 0.1 uM | Inhibition of ALK5 in mouse C2C12 cells assessed as decrease in TGFbeta1-induced Smad1/5 phosphorylation at 0.1 uM by Western blot method | 28103025 | ||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | LDN-193189 (DM3189) is a selective BMP signaling inhibitor, inhibits the ALK1, ALK2, ALK3 and ALK6 with IC50s of 0.8 nM, 0.8 nM, 5.3 nM and 16.7 nM in the kinase assay, respectively. LDN-193189 inhibits the transcriptional activity of the BMP type I receptors ALK2 and ALK3 with IC50s of 5 nM and 30 nM in C2C12 cells, respectively, exhibits 200-fold selectivity for BMP versus TGF-β. For cell testing, the water-soluble S7507 LDN-193189 2HCl is recommended.This product has poor solubility, animal experiments are available, cell experiments please choose carefully! | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | LDN193189 potently inhibits BMP4-mediated Smad1, Smad5 and Smad8 activation with IC50 of 5 nM, and efficiently inhibits transcriptional activity of the BMP type I receptors ALK2 and ALK3 with IC50 of 5 nM and 30 nM, respectively. Furthermore, LDN193189 also shows the inhibitory effect on the transcriptional activity induced by either constitutively active ALK2R206H or ALK2Q207D mutant proteins. [1] A recent study shows that LDN-193189 blocks the production of reactive oxygen species induced by oxidized LDL during atherogenesis in human aortic endothelial cells. [4] |
|||
|---|---|---|---|---|
| Kinase Assay | Alkaline phosphatase activity | |||
| C2C12 cells are seeded into 96-well plates at 2,000 cells per well in DMEM supplemented with 2% FBS. The wells are treated in quadruplicate with BMP ligands and LDN-193189 or vehicle. The cells are collected after 6 days in culture in 50 μL Tris-buffered saline and 1% Triton X-100. The lysates are added to p-nitro-phenylphosphate reagent in 96-well plates for 1 hours and then evaluated alkaline phosphatase activity (absorbance at 405 nm). Cell viability and quantity are measured by Cell Titer Aqueous One (absorbance at 490 nm), using replicate wells treated identically to those used for alkaline phosphatase measurements. | ||||
| Cell Research | Cell lines | PASMCs | ||
| Concentrations | 5 nM | |||
| Incubation Time | 12 h | |||
| Method | Cells were treated with various concnetrations of LDN-193189. |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | pSMAD1 / pSMAD5 / pSMAD8 / SMAD1 / SMAD5 / SMAD8 / ID1 / SMAD4 / PARP / Cleaved PARP |

|
31098401 | |
| Immunofluorescence | Tbx18 / Hcn4 |
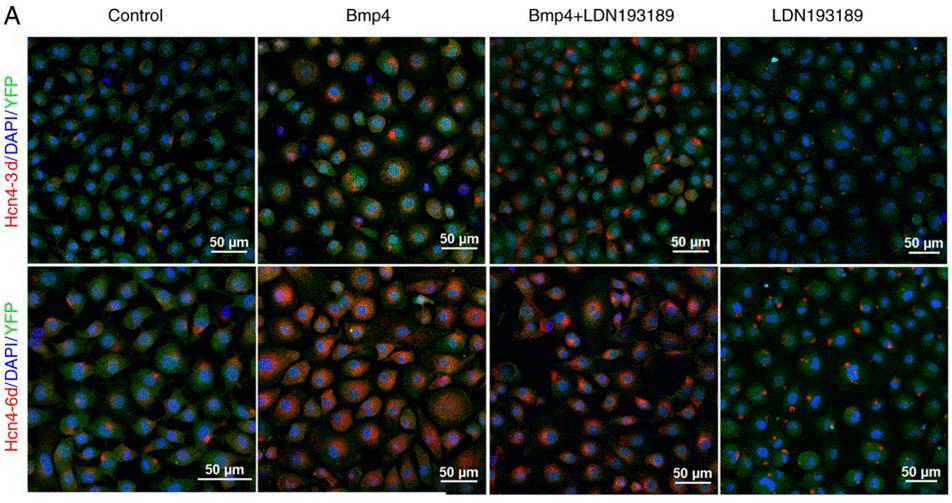
|
30906456 | |
| Growth inhibition assay | Cell viability |
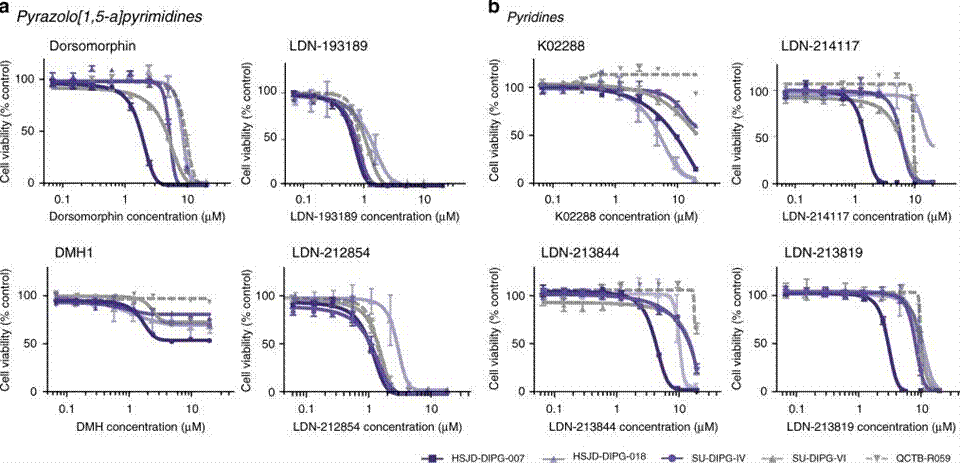
|
31098401 | |
| In Vivo | ||
| In vivo | In conditional caALK2-transgenic mice with Ad.Cre on on postnatal day 7 (P7), LDN-193189 (3 mg/kg i.p) leads to mild calcifications surrounding the left tibia and fibula first visible at P13, and prevents radiographic lesions at P15 without causing weight loss or growth retardation, spontaneous fractures, decreased bone density or behavioral abnormalities. [1] LDN193189 dorsalizes zebrafish embryos by inhibiting signaling pathways induced by bone morphogenetic protein (BMP)6 without effect on vascular development. [2] In PCa-118b tumor-bearing mice, LDN-193189 treatment attenuates tumor growth and reduces bone formation in the tumors. [3] In LDL receptor-deficient (LDLR-/-) mice, LDN-193189 potently inhibits development of atheroma. Moreover, LDN-193189 also exhibits the inhibitory effects on associated vascular inflammation, osteogenic activity, and calcification. [4] |
|
|---|---|---|
| Animal Research | Animal Models | Ad.Cre on P7 is injected into conditional caALK2–transgenic and wild-type mice. |
| Dosages | ≤3 mg/kg | |
| Administration | Administered via i.p. | |
|
Chemical Information & Solubility
| Molecular Weight | 406.48 | Formula | C25H22N6 |
| CAS No. | 1062368-24-4 | SDF | Download LDN-193189 SDF |
| Smiles | C1CN(CCN1)C2=CC=C(C=C2)C3=CN4C(=C(C=N4)C5=CC=NC6=CC=CC=C56)N=C3 | ||
| Storage (From the date of receipt) | |||
|
In vitro |
0.01M HCl : 2.5 mg/mL DMSO : Insoluble ( Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy LDN-193189 | LDN-193189 supplier | purchase LDN-193189 | LDN-193189 cost | LDN-193189 manufacturer | order LDN-193189 | LDN-193189 distributor