research use only
Pacritinib (SB1518) JAK2/FLT3 Inhibitor
Cat.No.S8057
Chemical Structure
Molecular Weight: 472.58
Quality Control
Products Often Used Together with Pacritinib (SB1518)
| Related Targets | EGFR STAT Pim |
|---|---|
| Other JAK Inhibitors | BMS-986165 (Deucravacitinib) AZD1480 WP1066 Momelotinib (CYT387) Filgotinib (GLPG0634) AT9283 Gandotinib (LY2784544) NVP-BSK805 2HCl TG101209 Cerdulatinib (PRT062070) hydrochloride |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MOLM14 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MOLM14 cells harboring FLT3-ITD mutant after 48 hrs by CellTiter-Glo assay, IC50 = 0.079 μM. | 27541357 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 0.29 μM. | 27541357 | ||
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by CellTiter-Glo assay, IC50 = 0.52 μM. | 27541357 | ||
| KMS-12-BM | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KMS-12-BM cells after 48 hrs by CellTiter-Glo assay, IC50 = 0.75 μM. | 27541357 | ||
| PC3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human PC3 cells after 48 hrs by CellTiter-Glo assay, IC50 = 0.77 μM. | 27541357 | ||
| Jurkat | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Jurkat cells after 48 hrs by CellTiter-Glo assay, IC50 = 0.839 μM. | 27541357 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by CellTiter-Glo assay, IC50 = 0.85 μM. | 27541357 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by MTT assay, IC50 = 0.88 μM. | 27541357 | ||
| Jurkat | Antiproliferative assay | Antiproliferative activity against human Jurkat cells, IC50 = 1.09 μM. | 27541357 | |||
| HEL 92.1.7 | Antiproliferative assay | 36 hrs | Antiproliferative activity against HEL 92.1.7 cells harboring JAK2 V617F mutant after 36 hrs by PrestoBlue dye based assay, IC50 = 1.17 μM. | 27541357 | ||
| OPM2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human OPM2 cells after 48 hrs by CellTiter-Glo assay, IC50 = 1.21 μM. | 27541357 | ||
| KHYG | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KHYG cells after 48 hrs by CellTiter-Glo assay, IC50 = 1.24 μM. | 27541357 | ||
| KG1 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KG1 cells after 48 hrs by CellTiter-Glo assay, IC50 = 1.48 μM. | 27541357 | ||
| NKYS | Antiproliferative assay | 48 hrs | Antiproliferative activity against human NKYS cells after 48 hrs by CellTiter-Glo assay, IC50 = 1.6 μM. | 27541357 | ||
| HCT116 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT116 cells after 48 hrs by CellTiter-Glo assay, IC50 = 1.69 μM. | 27541357 | ||
| HEL 92.1.7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against HEL 92.1.7 cells harboring JAK2 V617F mutant after 48 hrs by CellTiter-Glo assay, IC50 = 1.726 μM. | 27541357 | ||
| HL60 | Antiproliferative assay | Antiproliferative activity against human HL60 cells, IC50 = 1.78 μM. | 27541357 | |||
| PC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC3 cells after 72 hrs by MTT assay, IC50 = 2.41 μM. | 27541357 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by MTT assay, IC50 = 2.43 μM. | 27541357 | ||
| TAMH | Antiproliferative assay | 24 hrs | Antiproliferative activity against mouse TAMH cells after 24 hrs by CellTiter-Glo assay, IC50 = 3.68 μM. | 27541357 | ||
| MOLM14 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MOLM14 cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 0.079 μM. | 28953386 | ||
| KMS-12-BM | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KMS-12-BM cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 0.75 μM. | 28953386 | ||
| Jurkat | Antiproliferative assay | Antiproliferative activity against human Jurkat cells, IC50 = 1.09 μM. | 28953386 | |||
| HEL 92.1.7 | Antiproliferative assay | 36 hrs | Antiproliferative activity against human HEL 92.1.7 cells after 36 hrs by PrestoBlue dye based assay, IC50 = 1.17 μM. | 28953386 | ||
| OPM2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human OPM2 cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 1.21 μM. | 28953386 | ||
| KHYG | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KHYG cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 1.24 μM. | 28953386 | ||
| KG1 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human KG1 cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 1.48 μM. | 28953386 | ||
| NKYS | Antiproliferative assay | 48 hrs | Antiproliferative activity against human NKYS cells after 48 hrs by CellTiter-Glo luminescent assay, IC50 = 1.6 μM. | 28953386 | ||
| HL60 | Antiproliferative assay | Antiproliferative activity against human HL60 cells, IC50 = 1.78 μM. | 28953386 | |||
| AC10 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human AC10 cells assessed as cell viability after 24 hrs by CellTiter-Glo assay, IC50 = 2.02 μM. | 28953386 | ||
| TAMH | Cytotoxicity assay | 24 hrs | Cytotoxicity against TAMH cells assessed as cell viability after 24 hrs by CellTiter-Glo assay, IC50 = 3.68 μM. | 28953386 | ||
| KMS-12-BM | Function assay | 2 uM | 3 hrs | Inhibition of JAK2 in IL-6-stimulated human KMS-12-BM cells assessed as suppression of STAT3 phosphorylation at TY705 residue at 2 uM pretreated for 3 hrs followed by IL-6 stimulation by immunoblot method | 27541357 | |
| MOLM14 | Function assay | 0.1 uM | 3 hrs | Inhibition of JAK2 in IL-6-stimulated human MOLM14 cells assessed as suppression of STAT3 phosphorylation at TY705 residue at 0.1 uM pretreated for 3 hrs followed by IL-6 stimulation by immunoblot method | 27541357 | |
| HEL 92.1.7 | Function assay | 1 hr | Induction of JAK2 V617F mutant phosphorylation at Y1007/8 residues in HEL 92.1.7 cells after 1 hr by immunoblot method | 27541357 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 11 mg/mL
(23.27 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 472.58 | Formula | C28H32N4O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 937272-79-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | SB1518 | Smiles | C1CCN(C1)CCOC2=C3COCC=CCOCC4=CC(=CC=C4)C5=NC(=NC=C5)NC(=C3)C=C2 | ||
Mechanism of Action
| Features |
Dual JAK2/FLT3 inhibitor that has progressed to Phase III clinical trials for treatment of Myelofibrosis.
|
|---|---|
| Targets/IC50/Ki |
FLT3 (D835Y)
(Cell-free assay) 6 nM
JAK2 (V617F)
(Cell-free assay) 19 nM
FLT3
(Cell-free assay) 22 nM
JAK2
(Cell-free assay) 23 nM
TYK2
(Cell-free assay) 50 nM
JAK3
(Cell-free assay) 520 nM
|
| In vitro |
Pacritinib is a potent inhibitor of both wild-type JAK2 and JAK2V617F mutant (IC50= 19 nM) that is present in high frequencies among patients with MPD. Relative to JAK2, this compound is two-fold less potent against TYK2 (IC50= 50 nM), 23-fold less potent against JAK3 (IC50= 520 nM) and 56-fold less potent against JAK1 (IC50= 1280 nM). It effectively permeates cells to modulate signaling pathways downstream of JAK2, whether agonist activated or mutationally activated. This chemical induces apoptosis, cell cycle arrest and antiproliferative effects in JAK2WT- and JAK2V617F-dependent cells. It inhibits cell proliferation of Karpas 1106P and Ba/F3-JAK2V617F with IC50 of 348 and 160 nM, respectively. It inhibits endogenous colony growth derived from erythroid and myeloid progenitors with IC50 of 63 and 53 nM , respectively.
SB1518 also inhibits FLT3 and its mutant FLT3-D835Y(IC50= 6 nM ). This compound inhibits FLT3 phosphorylation and downstream STAT, MAPK and PI3K signaling in FLT3-internal-tandem duplication (ITD), FLT3-wt cells and primary AML blast cells. Its treatment leads to a dose-dependent decrease of pFLT3, pSTAT5, pERK1/2 and pAkt in FLT3-ITD harboring MV4-11 cells with IC50 of 80, 40, 33 and 29 nM , respectively. Treatment of the primary AML blast cells with this chemical for 3 h leads to a dose-dependent decrease of pFLT3, pSTAT3 and pSTAT5 with an IC50 below 0.5 μM. It induces apoptosis, cell cycle arrest and anti-proliferative effects in FLT3-mutant and FLT3-wt cells. It inhibits cell proliferation of FLT3-ITD-harboring cells MV4-11 and primary AML blast cells with IC50s of 47 nM and 0.19-1.3 μM, respectively.
|
| Kinase Assay |
kinase activity assays
|
|
All assays are carried out in 384-well white microtiter plates. Compounds are 4-fold serially diluted in 8 steps, starting from 10 μM. The reaction mixture consisted of 25 μL assay buffer (50 mM HEPES pH 7.5, 10 mM MgCl2, 5 mM MnCl2, 1 mM DTT, 0.1 mM Na3VO4, 5 mM β-glycerol phosphate). For FLT3 assays, the reaction contains 2.0 μg/mL FLT3 enzyme, 5 μM of poly(Glu,Tyr) substrate and 4 μM of ATP. For JAK1 assays, the reaction contains 2.5 μg/mL of JAK1 enzyme, 10 μM of poly(Glu,Ala,Tyr) substrate and 1.0 μM of ATP. For JAK2 assays, the reaction contained 0.35 μg/mL of JAK2 enzyme, 10 μM of poly (Glu,Ala,Tyr) substrate and 0.15 μM of ATP. For JAK3 assays, the reaction contained 3.5 μg/mL of JAK3 enzyme, 10 μM of poly (Glu,Ala,Tyr) substrate and 6.0 μM of ATP. For TYK2 assays, the reaction contained 2.5 μg/mL of TYK2 enzyme, 10 μM of poly (Glu,Ala,Tyr) substrate and 0.15 μM of ATP. The reaction is incubated at room temperature for 2 h prior to addition of 13 μL PKLight® detection reagent. After 10 min incubation luminescent signals are read on a multi-label plate reader.
|
|
| In vivo |
Pacritinib administrated at 150 mg/kg p.o. q.d. to JAK2V617F-dependent xenograft model, significantly ameliorates splenomegaly and hepatomegaly symptoms, with 60% normalization of spleen weight and 92% normalization of liver weight and is well tolerated without significant weight loss or any hematological toxicities, including thrombocytopenia and anemia. This compound induces dose-dependent inhibition of tumor growth of JAK2V617F-dependent SET-2 xenograft model (40% for 75 mg/kg and 61% for 150 mg/kg).
This chemical is efficacious in FLT3-ITD-bearing MV4-11 xenograft models. It treated once daily for 21 consecutive days, induces dose-dependent inhibition of tumor growth (38% for 25 mg/kg, 92% for 50 mg/kg and 121% for 100 mg/kg). Complete regression is observed in 3/10 and 8/8 mice for the 50 and 100 mg/kg/day groups, respectively.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | pFLT3 / FLT3 / pSTAT5 / STAT5 / GAPDH p-STAT3Y705 / STAT3 / ACTIN / PARP pSTAT3 Y705 / STAT3 / β-Tubulin pSTAT3 Y705 / STAT3 / Actin / MAPK / p-AKT S473 / AKT pFLT3(Y591) / pSTAT5(Y694) / pERK1、2 / pAkt(T308) / β-Actin pFLT3(Y591) / pSTAT5(Y694) / pERK1、2 / pAkt(T308) / β-Actin |

|
31102119 |
| Growth inhibition assay | Cell viability |
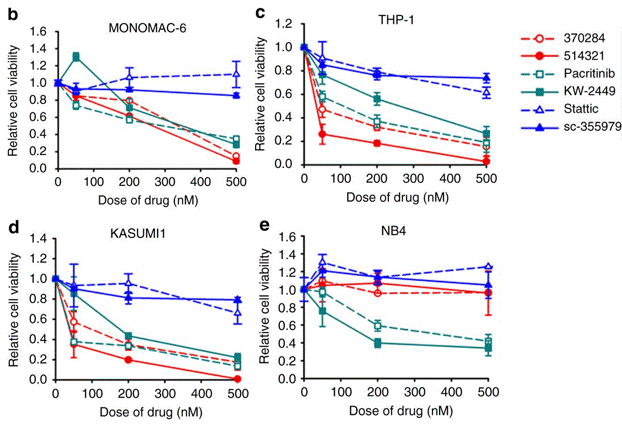
|
29235481 |
| IHC | HE staining of liver sections HE staining of skin grafts p-STAT3 / Ki-67 / mCD31 / VEGF-A / Bcl-2 |

|
29785143 |
| Immunofluorescence | Phalloidin TUNEL |

|
27334834 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06303193 | Not yet recruiting | Myelodysplastic Syndromes |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 1|Phase 2 |
| NCT06052618 | Not yet recruiting | KSHV Inflammatory Cytokine Syndrome (KICS)|Kaposi Sarcoma Herpesvirus -Associated Multicentric Castleman Disease |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 2 |
| NCT06159491 | Not yet recruiting | Chronic Myelomonocytic Leukemia |
Douglas Tremblay|Sobi Inc.|Icahn School of Medicine at Mount Sinai |
January 2 2024 | Phase 1|Phase 2 |
| NCT05531786 | Recruiting | Graft vs Host Disease |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
March 6 2023 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































