research use only
WHI-P154 JAK inhibitor
Cat.No.S2867
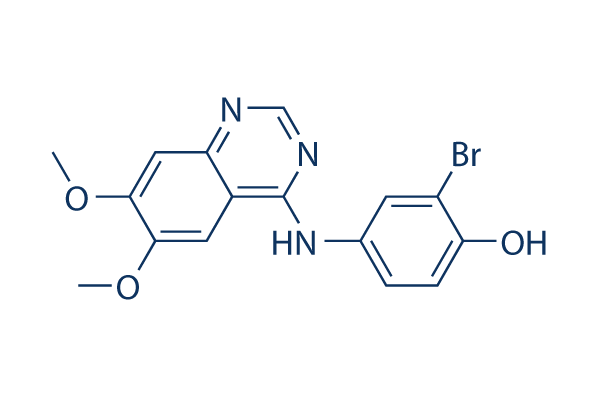
Chemical Structure
Molecular Weight: 376.2
Quality Control
| Related Targets | EGFR STAT Pim |
|---|---|
| Other JAK Inhibitors | BMS-986165 (Deucravacitinib) AZD1480 WP1066 Filgotinib (GLPG0634) Momelotinib (CYT387) AT9283 Gandotinib (LY2784544) Pacritinib (SB1518) TG101209 NVP-BSK805 2HCl |
Solubility
|
In vitro |
DMSO
: 75 mg/mL
(199.36 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 376.2 | Formula | C16H14BrN3O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 211555-04-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | COC1=C(C=C2C(=C1)C(=NC=N2)NC3=CC(=C(C=C3)O)Br)OC | ||
Mechanism of Action
| Targets/IC50/Ki |
EGFR
4 nM
VEGFR
100 nM
Src
100 nM
JAK3
1.8 μM
|
|---|---|
| In vitro |
WHI-P154 is first described as a JAK3 inhibitor that displays no activity at JAK1 or JAK2. This compound inhibits STAT1 activation, iNOS expression and NO production in macrophages in vitro. But it is proved that this chemical also inhibits other common kinases including EGFR, Src, Abl, VEGFR, MAPK and PI3-K and induces apoptosis in human glioblastoma cell lines. It inhibits glioblastoma cell adhesion and migration in the context of ECM. This compound exhibits significant cytotoxicity against U373 and U87 human glioblastoma cell lines, causing apoptotic cell death at micromolar concentrations. The in vitro antiglioblastoma activity of this chemical is amplified > 200-fold and rendered selective by conjugation to recombinant human epidermal growth factor (EGF). In vitro treatment with EGF-P154 results killing of glioblastoma cells at nanomolar concentrations with an IC50 of 813 nM, whereas no cytotoxicity against EGF-R-negative leukemia cellsis observed, even at concentrations as high as 100 mM.
|
| Kinase Assay |
Kinase assays
|
|
WHI-P154 is tested in kinase assays. The panel of kinases is selected to broadly cover the kinome, providing a good approximation of specificity. For all kinases, recombinant rat (IKKβ) or human (all others), full-length or GST-kinase domain fusion proteins, are used. This compound is inactive (concentration that inhibits response by 50% [IC50] > 30 μM) for the following kinases: AKT, AuroraA, cdk2, cdk6, CHK1, FGFR1, GSK3b, IKKb, IKKi, INSR, MAPK1, MAPKAP-K2, MASK, MET, PAK4, PDK1, PKCb, ROCK1, TaoK3, TrkA.
|
|
| In vivo |
The in vivo administration of EGF-P154 results in delayed tumor progression and improved tumor-free survival in a severe combined immunodeficient mouse glioblastoma xenograft model. Whereas none of the control mice remain alive tumor-free beyond 33 days (median tumor-free survival, 19 days) and all control mice have tumors that rapidly progress to reach an average size of > 500 mm3 by 58 days, 40% of mice treated for 10 consecutive days with 1 mg/kg/day this compound remain alive and free of detectable tumors for more than 58 days with a median tumor-free survival of 40 days. The tumors developing in the remaining 60% of the mice never reache a size > 50 mm3.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































