research use only
Ganetespib (STA-9090) HSP90 Inhibitor
Cat.No.S1159
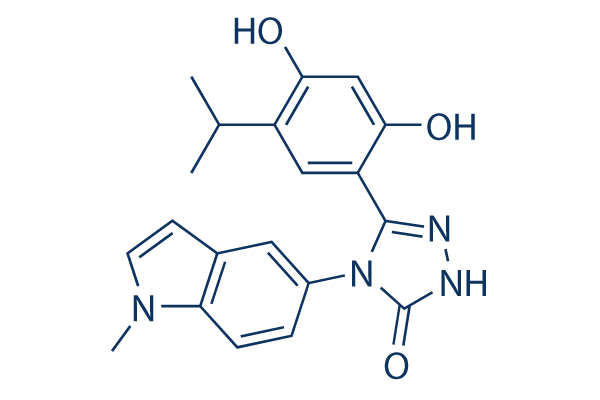
Chemical Structure
Molecular Weight: 364.4
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| NCI-H1975 | Growth Inhibition Assay | 72 h | IC50=8 nM | 22144665 | ||
| NCI-H1975 | Growth Inhibition Assay | 48 h | IC50=16 nM | 22144665 | ||
| Calu-6 | Growth Inhibition Assay | IC50=64 nM | 23012248 | |||
| Calu-1 | Growth Inhibition Assay | IC50=58 nM | 23012248 | |||
| H2122 | Growth Inhibition Assay | IC50=53 nM | 23012248 | |||
| A549 | Growth Inhibition Assay | IC50=43 nM | 23012248 | |||
| H358 | Growth Inhibition Assay | IC50=29 nM | 23012248 | |||
| H1734 | Growth Inhibition Assay | IC50=28 nM | 23012248 | |||
| H727 | Growth Inhibition Assay | IC50=28 nM | 23012248 | |||
| COR-L23 | Growth Inhibition Assay | IC50=22 nM | 23012248 | |||
| H1792 | Growth Inhibition Assay | IC50=20 nM | 23012248 | |||
| H2009 | Growth Inhibition Assay | IC50=19 nM | 23012248 | |||
| SK-LU-1 | Growth Inhibition Assay | IC50=18 nM | 23012248 | |||
| H2212 | Growth Inhibition Assay | IC50=17 nM | 23012248 | |||
| H441 | Growth Inhibition Assay | IC50=14 nM | 23012248 | |||
| H2030 | Growth Inhibition Assay | IC50=12 nM | 23012248 | |||
| H23 | Growth Inhibition Assay | IC50=11 nM | 23012248 | |||
| HOP-62 | Growth Inhibition Assay | IC50=11 nM | 23012248 | |||
| IA-LM | Growth Inhibition Assay | IC50=10 nM | 23012248 | |||
| H460 | Growth Inhibition Assay | IC50=8 nM | 23012248 | |||
| H157 | Growth Inhibition Assay | IC50=7 nM | 23012248 | |||
| H1355 | Growth Inhibition Assay | IC50=5 nM | 23012248 | |||
| VCaP | Growth Inhibition Assay | IC50=7 nM | 23152004 | |||
| LNCaP | Growth Inhibition Assay | IC50=8 nM | 23152004 | |||
| Ramos-RA1 | Growth Inhibition Assay | IC50=7.4 nM | 23303741 | |||
| Karpas-299 | Growth Inhibition Assay | IC50=9.6 nM | 23303741 | |||
| Kasumi-1 | Growth Inhibition Assay | IC50=5.8 nM | 23303741 | |||
| CCRF-CEM (2) | Growth Inhibition Assay | IC50=7.2 nM | 23303741 | |||
| CCRF-CEM (1) | Growth Inhibition Assay | IC50=12.5 nM | 23303741 | |||
| MOLT-4 | Growth Inhibition Assay | IC50=10.6 nM | 23303741 | |||
| RS4;11 | Growth Inhibition Assay | IC50=13.5 nM | 23303741 | |||
| COG-LL-317 | Growth Inhibition Assay | IC50=4.4 nM | 23303741 | |||
| NALM-6 | Growth Inhibition Assay | IC50=11.7 nM | 23303741 | |||
| CHLA-136 | Growth Inhibition Assay | IC50=23.2 nM | 23303741 | |||
| CHLA-90 | Growth Inhibition Assay | IC50=22.3 nM | 23303741 | |||
| NB-EBc1 | Growth Inhibition Assay | IC50=16.8 nM | 23303741 | |||
| NB-1643 | Growth Inhibition Assay | IC50=7.4 nM | 23303741 | |||
| SJ-GBM2 | Growth Inhibition Assay | IC50=12.9 nM | 23303741 | |||
| CHLA-258 | Growth Inhibition Assay | IC50=6.4 nM | 23303741 | |||
| CHLA-10 | Growth Inhibition Assay | IC50=5.7 nM | 23303741 | |||
| CHLA-9 | Growth Inhibition Assay | IC50=4.6 nM | 23303741 | |||
| TC-71 | Growth Inhibition Assay | IC50=4.5 nM | 23303741 | |||
| CHLA-266 | Growth Inhibition Assay | IC50=27.1 nM | 23303741 | |||
| BT-12 | Growth Inhibition Assay | IC50=14.3 nM | 23303741 | |||
| Rh30 | Growth Inhibition Assay | IC50=5.6 nM | 23303741 | |||
| Rh18 | Growth Inhibition Assay | IC50=6.2 nM | 23303741 | |||
| Rh41 | Growth Inhibition Assay | IC50=10.4 nM | 23303741 | |||
| RD | Growth Inhibition Assay | IC50=8 nM | 23303741 | |||
| K033 | Apoptosis Assay | 100 nM | 72 h | significantly induces apoptosis | 23418523 | |
| M23 | Apoptosis Assay | 100 nM | 72 h | significantly induces apoptosis | 23418523 | |
| K029 | Apoptosis Assay | 100 nM | 72 h | significantly induces apoptosis | 23418523 | |
| K028 | Apoptosis Assay | 100 nM | 72 h | significantly induces apoptosis | 23418523 | |
| K008 | Apoptosis Assay | 100 nM | 72 h | significantly induces apoptosis | 23418523 | |
| K033 | Function Assay | 250 nM | 24 h | induces a modest increase in G1 population | 23418523 | |
| M23 | Function Assay | 250 nM | 24 h | induces G1 and G2/M arrest | 23418523 | |
| K029 | Function Assay | 250 nM | 24 h | induces G1 arrest | 23418523 | |
| K028 | Function Assay | 250 nM | 24 h | induces G2 arrest | 23418523 | |
| K008 | Function Assay | 250 nM | 24 h | induces G2 arrest | 23418523 | |
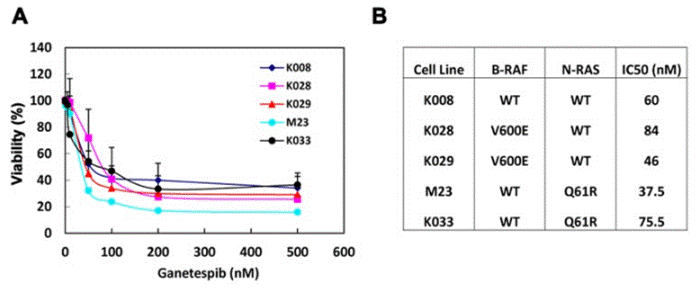
| K033 | Cell Viability Assay | IC50=75.5 nM | 23418523 | |||
| M23 | Cell Viability Assay | IC50=37.5 nM | 23418523 | |||
| K029 | Cell Viability Assay | IC50=46 nM | 23418523 | |||
| K028 | Cell Viability Assay | IC50=84 nM | 23418523 | |||
| K008 | Cell Viability Assay | IC50=60 nM | 23418523 | |||
| H3122 | Cell Viability Assay | 0-1000 nM | 72 h | IC50=10 nM | 23533265 | |
| H2228 | Cell Viability Assay | 0-1000 nM | 72 h | IC50=13 nM | 23533265 | |
| A1847 | Apoptosis Assay | 10-100 nM | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 | |
| OVCAR-8 | Apoptosis Assay | 10-100 nM | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 | |
| OVCAR-5 | Apoptosis Assay | 10-100 nM | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 | |
| SKOV-3 | Cell Viability Assay | 0-1000 nM | 72 h | inhibits cell viability dose dependently | 23900136 | |
| A1847 | Cell Viability Assay | 0-1000 nM | 72 h | inhibits cell viability dose dependently | 23900136 | |
| OVCAR-8 | Cell Viability Assay | 0-1000 nM | 72 h | inhibits cell viability dose dependently | 23900136 | |
| OVCAR-5 | Cell Viability Assay | 0-1000 nM | 72 h | inhibits cell viability dose dependently | 23900136 | |
| H146 | Function Assay | 30 nM | 72 h | induces persistent G2/M phase arrest | 24166505 | |
| GLC4 | Function Assay | 30 nM | 72 h | induces persistent G2/M phase arrest | 24166505 | |
| H82 | Function Assay | 30 nM | 72 h | induces persistent G2/M phase arrest | 24166505 | |
| AC3 | Growth Inhibition Assay | IC50=25.9 nM | 24166505 | |||
| H1173 | Growth Inhibition Assay | IC50=12.62 nM | 24166505 | |||
| H792 | Growth Inhibition Assay | IC50=45.07 nM | 24166505 | |||
| H620 | Growth Inhibition Assay | IC50=32.67 nM | 24166505 | |||
| N592 | Growth Inhibition Assay | IC50=14.12 nM | 24166505 | |||
| H526 | Growth Inhibition Assay | IC50=21.64 nM | 24166505 | |||
| H187 | Growth Inhibition Assay | IC50=24.99 nM | 24166505 | |||
| H146 | Growth Inhibition Assay | IC50=28.51 nM | 24166505 | |||
| H128 | Growth Inhibition Assay | IC50=69.55 nM | 24166505 | |||
| H69 | Growth Inhibition Assay | IC50=83.36 nM | 24166505 | |||
| GLC4 | Growth Inhibition Assay | IC50=20.47 nM | 24166505 | |||
| H82 | Growth Inhibition Assay | IC50=30.27 nM | 24166505 | |||
| MDA-MB-231 | Function Assay | 100 nM | 24 h | inhibits the migratory and invasive capacity | 24173541 | |
| BT-20 | Function Assay | 100/250 nM | 24 h | resulted in a dose-dependent destabilization of EGFR, IGF-IR, MET, and CRAF | 24173541 | |
| MDA-MB-435 | Function Assay | 100 nM | 30 min | inhibits accumulation of HIF-1α | 24248265 | |
| MDA-MB-231 | Function Assay | 100 nM | 30 min | inhibits accumulation of HIF-1α | 24248265 | |
| HCC2998 | Growth Inhibition Assay | IC50=128 nM | 24682747 | |||
| SK-CO-1 | Growth Inhibition Assay | IC50=81 nM | 24682747 | |||
| LS-123 | Growth Inhibition Assay | IC50=73 nM | 24682747 | |||
| SNU-C2B | Growth Inhibition Assay | IC50=45 nM | 24682747 | |||
| LS-1034 | Growth Inhibition Assay | IC50=31 nM | 24682747 | |||
| LoVo | Growth Inhibition Assay | IC50=22 nM | 24682747 | |||
| COLO-678 | Growth Inhibition Assay | IC50=21 nM | 24682747 | |||
| NCI-H747 | Growth Inhibition Assay | IC50=17 nM | 24682747 | |||
| COLO-205 | Growth Inhibition Assay | IC50=14 nM | 24682747 | |||
| HCT 116 | Growth Inhibition Assay | IC50=14 nM | 24682747 | |||
| HuTu-80 | Growth Inhibition Assay | IC50=13 nM | 24682747 | |||
| HCT-15 | Growth Inhibition Assay | IC50=8 nM | 24682747 | |||
| SW620 | Growth Inhibition Assay | IC50=8 nM | 24682747 | |||
| LS-411 N | Growth Inhibition Assay | IC50=5 nM | 24682747 | |||
| RKO | Growth Inhibition Assay | IC50=4 nM | 24682747 | |||
| SW780 | Growth Inhibition Assay | IC50=3451 nM | 24784839 | |||
| RT4 | Growth Inhibition Assay | IC50=1733 nM | 24784839 | |||
| TCCSUP | Growth Inhibition Assay | IC50=142 nM | 24784839 | |||
| MGH-U3 | Growth Inhibition Assay | IC50=53 nM | 24784839 | |||
| HT-1197 | Growth Inhibition Assay | IC50=53 nM | 24784839 | |||
| 5637 | Growth Inhibition Assay | IC50=44 nM | 24784839 | |||
| 35612 | Growth Inhibition Assay | IC50=38 nM | 24784839 | |||
| KU-19-19 | Growth Inhibition Assay | IC50=36 nM | 24784839 | |||
| LB831-BLC | Growth Inhibition Assay | IC50=34 nM | 24784839 | |||
| UM-UC3 | Growth Inhibition Assay | IC50=33 nM | 24784839 | |||
| 647-V | Growth Inhibition Assay | IC50=27 nM | 24784839 | |||
| HT-1376 | Growth Inhibition Assay | IC50=21 nM | 24784839 | |||
| J82 | Growth Inhibition Assay | IC50=18 nM | 24784839 | |||
| BFTC | Growth Inhibition Assay | IC50=17 nM | 24784839 | |||
| SCaBER | Growth Inhibition Assay | IC50=10 nM | 24784839 | |||
| 639-V | Growth Inhibition Assay | IC50=10 nM | 24784839 | |||
| RT112 | Growth Inhibition Assay | IC50=9 nM | 24784839 | |||
| T24 | Growth Inhibition Assay | IC50=7 nM | 24784839 | |||
| SW-1710 | Growth Inhibition Assay | IC50=6 nM | 24784839 | |||
| DSH1 | Growth Inhibition Assay | IC50=6 nM | 24784839 | |||
| CAL27 | Cytoxicity Assay | 10/50 nM | 24 h | decreases cell proliferation dose dependently | 25205430 | |
| Detroit562 | Cytoxicity Assay | 10/50 nM | 24 h | decreases cell proliferation dose dependently | 25205430 | |
| FUDA | Cytoxicity Assay | 10/50 nM | 24 h | decreases cell proliferation dose dependently | 25205430 | |
| SCC25 | Cytoxicity Assay | 10/50 nM | 24 h | decreases cell proliferation dose dependently | 25205430 | |
| HT-29 | Function Assay | 50nM | 24 h | DMSO | induced G0/G1 arrest | 25210794 |
| HCT-116 | Function Assay | 50nM | 24 h | DMSO | induced G0/G1 arrest | 25210794 |
| MGC-803 | Function Assay | 0.1-1000 nM | 24 h | induces G2/M cell-cycle arrest | 25590805 | |
| MKN-28 | Cell Viability Assay | 0.1-1000 nM | 72 h | inhibits cell viability dose dependently | 25590805 | |
| SGC-7901 | Cell Viability Assay | 0.1-1000 nM | 72 h | inhibits cell viability dose dependently | 25590805 | |
| MGC-803 | Cell Viability Assay | 0.1-1000 nM | 72 h | inhibits cell viability dose dependently | 25590805 | |
| MV411 | Apoptosis Assay | 30/80/150/250 nM | 24/48/72 h | induces dose dependant induction of apoptosis | 25882550 | |
| HL60 | Apoptosis Assay | 30/80/150/250 nM | 24/48/72 h | induces dose dependant induction of apoptosis | 25882550 | |
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 40 mg/mL
(109.76 mM)
Ethanol : 9 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 364.4 | Formula | C20H20N4O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 888216-25-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC(C)C1=C(C=C(C(=C1)C2=NNC(=O)N2C3=CC4=C(C=C3)N(C=C4)C)O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
HSP90
(OSA 8 cells) 4 nM
|
|---|---|
| In vitro |
The 50% inhibitory concentrations (IC50) for Ganetespib (STA-9090) against malignant mast cell lines are 10-50 times lower than that for 17-AAG, indicating that triazolone class of HSP90 inhibitors likely exhibits greater potency than geldanamycin based inhibitors. This compound inhibits MG63 cell lines with IC50 of 43 nM. It binds to the ATP-binding domain at the N-terminus of Hsp90 and serves as a potent Hsp90 inhibitor by causing degradation of multiple oncogenic Hsp90 client proteins including HER2/neu, mutated EGFR, Akt, c-Kit, IGF-1R, PDGFRα, Jak1, Jak2, STAT3, STAT5, HIF-1α, CDC2 and c-Met as well as Wilms' tumor 1. At low nanomolar concentrations, it potently arrests cell proliferation and induces apoptosis in a wide variety of human cancer cell lines, including many receptor tyrosine kinase inhibitor- and tanespimycin-resistant cell lines. It exhibits potent cytotoxicity in a range of solid and hematologic tumor cell lines, including those that express mutated kinases that confer resistance to small-molecule tyrosine kinase inhibitors. Its treatment rapidly caused the degradation of known Hsp90 client proteins, exhibits superior potency to the ansamycin inhibitor 17-AAG, and shows sustained activity even with short exposure times. In anohter study, it induces apoptosis of malignant canine mast cell lines. It is active at significantly lower concentrations for C2 and BR canine malignant mast cells with IC50 of 19 and 4 nM, respectively, while 17-AAG inhibits C2 and BR canine malignant mast cells with IC50 of 958 and 44 nM, respectively. Both the expression of WT and mutant Kit are downregulated by 100 nM of this compound after 24 hours in all lines treated including C2 and BMCMCs cells. However, no effects on PI3K or HSP90 expression are observed following treatment with it.
|
| In vivo |
Administration of Ganetespib (STA-9090) leads to significant tumor shrinkage in several tumor xenograft models in mice and appears to be less toxic. Furthermore, this compound demonstrated better tumor penetration compared with tanespimycin. It inhibits in vivo tumor growth in both malignant mast cell and OSA xenograft models. Ganetespib significantly inhibits tumor growth when dosed with two repeating cycles of 25 mg/kg/day for 3 days, with a %T/C value of 18. It is well-tolerated, with the vehicle and Ganetespib groups having average bodyweight changes relative to the start of the study of +0.3% and -8.1% on day 17, respectively.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
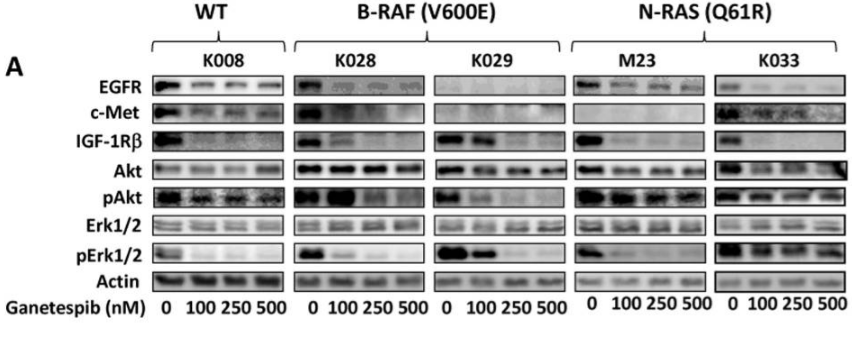
| Western blot | EGFR / c-Met / IGF-1Rβ/ Akt / p-Akt / ERK / p-ERK p27(Kip1) / p21 (Cip1) / Cyclin D1 / Cyclin E / Cyclin B1 / CDK1 / CDK2 / CDK4 Survivin / Bcl-2 / Bcl-xl / Mcl-1 B-RAF / C-RAF / N-RAS HER2 / p-STAT3 / BIM CDK1 / Cyclin D1 / Cyclin B1 / p27 c-PARP / c-caspase 3 / caspase 8 / c-caspase 8 / caspase 9 / c-caspase 9 ErbB2 / pErbB2 / Src / pSrc / mTOR / pmTOR / Bad / pBad / GSK3 / pGSK3 Wee1 / p-Wee1 / Chk1 / p-Chk1 |
|
23418523 |
| Growth inhibition assay | Cell viability |
|
23418523 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02192541 | Terminated | Neoplasms |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
December 2 2014 | Phase 1 |
| NCT02008877 | Completed | Malignant Peripheral Nerve Sheath Tumors (MPNST)|Sarcoma |
Sarcoma Alliance for Research through Collaboration|Synta Pharmaceuticals Corp.|United States Department of Defense |
December 2013 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Does this inhibitor inhibit both isoforms of HSP90?
Answer:
We don't have the information now and it is not very clear in the literature either. From following two references, it indicates that it might be specific to the alpha form “It binds to the ATP binding site of Hsp90 alpha with a Kd of 110 nM” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3477583/






































