research use only
Erastin Ferroptosis inducer
Cat.No.S7242
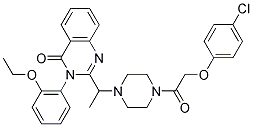
Chemical Structure
Molecular Weight: 547.04
Quality Control
Products Often Used Together with Erastin
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other Ferroptosis Inhibitors | Imidazole Ketone Erastin (IKE) FIN56 UAMC-3203 iFSP1 SRS11-92 icFSP1 SRS16-86 N6F11 Bufotalin Erastin2 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human BJeH cells | Function assay | 6 h | Induction of reactive oxygen species production in human BJeH cells expressing wild type RAS after 6 hrs by DCF-based flow cytometric analysis | 22832321 | ||
| human BJeLR cells | Cytotoxic assay | 10 μM | 12 h | Cytotoxicity against human BJeLR cells expressing RAS G12V mutant at 10 uM at 12 hrs by trypan blue staining | 22832321 | |
| human BJ cells | Function assay | 4.6 μM | Increase in intracellular oxidative species in human BJ cells expressing TERT, LT, ST and RAS G12V mutant genes cells at 4.6 uM | 17568748 | ||
| human BJ cells | Function assay | 9 μM | Induction of cell death in human BJ cells expressing TERT, LT, ST and RAS G12V mutant genes cells at 9 uM. | 17568748 | ||
| human BJ cells | Function assay | Induction of cell death in human BJ cells expressing TERT, LT, ST and RAS G12V mutant genes in presence of U0126 by trypan blue exclusion method, IC50=31.2 μM. | 17568748 | |||
| human A673 cells | Function assay | Induction of cell death in human A673 cells, EC50=30 μM. | 17568748 | |||
| human MX2 cells | Function assay | Induction of cell death in human MX2 cells, EC50=18 μM. | 17568748 | |||
| human HOS cells | Function assay | Induction of cell death in human HOS cells , EC50=17 μM. | 17568748 | |||
| human Hs925.T cells | Function assay | Induction of cell death in human Hs925.T cells, EC50=17 μM. | 17568748 | |||
| human Hs51.T cells | Function assay | Induction of cell death in human Hs51.T cells, EC50=12 μM. | 17568748 | |||
| human EWS502 cells | Function assay | Induction of cell death in human EWS502 cells, EC50=10 μM. | 17568748 | |||
| human BJ cells | Function assay | Induction of cell death in TERT expressing human BJ cells, EC50=10 μM. | 17568748 | |||
| human TC71 cells | Function assay | Induction of cell death in human TC71 cells, EC50=10 μM. | 17568748 | |||
| human U937 cells | Function assay | Induction of cell death in human U937 cells, EC50=10 μM. | 17568748 | |||
| human SK-N-MC cells | Function assay | Induction of cell death in human SK-N-MC cells, EC50=10 μM. | 17568748 | |||
| human TC32 cells | Function assay | Induction of cell death in human TC32 cells | 17568748 | |||
| human U2OS cells | Function assay | Induction of cell death in human U2OS cells, EC50=6 μM. | 17568748 | |||
| human LNCaP cells | Function assay | Induction of cell death in human LNCaP cells, EC50=6 μM. | 17568748 | |||
| human Calu1 cells | Function assay | Inhibition of human Calu1 cells expressing KRAS with activating mutations by trypan blue exclusion assay, IC50=4 μM. | 17568748 | |||
| human SKUT cells | Function assay | Induction of cell death in human SKUT cells, EC50=4 μM. | 17568748 | |||
| human MES-SA cells | Function assay | Induction of cell death in human MES-SA cells, EC50=3 μM. | 17568748 | |||
| human SVR cells | Function assay | Induction of cell death in human SVR cells, EC50=2.5 μM. | 17568748 | |||
| human HT1080 cells | Function assay | Induction of cell death in human HT1080 cells in presence of PD-98059 by trypan blue exclusion method, IC50=1 μM. | 17568748 | |||
| human BJ cells | Function assay | Induction of cell death in human BJ cells expressing TERT, LT, ST and RAS G12V mutant genes cells in presence of PD-98059 by trypan blue exclusion method, IC50=0.9 μM. | 17568748 | |||
| human HeLa cells | Function assay | Induction of cell death in human HeLa cells, EC50=0.6 μM. | 17568748 | |||
| human CCF-STTG1 cells | Function assay | Inhibition of Xct in human CCF-STTG1 cells assessed as glutamate release after 2 hrs by fluorometry, IC50=0.2 μM. | 26231156 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 25 mg/mL
(45.7 mM)
Warmed with 50°C water bath;
Ultrasonicated;
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 547.04 | Formula | C30H31ClN4O4 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 571203-78-6 | Download SDF | Storage of Stock Solutions | Solutions are unstable. Prepare fresh or purchase small, pre-packaged sizes. Repackage upon receipt. | |
| Synonyms | N/A | Smiles | CCOC1=CC=CC=C1N2C(=O)C3=CC=CC=C3N=C2C(C)N4CCN(CC4)C(=O)COC5=CC=C(C=C5)Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Ferroptosis
|
|---|---|
| In vitro |
Erastin is selectively lethal to oncogenic RAS-mutant cell lines, and triggers a unique iron-dependent form of non-apoptotic cell death called ferroptosis. This compound binds directly to VDAC2 and causes mitochondrial damage via ROS production in an NADH-dependent manner, which induces cell death in some tumor cells harbouring activating mutations in the RAS-RAF-MEK pathway. In addition, this chemical, via inducing ROS-mediated CID (Caspase-independent cell death), strongly enhances the effect in WT EGFR cells. |
| In vivo |
Erastin is a ferroptosis activator by inhibiting voltage-dependent anion channels (VDAC2/VDAC3). This compound also inhibits cystine-glutamate antiporter (xCT). |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
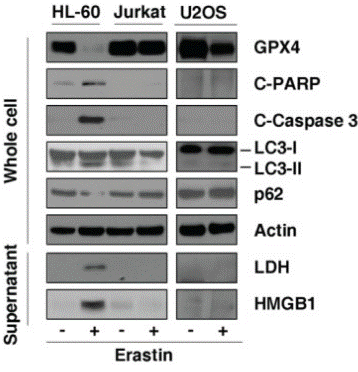
| Western blot | GPX4 / cleaved-PARP / cleaved-caspase3 / LC3 / p62 / LDH / HMGB1 TfR1 / p-JNK / JNK / p-P38 HSPA5 / p-EIF2AK3 GRP78 |
|
27308510 |
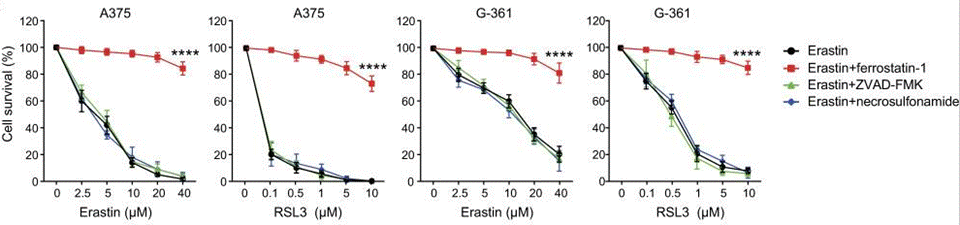
| Growth inhibition assay | Cell survival |
|
29348676 |
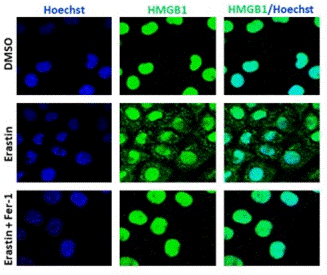
| Immunofluorescence | HMGB1 |
|
31105999 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































