
- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-DYKDDDDK Tag magnetic beads
- Anti-DYKDDDDK Tag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Wnt/beta-catenin
Isoform-selective Products
Signaling Pathway
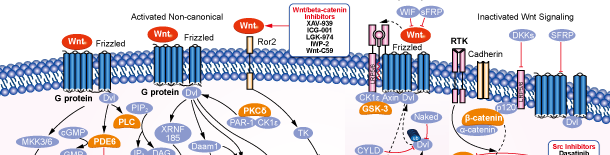
Wnt/beta-catenin Products
- All (66)
- Wnt/beta-catenin Inhibitors (34)
- Wnt/beta-catenin Antibodie (1)
- Wnt/beta-catenin Activators (11)
- Wnt/beta-catenin Antagonists (3)
- Wnt/beta-catenin Agonists (3)
- Wnt/beta-catenin Modulators (3)
- New Wnt/beta-catenin Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
| S1263 | Laduviglusib (CHIR-99021) | Laduviglusib (CHIR-99021, CT99021) is a GSK-3α and GSK-3β inhibitor with IC50 of 10 nM and 6.7 nM, respectively. CHIR99201 does not exhibit cross-reactivity against cyclin-dependent kinases (CDKs) and shows a 350-fold selectivity toward GSK-3β compared to CDKs. CHIR99021 functions as a Wnt/β-catenin activator and induces autophagy. |
|
|
| S2924 | Laduviglusib (CHIR-99021) HCl | Laduviglusib (CHIR-99021; CT99021) HCl is hydrochloride of CHIR-99021, which is a GSK-3α/β inhibitor with IC50 of 10 nM/6.7 nM; CHIR-99021 shows greater than 500-fold selectivity for GSK-3 versus its closest homologs Cdc2 and ERK2. CHIR-99021 is a potent pharmacological activators of the Wnt/beta-catenin signaling pathway. CHIR-99021 significantly rescues light-induced autophagy and augments GR, RORα and autophagy-related proteins. |
|
|
| S1180 | XAV-939 | XAV-939 (NVP-XAV939) selectively inhibits Wnt/β-catenin-mediated transcription through tankyrase1/2 inhibition with IC50 of 11 nM/4 nM in cell-free assays, regulates axin levels and does not affect CRE, NF-κB or TGF-β. |
|
|
| S2662 | ICG-001 | ICG-001 antagonizes Wnt/β-catenin/TCF-mediated transcription and specifically binds to CREB-binding protein (CBP) with IC50 of 3 μM, but is not the related transcriptional coactivator p300. ICG-001 induces apoptosis. |
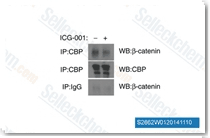
|
|
| S7086 | IWR-1-endo | IWR-1-endo (endo-IWR 1, IWR-1) is a Wnt pathway inhibitor with IC50 of 180 nM in L-cells expressing Wnt3A, induces Axin2 protein levels and promotes β-catenin phosphorylation by stabilizing Axin-scaffolded destruction complexes. |
|
|
| S7085 | IWP-2 | IWP-2 is an inhibitor of Wnt processing and secretion with IC50 of 27 nM in a cell-free assay, selective blockage of Porcn-mediated Wnt palmitoylation, does not affect Wnt/β-catenin in general and displays no effect against Wnt-stimulated cellular responses. IWP-2 specifically inhibits CK1δ. |
|
|
| S2788 | Capmatinib | Capmatinib is a novel, ATP-competitive inhibitor of c-MET with IC50 of 0.13 nM in a cell-free assay, inactive against RONβ, as well as EGFR and HER-3. Capmatinib (INCB28060) inhibits Wnt/β-catenin and EMT signaling pathways and induces apoptosis in diffuse gastric cancer positive for c-MET amplification. Phase 1. |
|
|
| S8320 | SKL2001 | SKL2001 is a novel agonist of the Wnt/β-catenin pathway. It disrupts the Axin/β-catenin interaction. |

|
|
| S8178 | BML-284 (Wnt agonist 1) | BML-284 (Wnt agonist 1) is a cell permeable Wnt signaling pathway activator, which induces β-catenin- and TCF-dependent transcriptional activity with EC50 of 0.7 μM. |
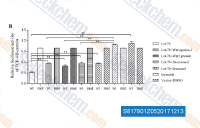
|
|
| S8968 | Foscenvivint (PRI-724) | Foscenvivint (PRI-724) is a potent and specific inhibitor that disrupts the interaction of β-catenin and CBP. | ||
| S8129 | Salinomycin (from Streptomyces albus) | Salinomycin, traditionally used as an anti-coccidial drug, has recently been shown to possess anti-cancer and anti-cancer stem cell (CSC) effects. |
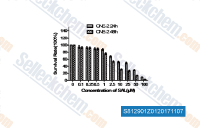
|
|
| S7484 | FH535 | FH535 is a Wnt/β-catenin signaling inhibitor and also a dual PPARγ and PPARδ antagonist. |
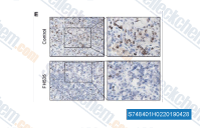
|
|
| S8429 | PNU-74654 | PNU-74654 disrupts the Wnt signaling pathway via inhibition of the interaction (KD50=450 nM) between β-catenin and Tcf4. | ||
| S8474 | LF3 | LF3 is a specific inhibitor of canonical Wnt signaling by disrupting the interaction between β-catenin and TCF4 with an IC50 less than 2 μM. | ||
| S8327 | KYA1797K | KYA1797K is a highly potent and selective Wnt/β-catenin inhibitor with IC50 of 0.75 µM (TOPflash assay). |

|
|
| S7954 | CP21R7 | CP21R7 is a potent and selective GSK-3β inhibitor that can potently activate canonical Wnt signalling. | ||
| S7096 | KY02111 | KY02111 promotes differentiation of hPSCs to cardiomyocytes by inhibiting Wnt signaling, may act downstream of APC and GSK3β. |
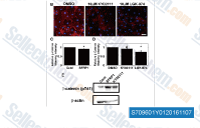
|
|
| S8647 | iCRT3 | iCRT3 is an antagonist of Wnt/β-catenin signaling with an IC50 of 8.2 nM in the Wnt responsive STF16-luc reporter assays. | ||
| S5992 | Heparan Sulfate | Heparan sulfate (HS, Heparitin sulfate, Alpha-idosane, HHS 5, N-Acetylheparan Sulfate, Suleparoid, Tavidan), a constituent of HS proteoglycans (HSPGs), is a linear polysaccharide present on the cell surface. Heparan sulfate influences the binding affinity of intestinal epithelium cells (IECs) to Wnt, thereby promoting activation of canonical Wnt signaling and facilitating regeneration of small intestinal crypts after epithelial injury. | ||
| S7421 | CGP 57380 | CGP 57380 is a potent MNK1 inhibitor with IC50 of 2.2 μM, exhibiting no inhibitory activity on p38, JNK1, ERK1 and -2, PKC, or c-Src-like kinases. CGP 57380 upregulates β-catenin and potentiates radiation-induced apoptosis. |

|
|
| S8761 | Adavivint (SM04690) | Adavivint (SM04690,Lorecivivint) is a potent and specific inhibitor of canonical Wnt signaling with an EC50 of 19.5 nM for inhibiting the TCF/LEF reporter. It is ∼150- to 500-fold more potent than the other known Wnt inhibitors across multiple cellular assays. | ||
| S6901 | MSAB | MSAB is a selective inhibitor of Wnt/β-catenin signaling that binds to β-catenin, promoting its degradation, and specifically downregulates Wnt/β-catenin target genes. MSAB shows potent anti-tumor effects. | ||
| S3842 | Isoquercitrin | Isoquercitrin (Hirsutrin, 3-Glucosylquercetin, Quercetin 3-o-glucopyranoside), a flavonoid compound with anticancer activity isolated from Bidens bipinnata L, is an inhibitor of Wnt/β-catenin that acts downstream of the β-catenin nuclear translocation. | ||
| S8392 | NCB-0846 | NCB-0846 is a novel, orally small-molecule Wnt inhibitor that inhibits TNIK (TRAF2 and NCK-Interacting Kinase, MAP4K7) with an IC50 value of 21 nM. | ||
| S8248 | IQ-1 | IQ-1 enables Wnt/β-catenin-driven expansion of mouse ESCs and prevents spontaneous differentiation, Maintains embryonic stem cell (ESC) pluripotency in Wnt3a-treated cells and prevents PP2A/Nkd interaction. | ||
| S8704 | iCRT14 | iCRT14 is a β-catenin/Tcf inhibitor with a Ki value of 54 ± 5.2 μM in homogeneous fluorescence polarization (FP) assay. | ||
| S7981 | CCT251545 | CCT251545 is a potent, orally bioavailable inhibitor of WNT signaling with IC50 of 5 nM in 7dF3 cells. CCT251545 also act as a selective chemical probe for exploring the role of CDK8 and CDK19 in human disease. | ||
| S7490 | WIKI4 | WIKI4 is a novel Tankyrase inhibitor with IC50 of 15 nM for TNKS2, and leads to inhibition of Wnt/beta-catenin signaling. | ||
| S7914 | Isoxazole 9 (ISX-9) | Isoxazole 9 (Isx-9) is a synthetic promotor of adult neurogenesis by triggering neuronal differentiation of adult neural stem/precursor cells (NSPCs). Isoxazole 9 (Isx-9) activates multiple pathways including TGF-β induced epithelial–mesenchymal transition (EMT) signaling, canonical and non-canonical Wnt signaling at different stages of cardiac differentiation. | ||
| S6745 | JW55 | JW55 is a potent and selective inhibitor of the canonical Wnt pathway that functions via inhibition of the PARP domain of tankyrase 1 and tankyrase 2 (TNKS1/2). | ||
| S3630 | Methyl Vanillate | Methyl Vanillate, one of the ingredients in Hovenia dulcis Thunb, activates the Wnt/β-catenin pathway and induces osteoblast differentiation in vitro. | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S9965 | M435-1279 | M435-1279 is a UBE2T inhibitor, inhibiting the Wnt/β-catenin signaling pathway hyperactivation through blocking UBE2T-mediated degradation of RACK1. | ||
| S6898 | RCM-1 | RCM-1 is a nontoxic inhibitor of Forkhead box M1 (FOXM1) that suppresses goblet cell metaplasia and prevents IL-13 and STAT6 signaling in allergen-exposed mice. RCM-1 decreases carcinogenesis and nuclear β-catenin. | ||
| S5815 | WAY-316606 | WAY-316606, a specific antagonist of SFRP1, functions as an activator of canonical Wnt/β-catenin signalling in the human hair bulb. | ||
| S9828 | WAY-262611 | WAY-262611 (BML-WN110, Compound 5) is a beta-catenin (β-Catenin) agonist that increases bone formation rate with EC50 of 0.63 μM in TCF-Luciferase assay. WAY-262611 is also an inhibitor of dickkopf 1 (DKK1). | ||
| E1731 | JW74 | JW74 is a specific inhibitor of the canonical Wnt signaling with IC50 790 nM in the ST-Luc assay. It inhibit the growth of tumor cells in both mouse xenograft model of colorectal cancer and in ApcMin mice. | ||
| S6961 | Foxy-5 | Foxy-5, a Wnt-5a mimicking hexapeptide, is a WNT signalling pathway modulator potentially for the treatment of metastatic breast cancer, prostate cancer and colorectal cancer. | ||
| F0012 | Anti-β-Actin Rabbit Antibody [C21N23] | ACTB,beta Actin,beta Actin Loading Control,beta-Actin,β-Actin | ||
| F0336 | Anti-Non-phospho β-Catenin (S33/37/T41) Rabbit Antibody [E13A9] | |||
| F0487 | Anti-Non-phospho β-Catenin (Ser45) Rabbit Antibody [K20N13] | |||
| F1056New | Anti-Wnt3a Rabbit Antibody [C3D14] | Proto-oncogene Int-4 homolog, WNT3 | ||
| F0613 | Anti-Wnt5a/b Rabbit Antibody [K19B14] | Wnt-5b, WNT5B, Wnt-5a, WNT5A | ||
| F0494New | Anti-Phospho-β-Catenin (Ser552) Rabbit Antibody [K9A16] | |||
| F0265New | Anti-TCF4/TCF7L2 Rabbit Antibody [P1D24] | TCF-4,TCF4/TCF7L2,TCF7L2 | ||
| F1661New | Anti-DKK1 Rabbit Antibody [K16J6] | DKK1,Dkk-1 | ||
| F0534 | Anti-TCF3/TCF7L1 Rabbit Antibody [G18A14] | |||
| F2477New | Anti-Wnt2/IRP Rabbit Antibody [B13H8] | |||
| S3223 | L-Quebrachitol | L-Quebrachitol (L-QCT), a natural product isolated from many plants, promotes proliferation and cell DNA synthesis. L-Quebrachitol upregulates bone morphogenetic protein-2 (BMP-2) and runt-related transcription factor-2 (Runx2) and regulatory genes associated with mitogen-activated protein kinase (MAPK) and Wnt/β-catenin signaling pathway, while down-regulating the receptor activator of the nuclear factor-κB(NF-κB) ligand (RANKL) mRNA level. | ||
| E1486 | Zamaporvint (RXC004) | Zamaporvint (RXC004) is an orally active and selective inhibitor of Wnt. It targets membrane-bound o-acyltransferase Porcupine and inhibits Wnt ligand palmitoylation, secretion, and pathway activation. RXC004 exhibits potent antiproliferative effects in Wnt ligand-dependent colorectal and pancreatic cell lines and possesses multiple antitumor mechanisms. | ||
| E1069 | M2912 | M2912 (MSC2504877) is a very potent TNKS1/TNKS2 inhibitor (IC50=0.6 nM for TNKS1) with exquisite selectivity over other PARP family enzymes and favorable compound properties. This inhibitor potently modulates the Wnt/β-catenin pathway by elevating the levels of axin2 (EC50=17 nM) and tankyrase in DLD1 cells in a dose-dependent manner resulting in reduced cellular Wnt reporter activity. | ||
| S0733 | Tegatrabetan (BC-2059) | Tegatrabetan (BC2059,Tegavivint) is an antagonist of β-Catenin. Tegatrabetan (BC2059) treatment disrupts the binding of β-catenin with the scaffold protein transducin β-like 1 (TBL1) and proteasomal degradation and declines in the nuclear levels of β-catenin. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| E0769 | Ginsenoside Rh4 | Ginsenoside Rh4 is an important active ingredient of traditional Chinese medicine ginseng, which has been shown to inhibit Wnt/β-Catenin, JAK2/STAT3, TGF-β/Smad2/3 and other signaling pathways. | ||
| S8462New | Toxoflavin | PKF118-310 is a potent antagonist of transcription factor 4 (TCF4)/β-catenin signaling, and also an inhibitor of KDM4A . It exhibits anticancer activity and can be used in solid tumor research. | ||
| A2489 | Vantictumab (Anti-FZD) | Vantictumab (Anti-FZD) is a monoclonal antibody targeting the Wnt signaling pathway with potential antineoplastic activity. Vantictumab binds to certain receptors in the Wnt signaling pathway thereby preventing the activation of the Wnt signaling pathway. This results in an inhibition of cancer stem cell (CSC) activity and a subsequent inhibition of cancer cell proliferation. MW: 145.5 KD. | ||
| E0072 | Indirubin-3′-oxime | Indirubin-3′-oxime (IDR3O, I3O) is an indirubin analogue that shows favorable inhibitory activity targeting GSK-3β and CDKs. Indirubin-3′-oxime also inhibits JNKs with IC50s of 0.8 μM, 1.4 μM, and 1.0 μM for JNK1, JNK2, and JNK3, respectively. Indirubin-3′-oxime activates Wnt/β-catenin signaling and inhibits adipocyte differentiation and obesity. | ||
| S8233 | Prodigiosin | Prodigiosin is a natural red pigment produced by numerous bacterial species which can inhibit Wnt/β-catenin pathway. | ||
| E4800New | BML-284 | BML-284 (Wnt Agonist) is a potent and cell-permeable activator of Wnt signaling that induces TCF-dependent transcriptional activity with an EC50 value of 0.7 µM. BML-284 also acts as an agonist of β-catenin that effectively activates direct Wnt-β-catenin signaling to induce adipocyte dedifferentiation while circumventing the apoptotic effect in the hypertonic treatment. | ||
| E6057New | Pamidronate disodium pentahydrate | Pamidronate disodium pentahydrate is a nitrogen-containing bisphosphonate and used to prevent bone loss due to steroid use like glucocorticoid-induced low bone mineral density in children or to inhibit calcium release from bone by impairing osteoclast-mediated bone resorption. It also inhibits the Wnt/β-catenin signaling pathway that controls osteogenic differentiation in bone marrow mesenchymal stem cells (BMMSCs). | ||
| S6085 | KY19382 (A3051) | KY19382 (A3051) is a Wnt/β-catenin signalling activator through inhibitory effects on both CXXC5–DVL interaction and GSK3β activity with IC50s of 19 nM and 10 nM, respectively. | ||
| S9416 | Triptonide | Triptonide, isolated from the roots of Tripterygium wilfordii, can effectively inhibit canonical Wnt/β-catenin signaling by targeting the downstream C-terminal transcription domain of β-catenin or a nuclear component associated with β-catenin. It is also an antineoplastic agent, an anti-inflammatory agent and an immunosuppressive agent. | ||
| E0033 | IWP-4 | IWP-4 is a potent Wnt/β-catenin signaling pathway inhibitor with an IC50 of 25 nM. |
||
| E2376 | KY-05009 | KY-05009 is an ATP-competitive Traf2- and Nck-interacting kinase (TNIK) inhibitor with a Ki of 100 nM, which inhibits the protein expression of TNIK and transcriptional activity of Wnt target genes and induces apoptosis in cancer cells. | ||
| S0458 | KY1220 | KY1220 is a small molecule that destabilizes both β-catenin and Ras, via targeting the Wnt/β-catenin pathway with IC50 of 2.1 μM in HEK293 reporter cells. | ||
| F0297New | Anti-Phospho-β-Catenin (Ser675) Rabbit Antibody [G12L16] | |||
| S1180 | XAV-939 | XAV-939 (NVP-XAV939) selectively inhibits Wnt/β-catenin-mediated transcription through tankyrase1/2 inhibition with IC50 of 11 nM/4 nM in cell-free assays, regulates axin levels and does not affect CRE, NF-κB or TGF-β. |

|
|
| S2662 | ICG-001 | ICG-001 antagonizes Wnt/β-catenin/TCF-mediated transcription and specifically binds to CREB-binding protein (CBP) with IC50 of 3 μM, but is not the related transcriptional coactivator p300. ICG-001 induces apoptosis. |

|
|
| S7086 | IWR-1-endo | IWR-1-endo (endo-IWR 1, IWR-1) is a Wnt pathway inhibitor with IC50 of 180 nM in L-cells expressing Wnt3A, induces Axin2 protein levels and promotes β-catenin phosphorylation by stabilizing Axin-scaffolded destruction complexes. |

|
|
| S7085 | IWP-2 | IWP-2 is an inhibitor of Wnt processing and secretion with IC50 of 27 nM in a cell-free assay, selective blockage of Porcn-mediated Wnt palmitoylation, does not affect Wnt/β-catenin in general and displays no effect against Wnt-stimulated cellular responses. IWP-2 specifically inhibits CK1δ. |

|
|
| S2788 | Capmatinib | Capmatinib is a novel, ATP-competitive inhibitor of c-MET with IC50 of 0.13 nM in a cell-free assay, inactive against RONβ, as well as EGFR and HER-3. Capmatinib (INCB28060) inhibits Wnt/β-catenin and EMT signaling pathways and induces apoptosis in diffuse gastric cancer positive for c-MET amplification. Phase 1. |

|
|
| S8968 | Foscenvivint (PRI-724) | Foscenvivint (PRI-724) is a potent and specific inhibitor that disrupts the interaction of β-catenin and CBP. | ||
| S8129 | Salinomycin (from Streptomyces albus) | Salinomycin, traditionally used as an anti-coccidial drug, has recently been shown to possess anti-cancer and anti-cancer stem cell (CSC) effects. |

|
|
| S7484 | FH535 | FH535 is a Wnt/β-catenin signaling inhibitor and also a dual PPARγ and PPARδ antagonist. |

|
|
| S8429 | PNU-74654 | PNU-74654 disrupts the Wnt signaling pathway via inhibition of the interaction (KD50=450 nM) between β-catenin and Tcf4. | ||
| S8474 | LF3 | LF3 is a specific inhibitor of canonical Wnt signaling by disrupting the interaction between β-catenin and TCF4 with an IC50 less than 2 μM. | ||
| S8327 | KYA1797K | KYA1797K is a highly potent and selective Wnt/β-catenin inhibitor with IC50 of 0.75 µM (TOPflash assay). |

|
|
| S7096 | KY02111 | KY02111 promotes differentiation of hPSCs to cardiomyocytes by inhibiting Wnt signaling, may act downstream of APC and GSK3β. |

|
|
| S8761 | Adavivint (SM04690) | Adavivint (SM04690,Lorecivivint) is a potent and specific inhibitor of canonical Wnt signaling with an EC50 of 19.5 nM for inhibiting the TCF/LEF reporter. It is ∼150- to 500-fold more potent than the other known Wnt inhibitors across multiple cellular assays. | ||
| S6901 | MSAB | MSAB is a selective inhibitor of Wnt/β-catenin signaling that binds to β-catenin, promoting its degradation, and specifically downregulates Wnt/β-catenin target genes. MSAB shows potent anti-tumor effects. | ||
| S3842 | Isoquercitrin | Isoquercitrin (Hirsutrin, 3-Glucosylquercetin, Quercetin 3-o-glucopyranoside), a flavonoid compound with anticancer activity isolated from Bidens bipinnata L, is an inhibitor of Wnt/β-catenin that acts downstream of the β-catenin nuclear translocation. | ||
| S8392 | NCB-0846 | NCB-0846 is a novel, orally small-molecule Wnt inhibitor that inhibits TNIK (TRAF2 and NCK-Interacting Kinase, MAP4K7) with an IC50 value of 21 nM. | ||
| S8248 | IQ-1 | IQ-1 enables Wnt/β-catenin-driven expansion of mouse ESCs and prevents spontaneous differentiation, Maintains embryonic stem cell (ESC) pluripotency in Wnt3a-treated cells and prevents PP2A/Nkd interaction. | ||
| S8704 | iCRT14 | iCRT14 is a β-catenin/Tcf inhibitor with a Ki value of 54 ± 5.2 μM in homogeneous fluorescence polarization (FP) assay. | ||
| S7981 | CCT251545 | CCT251545 is a potent, orally bioavailable inhibitor of WNT signaling with IC50 of 5 nM in 7dF3 cells. CCT251545 also act as a selective chemical probe for exploring the role of CDK8 and CDK19 in human disease. | ||
| S7490 | WIKI4 | WIKI4 is a novel Tankyrase inhibitor with IC50 of 15 nM for TNKS2, and leads to inhibition of Wnt/beta-catenin signaling. | ||
| S6745 | JW55 | JW55 is a potent and selective inhibitor of the canonical Wnt pathway that functions via inhibition of the PARP domain of tankyrase 1 and tankyrase 2 (TNKS1/2). | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S9965 | M435-1279 | M435-1279 is a UBE2T inhibitor, inhibiting the Wnt/β-catenin signaling pathway hyperactivation through blocking UBE2T-mediated degradation of RACK1. | ||
| S6898 | RCM-1 | RCM-1 is a nontoxic inhibitor of Forkhead box M1 (FOXM1) that suppresses goblet cell metaplasia and prevents IL-13 and STAT6 signaling in allergen-exposed mice. RCM-1 decreases carcinogenesis and nuclear β-catenin. | ||
| E1731 | JW74 | JW74 is a specific inhibitor of the canonical Wnt signaling with IC50 790 nM in the ST-Luc assay. It inhibit the growth of tumor cells in both mouse xenograft model of colorectal cancer and in ApcMin mice. | ||
| E1486 | Zamaporvint (RXC004) | Zamaporvint (RXC004) is an orally active and selective inhibitor of Wnt. It targets membrane-bound o-acyltransferase Porcupine and inhibits Wnt ligand palmitoylation, secretion, and pathway activation. RXC004 exhibits potent antiproliferative effects in Wnt ligand-dependent colorectal and pancreatic cell lines and possesses multiple antitumor mechanisms. | ||
| E1069 | M2912 | M2912 (MSC2504877) is a very potent TNKS1/TNKS2 inhibitor (IC50=0.6 nM for TNKS1) with exquisite selectivity over other PARP family enzymes and favorable compound properties. This inhibitor potently modulates the Wnt/β-catenin pathway by elevating the levels of axin2 (EC50=17 nM) and tankyrase in DLD1 cells in a dose-dependent manner resulting in reduced cellular Wnt reporter activity. | ||
| S5554 | Lanatoside C | Lanatoside C is a cardiac glycoside with antiviral and anti-tumor activity. Lanatoside C induces G2/M cell cycle arrest and induces autophagy and apoptosis via attenuating MAPK, Wnt, JAK-STAT, and PI3K/AKT/mTOR signaling pathways. | ||
| E0769 | Ginsenoside Rh4 | Ginsenoside Rh4 is an important active ingredient of traditional Chinese medicine ginseng, which has been shown to inhibit Wnt/β-Catenin, JAK2/STAT3, TGF-β/Smad2/3 and other signaling pathways. | ||
| S8233 | Prodigiosin | Prodigiosin is a natural red pigment produced by numerous bacterial species which can inhibit Wnt/β-catenin pathway. | ||
| E6057New | Pamidronate disodium pentahydrate | Pamidronate disodium pentahydrate is a nitrogen-containing bisphosphonate and used to prevent bone loss due to steroid use like glucocorticoid-induced low bone mineral density in children or to inhibit calcium release from bone by impairing osteoclast-mediated bone resorption. It also inhibits the Wnt/β-catenin signaling pathway that controls osteogenic differentiation in bone marrow mesenchymal stem cells (BMMSCs). | ||
| S9416 | Triptonide | Triptonide, isolated from the roots of Tripterygium wilfordii, can effectively inhibit canonical Wnt/β-catenin signaling by targeting the downstream C-terminal transcription domain of β-catenin or a nuclear component associated with β-catenin. It is also an antineoplastic agent, an anti-inflammatory agent and an immunosuppressive agent. | ||
| E0033 | IWP-4 | IWP-4 is a potent Wnt/β-catenin signaling pathway inhibitor with an IC50 of 25 nM. |
||
| E2376 | KY-05009 | KY-05009 is an ATP-competitive Traf2- and Nck-interacting kinase (TNIK) inhibitor with a Ki of 100 nM, which inhibits the protein expression of TNIK and transcriptional activity of Wnt target genes and induces apoptosis in cancer cells. | ||
| A2489 | Vantictumab (Anti-FZD) | Vantictumab (Anti-FZD) is a monoclonal antibody targeting the Wnt signaling pathway with potential antineoplastic activity. Vantictumab binds to certain receptors in the Wnt signaling pathway thereby preventing the activation of the Wnt signaling pathway. This results in an inhibition of cancer stem cell (CSC) activity and a subsequent inhibition of cancer cell proliferation. MW: 145.5 KD. | ||
| S1263 | Laduviglusib (CHIR-99021) | Laduviglusib (CHIR-99021, CT99021) is a GSK-3α and GSK-3β inhibitor with IC50 of 10 nM and 6.7 nM, respectively. CHIR99201 does not exhibit cross-reactivity against cyclin-dependent kinases (CDKs) and shows a 350-fold selectivity toward GSK-3β compared to CDKs. CHIR99021 functions as a Wnt/β-catenin activator and induces autophagy. |

|
|
| S2924 | Laduviglusib (CHIR-99021) HCl | Laduviglusib (CHIR-99021; CT99021) HCl is hydrochloride of CHIR-99021, which is a GSK-3α/β inhibitor with IC50 of 10 nM/6.7 nM; CHIR-99021 shows greater than 500-fold selectivity for GSK-3 versus its closest homologs Cdc2 and ERK2. CHIR-99021 is a potent pharmacological activators of the Wnt/beta-catenin signaling pathway. CHIR-99021 significantly rescues light-induced autophagy and augments GR, RORα and autophagy-related proteins. |

|
|
| S8178 | BML-284 (Wnt agonist 1) | BML-284 (Wnt agonist 1) is a cell permeable Wnt signaling pathway activator, which induces β-catenin- and TCF-dependent transcriptional activity with EC50 of 0.7 μM. |

|
|
| S7954 | CP21R7 | CP21R7 is a potent and selective GSK-3β inhibitor that can potently activate canonical Wnt signalling. | ||
| S7421 | CGP 57380 | CGP 57380 is a potent MNK1 inhibitor with IC50 of 2.2 μM, exhibiting no inhibitory activity on p38, JNK1, ERK1 and -2, PKC, or c-Src-like kinases. CGP 57380 upregulates β-catenin and potentiates radiation-induced apoptosis. |

|
|
| S7914 | Isoxazole 9 (ISX-9) | Isoxazole 9 (Isx-9) is a synthetic promotor of adult neurogenesis by triggering neuronal differentiation of adult neural stem/precursor cells (NSPCs). Isoxazole 9 (Isx-9) activates multiple pathways including TGF-β induced epithelial–mesenchymal transition (EMT) signaling, canonical and non-canonical Wnt signaling at different stages of cardiac differentiation. | ||
| S3630 | Methyl Vanillate | Methyl Vanillate, one of the ingredients in Hovenia dulcis Thunb, activates the Wnt/β-catenin pathway and induces osteoblast differentiation in vitro. | ||
| S3223 | L-Quebrachitol | L-Quebrachitol (L-QCT), a natural product isolated from many plants, promotes proliferation and cell DNA synthesis. L-Quebrachitol upregulates bone morphogenetic protein-2 (BMP-2) and runt-related transcription factor-2 (Runx2) and regulatory genes associated with mitogen-activated protein kinase (MAPK) and Wnt/β-catenin signaling pathway, while down-regulating the receptor activator of the nuclear factor-κB(NF-κB) ligand (RANKL) mRNA level. | ||
| E0072 | Indirubin-3′-oxime | Indirubin-3′-oxime (IDR3O, I3O) is an indirubin analogue that shows favorable inhibitory activity targeting GSK-3β and CDKs. Indirubin-3′-oxime also inhibits JNKs with IC50s of 0.8 μM, 1.4 μM, and 1.0 μM for JNK1, JNK2, and JNK3, respectively. Indirubin-3′-oxime activates Wnt/β-catenin signaling and inhibits adipocyte differentiation and obesity. | ||
| E4800New | BML-284 | BML-284 (Wnt Agonist) is a potent and cell-permeable activator of Wnt signaling that induces TCF-dependent transcriptional activity with an EC50 value of 0.7 µM. BML-284 also acts as an agonist of β-catenin that effectively activates direct Wnt-β-catenin signaling to induce adipocyte dedifferentiation while circumventing the apoptotic effect in the hypertonic treatment. | ||
| S6085 | KY19382 (A3051) | KY19382 (A3051) is a Wnt/β-catenin signalling activator through inhibitory effects on both CXXC5–DVL interaction and GSK3β activity with IC50s of 19 nM and 10 nM, respectively. | ||
| S8647 | iCRT3 | iCRT3 is an antagonist of Wnt/β-catenin signaling with an IC50 of 8.2 nM in the Wnt responsive STF16-luc reporter assays. | ||
| S0733 | Tegatrabetan (BC-2059) | Tegatrabetan (BC2059,Tegavivint) is an antagonist of β-Catenin. Tegatrabetan (BC2059) treatment disrupts the binding of β-catenin with the scaffold protein transducin β-like 1 (TBL1) and proteasomal degradation and declines in the nuclear levels of β-catenin. | ||
| S8462New | Toxoflavin | PKF118-310 is a potent antagonist of transcription factor 4 (TCF4)/β-catenin signaling, and also an inhibitor of KDM4A . It exhibits anticancer activity and can be used in solid tumor research. | ||
| S8320 | SKL2001 | SKL2001 is a novel agonist of the Wnt/β-catenin pathway. It disrupts the Axin/β-catenin interaction. |

|
|
| S5815 | WAY-316606 | WAY-316606, a specific antagonist of SFRP1, functions as an activator of canonical Wnt/β-catenin signalling in the human hair bulb. | ||
| S9828 | WAY-262611 | WAY-262611 (BML-WN110, Compound 5) is a beta-catenin (β-Catenin) agonist that increases bone formation rate with EC50 of 0.63 μM in TCF-Luciferase assay. WAY-262611 is also an inhibitor of dickkopf 1 (DKK1). | ||
| S5992 | Heparan Sulfate | Heparan sulfate (HS, Heparitin sulfate, Alpha-idosane, HHS 5, N-Acetylheparan Sulfate, Suleparoid, Tavidan), a constituent of HS proteoglycans (HSPGs), is a linear polysaccharide present on the cell surface. Heparan sulfate influences the binding affinity of intestinal epithelium cells (IECs) to Wnt, thereby promoting activation of canonical Wnt signaling and facilitating regeneration of small intestinal crypts after epithelial injury. | ||
| S6961 | Foxy-5 | Foxy-5, a Wnt-5a mimicking hexapeptide, is a WNT signalling pathway modulator potentially for the treatment of metastatic breast cancer, prostate cancer and colorectal cancer. | ||
| S0458 | KY1220 | KY1220 is a small molecule that destabilizes both β-catenin and Ras, via targeting the Wnt/β-catenin pathway with IC50 of 2.1 μM in HEK293 reporter cells. | ||
| F1056New | Anti-Wnt3a Rabbit Antibody [C3D14] | Proto-oncogene Int-4 homolog, WNT3 | ||
| F0494New | Anti-Phospho-β-Catenin (Ser552) Rabbit Antibody [K9A16] | |||
| F0265New | Anti-TCF4/TCF7L2 Rabbit Antibody [P1D24] | TCF-4,TCF4/TCF7L2,TCF7L2 | ||
| F1661New | Anti-DKK1 Rabbit Antibody [K16J6] | DKK1,Dkk-1 | ||
| F2477New | Anti-Wnt2/IRP Rabbit Antibody [B13H8] | |||
| S8462New | Toxoflavin | PKF118-310 is a potent antagonist of transcription factor 4 (TCF4)/β-catenin signaling, and also an inhibitor of KDM4A . It exhibits anticancer activity and can be used in solid tumor research. | ||
| E4800New | BML-284 | BML-284 (Wnt Agonist) is a potent and cell-permeable activator of Wnt signaling that induces TCF-dependent transcriptional activity with an EC50 value of 0.7 µM. BML-284 also acts as an agonist of β-catenin that effectively activates direct Wnt-β-catenin signaling to induce adipocyte dedifferentiation while circumventing the apoptotic effect in the hypertonic treatment. | ||
| E6057New | Pamidronate disodium pentahydrate | Pamidronate disodium pentahydrate is a nitrogen-containing bisphosphonate and used to prevent bone loss due to steroid use like glucocorticoid-induced low bone mineral density in children or to inhibit calcium release from bone by impairing osteoclast-mediated bone resorption. It also inhibits the Wnt/β-catenin signaling pathway that controls osteogenic differentiation in bone marrow mesenchymal stem cells (BMMSCs). | ||
| F0297New | Anti-Phospho-β-Catenin (Ser675) Rabbit Antibody [G12L16] |
Choose Selective Wnt/beta-catenin Inhibitors
Tags: Wnt inhibition | Wnt signaling | Wnt pathway | Wnt cancer | beta-catenin signaling | beta-catenin pathway | beta-catenin cancer | Wnt beta-catenin pathway | Wnt activation | Wnt tumor | beta-catenin activation | beta-catenin tumor | beta-catenin phosphorylation | Wnt activity | β-catenin signaling | beta-catenin mutation | Wnt assay | Wnt mutation | Wnt drug | beta-catenin assay | Wnt inhibitors clinical trials | Wnt inhibitor review







































