- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
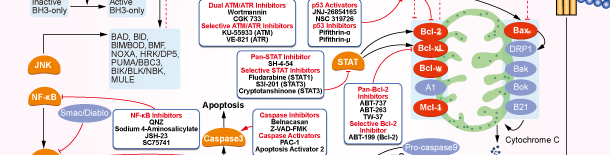
Bcl-2
Bcl-2 Products
- All (57)
- Bcl-2 Inhibitors (38)
- Bcl-2 Activators (8)
- Bcl-2 Antagonists (5)
- Bcl-2 Modulators (5)
- New Bcl-2 Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
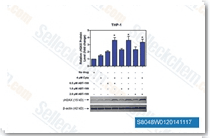
| S8048 | Venetoclax (ABT-199) | Venetoclax (ABT-199, GDC-0199) is a Bcl-2-selective inhibitor with Ki of <0.01 nM in cell-free assays, >4800-fold more selective versus Bcl-xL and Bcl-w, and no activity to Mcl-1. Venetoclax is reported to induce cell growth suppression, apoptosis, cell cycle arrest, and autophagy in triple negative breast cancer MDA-MB-231 cells. Phase 3. |
|
|
| S1002 | ABT-737 | ABT-737 is a BH3 mimetic inhibitor of Bcl-xL, Bcl-2 and Bcl-w with EC50 of 78.7 nM, 30.3 nM and 197.8 nM in cell-free assays, respectively; no inhibition observed against Mcl-1, Bcl-B or Bfl-1. ABT-737 induces mitochondrial pathway apoptosis and mitophagy. Phase 2. |

|
|
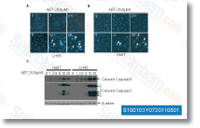
| S1001 | Navitoclax (ABT-263) | Navitoclax (ABT-263) is a potent inhibitor of Bcl-xL, Bcl-2 and Bcl-w with Ki of ≤ 0.5 nM, ≤1 nM and ≤1 nM in cell-free assays, but binds more weakly to Mcl-1 and A1. Phase 2. |
|
|
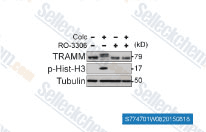
| S7747 | Ro-3306 | RO-3306 is an ATP-competitive, and selective CDK1 inhibitor with Ki of 20 nM, >15-fold selectivity against a diverse panel of human kinases. RO-3306 enhances p53-mediated Bax activation and mitochondrial apoptosis. |
|
|
| S1057 | Obatoclax Mesylate (GX15-070) | Obatoclax Mesylate (GX15-070) is an antagonist of Bcl-2 with Ki of 0.22 μM in a cell-free assay, can assist in overcoming MCL-1 mediated resistance to apoptosis. |
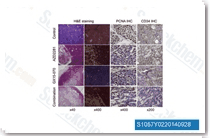
|
|
| S8383 | S63845 | S63845 is a new, selective MCL-1 inhibitor with the Kd value of 0.19 nM and has no discernible binding to the other BCL-2 members, BCL-2 or BCL-XL. | ||
| S7790 | A-1210477 | A-1210477 is a potent and selective MCL-1 inhibitor with Ki and IC50 of 0.454 nM and 26.2 nM, respectively, >100-fold selectivity over other Bcl-2 family members. |
|
|
| S1121 | TW-37 | TW-37 is a novel nonpeptide inhibitor to recombinant Bcl-2, Bcl-xL and Mcl-1 with Ki of 0.29 μM, 1.11 μM and 0.26 μM in cell-free assays, respectively. |
|
|
| S7801 | A-1331852 | A-1331852 is a potent and selectiveBCL-XL inhibitor with Ki value less than 0.01 nM for BCL-XL and 6 nM, 4 nM, 142 nM for Bcl-2, Bcl-W, MCL-1 respectively. It may be useful in the treatment of cancer, immune and autoimmune diseases. |
|
|
| S7800 | A-1155463 Dihydrochloride | A-1155463 Dihydrochloride, a highly potent and selective BCL-XL inhibitor, shows picomolar binding affinity to BCL-XL, and >1000-fold weaker binding to BCL-2 and related proteins BCL-W(Ki=19 nM) and MCL-1(Ki>440 nM). |
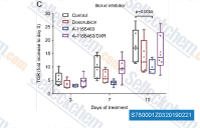
|
|
| S2606 | Mifepristone | Mifepristone is a remarkably active antagonist of progesterone receptor and glucocorticoid receptor with IC50 of 0.2 nM and 2.6 nM, respectively. Mifepristone promotes cell autophagy and apoptosis, decreases Bcl-2 level and increases Beclin1 level, accompanied by weakened interaction between Bcl-2 and Beclin1. |

|
|
| S7531 | UMI-77 | UMI-77 is a selective Mcl-1 inhibitor with Ki of 490 nM, showing selectivity over other members of Bcl-2 family. |

|
|
| S2812 | (R)-(-)-Gossypol (AT-101) acetic acid | (R)-(-)-Gossypol (AT-101) acetic acid, the R-(-) enantiomer of Gossypol acetic acid, binds with Bcl-2, Bcl-xL and Mcl-1 with Ki of 0.32 μM, 0.48 μM and 0.18 μM in cell-free assays; does not inhibit BIR3 domain and BID. AT-101 simultaneously triggers apoptosis and a cytoprotective type of autophagy. Phase 2. |
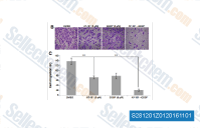
|
|
| S8643 | AZD5991 | AZD5991 is a macrocyclic MCL-1 inhibitor with sub-nanomolar affinity for MCL-1 (Ki = 0.13 nM). The binding affinity of AZD5991 is about 25-fold lower for mouse Mcl-1 vs. human Mcl-1 but only four-fold lower for rat Mcl-1. | ||
| S8061 | Sabutoclax | Sabutoclax (BI-97C1) is a pan-Bcl-2 inhibitor, including Bcl-xL, Bcl-2, Mcl-1 and Bfl-1 with IC50 of 0.31 μM, 0.32 μM, 0.20 μM and 0.62 μM, respectively. |
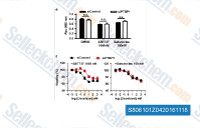
|
|
| S7100 | WEHI-539 | WEHI-539 has high affinity (IC50=1.1 nM) and selectivity for BCL-XL and potently kills cells by selectively antagonizing its prosurvival activity. It has more than a 400-fold higher affinity for BCL-XL versus other prosurvival BCL-2 family members. | ||
| S1071 | HA14-1 | HA14-1 is a non-peptidic ligand of a Bcl-2 surface pocket with IC50 of ~9 μM. |
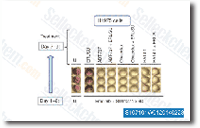
|
|
| S8836 | MIK665 (S64315) | MIK665 (S64315) is an inhibitor of induced myeloid leukemia cell differentiation protein Mcl-1 with Ki value of 1.2 nM and has potential pro-apoptotic and antineoplastic activities. | ||
| S6709 | Obatoclax (GX15-070) | Obatoclax (GX15-070) is an antagonist of Bcl-2 with an Ki of 0.22 μM in a cell-free assay, can assist in overcoming MCL-1 mediated resistance to apoptosis. |
||
| S8820 | Unesbulin (PTC596) | Unesbulin (PTC596) is a second-generation BMI-1 inhibitor that accelerates BMI-1 degradation. PTC596 downregulates MCL-1 and induces p53-independent mitochondrial apoptosis. IC50 values at 72 hours ranged from 68 to 340 nM in mantle cell lymphoma (MCL) cell lines. | ||
| S2448 | Gambogic Acid | Gambogic Acid (Guttatic Acid, Guttic Acid, Beta-Guttiferrin) activates caspases with EC50 of 0.78-1.64 μM and competitively inhibits Bcl-XL, Bcl-2, Bcl-W, Bcl-B, Bfl-1 and Mcl-1 with IC50 of 1.47, 1.21, 2.02, 0.66, 1.06 and 0.79 μM, respectively. | ||
| S7126 | Marinopyrrole A (Maritoclax) | Marinopyrrole A (Maritoclax) is a selective Mcl-1 antagonist. It binds to Mcl-1, but not Bcl-XL, and targets Mcl-1 for proteasomal degradation. Maritoclax disrupts the interaction between Bim and Mcl-1 with an IC50 of 10.1 μM. | ||
| S8865 | BAI1 | BAI1 is a direct allosteric inhibitor of BAX with a dissociation constant (Kd) of 15.0 ± 4 μM. | ||
| S6852 | Gossypol | Gossypol (BL 193) is an orally-active polyphenol isolated from cotton seeds and roots. Gossypol is a potent inhibitor of 5α-reductase 1 and 3α-hydroxysteroid dehydrogenase with IC50 of 3.33 μM and 0.52 μM in cell-free assay, respectively. Gossypol also inhibits the binding of BH3 peptide to Bcl protein with IC50 of 0.4 μM and 10 μM for Bcl-XL and Bcl-2, respectively. Gossypol induces apoptosis and cell growth inhibition in various cancer cells. | ||
| E2926New | A-1155463 | A-1155463 is a highly potent and selective BCL-XL inhibitor with EC50 of 65 nM in H146 cells. | ||
| S8650 | BTSA1 | BTSA1 is a pharmacologically optimized BAX activator that binds with high affinity and specificity to the N-terminal activation site and induces conformational changes to BAX leading to BAX-mediated apoptosis. It effectively promotes apoptosis in leukemia cell lines and patient samples while sparing healthy cells. | ||
| S8759 | S55746 | S55746 (S 055746,BCL201) is a novel, orally active BCL-2 specific inhibitor (Ki = 1.3 nM) with poor affinity for BCL-XL and no significant binding to MCL-1, BFL-1 (BCL2A1/A1). The selectivity of S55746 for BCL-2 versus BCL-XL ranges from ~70 to 400 folds. | ||
| S7105 | BAM7 | BAM 7 is a direct and selective activator of proapoptotic Bax with EC50 of 3.3 μM. | ||
| S3275 | Senkyunolide I | Senkyunolide I (SEI, SENI) is an orally active compound isolated from Ligusticum chuanxiong with analgesic, anti-migraine, neuroprotective, anti-oxidation and anti-apoptosis activities. Senkyunolide I (SEI, SENI) up-regulates the phosphorylation of Erk1/2 and induces Nrf2 nuclear translocation with enhanced HO-1 and NQO1 expressions. Senkyunolide I (SEI, SENI) promotes the ratio of Bcl-2/Bax and inhibits the expressions of cleaved caspase 3 and caspase 9. | ||
| S5967 | Berberine chloride hydrate | Berberine (Natural Yellow 18) chloride hydrate is a quaternary ammonium salt from the group of isoquinoline alkaloids. Berberine activates caspase 3 and caspase 8, cleavage of poly ADP-ribose polymerase (PARP) and the release of cytochrome c. Berberine chloride decreases the expression of c-IAP1, Bcl-2 and Bcl-XL. Berberine chloride induces apoptosis with sustained phosphorylation of JNK and p38 MAPK, as well as generation of the ROS. Berberine chloride is a dual topoisomerase I and II inhibitor. Berberine chloride is also a potential autophagy modulator. | ||
| S8199 | Ruxotemitide (LTX 315) | Ruxotemitide (LTX 315) is the oncolytic peptide that kills cancer cells through Bax/Bak-regulated mitochondrial membrane permeabilization. | ||
| S7849 | BDA-366 | BDA-366 is a small-molecule Bcl2-BH4 domain antagonist and binds BH4 with high affinity and selectivity. It directly binds to Bcl2 with high binding affinity (Ki =3.3 ± 0.73 nM). | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S0563 | 10-Deacetyl-7-xylosyl paclitaxel | 10-Deacetyl-7-xylosyl paclitaxel (10-Deacetyl-7-xylosyltaxol, 7-xylosyl-10-deacetylpaclitaxel), a derivative of paclitaxel and naturally occurring xyloside isolated from Taxus chinensis, causes significant mitotic arrest in PC-3 cells followed by up-regulating expression of pro-apoptotic Bax and Bad protein, as well as down-regulating expression of anti-apoptotic Bcl-2 and Bcl-XL , which leads to a disturbance of the mitochondrial membrane permeability and to the activation of caspase-9. | ||
| E0124 | Chelerythrine | Chelerythrine (Toddaline, Broussonpapyrine) is a potent, selective antagonist of PKC with an IC50 of 0.66 μM. Chelerythrine also inhibits the BclXL-Bak BH3 peptide binding with an IC50 of 1.5 μM. Chelerythrine shows antitumor, antidiabetic and anti-inflammatory activity. |
||
| E3037 | Solanum lyratum Extract | Solanum lyratum Extract (300 μg/ml) increases Bax levels and decreases Bcl-2 levels, which cause the loss of mitochondrial membrane potential (Δæm) followed by cytochrome C release and caspase-9 and -3 activation, finally leading to apoptosis. Solanum lyratum Extract (300 μg/ml) also promotes p53 and p27, but decreases the levels of cyclin B1 thus causing S-phase arrest. | ||
| S3224 | Cinobufagin | Cinobufagin (Cinobufagine), an active ingredient of Venenum Bufonis, inhibits tumor development. Cinobufagin increases ATM and Chk2 and decreases CDC25C, CDK1, and cyclin B. Cinobufagin inhibits PI3K, AKT and Bcl-2 while increases levels of cleaved caspase-9 and caspase-3. Thus, Cinobufagin induces cell cycle arrest at the G2/M phase and apoptosis. | ||
| S6895 | TCPOBOP | TCPOBOP is a constitutive androstane receptor (CAR) agonist. TCPOBOP attenuates Fas-induced murine liver injury by altering Bcl-2 proteins. | ||
| S9970 | APG-2575 (lisaftoclax) | APG-2575 (lisaftoclax) is a dual Bcl-2 and Bcl-xl inhibitor with IC50 values of 2 nM and 5.9 nM for Bcl-2 and Bcl-xl, respectively. | ||
| S3245 | Nodakenetin | Nodakenetin (NANI), a plant-derived coumarin isolated from Angelica decursiva, inhibits α-glucosidase, PTP1B, rat lens aldose reductase (RLAR), AChE, BChE, and β-site amyloid precursor protein cleaving enzyme 1 (BACE1). Nodakenetin alters the protein expression of Bax and Bcl-2, and prompts mitochondrial apoptosis. Nodakenetin exhibits anti-tumor activity. | ||
| S3267 | Nicotiflorin (Kaempferol-3-O-rutinoside) | Nicotiflorin (Kaempferol-3-O-rutinoside), a flavonoid extracted from Carthamus tinctorius, alters the shape and structure of injured neurons, decreases the number of apoptotic cells, down-regulates expression of p-JAK2, p-STAT3, caspase-3, and Bax and decreases Bax immunoredactivity, and increases Bcl-2 protein expression and immunoreactivity. | ||
| S3243 | Zeaxanthin | Zeaxanthin, the carotenoid alcohol participates in the xanthophyll cycle, activates the extrinsic apoptosis pathway which induces apoptosis on uveal melanoma cells with IC50 value 40.8 µM. | ||
| E2354 | Valepotriate | Valepotriate, an unstable iridoid isolated from Valeriana jatamansi Jones, has anti-epileptic by significantly increasing the expression of GABAA, glutamic acid decarboxylase 65, and Bcl-2 and reduce the expression of caspase-3. | ||
| E2028 | Humanin | Humanin (human) (1-24-Protein humanin (human)), a small mitochondrial-derived cytoprotective polypeptide encoded by mtDNA, exhibits protective effects in several cell types against cellular stress conditions and apoptosis through regulating various signaling mechanisms, such as JAK/STAT pathway and interaction of BCL-2 family of protein. | ||
| E0011 | Linderalactone | Linderalactone inhibits human lung cancer growth by modulating the expression of apoptosis-related proteins (Bax and Bcl-2) with an IC50 of 15 µM in A-549 cells. Linderalactone induces G2/M cell cycle arrest and could also suppress the JAK/STAT signalling pathway. Linderalactone can be isolated from Radix linderae. | ||
| S9276 | Alisol B | Alisol B, a triterpene from Alismatis rhizoma, induces Bax up-regulation and nuclear translocation, the activation of initiator caspase-8 and caspase-9, and executor caspase-3, suggesting the involvement of both extrinsic and intrinsic apoptosis pathways. | ||
| S2271 | Berberine chloride | Berberine chloride is a quaternary ammonium salt from the group of isoquinoline alkaloids. Berberine activates caspase 3 and caspase 8, cleavage of poly ADP-ribose polymerase (PARP) and the release of cytochrome c. Berberine chloride decreases the expression of c-IAP1, Bcl-2 and Bcl-XL. Berberine chloride induces apoptosis with sustained phosphorylation of JNK and p38 MAPK, as well as generation of the ROS. Berberine chloride is a dual topoisomerase I and II inhibitor. Berberine chloride is also a potential autophagy modulator. | ||
| S5600 | Flavokawain A | Flavokawain A, extracted from kava, is an apoptotic inducers and anticarcinogenic agent. Flavokawain A can down-regulation of antiapoptotic proteins, such as XIAP, survivin, and Bcl-xL, thereby changing the balance between apoptotic and antiapoptotic molecules and then induce cell death in tumor cells. | ||
| S8177 | BH3I-1 | BH3I-1 is a Bcl-XL-BH3 domain interaction inhibitor with Ki of 2.4 μM (by fluorescence polarization ).It is a selective inhibitor of Bcl-2 family proteins. | ||
| S8924 | DT2216 | DT2216 is a potent and selective degrader of BCL-XL based on PROTAC technology. DT2216 inhibits various BCL-XL-dependent leukemia and cancer cells but considerably less toxic to platelets. | ||
| S6004 | CCI-007 | CCI-007 is a novel small molecule with cytotoxic activity against infant leukemia with MLL rearrangements. | ||
| S9665 | Motixafortide (BL-8040) | Motixafortide (BL-8040, BKT140, TF 14016, 4-fluorobenzoyl, 4F-benzoyl-TN14003, T140) is an antagonist of CXCR4 with IC50 of ~1 nM. BL-8040 induces the apoptosis of AML blasts by down-regulating ERK, BCL-2, MCL-1 and cyclin-D1 via altered miR-15a/16-1 expression. | ||
| S8758 | VU661013 | VU661013 is a novel, potent, selective MCL1 inhibitor with Ki of 97 ± 30 pM of human MCL-1 in a TR-FRET assay. However, VU661013 does not significantly inhibit BCL-xL or BCL-2 with Ki > 40 μM or = 0.73 μM. VU661013 de-stabilizes BIM/MCL-1 association, leads to apoptosis in AML. | ||
| S5550 | Ethyl gallate | Ethyl gallat (Phyllemblin, gallic acid ethyl ester), which could be found naturally in a variety of plant sources, is a food additive with antimicrobial activity. Ethyl gallat activates the death receptor-dependent pathway of apoptosis by enhancing the expression of caspases-8, -9, and -3 and the Bcl-2 interacting domain (Bid). | ||
| S6990 | PhytohemagglutininPhytohemagglutinin (PHA) | PhytohemagglutininPhytohemagglutinin (PHA) is expressed in Pichia pastoris using native signal peptides, or the Saccharomyces alpha-factor preprosequence, to direct proteins into the secretory pathway. Phytohemagglutinin induces apoptosis in human HEp-2 carcinoma cells via increasing proapoptotic protein Bax and activating caspases-3. | ||
| E2663 | BT2 | BT2 is a novel branched-chain α-ketoacid dehydrogenase complex kinase (BDK) inhibitor wieh an IC50 of 3.19 μM. | ||
| E3020 | Hedyotic Diffusa Extract | Hedyotic Diffusa Extract is extracted from Hedyotic diffusa, of which polysaccharides inhibit the growth of CNE2 cells in a dose-and-time-dependent way, the mechanism may involve induction of cell apoptosis, which is associated with the activation of Bax and caspase-3 protein and the down-regulation of Bcl-2 protein expression. | ||
| S8048 | Venetoclax (ABT-199) | Venetoclax (ABT-199, GDC-0199) is a Bcl-2-selective inhibitor with Ki of <0.01 nM in cell-free assays, >4800-fold more selective versus Bcl-xL and Bcl-w, and no activity to Mcl-1. Venetoclax is reported to induce cell growth suppression, apoptosis, cell cycle arrest, and autophagy in triple negative breast cancer MDA-MB-231 cells. Phase 3. |

|
|
| S1002 | ABT-737 | ABT-737 is a BH3 mimetic inhibitor of Bcl-xL, Bcl-2 and Bcl-w with EC50 of 78.7 nM, 30.3 nM and 197.8 nM in cell-free assays, respectively; no inhibition observed against Mcl-1, Bcl-B or Bfl-1. ABT-737 induces mitochondrial pathway apoptosis and mitophagy. Phase 2. |

|
|
| S1001 | Navitoclax (ABT-263) | Navitoclax (ABT-263) is a potent inhibitor of Bcl-xL, Bcl-2 and Bcl-w with Ki of ≤ 0.5 nM, ≤1 nM and ≤1 nM in cell-free assays, but binds more weakly to Mcl-1 and A1. Phase 2. |

|
|
| S8383 | S63845 | S63845 is a new, selective MCL-1 inhibitor with the Kd value of 0.19 nM and has no discernible binding to the other BCL-2 members, BCL-2 or BCL-XL. | ||
| S7790 | A-1210477 | A-1210477 is a potent and selective MCL-1 inhibitor with Ki and IC50 of 0.454 nM and 26.2 nM, respectively, >100-fold selectivity over other Bcl-2 family members. |

|
|
| S1121 | TW-37 | TW-37 is a novel nonpeptide inhibitor to recombinant Bcl-2, Bcl-xL and Mcl-1 with Ki of 0.29 μM, 1.11 μM and 0.26 μM in cell-free assays, respectively. |

|
|
| S7801 | A-1331852 | A-1331852 is a potent and selectiveBCL-XL inhibitor with Ki value less than 0.01 nM for BCL-XL and 6 nM, 4 nM, 142 nM for Bcl-2, Bcl-W, MCL-1 respectively. It may be useful in the treatment of cancer, immune and autoimmune diseases. |

|
|
| S7800 | A-1155463 Dihydrochloride | A-1155463 Dihydrochloride, a highly potent and selective BCL-XL inhibitor, shows picomolar binding affinity to BCL-XL, and >1000-fold weaker binding to BCL-2 and related proteins BCL-W(Ki=19 nM) and MCL-1(Ki>440 nM). |

|
|
| S2606 | Mifepristone | Mifepristone is a remarkably active antagonist of progesterone receptor and glucocorticoid receptor with IC50 of 0.2 nM and 2.6 nM, respectively. Mifepristone promotes cell autophagy and apoptosis, decreases Bcl-2 level and increases Beclin1 level, accompanied by weakened interaction between Bcl-2 and Beclin1. |

|
|
| S7531 | UMI-77 | UMI-77 is a selective Mcl-1 inhibitor with Ki of 490 nM, showing selectivity over other members of Bcl-2 family. |

|
|
| S2812 | (R)-(-)-Gossypol (AT-101) acetic acid | (R)-(-)-Gossypol (AT-101) acetic acid, the R-(-) enantiomer of Gossypol acetic acid, binds with Bcl-2, Bcl-xL and Mcl-1 with Ki of 0.32 μM, 0.48 μM and 0.18 μM in cell-free assays; does not inhibit BIR3 domain and BID. AT-101 simultaneously triggers apoptosis and a cytoprotective type of autophagy. Phase 2. |

|
|
| S8643 | AZD5991 | AZD5991 is a macrocyclic MCL-1 inhibitor with sub-nanomolar affinity for MCL-1 (Ki = 0.13 nM). The binding affinity of AZD5991 is about 25-fold lower for mouse Mcl-1 vs. human Mcl-1 but only four-fold lower for rat Mcl-1. | ||
| S8061 | Sabutoclax | Sabutoclax (BI-97C1) is a pan-Bcl-2 inhibitor, including Bcl-xL, Bcl-2, Mcl-1 and Bfl-1 with IC50 of 0.31 μM, 0.32 μM, 0.20 μM and 0.62 μM, respectively. |

|
|
| S1071 | HA14-1 | HA14-1 is a non-peptidic ligand of a Bcl-2 surface pocket with IC50 of ~9 μM. |

|
|
| S8836 | MIK665 (S64315) | MIK665 (S64315) is an inhibitor of induced myeloid leukemia cell differentiation protein Mcl-1 with Ki value of 1.2 nM and has potential pro-apoptotic and antineoplastic activities. | ||
| S8820 | Unesbulin (PTC596) | Unesbulin (PTC596) is a second-generation BMI-1 inhibitor that accelerates BMI-1 degradation. PTC596 downregulates MCL-1 and induces p53-independent mitochondrial apoptosis. IC50 values at 72 hours ranged from 68 to 340 nM in mantle cell lymphoma (MCL) cell lines. | ||
| S2448 | Gambogic Acid | Gambogic Acid (Guttatic Acid, Guttic Acid, Beta-Guttiferrin) activates caspases with EC50 of 0.78-1.64 μM and competitively inhibits Bcl-XL, Bcl-2, Bcl-W, Bcl-B, Bfl-1 and Mcl-1 with IC50 of 1.47, 1.21, 2.02, 0.66, 1.06 and 0.79 μM, respectively. | ||
| S8865 | BAI1 | BAI1 is a direct allosteric inhibitor of BAX with a dissociation constant (Kd) of 15.0 ± 4 μM. | ||
| S6852 | Gossypol | Gossypol (BL 193) is an orally-active polyphenol isolated from cotton seeds and roots. Gossypol is a potent inhibitor of 5α-reductase 1 and 3α-hydroxysteroid dehydrogenase with IC50 of 3.33 μM and 0.52 μM in cell-free assay, respectively. Gossypol also inhibits the binding of BH3 peptide to Bcl protein with IC50 of 0.4 μM and 10 μM for Bcl-XL and Bcl-2, respectively. Gossypol induces apoptosis and cell growth inhibition in various cancer cells. | ||
| E2926New | A-1155463 | A-1155463 is a highly potent and selective BCL-XL inhibitor with EC50 of 65 nM in H146 cells. | ||
| S8759 | S55746 | S55746 (S 055746,BCL201) is a novel, orally active BCL-2 specific inhibitor (Ki = 1.3 nM) with poor affinity for BCL-XL and no significant binding to MCL-1, BFL-1 (BCL2A1/A1). The selectivity of S55746 for BCL-2 versus BCL-XL ranges from ~70 to 400 folds. | ||
| S5967 | Berberine chloride hydrate | Berberine (Natural Yellow 18) chloride hydrate is a quaternary ammonium salt from the group of isoquinoline alkaloids. Berberine activates caspase 3 and caspase 8, cleavage of poly ADP-ribose polymerase (PARP) and the release of cytochrome c. Berberine chloride decreases the expression of c-IAP1, Bcl-2 and Bcl-XL. Berberine chloride induces apoptosis with sustained phosphorylation of JNK and p38 MAPK, as well as generation of the ROS. Berberine chloride is a dual topoisomerase I and II inhibitor. Berberine chloride is also a potential autophagy modulator. | ||
| E0124 | Chelerythrine | Chelerythrine (Toddaline, Broussonpapyrine) is a potent, selective antagonist of PKC with an IC50 of 0.66 μM. Chelerythrine also inhibits the BclXL-Bak BH3 peptide binding with an IC50 of 1.5 μM. Chelerythrine shows antitumor, antidiabetic and anti-inflammatory activity. |
||
| S3224 | Cinobufagin | Cinobufagin (Cinobufagine), an active ingredient of Venenum Bufonis, inhibits tumor development. Cinobufagin increases ATM and Chk2 and decreases CDC25C, CDK1, and cyclin B. Cinobufagin inhibits PI3K, AKT and Bcl-2 while increases levels of cleaved caspase-9 and caspase-3. Thus, Cinobufagin induces cell cycle arrest at the G2/M phase and apoptosis. | ||
| S6895 | TCPOBOP | TCPOBOP is a constitutive androstane receptor (CAR) agonist. TCPOBOP attenuates Fas-induced murine liver injury by altering Bcl-2 proteins. | ||
| S9970 | APG-2575 (lisaftoclax) | APG-2575 (lisaftoclax) is a dual Bcl-2 and Bcl-xl inhibitor with IC50 values of 2 nM and 5.9 nM for Bcl-2 and Bcl-xl, respectively. | ||
| S3243 | Zeaxanthin | Zeaxanthin, the carotenoid alcohol participates in the xanthophyll cycle, activates the extrinsic apoptosis pathway which induces apoptosis on uveal melanoma cells with IC50 value 40.8 µM. | ||
| E2354 | Valepotriate | Valepotriate, an unstable iridoid isolated from Valeriana jatamansi Jones, has anti-epileptic by significantly increasing the expression of GABAA, glutamic acid decarboxylase 65, and Bcl-2 and reduce the expression of caspase-3. | ||
| E0011 | Linderalactone | Linderalactone inhibits human lung cancer growth by modulating the expression of apoptosis-related proteins (Bax and Bcl-2) with an IC50 of 15 µM in A-549 cells. Linderalactone induces G2/M cell cycle arrest and could also suppress the JAK/STAT signalling pathway. Linderalactone can be isolated from Radix linderae. | ||
| S2271 | Berberine chloride | Berberine chloride is a quaternary ammonium salt from the group of isoquinoline alkaloids. Berberine activates caspase 3 and caspase 8, cleavage of poly ADP-ribose polymerase (PARP) and the release of cytochrome c. Berberine chloride decreases the expression of c-IAP1, Bcl-2 and Bcl-XL. Berberine chloride induces apoptosis with sustained phosphorylation of JNK and p38 MAPK, as well as generation of the ROS. Berberine chloride is a dual topoisomerase I and II inhibitor. Berberine chloride is also a potential autophagy modulator. | ||
| S5600 | Flavokawain A | Flavokawain A, extracted from kava, is an apoptotic inducers and anticarcinogenic agent. Flavokawain A can down-regulation of antiapoptotic proteins, such as XIAP, survivin, and Bcl-xL, thereby changing the balance between apoptotic and antiapoptotic molecules and then induce cell death in tumor cells. | ||
| S8177 | BH3I-1 | BH3I-1 is a Bcl-XL-BH3 domain interaction inhibitor with Ki of 2.4 μM (by fluorescence polarization ).It is a selective inhibitor of Bcl-2 family proteins. | ||
| S8924 | DT2216 | DT2216 is a potent and selective degrader of BCL-XL based on PROTAC technology. DT2216 inhibits various BCL-XL-dependent leukemia and cancer cells but considerably less toxic to platelets. | ||
| S6004 | CCI-007 | CCI-007 is a novel small molecule with cytotoxic activity against infant leukemia with MLL rearrangements. | ||
| S9665 | Motixafortide (BL-8040) | Motixafortide (BL-8040, BKT140, TF 14016, 4-fluorobenzoyl, 4F-benzoyl-TN14003, T140) is an antagonist of CXCR4 with IC50 of ~1 nM. BL-8040 induces the apoptosis of AML blasts by down-regulating ERK, BCL-2, MCL-1 and cyclin-D1 via altered miR-15a/16-1 expression. | ||
| S8758 | VU661013 | VU661013 is a novel, potent, selective MCL1 inhibitor with Ki of 97 ± 30 pM of human MCL-1 in a TR-FRET assay. However, VU661013 does not significantly inhibit BCL-xL or BCL-2 with Ki > 40 μM or = 0.73 μM. VU661013 de-stabilizes BIM/MCL-1 association, leads to apoptosis in AML. | ||
| E2663 | BT2 | BT2 is a novel branched-chain α-ketoacid dehydrogenase complex kinase (BDK) inhibitor wieh an IC50 of 3.19 μM. | ||
| E3020 | Hedyotic Diffusa Extract | Hedyotic Diffusa Extract is extracted from Hedyotic diffusa, of which polysaccharides inhibit the growth of CNE2 cells in a dose-and-time-dependent way, the mechanism may involve induction of cell apoptosis, which is associated with the activation of Bax and caspase-3 protein and the down-regulation of Bcl-2 protein expression. | ||
| S7747 | Ro-3306 | RO-3306 is an ATP-competitive, and selective CDK1 inhibitor with Ki of 20 nM, >15-fold selectivity against a diverse panel of human kinases. RO-3306 enhances p53-mediated Bax activation and mitochondrial apoptosis. |

|
|
| S8650 | BTSA1 | BTSA1 is a pharmacologically optimized BAX activator that binds with high affinity and specificity to the N-terminal activation site and induces conformational changes to BAX leading to BAX-mediated apoptosis. It effectively promotes apoptosis in leukemia cell lines and patient samples while sparing healthy cells. | ||
| S7105 | BAM7 | BAM 7 is a direct and selective activator of proapoptotic Bax with EC50 of 3.3 μM. | ||
| S3275 | Senkyunolide I | Senkyunolide I (SEI, SENI) is an orally active compound isolated from Ligusticum chuanxiong with analgesic, anti-migraine, neuroprotective, anti-oxidation and anti-apoptosis activities. Senkyunolide I (SEI, SENI) up-regulates the phosphorylation of Erk1/2 and induces Nrf2 nuclear translocation with enhanced HO-1 and NQO1 expressions. Senkyunolide I (SEI, SENI) promotes the ratio of Bcl-2/Bax and inhibits the expressions of cleaved caspase 3 and caspase 9. | ||
| S3238 | Resibufogenin | Resibufogenin (Bufogenin, Recibufogenin), a component of huachansu with anticancer effect, triggers necroptosis through upregulating receptor-interacting protein kinase 3 (RIP3) and phosphorylating mixed lineage kinase domain-like protein at Ser358. Resibufogenin exerts cytotoxic effect by inducing reactive oxygen species (ROS) accumulation. Resibufogenin induces apoptosis and caspase-3 and caspase-8 activity. Resibufogenin increases Bax/Bcl-2 expression, and suppresses cyclin D1, cyclin E, PI3K, p-AKT, p-GSK3β and β-catenin protein expression. | ||
| S9276 | Alisol B | Alisol B, a triterpene from Alismatis rhizoma, induces Bax up-regulation and nuclear translocation, the activation of initiator caspase-8 and caspase-9, and executor caspase-3, suggesting the involvement of both extrinsic and intrinsic apoptosis pathways. | ||
| S5550 | Ethyl gallate | Ethyl gallat (Phyllemblin, gallic acid ethyl ester), which could be found naturally in a variety of plant sources, is a food additive with antimicrobial activity. Ethyl gallat activates the death receptor-dependent pathway of apoptosis by enhancing the expression of caspases-8, -9, and -3 and the Bcl-2 interacting domain (Bid). | ||
| S6990 | PhytohemagglutininPhytohemagglutinin (PHA) | PhytohemagglutininPhytohemagglutinin (PHA) is expressed in Pichia pastoris using native signal peptides, or the Saccharomyces alpha-factor preprosequence, to direct proteins into the secretory pathway. Phytohemagglutinin induces apoptosis in human HEp-2 carcinoma cells via increasing proapoptotic protein Bax and activating caspases-3. | ||
| S1057 | Obatoclax Mesylate (GX15-070) | Obatoclax Mesylate (GX15-070) is an antagonist of Bcl-2 with Ki of 0.22 μM in a cell-free assay, can assist in overcoming MCL-1 mediated resistance to apoptosis. |

|
|
| S7100 | WEHI-539 | WEHI-539 has high affinity (IC50=1.1 nM) and selectivity for BCL-XL and potently kills cells by selectively antagonizing its prosurvival activity. It has more than a 400-fold higher affinity for BCL-XL versus other prosurvival BCL-2 family members. | ||
| S6709 | Obatoclax (GX15-070) | Obatoclax (GX15-070) is an antagonist of Bcl-2 with an Ki of 0.22 μM in a cell-free assay, can assist in overcoming MCL-1 mediated resistance to apoptosis. |
||
| S7126 | Marinopyrrole A (Maritoclax) | Marinopyrrole A (Maritoclax) is a selective Mcl-1 antagonist. It binds to Mcl-1, but not Bcl-XL, and targets Mcl-1 for proteasomal degradation. Maritoclax disrupts the interaction between Bim and Mcl-1 with an IC50 of 10.1 μM. | ||
| S7849 | BDA-366 | BDA-366 is a small-molecule Bcl2-BH4 domain antagonist and binds BH4 with high affinity and selectivity. It directly binds to Bcl2 with high binding affinity (Ki =3.3 ± 0.73 nM). | ||
| S8199 | Ruxotemitide (LTX 315) | Ruxotemitide (LTX 315) is the oncolytic peptide that kills cancer cells through Bax/Bak-regulated mitochondrial membrane permeabilization. | ||
| S0563 | 10-Deacetyl-7-xylosyl paclitaxel | 10-Deacetyl-7-xylosyl paclitaxel (10-Deacetyl-7-xylosyltaxol, 7-xylosyl-10-deacetylpaclitaxel), a derivative of paclitaxel and naturally occurring xyloside isolated from Taxus chinensis, causes significant mitotic arrest in PC-3 cells followed by up-regulating expression of pro-apoptotic Bax and Bad protein, as well as down-regulating expression of anti-apoptotic Bcl-2 and Bcl-XL , which leads to a disturbance of the mitochondrial membrane permeability and to the activation of caspase-9. | ||
| S3245 | Nodakenetin | Nodakenetin (NANI), a plant-derived coumarin isolated from Angelica decursiva, inhibits α-glucosidase, PTP1B, rat lens aldose reductase (RLAR), AChE, BChE, and β-site amyloid precursor protein cleaving enzyme 1 (BACE1). Nodakenetin alters the protein expression of Bax and Bcl-2, and prompts mitochondrial apoptosis. Nodakenetin exhibits anti-tumor activity. | ||
| S3267 | Nicotiflorin (Kaempferol-3-O-rutinoside) | Nicotiflorin (Kaempferol-3-O-rutinoside), a flavonoid extracted from Carthamus tinctorius, alters the shape and structure of injured neurons, decreases the number of apoptotic cells, down-regulates expression of p-JAK2, p-STAT3, caspase-3, and Bax and decreases Bax immunoredactivity, and increases Bcl-2 protein expression and immunoreactivity. | ||
| E2028 | Humanin | Humanin (human) (1-24-Protein humanin (human)), a small mitochondrial-derived cytoprotective polypeptide encoded by mtDNA, exhibits protective effects in several cell types against cellular stress conditions and apoptosis through regulating various signaling mechanisms, such as JAK/STAT pathway and interaction of BCL-2 family of protein. | ||
| E2926New | A-1155463 | A-1155463 is a highly potent and selective BCL-XL inhibitor with EC50 of 65 nM in H146 cells. |
Choose Selective Bcl-2 Inhibitors
Tags: Bcl-2 apoptosis | Bcl-2 inhibition | Bcl-2 protein | Bax apoptosis | Bak protein | Bax protein | Bcl-2 cancer | Bcl-2 expression | Bcl-2 lymphoma | Bcl-2 protein family | Bak apoptosis | Bcl-2 pathway | Bcl-2 phosphorylation | Mcl-1 apoptosis | Bcl-xL apoptosis | Bcl-2 apoptosis pathway | Bcl-2 activation | Bcl-xL protein | Bcl-2 cleavage | Bcl-2 inhibitor clinical trial | Bcl-2 inhibitor review