| S1267 |
Vemurafenib (PLX4032)
|
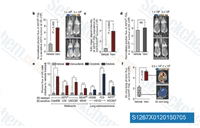
Vemurafenib (PLX4032, RG7204, RO5185426) is a novel and potent inhibitor of B-RafV600E with IC50 of 31 nM in cell-free assay, exhibiting 10-fold selectivity for B-RafV600E over wild-type B-Raf in enzymatic assays, and the cellular selectivity can exceed 100-fold. This compound also induces autophagy.
|
-
Nat Commun, 2025, 16(1):512
-
Int J Biol Sci, 2025, 21(11):4961-4978
-
Cells, 2025, 14(12)913
|
|
| S7397 |
Sorafenib (BAY 43-9006)
|
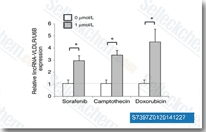
Sorafenib is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. Sorafenib inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. Sorafenib induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity.
|
-
Mol Cancer, 2025, 24(1):34
-
Nat Commun, 2025, 16(1):509
-
Adv Sci (Weinh), 2025, 12(30):e04372
|
|
| S2807 |
Dabrafenib (GSK2118436)
|
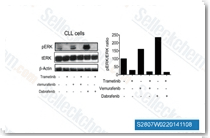
Dabrafenib is a mutant BRAFV600E specific inhibitor with IC50 of 0.7 nM in cell-free assays, with 7- and 9-fold less potency against B-Raf(wt) and c-Raf, respectively.
|
-
Nat Commun, 2025, 16(1):512
-
J Clin Invest, 2025, e191781
-
Cell Rep Med, 2025, 6(6):102183
|
|
| S1040 |
Sorafenib tosylate
|
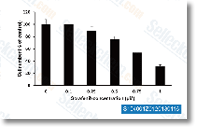
Sorafenib tosylate is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. This compound inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. It induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity.
|
-
Int J Oncol, 2025, 67(3)72
-
Nature, 2024, 629(8013):927-936
-
Cell Mol Life Sci, 2024, 81(1):238
|
|
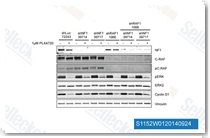
| S1152 |
PLX-4720
|
PLX4720 is a potent and selective inhibitor of B-RafV600E with IC50 of 13 nM in a cell-free assay, equally potent to c-Raf-1(Y340D and Y341D mutations), 10-fold selectivity for B-RafV600E than wild-type B-Raf.
|
-
J Clin Invest, 2025, 135(18)e178446
-
Cancer Res, 2025, 10.1158/0008-5472.CAN-24-3267
-
Cell Prolif, 2025, e70086.
|
|
| S5069 |
Dabrafenib Mesylate
|
Dabrafenib Mesylate (GSK2118436) is the mesylate salt form of dabrafenib, an orally bioavailable inhibitor of B-raf (BRAF) protein with IC50s of 0.8 nM, 3.2 nM and 5 nM for B-Raf (V600E), B-Raf (WT) and C-Raf, respectively.
|
-
EMBO Mol Med, 2024, 10.1038/s44321-024-00106-1
-
iScience, 2024, 27(10):110862
-
Cells, 2023, 12(4)664
|
|
| S7842 |
LY3009120
|
LY3009120 (DP-4978) is a potent pan-Raf inhibitor with IC50 of 44 nM, 31-47 nM, and 42 nM for A-raf, B-Raf, and C-Raf in A375 cells, respectively. This compound induces autophagy. Phase 1.
|
-
Cancer Res, 2025, 10.1158/0008-5472.CAN-24-3819
-
J Biol Chem, 2025, 301(8):110454
-
Front Biosci (Landmark Ed), 2025, 30(8):39944
|

|
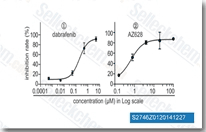
| S2746 |
AZ 628
|
AZ628 is a new pan-Raf inhibitor for BRAF, BRAFV600E, and c-Raf-1 with IC50 of 105 nM, 34 nM and 29 nM in cell-free assays, also inhibits VEGFR2, DDR2, Lyn, Flt1, FMS, etc. AZ628 induces apoptosis.
|
-
J Biol Chem, 2025, 301(8):110454
-
PLoS One, 2024, 19(1):e0295629
-
Cell Genom, 2024, 4(2):100487
|
|
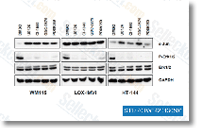
| S1104 |
GDC-0879
|
GDC-0879 (AR-00341677) is a novel, potent, and selective B-Raf inhibitor with IC50 of 0.13 nM in A375 and Colo205 cells with activity against c-Raf as well; no inhibition known to other protein kinases.
|
-
J Biol Chem, 2025, 301(8):110454
-
bioRxiv, 2025, 2025.04.29.651188
-
Nature, 2024, 629(8013):927-936
|
|
| S2220 |
SB590885
|
SB590885 is a potent B-Raf inhibitor with Ki of 0.16 nM in a cell-free assay, 11-fold greater selectivity for B-Raf over c-Raf, no inhibition to other human kinases.
|
-
Commun Biol, 2025, 8(1):125
-
Nat Commun, 2024, 15(1):668
-
Mol Cell, 2024, 84(14):2665-2681.e13
|

|
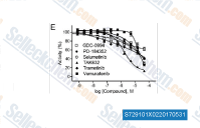
| S7291 |
TAK-632
|
TAK-632 is a potent pan-Raf inhibitor with IC50 of 8.3 nM and 1.4 nM for B-Raf(wt) and C-Raf in cell-free assays, respectively, showing less or no inhibition against other tested kinases.
|
-
bioRxiv, 2025, 2025.04.29.651188
-
Cell Rep, 2024, 43(9):114710
-
Cell Rep, 2024, 43(7):114471
|
|
| S2161 |
RAF265 (CHIR-265)
|
RAF265 (CHIR-265) is a potent selective inhibitor of C-Raf/B-Raf/B-Raf V600E with IC50 of 3-60 nM, and exhibits potent inhibition on VEGFR2 phosphorylation with EC50 of 30 nM in cell-free assays. This compound induces cell cycle arrest and apoptosis. Phase 2.
|
-
bioRxiv, 2025, 2025.04.29.651188
-
Cancer Cell, 2022, S1535-6108(22)00312-9
-
iScience, 2022, 25(10):105182
|

|
| S2872 |
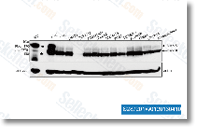
GW5074
|
GW5074 is a potent and selective c-Raf inhibitor with IC50 of 9 nM, no effect on the activities of JNK1/2/3, MEK1, MKK6/7, CDK1/2, c-Src, p38 MAP, VEGFR2 or c-Fms is noted. This compound inhibits LK-induced apoptosis.
|
-
Cancers (Basel), 2025, 17(18)2958
-
Aging Cell, 2024, e14320.
-
University of Central Florida, 2022,
|
|
| S7108 |
Encorafenib (LGX818)
|
Encorafenib (LGX818) is a highly potent RAF inhibitor with selective anti-proliferative and apoptotic activity in cells expressing B-RAF(V600E) with EC50 of 4 nM. This compound has reached Phase 3.
|
-
Cell Rep Med, 2025, 6(2):101943
-
J Immunother Cancer, 2025, 13(1)e010460
-
Oncogenesis, 2025, 14(1):27
|

|
| S7170 |
Avutometinib (Ro 5126766)
|
Avutometinib(RO5126766,CH5126766,VS 6766, CKI-27, R-7304, RG-7304) is a dual RAF/MEK inhibitor with IC50 of 8.2 nM,19 nM, 56 nM, and 160 nM for BRAF V600E, BRAF, CRAF, and MEK1, respectively. Phase 1.
|
-
Cancer Chemother Pharmacol, 2025, 95(1):78
-
Nat Biomed Eng, 2024, 10.1038/s41551-024-01273-9
-
Cell Rep Med, 2024, 5(11):101818
|
|
| S7964 |
PLX7904
|
PLX7904, also known as PB04, is a potent and selective paradox-breaker RAF inhibitor. It is able to efficiently inhibit activation of ERK1/2 in mutant BRAF melanoma cells but does not hyperactivate ERK1/2 in mutant RAS-expressing cells.
|
-
Nat Commun, 2022, 13(1):4109
-
J Invest Dermatol, 2022, S0022-202X(22)00096-3
-
Mol Cancer Ther, 2020, 25;molcanther.1021.2019
|
|
| S7965 |
Plixorafenib (PLX8394)
|
Plixorafenib (PLX8394) is a next-generation, orally available, small-molecule BRAF inhibitor with IC50 values of 3.8 nM, 14 nM and 23 nM for BRAF(V600E), WT BRAF and CRAF respectively. It has potential antineoplastic activity.
|
-
J Biol Chem, 2025, 301(8):110454
-
Nature, 2024, 629(8013):927-936
-
Cell Rep, 2024, 43(10):114829
|
|
| S2720 |
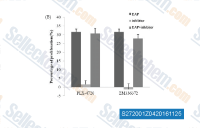
ZM 336372
|
ZM 336372 (Zinc00581684) is a potent and selective c-Raf inhibitor with IC50 of 70 nM, 10-fold selectivity over B-RAF, no inhibition to PKA/B/C, AMPK, p70S6, etc.
|
-
Clin Cancer Res, 2021, 10.1158/1078-0432.CCR-21-2660
-
Cell Metab, 2020, 5;31(5):892-908 e11
-
Exp Gerontol, 2019, 126:110691
|
|
| S8015 |
Agerafenib (CEP-32496)
|
Agerafenib (CEP-32496) is a highly potent inhibitor of BRAF(V600E/WT) and c-Raf with Kd of 14 nM/36 nM and 39 nM, respectively. It also shows potent activity against Abl-1, c-Kit, Ret (c-Ret), PDGFRβ and VEGFR2, but has insignificant affinity for MEK-1, MEK-2, ERK-1 and ERK-2. This compound is currently in Phase 1/2 clinical trials.
|
-
Mol Pharmacol, 2021, 99(6):435-447
-
Dis Model Mech, 2020, dmm.047779
-
Cancer Immunol Res, 2019,
|
|
| S8745 |
Naporafenib (LXH254)
|
Naporafenib (LXH254) is a type II ATP-competitive inhibitor that, at picomolar concentrations, inhibits both B- and CRAF kinase activities with a high degree of selectivity against a panel of 456 human kinases and in cell-based assays.
|
-
bioRxiv, 2025, 2025.06.10.658786
-
Cell Commun Signal, 2024, 22(1):428
-
Sci Adv, 2023, 9(35):eade7486
|
|
| S7926 |
Lifirafenib (BGB-283)
|
Lifirafenib (BGB-283, Beigene-283) potently inhibits RAF family kinases and EGFR activities in biochemical assays, with IC50 values of 23, 29 and 495 nM for the recombinant BRAFV600E kinase domain, EGFR and EGFR T790M/L858R mutant.
|
-
Sci Adv, 2023, 9(35):eade7486
-
Mol Cancer Ther, 2022, MCT-22-0004
-
Nat Commun, 2020, 11(1):3157
|
|
| S7121 |
Tovorafenib (MLN2480)
|
Tovorafenib (MLN2480, BIIB-024, TAK580, AMG-2112819, BSK1369, DAY-101) is an oral, selective pan-Raf kinase inhibitor in chinical trials.
|
-
bioRxiv, 2025, 2025.04.29.651188
-
Sci Adv, 2023, 9(35):eade7486
-
Int J Hematol, 2021, 10.1007/s12185-021-03244-1
|
|
| S8853 |
Belvarafenib
|
Belvarafenib is a selective and orally bioavailable pan-RAF kinase inhibitor with IC50 values of 41 nM, 7 nM and 2 nM for BRAF WT, BRAF(V600E) and CRAF kinases, respectively.
|
-
Sci Adv, 2023, 9(35):eade7486
-
SLAS Discov, 2022, S2472-5552(22)12517-7
-
Clin Cancer Res, 2021, 10.1158/1078-0432.CCR-21-2660
|
|
| S6660 |
B-Raf inhibitor 1 (Compound 13) dihydrochloride
|
B-Raf inhibitor 1 (Compound 13) is a type IIA Raf inhibitor which bind to the DFG-out conformation with ki of 1 nM,1 nM,and 0.3 nM for B-Raf(WT), B-Raf(V600E) and C-Raf, respectively.
|
-
Biomedicines, 2023, 11(4)1013
-
Osteoarthritis Cartilage, 2022, 30-11:1443-1454
-
Eur J Med Chem, 2017, 140:510-527
|
|
| S8690 |
RAF709
|
RAF709 is a potent inhibitor of B/C RAF kinase with almost equivalent IC50 values of 0.4 nM for B-RAF and C-RAF, showing a high level of selectivity, demonstrating greater than 99% on-target binding to BRAF, BRAFV600E, and CRAF at 1 μM and very few off-targets with DDR1 (>99%), DDR2 (86%), FRK (92%), and PDGFRb (96%), the only kinases with binding >80% at 1 μM.
|
-
bioRxiv, 2025, 2025.04.29.651188
-
Nature, 2024, 629(8013):927-936
-
Clin Cancer Res, 2020, 20 pii: clincanres
|
|
| S7743 |
CCT196969
|
CCT196969 is a novel orally available, pan-RAF inhibitor with anti-SRC activity. It also inhibits SRC, LCK, and the p38 MAPKs.
|
-
Nat Commun, 2022, 13(1):7113
-
Cell Rep, 2019, 29(3):573-588
|
|
| E1748 |
HG6-64-1
|
HG6-64-1 (HMSL 10017-101-1) is a highly potent and selective inhibitor of B-Raf with an IC50 of 0.09 μM on B-raf V600E transformed Ba/F3 cells.
|
|
|
| E1682New |
Tinlorafenib
|
Tinlorafenib (PF-07284890, ARRY-461) is a potent, brain-penetrant inhibitor of BRAF targeting V600E/V600K mutations with IC50 values of 4.25 nM and 2.7 nM, respectively. It also effectively inhibits ERK phosphorylation and represents a new class of selective BRAF inhibitors for brain tumors.
|
|
|
| E2505 |
TBAP-001
|
TBAP-001 (Synthesis 13), extracted from patent WO2015075483A1, is a pan-Raf kinase inhibitor, with an IC50 of 62 nM in BRAF V600E kinase assay.
|
|
|
| E8290New |
Flezurafenib
|
Flezurafenib is an oral inhibitor of pan-RAF with potential antineoplastic activity.
|
|
|
| E1079 |
GNE-9815
|
GNE-9815 is a highly selective, pan-Raf inhibitor with good oral bioavailability. This compound exhibits Ki values of 0.062 and 0.19 nM for c-Raf and b-Raf, respectively.
|
|
|
| S6538 |
B-Raf IN 1
|
B-Raf IN 1 is an inhibitor of Raf wih IC50 values of 24 nM and 25 nM for B-Raf and C-Raf respectively. It is selective over 13 other kinases, including PKCα, IKKβ, and PI3Kα.
|
|
|
| E2651 |
Raf inhibitor 1
|
Raf inhibitor 1 is a potent Raf kinase inhibitor with Kis of 1 nM, 1 nM, and 0.3 nM for B-Raf WT, B-Raf V600E, and C-Raf, respectively.
|
|
|
| S9621 |
Donafenib (Sorafenib D3)
|
Donafenib (Sorafenib D3, Bay 43-9006 D3, CM-4307) is the deuterium labeled Sorafenib. Sorafenib is a multikinase inhibitor IC50s of 6 nM, 15 nM, 20 nM and 22 nM for Raf-1, mVEGFR-2, mVEGFR-3 and B-RAF, respectively.
|
|
|
| E4681 |
Claturafenib (PF-07799933)
|
Claturafenib (PF-07799933) is a brain-penetrant, selective, pan-mutant inhibitor of BRAF. It exhibits broad inhibition of pERK levels in HT29 cells with an IC50 value of 1.6 nM. This compound displays antitumor activity against BRAFV600- and non-V600-mutant cancers preclinically and in treatment-refractory patients.
|
|
|
| S6680 |
L-779450
|
L-779450 is a highly potent low nanomolar inhibitor of B-raf with IC50 of 10 nM and Kd of 2.4 nM.
|
-
Sci Adv, 2025, 11(4):eadq2395
|
|
| E1965 |
NST-628
|
NST-628 is a potent and well-tolerated single-agent inhibitor of both RAF and MEK. It acts as a pan-RAF–MEK molecular glue that blocks the phosphorylation and activation of MEK by RAF.
|
|
|
| E4599 |
BGB-3245(Brimarafenib)
|
BGB-3245 (Brimarafenib) is an inhibitor of Raf kinase. This compound displays potent effects against different RAF isoforms, including BRAF Class I/II/III mutations, BRAF fusions, and heterodimerization with CRAF in preclinical models. It also exhibits antineoplastic effects.
|
|
|
| E0934 |
Raf inhibitor 2
|
Raf inhibitor 2 is a potent Raf kinase inhibitor with an IC50 of <1.0 μM.
|
|
|
| S8755 |
AZ304
|
AZ304 is a synthetic inhibitor designed to interact with the ATP-binding site of wild type and V600E mutant BRAF with IC50 values of 79 nM and 38 nM, respectively. It also inhibits CRAF, p38 and CSF1R at sub 100 nM potencies.
|
|
|
| E1649 |
Exarafenib
|
Exarafenib(KIN-002787, KIN-2787, RAF/KIN_2787) is an orally-available, selective inhibitor of pan-RAF. Exarafenib is effective in RAF-dependent cancers, including all classes of BRAF alterations. Exarafenib suppresses MAPK signaling in RAF-dependent melanoma cell lines. KIN-2787 exhibits low nanomolar to picomolar potency against RAF1, BRAF, and ARAF with an IC50 of 0.06-3.46 nM with minimal activity towards non-RAF kinases.
|
|
|
| S1178 |
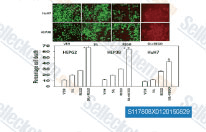
Regorafenib (BAY 73-4506)
|
Regorafenib is a multi-target inhibitor for VEGFR1, VEGFR2, VEGFR3, PDGFRβ, Kit (c-Kit), RET (c-RET) and Raf-1 with IC50 of 13 nM/4.2 nM/46 nM, 22 nM, 7 nM, 1.5 nM and 2.5 nM in cell-free assays, respectively. Regorafenib induces autophagy.
|
-
Nat Commun, 2025, 16(1):509
-
Cell Death Differ, 2025, 10.1038/s41418-025-01536-1
-
Cell Mol Gastroenterol Hepatol, 2025, S2352-345X(25)00026-8
|
|
| S1574 |
Doramapimod (BIRB 796)
|
Doramapimod (BIRB 796) is a pan-p38 MAPK inhibitor with IC50 of 38 nM, 65 nM, 200 nM and 520 nM for p38α/β/γ/δ in cell-free assays. It binds p38α with Kd of 0.1 nM in THP-1 cells, showing 330-fold greater selectivity versus JNK2, weak inhibition for c-RAF, Fyn and Lck, and insignificant inhibition of ERK-1, SYK, IKK2.
|
-
Nat Cancer, 2025, 6(2):259-277
-
Nat Commun, 2025, 16(1):3319
-
Nat Commun, 2025, 16(1):1834
|

|
| S5077 |
Regorafenib (BAY-734506) Monohydrate
|
Regorafenib (BAY-734506, Fluoro-sorafenib, Resihance, Stivarga, regorafaenib monohydrate) Monohydrate is a novel oral multikinase inhibitor with IC50 values of 13, 4.2, 46, 22, 7, 1.5, 2.5, 28, 19 nM for VEGFR1, murine VEGFR2, murine VEGFR3, PDGFR-β, Kit (c-Kit), RET (c-RET), RAF-1, B-RAF and B-RAF(V600E) respectively.
|
-
Nature, 2024, 629(8011):450-457
-
iScience, 2024, 27(10):110862
-
STAR Protoc, 2024, 5(2):103090
|
|
| S4947 |
Regorafenib Hydrochloride
|
Regorafenib (Stivarga, BAY 73-4506) Hydrochloride is a multi-target inhibitor for VEGFR1, Murine VEGFR2/3, PDGFRβ, Kit (c-Kit), RET (c-RET) and Raf-1 with IC50 of 13 nM, 4.2 nM/46 nM, 22 nM, 7 nM, 1.5 nM and 2.5 nM, respectively.
|
-
Cancers (Basel), 2023, 15(18)4477
-
Cancers (Basel), 2023, 15(18)4477
-
J Clin Med, 2023, 12(23)7267
|
|
| S2202 |
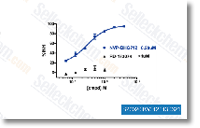
NVP-BHG712
|
NVP-BHG712 is a specific EphB4 inhibitor with ED50 of 25 nM that discriminates between VEGFR and EphB4 inhibition; this compound also shows activity against c-Raf, c-Src and c-Abl with IC50 of 0.395 μM, 1.266 μM and 1.667 μM, respectively.
|
-
BMC Oral Health, 2025, 25(1):118
-
Nat Commun, 2024, 15(1):5894
-
Apoptosis, 2024, 10.1007/s10495-024-02042-4
|
|
| S8189 |
BAW2881 (NVP-BAW2881)
|
BAW2881 (NVP-BAW2881) is a novel vascular endothelial growth factor (VEGF) receptor tyrosine-kinase inhibitor that potently inhibits VEGFR1-3 at 1.0-4.3 nanomolar (nM) concentrations and inhibits PDGFRβ, c-Kit, and RET (c-RET) at 45-72 nM concentrations.
|
-
J Transl Med, 2025, 23(1):985
-
JCI Insight, 2021, 143285
-
J Cell Biochem, 2019, 120(4):5583-5596
|
|
| S6905 |
MCP110
|
MCP110 is an inhibitor of Ras/Raf-1 interaction. This compound disrupts the interaction of activated Ras with Raf and is potential for the treatment of human tumors.
|
|
|
| S1267 |
Vemurafenib (PLX4032)
|
Vemurafenib (PLX4032, RG7204, RO5185426) is a novel and potent inhibitor of B-RafV600E with IC50 of 31 nM in cell-free assay, exhibiting 10-fold selectivity for B-RafV600E over wild-type B-Raf in enzymatic assays, and the cellular selectivity can exceed 100-fold. This compound also induces autophagy. |
- Nat Commun, 2025, 16(1):512
- Int J Biol Sci, 2025, 21(11):4961-4978
- Cells, 2025, 14(12)913
|

|
| S7397 |
Sorafenib (BAY 43-9006)
|
Sorafenib is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. Sorafenib inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. Sorafenib induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity. |
- Mol Cancer, 2025, 24(1):34
- Nat Commun, 2025, 16(1):509
- Adv Sci (Weinh), 2025, 12(30):e04372
|

|
| S2807 |
Dabrafenib (GSK2118436)
|
Dabrafenib is a mutant BRAFV600E specific inhibitor with IC50 of 0.7 nM in cell-free assays, with 7- and 9-fold less potency against B-Raf(wt) and c-Raf, respectively. |
- Nat Commun, 2025, 16(1):512
- J Clin Invest, 2025, e191781
- Cell Rep Med, 2025, 6(6):102183
|

|
| S1040 |
Sorafenib tosylate
|
Sorafenib tosylate is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. This compound inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. It induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity. |
- Int J Oncol, 2025, 67(3)72
- Nature, 2024, 629(8013):927-936
- Cell Mol Life Sci, 2024, 81(1):238
|

|
| S1152 |
PLX-4720
|
PLX4720 is a potent and selective inhibitor of B-RafV600E with IC50 of 13 nM in a cell-free assay, equally potent to c-Raf-1(Y340D and Y341D mutations), 10-fold selectivity for B-RafV600E than wild-type B-Raf. |
- J Clin Invest, 2025, 135(18)e178446
- Cancer Res, 2025, 10.1158/0008-5472.CAN-24-3267
- Cell Prolif, 2025, e70086.
|

|
| S5069 |
Dabrafenib Mesylate
|
Dabrafenib Mesylate (GSK2118436) is the mesylate salt form of dabrafenib, an orally bioavailable inhibitor of B-raf (BRAF) protein with IC50s of 0.8 nM, 3.2 nM and 5 nM for B-Raf (V600E), B-Raf (WT) and C-Raf, respectively. |
- EMBO Mol Med, 2024, 10.1038/s44321-024-00106-1
- iScience, 2024, 27(10):110862
- Cells, 2023, 12(4)664
|
|
| S7842 |
LY3009120
|
LY3009120 (DP-4978) is a potent pan-Raf inhibitor with IC50 of 44 nM, 31-47 nM, and 42 nM for A-raf, B-Raf, and C-Raf in A375 cells, respectively. This compound induces autophagy. Phase 1. |
- Cancer Res, 2025, 10.1158/0008-5472.CAN-24-3819
- J Biol Chem, 2025, 301(8):110454
- Front Biosci (Landmark Ed), 2025, 30(8):39944
|

|
| S2746 |
AZ 628
|
AZ628 is a new pan-Raf inhibitor for BRAF, BRAFV600E, and c-Raf-1 with IC50 of 105 nM, 34 nM and 29 nM in cell-free assays, also inhibits VEGFR2, DDR2, Lyn, Flt1, FMS, etc. AZ628 induces apoptosis. |
- J Biol Chem, 2025, 301(8):110454
- PLoS One, 2024, 19(1):e0295629
- Cell Genom, 2024, 4(2):100487
|

|
| S1104 |
GDC-0879
|
GDC-0879 (AR-00341677) is a novel, potent, and selective B-Raf inhibitor with IC50 of 0.13 nM in A375 and Colo205 cells with activity against c-Raf as well; no inhibition known to other protein kinases. |
- J Biol Chem, 2025, 301(8):110454
- bioRxiv, 2025, 2025.04.29.651188
- Nature, 2024, 629(8013):927-936
|

|
| S2220 |
SB590885
|
SB590885 is a potent B-Raf inhibitor with Ki of 0.16 nM in a cell-free assay, 11-fold greater selectivity for B-Raf over c-Raf, no inhibition to other human kinases. |
- Commun Biol, 2025, 8(1):125
- Nat Commun, 2024, 15(1):668
- Mol Cell, 2024, 84(14):2665-2681.e13
|

|
| S7291 |
TAK-632
|
TAK-632 is a potent pan-Raf inhibitor with IC50 of 8.3 nM and 1.4 nM for B-Raf(wt) and C-Raf in cell-free assays, respectively, showing less or no inhibition against other tested kinases. |
- bioRxiv, 2025, 2025.04.29.651188
- Cell Rep, 2024, 43(9):114710
- Cell Rep, 2024, 43(7):114471
|

|
| S2161 |
RAF265 (CHIR-265)
|
RAF265 (CHIR-265) is a potent selective inhibitor of C-Raf/B-Raf/B-Raf V600E with IC50 of 3-60 nM, and exhibits potent inhibition on VEGFR2 phosphorylation with EC50 of 30 nM in cell-free assays. This compound induces cell cycle arrest and apoptosis. Phase 2. |
- bioRxiv, 2025, 2025.04.29.651188
- Cancer Cell, 2022, S1535-6108(22)00312-9
- iScience, 2022, 25(10):105182
|

|
| S2872 |
GW5074
|
GW5074 is a potent and selective c-Raf inhibitor with IC50 of 9 nM, no effect on the activities of JNK1/2/3, MEK1, MKK6/7, CDK1/2, c-Src, p38 MAP, VEGFR2 or c-Fms is noted. This compound inhibits LK-induced apoptosis. |
- Cancers (Basel), 2025, 17(18)2958
- Aging Cell, 2024, e14320.
- University of Central Florida, 2022,
|

|
| S7108 |
Encorafenib (LGX818)
|
Encorafenib (LGX818) is a highly potent RAF inhibitor with selective anti-proliferative and apoptotic activity in cells expressing B-RAF(V600E) with EC50 of 4 nM. This compound has reached Phase 3. |
- Cell Rep Med, 2025, 6(2):101943
- J Immunother Cancer, 2025, 13(1)e010460
- Oncogenesis, 2025, 14(1):27
|

|
| S7170 |
Avutometinib (Ro 5126766)
|
Avutometinib(RO5126766,CH5126766,VS 6766, CKI-27, R-7304, RG-7304) is a dual RAF/MEK inhibitor with IC50 of 8.2 nM,19 nM, 56 nM, and 160 nM for BRAF V600E, BRAF, CRAF, and MEK1, respectively. Phase 1.
|
- Cancer Chemother Pharmacol, 2025, 95(1):78
- Nat Biomed Eng, 2024, 10.1038/s41551-024-01273-9
- Cell Rep Med, 2024, 5(11):101818
|
|
| S7964 |
PLX7904
|
PLX7904, also known as PB04, is a potent and selective paradox-breaker RAF inhibitor. It is able to efficiently inhibit activation of ERK1/2 in mutant BRAF melanoma cells but does not hyperactivate ERK1/2 in mutant RAS-expressing cells. |
- Nat Commun, 2022, 13(1):4109
- J Invest Dermatol, 2022, S0022-202X(22)00096-3
- Mol Cancer Ther, 2020, 25;molcanther.1021.2019
|
|
| S7965 |
Plixorafenib (PLX8394)
|
Plixorafenib (PLX8394) is a next-generation, orally available, small-molecule BRAF inhibitor with IC50 values of 3.8 nM, 14 nM and 23 nM for BRAF(V600E), WT BRAF and CRAF respectively. It has potential antineoplastic activity. |
- J Biol Chem, 2025, 301(8):110454
- Nature, 2024, 629(8013):927-936
- Cell Rep, 2024, 43(10):114829
|
|
| S2720 |
ZM 336372
|
ZM 336372 (Zinc00581684) is a potent and selective c-Raf inhibitor with IC50 of 70 nM, 10-fold selectivity over B-RAF, no inhibition to PKA/B/C, AMPK, p70S6, etc. |
- Clin Cancer Res, 2021, 10.1158/1078-0432.CCR-21-2660
- Cell Metab, 2020, 5;31(5):892-908 e11
- Exp Gerontol, 2019, 126:110691
|

|
| S8015 |
Agerafenib (CEP-32496)
|
Agerafenib (CEP-32496) is a highly potent inhibitor of BRAF(V600E/WT) and c-Raf with Kd of 14 nM/36 nM and 39 nM, respectively. It also shows potent activity against Abl-1, c-Kit, Ret (c-Ret), PDGFRβ and VEGFR2, but has insignificant affinity for MEK-1, MEK-2, ERK-1 and ERK-2. This compound is currently in Phase 1/2 clinical trials. |
- Mol Pharmacol, 2021, 99(6):435-447
- Dis Model Mech, 2020, dmm.047779
- Cancer Immunol Res, 2019,
|
|
| S8745 |
Naporafenib (LXH254)
|
Naporafenib (LXH254) is a type II ATP-competitive inhibitor that, at picomolar concentrations, inhibits both B- and CRAF kinase activities with a high degree of selectivity against a panel of 456 human kinases and in cell-based assays. |
- bioRxiv, 2025, 2025.06.10.658786
- Cell Commun Signal, 2024, 22(1):428
- Sci Adv, 2023, 9(35):eade7486
|
|
| S7926 |
Lifirafenib (BGB-283)
|
Lifirafenib (BGB-283, Beigene-283) potently inhibits RAF family kinases and EGFR activities in biochemical assays, with IC50 values of 23, 29 and 495 nM for the recombinant BRAFV600E kinase domain, EGFR and EGFR T790M/L858R mutant. |
- Sci Adv, 2023, 9(35):eade7486
- Mol Cancer Ther, 2022, MCT-22-0004
- Nat Commun, 2020, 11(1):3157
|
|
| S7121 |
Tovorafenib (MLN2480)
|
Tovorafenib (MLN2480, BIIB-024, TAK580, AMG-2112819, BSK1369, DAY-101) is an oral, selective pan-Raf kinase inhibitor in chinical trials. |
- bioRxiv, 2025, 2025.04.29.651188
- Sci Adv, 2023, 9(35):eade7486
- Int J Hematol, 2021, 10.1007/s12185-021-03244-1
|
|
| S8853 |
Belvarafenib
|
Belvarafenib is a selective and orally bioavailable pan-RAF kinase inhibitor with IC50 values of 41 nM, 7 nM and 2 nM for BRAF WT, BRAF(V600E) and CRAF kinases, respectively. |
- Sci Adv, 2023, 9(35):eade7486
- SLAS Discov, 2022, S2472-5552(22)12517-7
- Clin Cancer Res, 2021, 10.1158/1078-0432.CCR-21-2660
|
|
| S6660 |
B-Raf inhibitor 1 (Compound 13) dihydrochloride
|
B-Raf inhibitor 1 (Compound 13) is a type IIA Raf inhibitor which bind to the DFG-out conformation with ki of 1 nM,1 nM,and 0.3 nM for B-Raf(WT), B-Raf(V600E) and C-Raf, respectively. |
- Biomedicines, 2023, 11(4)1013
- Osteoarthritis Cartilage, 2022, 30-11:1443-1454
- Eur J Med Chem, 2017, 140:510-527
|
|
| S8690 |
RAF709
|
RAF709 is a potent inhibitor of B/C RAF kinase with almost equivalent IC50 values of 0.4 nM for B-RAF and C-RAF, showing a high level of selectivity, demonstrating greater than 99% on-target binding to BRAF, BRAFV600E, and CRAF at 1 μM and very few off-targets with DDR1 (>99%), DDR2 (86%), FRK (92%), and PDGFRb (96%), the only kinases with binding >80% at 1 μM. |
- bioRxiv, 2025, 2025.04.29.651188
- Nature, 2024, 629(8013):927-936
- Clin Cancer Res, 2020, 20 pii: clincanres
|
|
| S7743 |
CCT196969
|
CCT196969 is a novel orally available, pan-RAF inhibitor with anti-SRC activity. It also inhibits SRC, LCK, and the p38 MAPKs. |
- Nat Commun, 2022, 13(1):7113
- Cell Rep, 2019, 29(3):573-588
|
|
| E1748 |
HG6-64-1
|
HG6-64-1 (HMSL 10017-101-1) is a highly potent and selective inhibitor of B-Raf with an IC50 of 0.09 μM on B-raf V600E transformed Ba/F3 cells. |
|
|
| E1682New |
Tinlorafenib
|
Tinlorafenib (PF-07284890, ARRY-461) is a potent, brain-penetrant inhibitor of BRAF targeting V600E/V600K mutations with IC50 values of 4.25 nM and 2.7 nM, respectively. It also effectively inhibits ERK phosphorylation and represents a new class of selective BRAF inhibitors for brain tumors. |
|
|
| E2505 |
TBAP-001
|
TBAP-001 (Synthesis 13), extracted from patent WO2015075483A1, is a pan-Raf kinase inhibitor, with an IC50 of 62 nM in BRAF V600E kinase assay. |
|
|
| E8290New |
Flezurafenib
|
Flezurafenib is an oral inhibitor of pan-RAF with potential antineoplastic activity. |
|
|
| E1079 |
GNE-9815
|
GNE-9815 is a highly selective, pan-Raf inhibitor with good oral bioavailability. This compound exhibits Ki values of 0.062 and 0.19 nM for c-Raf and b-Raf, respectively. |
|
|
| S6538 |
B-Raf IN 1
|
B-Raf IN 1 is an inhibitor of Raf wih IC50 values of 24 nM and 25 nM for B-Raf and C-Raf respectively. It is selective over 13 other kinases, including PKCα, IKKβ, and PI3Kα. |
|
|
| E2651 |
Raf inhibitor 1
|
Raf inhibitor 1 is a potent Raf kinase inhibitor with Kis of 1 nM, 1 nM, and 0.3 nM for B-Raf WT, B-Raf V600E, and C-Raf, respectively. |
|
|
| S9621 |
Donafenib (Sorafenib D3)
|
Donafenib (Sorafenib D3, Bay 43-9006 D3, CM-4307) is the deuterium labeled Sorafenib. Sorafenib is a multikinase inhibitor IC50s of 6 nM, 15 nM, 20 nM and 22 nM for Raf-1, mVEGFR-2, mVEGFR-3 and B-RAF, respectively. |
|
|
| E4681 |
Claturafenib (PF-07799933)
|
Claturafenib (PF-07799933) is a brain-penetrant, selective, pan-mutant inhibitor of BRAF. It exhibits broad inhibition of pERK levels in HT29 cells with an IC50 value of 1.6 nM. This compound displays antitumor activity against BRAFV600- and non-V600-mutant cancers preclinically and in treatment-refractory patients. |
|
|
| S6680 |
L-779450
|
L-779450 is a highly potent low nanomolar inhibitor of B-raf with IC50 of 10 nM and Kd of 2.4 nM. |
- Sci Adv, 2025, 11(4):eadq2395
|
|
| E1965 |
NST-628
|
NST-628 is a potent and well-tolerated single-agent inhibitor of both RAF and MEK. It acts as a pan-RAF–MEK molecular glue that blocks the phosphorylation and activation of MEK by RAF. |
|
|
| E4599 |
BGB-3245(Brimarafenib)
|
BGB-3245 (Brimarafenib) is an inhibitor of Raf kinase. This compound displays potent effects against different RAF isoforms, including BRAF Class I/II/III mutations, BRAF fusions, and heterodimerization with CRAF in preclinical models. It also exhibits antineoplastic effects. |
|
|
| E0934 |
Raf inhibitor 2
|
Raf inhibitor 2 is a potent Raf kinase inhibitor with an IC50 of <1.0 μM. |
|
|
| S8755 |
AZ304
|
AZ304 is a synthetic inhibitor designed to interact with the ATP-binding site of wild type and V600E mutant BRAF with IC50 values of 79 nM and 38 nM, respectively. It also inhibits CRAF, p38 and CSF1R at sub 100 nM potencies. |
|
|
| E1649 |
Exarafenib
|
Exarafenib(KIN-002787, KIN-2787, RAF/KIN_2787) is an orally-available, selective inhibitor of pan-RAF. Exarafenib is effective in RAF-dependent cancers, including all classes of BRAF alterations. Exarafenib suppresses MAPK signaling in RAF-dependent melanoma cell lines. KIN-2787 exhibits low nanomolar to picomolar potency against RAF1, BRAF, and ARAF with an IC50 of 0.06-3.46 nM with minimal activity towards non-RAF kinases. |
|
|
| S1178 |
Regorafenib (BAY 73-4506)
|
Regorafenib is a multi-target inhibitor for VEGFR1, VEGFR2, VEGFR3, PDGFRβ, Kit (c-Kit), RET (c-RET) and Raf-1 with IC50 of 13 nM/4.2 nM/46 nM, 22 nM, 7 nM, 1.5 nM and 2.5 nM in cell-free assays, respectively. Regorafenib induces autophagy. |
- Nat Commun, 2025, 16(1):509
- Cell Death Differ, 2025, 10.1038/s41418-025-01536-1
- Cell Mol Gastroenterol Hepatol, 2025, S2352-345X(25)00026-8
|

|
| S1574 |
Doramapimod (BIRB 796)
|
Doramapimod (BIRB 796) is a pan-p38 MAPK inhibitor with IC50 of 38 nM, 65 nM, 200 nM and 520 nM for p38α/β/γ/δ in cell-free assays. It binds p38α with Kd of 0.1 nM in THP-1 cells, showing 330-fold greater selectivity versus JNK2, weak inhibition for c-RAF, Fyn and Lck, and insignificant inhibition of ERK-1, SYK, IKK2. |
- Nat Cancer, 2025, 6(2):259-277
- Nat Commun, 2025, 16(1):3319
- Nat Commun, 2025, 16(1):1834
|

|
| S5077 |
Regorafenib (BAY-734506) Monohydrate
|
Regorafenib (BAY-734506, Fluoro-sorafenib, Resihance, Stivarga, regorafaenib monohydrate) Monohydrate is a novel oral multikinase inhibitor with IC50 values of 13, 4.2, 46, 22, 7, 1.5, 2.5, 28, 19 nM for VEGFR1, murine VEGFR2, murine VEGFR3, PDGFR-β, Kit (c-Kit), RET (c-RET), RAF-1, B-RAF and B-RAF(V600E) respectively. |
- Nature, 2024, 629(8011):450-457
- iScience, 2024, 27(10):110862
- STAR Protoc, 2024, 5(2):103090
|
|
| S4947 |
Regorafenib Hydrochloride
|
Regorafenib (Stivarga, BAY 73-4506) Hydrochloride is a multi-target inhibitor for VEGFR1, Murine VEGFR2/3, PDGFRβ, Kit (c-Kit), RET (c-RET) and Raf-1 with IC50 of 13 nM, 4.2 nM/46 nM, 22 nM, 7 nM, 1.5 nM and 2.5 nM, respectively. |
- Cancers (Basel), 2023, 15(18)4477
- Cancers (Basel), 2023, 15(18)4477
- J Clin Med, 2023, 12(23)7267
|
|
| S2202 |
NVP-BHG712
|
NVP-BHG712 is a specific EphB4 inhibitor with ED50 of 25 nM that discriminates between VEGFR and EphB4 inhibition; this compound also shows activity against c-Raf, c-Src and c-Abl with IC50 of 0.395 μM, 1.266 μM and 1.667 μM, respectively. |
- BMC Oral Health, 2025, 25(1):118
- Nat Commun, 2024, 15(1):5894
- Apoptosis, 2024, 10.1007/s10495-024-02042-4
|

|
| S8189 |
BAW2881 (NVP-BAW2881)
|
BAW2881 (NVP-BAW2881) is a novel vascular endothelial growth factor (VEGF) receptor tyrosine-kinase inhibitor that potently inhibits VEGFR1-3 at 1.0-4.3 nanomolar (nM) concentrations and inhibits PDGFRβ, c-Kit, and RET (c-RET) at 45-72 nM concentrations. |
- J Transl Med, 2025, 23(1):985
- JCI Insight, 2021, 143285
- J Cell Biochem, 2019, 120(4):5583-5596
|
|
| S6905 |
MCP110
|
MCP110 is an inhibitor of Ras/Raf-1 interaction. This compound disrupts the interaction of activated Ras with Raf and is potential for the treatment of human tumors. |
|
|








































































