- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Magnetic Separator
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Sorafenib
Synonyms: NSC-724772,BAY 43-9006
Sorafenib is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. Sorafenib inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. Sorafenib induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity.
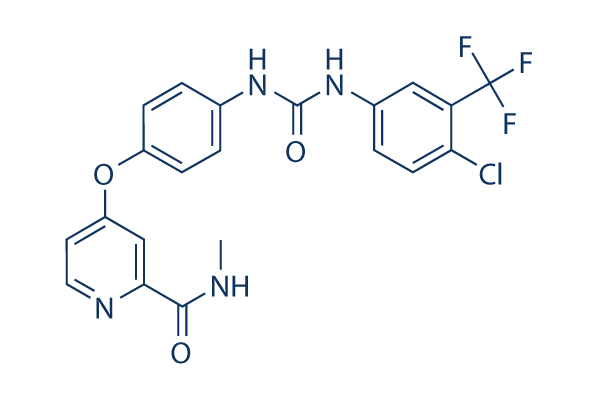
Sorafenib Chemical Structure
CAS No. 284461-73-0
Purity & Quality Control
Batch:
Purity:
99.91%
99.91
Sorafenib Related Products
| Related Targets | C-Raf/Raf-1 B-Raf A-raf | Click to Expand |
|---|---|---|
| Related Products | PLX-4720 LY3009120 AZ 628 GDC-0879 SB590885 TAK-632 RAF265 (CHIR-265) GW5074 Avutometinib PLX7904 Plixorafenib (PLX8394) ZM 336372 Naporafenib (LXH254) Lifirafenib (BGB-283) Agerafenib (CEP-32496) Tovorafenib (MLN2480) Belvarafenib B-Raf inhibitor 1 (Compound 13) dihydrochloride RAF709 CCT196969 | Click to Expand |
| Related Compound Libraries | Kinase Inhibitor Library MAPK Inhibitor Library Cell Cycle compound library TGF-beta/Smad compound library Anti-alzheimer Disease Compound Library | Click to Expand |
Signaling Pathway
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MV-4-11 | Growth Inhibition Assay | IC50=0.00000303 μM | SANGER | |||
| MONO-MAC-6 | Growth Inhibition Assay | IC50=0.00418 μM | SANGER | |||
| ALL-PO | Growth Inhibition Assay | IC50=0.03184 μM | SANGER | |||
| NKM-1 | Growth Inhibition Assay | IC50=0.07416 μM | SANGER | |||
| CGTH-W-1 | Growth Inhibition Assay | IC50=0.25022 μM | SANGER | |||
| BB65-RCC | Growth Inhibition Assay | IC50=0.47073 μM | SANGER | |||
| NOS-1 | Growth Inhibition Assay | IC50=0.5636 μM | SANGER | |||
| SH-4 | Growth Inhibition Assay | IC50=0.65613 μM | SANGER | |||
| HOP-62 | Growth Inhibition Assay | IC50=0.85088 μM | SANGER | |||
| HCC2998 | Growth Inhibition Assay | IC50=0.88818 μM | SANGER | |||
| GDM-1 | Growth Inhibition Assay | IC50=0.90698 μM | SANGER | |||
| KM12 | Growth Inhibition Assay | IC50=1.02098 μM | SANGER | |||
| LB2518-MEL | Growth Inhibition Assay | IC50=1.20809 μM | SANGER | |||
| NCI-H1436 | Growth Inhibition Assay | IC50=1.21678 μM | SANGER | |||
| EM-2 | Growth Inhibition Assay | IC50=1.35578 μM | SANGER | |||
| LAMA-84 | Growth Inhibition Assay | IC50=1.37648 μM | SANGER | |||
| KG-1 | Growth Inhibition Assay | IC50=1.47935 μM | SANGER | |||
| A388 | Growth Inhibition Assay | IC50=1.59165 μM | SANGER | |||
| no-10 | Growth Inhibition Assay | IC50=1.61726 μM | SANGER | |||
| SF126 | Growth Inhibition Assay | IC50=1.63812 μM | SANGER | |||
| MEG-01 | Growth Inhibition Assay | IC50=1.8098 μM | SANGER | |||
| A3-KAW | Growth Inhibition Assay | IC50=1.8842 μM | SANGER | |||
| D-247MG | Growth Inhibition Assay | IC50=2.1448 μM | SANGER | |||
| OVCAR-4 | Growth Inhibition Assay | IC50=2.21393 μM | SANGER | |||
| NCI-SNU-1 | Growth Inhibition Assay | IC50=2.3162 μM | SANGER | |||
| NCI-H2171 | Growth Inhibition Assay | IC50=2.39764 μM | SANGER | |||
| SIG-M5 | Growth Inhibition Assay | IC50=2.42242 μM | SANGER | |||
| BE-13 | Growth Inhibition Assay | IC50=2.69609 μM | SANGER | |||
| K052 | Growth Inhibition Assay | IC50=2.74616 μM | SANGER | |||
| L-540 | Growth Inhibition Assay | IC50=2.75789 μM | SANGER | |||
| KMOE-2 | Growth Inhibition Assay | IC50=2.8135 μM | SANGER | |||
| MFH-ino | Growth Inhibition Assay | IC50=2.92185 μM | SANGER | |||
| HL-60 | Growth Inhibition Assay | IC50=3.06299 μM | SANGER | |||
| HCC2218 | Growth Inhibition Assay | IC50=3.12003 μM | SANGER | |||
| TE-5 | Growth Inhibition Assay | IC50=3.13162 μM | SANGER | |||
| MZ1-PC | Growth Inhibition Assay | IC50=3.47509 μM | SANGER | |||
| MRK-nu-1 | Growth Inhibition Assay | IC50=3.61468 μM | SANGER | |||
| MZ7-mel | Growth Inhibition Assay | IC50=3.66099 μM | SANGER | |||
| BC-1 | Growth Inhibition Assay | IC50=3.7402 μM | SANGER | |||
| ST486 | Growth Inhibition Assay | IC50=3.83673 μM | SANGER | |||
| KS-1 | Growth Inhibition Assay | IC50=3.88198 μM | SANGER | |||
| SK-NEP-1 | Growth Inhibition Assay | IC50=4.16815 μM | SANGER | |||
| BC-3 | Growth Inhibition Assay | IC50=4.23391 μM | SANGER | |||
| NCI-H1581 | Growth Inhibition Assay | IC50=4.28798 μM | SANGER | |||
| MHH-PREB-1 | Growth Inhibition Assay | IC50=4.40484 μM | SANGER | |||
| NOMO-1 | Growth Inhibition Assay | IC50=4.48905 μM | SANGER | |||
| QIMR-WIL | Growth Inhibition Assay | IC50=5.07294 μM | SANGER | |||
| SF539 | Growth Inhibition Assay | IC50=5.13227 μM | SANGER | |||
| TE-12 | Growth Inhibition Assay | IC50=5.24929 μM | SANGER | |||
| NCI-H510A | Growth Inhibition Assay | IC50=5.41685 μM | SANGER | |||
| JAR | Growth Inhibition Assay | IC50=5.50824 μM | SANGER | |||
| no-11 | Growth Inhibition Assay | IC50=5.73568 μM | SANGER | |||
| BV-173 | Growth Inhibition Assay | IC50=5.95682 μM | SANGER | |||
| SR | Growth Inhibition Assay | IC50=6.00678 μM | SANGER | |||
| MOLT-16 | Growth Inhibition Assay | IC50=6.25266 μM | SANGER | |||
| MZ2-MEL | Growth Inhibition Assay | IC50=6.31839 μM | SANGER | |||
| SW954 | Growth Inhibition Assay | IC50=6.45866 μM | SANGER | |||
| ML-2 | Growth Inhibition Assay | IC50=6.52849 μM | SANGER | |||
| OCI-AML2 | Growth Inhibition Assay | IC50=6.61062 μM | SANGER | |||
| SIMA | Growth Inhibition Assay | IC50=7.00101 μM | SANGER | |||
| DOHH-2 | Growth Inhibition Assay | IC50=7.05676 μM | SANGER | |||
| 697 | Growth Inhibition Assay | IC50=7.05989 μM | SANGER | |||
| NB1 | Growth Inhibition Assay | IC50=7.40407 μM | SANGER | |||
| D-392MG | Growth Inhibition Assay | IC50=7.62663 μM | SANGER | |||
| ES8 | Growth Inhibition Assay | IC50=7.76503 μM | SANGER | |||
| RPMI-8226 | Growth Inhibition Assay | IC50=7.84511 μM | SANGER | |||
| IST-MEL1 | Growth Inhibition Assay | IC50=8.40002 μM | SANGER | |||
| NB14 | Growth Inhibition Assay | IC50=8.63133 μM | SANGER | |||
| HD-MY-Z | Growth Inhibition Assay | IC50=8.63746 μM | SANGER | |||
| TE-10 | Growth Inhibition Assay | IC50=8.76353 μM | SANGER | |||
| LC-1F | Growth Inhibition Assay | IC50=9.10834 μM | SANGER | |||
| OS-RC-2 | Growth Inhibition Assay | IC50=9.11243 μM | SANGER | |||
| NCI-SNU-16 | Growth Inhibition Assay | IC50=9.21026 μM | SANGER | |||
| SHP-77 | Growth Inhibition Assay | IC50=9.71662 μM | SANGER | |||
| A4-Fuk | Growth Inhibition Assay | IC50=9.7561 μM | SANGER | |||
| NB6 | Growth Inhibition Assay | IC50=9.76029 μM | SANGER | |||
| JiyoyeP-2003 | Growth Inhibition Assay | IC50=10.4745 μM | SANGER | |||
| DMS-114 | Growth Inhibition Assay | IC50=10.5441 μM | SANGER | |||
| NB7 | Growth Inhibition Assay | IC50=10.7526 μM | SANGER | |||
| NCI-H747 | Growth Inhibition Assay | IC50=11.1216 μM | SANGER | |||
| HH | Growth Inhibition Assay | IC50=11.3876 μM | SANGER | |||
| EW-18 | Growth Inhibition Assay | IC50=11.9044 μM | SANGER | |||
| CHP-126 | Growth Inhibition Assay | IC50=11.9738 μM | SANGER | |||
| NTERA-S-cl-D1 | Growth Inhibition Assay | IC50=12.0278 μM | SANGER | |||
| DEL | Growth Inhibition Assay | IC50=12.0985 μM | SANGER | |||
| LU-139 | Growth Inhibition Assay | IC50=12.5413 μM | SANGER | |||
| P30-OHK | Growth Inhibition Assay | IC50=12.5479 μM | SANGER | |||
| NCI-H1522 | Growth Inhibition Assay | IC50=12.746 μM | SANGER | |||
| NCI-H1299 | Growth Inhibition Assay | IC50=13.2911 μM | SANGER | |||
| UACC-257 | Growth Inhibition Assay | IC50=13.5126 μM | SANGER | |||
| Calu-6 | Growth Inhibition Assay | IC50=13.6046 μM | SANGER | |||
| NCI-H1882 | Growth Inhibition Assay | IC50=13.8555 μM | SANGER | |||
| BB30-HNC | Growth Inhibition Assay | IC50=14.0609 μM | SANGER | |||
| ES1 | Growth Inhibition Assay | IC50=14.1551 μM | SANGER | |||
| NCI-H1694 | Growth Inhibition Assay | IC50=14.4811 μM | SANGER | |||
| IST-SL1 | Growth Inhibition Assay | IC50=14.9616 μM | SANGER | |||
| ECC4 | Growth Inhibition Assay | IC50=15.0558 μM | SANGER | |||
| MDA-MB-134-VI | Growth Inhibition Assay | IC50=15.4131 μM | SANGER | |||
| SCH | Growth Inhibition Assay | IC50=15.4728 μM | SANGER | |||
| SK-N-FI | Growth Inhibition Assay | IC50=15.6534 μM | SANGER | |||
| HDLM-2 | Growth Inhibition Assay | IC50=16.0714 μM | SANGER | |||
| Ramos-2G6-4C10 | Growth Inhibition Assay | IC50=16.1297 μM | SANGER | |||
| EW-24 | Growth Inhibition Assay | IC50=16.1661 μM | SANGER | |||
| NCI-H2141 | Growth Inhibition Assay | IC50=16.189 μM | SANGER | |||
| LC4-1 | Growth Inhibition Assay | IC50=16.6119 μM | SANGER | |||
| HT-144 | Growth Inhibition Assay | IC50=17.006 μM | SANGER | |||
| SK-MEL-1 | Growth Inhibition Assay | IC50=17.0072 μM | SANGER | |||
| SCC-15 | Growth Inhibition Assay | IC50=17.1638 μM | SANGER | |||
| C8166 | Growth Inhibition Assay | IC50=17.6833 μM | SANGER | |||
| GOTO | Growth Inhibition Assay | IC50=17.8344 μM | SANGER | |||
| COR-L279 | Growth Inhibition Assay | IC50=18.1362 μM | SANGER | |||
| K-562 | Growth Inhibition Assay | IC50=18.7143 μM | SANGER | |||
| ES3 | Growth Inhibition Assay | IC50=18.8041 μM | SANGER | |||
| LU-165 | Growth Inhibition Assay | IC50=19.7008 μM | SANGER | |||
| KM-H2 | Growth Inhibition Assay | IC50=20.3184 μM | SANGER | |||
| RL | Growth Inhibition Assay | IC50=20.9692 μM | SANGER | |||
| EW-3 | Growth Inhibition Assay | IC50=21.1889 μM | SANGER | |||
| A101D | Growth Inhibition Assay | IC50=21.3752 μM | SANGER | |||
| HUTU-80 | Growth Inhibition Assay | IC50=21.3946 μM | SANGER | |||
| NCI-H23 | Growth Inhibition Assay | IC50=21.3992 μM | SANGER | |||
| PF-382 | Growth Inhibition Assay | IC50=21.4403 μM | SANGER | |||
| LB373-MEL-D | Growth Inhibition Assay | IC50=21.5615 μM | SANGER | |||
| TE-8 | Growth Inhibition Assay | IC50=21.6394 μM | SANGER | |||
| TE-9 | Growth Inhibition Assay | IC50=21.8513 μM | SANGER | |||
| Daudi | Growth Inhibition Assay | IC50=21.9304 μM | SANGER | |||
| D-542MG | Growth Inhibition Assay | IC50=22.0256 μM | SANGER | |||
| U-698-M | Growth Inhibition Assay | IC50=22.4603 μM | SANGER | |||
| ES6 | Growth Inhibition Assay | IC50=22.7366 μM | SANGER | |||
| DU-4475 | Growth Inhibition Assay | IC50=23.8897 μM | SANGER | |||
| ECC12 | Growth Inhibition Assay | IC50=24.2803 μM | SANGER | |||
| C2BBe1 | Growth Inhibition Assay | IC50=24.3239 μM | SANGER | |||
| IST-SL2 | Growth Inhibition Assay | IC50=24.4362 μM | SANGER | |||
| DJM-1 | Growth Inhibition Assay | IC50=24.5221 μM | SANGER | |||
| DMS-153 | Growth Inhibition Assay | IC50=24.8614 μM | SANGER | |||
| NB13 | Growth Inhibition Assay | IC50=25.0265 μM | SANGER | |||
| SK-N-DZ | Growth Inhibition Assay | IC50=26.3414 μM | SANGER | |||
| COR-L88 | Growth Inhibition Assay | IC50=26.5796 μM | SANGER | |||
| LU-65 | Growth Inhibition Assay | IC50=26.8535 μM | SANGER | |||
| TGBC1TKB | Growth Inhibition Assay | IC50=26.9828 μM | SANGER | |||
| THP-1 | Growth Inhibition Assay | IC50=27.2141 μM | SANGER | |||
| ONS-76 | Growth Inhibition Assay | IC50=27.332 μM | SANGER | |||
| LC-2-ad | Growth Inhibition Assay | IC50=27.6231 μM | SANGER | |||
| EW-13 | Growth Inhibition Assay | IC50=29.1746 μM | SANGER | |||
| MS-1 | Growth Inhibition Assay | IC50=30.7278 μM | SANGER | |||
| NCI-H2227 | Growth Inhibition Assay | IC50=30.9806 μM | SANGER | |||
| LXF-289 | Growth Inhibition Assay | IC50=31.4492 μM | SANGER | |||
| MC116 | Growth Inhibition Assay | IC50=32.0826 μM | SANGER | |||
| EVSA-T | Growth Inhibition Assay | IC50=32.2585 μM | SANGER | |||
| CTB-1 | Growth Inhibition Assay | IC50=33.1101 μM | SANGER | |||
| COLO-320-HSR | Growth Inhibition Assay | IC50=33.1603 μM | SANGER | |||
| NCI-H2196 | Growth Inhibition Assay | IC50=33.2557 μM | SANGER | |||
| LB2241-RCC | Growth Inhibition Assay | IC50=33.3135 μM | SANGER | |||
| LS-513 | Growth Inhibition Assay | IC50=33.8638 μM | SANGER | |||
| LP-1 | Growth Inhibition Assay | IC50=33.9956 μM | SANGER | |||
| A253 | Growth Inhibition Assay | IC50=34.2296 μM | SANGER | |||
| SK-MM-2 | Growth Inhibition Assay | IC50=34.9451 μM | SANGER | |||
| NCI-H1963 | Growth Inhibition Assay | IC50=35.3072 μM | SANGER | |||
| MMAC-SF | Growth Inhibition Assay | IC50=35.8785 μM | SANGER | |||
| LB831-BLC | Growth Inhibition Assay | IC50=36.0654 μM | SANGER | |||
| WSU-NHL | Growth Inhibition Assay | IC50=36.164 μM | SANGER | |||
| CESS | Growth Inhibition Assay | IC50=36.2848 μM | SANGER | |||
| NEC8 | Growth Inhibition Assay | IC50=36.5835 μM | SANGER | |||
| KNS-42 | Growth Inhibition Assay | IC50=37.1237 μM | SANGER | |||
| MHH-CALL-2 | Growth Inhibition Assay | IC50=37.1821 μM | SANGER | |||
| K5 | Growth Inhibition Assay | IC50=38.43 μM | SANGER | |||
| CP66-MEL | Growth Inhibition Assay | IC50=39.0733 μM | SANGER | |||
| OPM-2 | Growth Inhibition Assay | IC50=39.8432 μM | SANGER | |||
| IST-MES1 | Growth Inhibition Assay | IC50=40.3096 μM | SANGER | |||
| EC-GI-10 | Growth Inhibition Assay | IC50=41.5805 μM | SANGER | |||
| CTV-1 | Growth Inhibition Assay | IC50=42.8406 μM | SANGER | |||
| DG-75 | Growth Inhibition Assay | IC50=43.7595 μM | SANGER | |||
| KNS-81-FD | Growth Inhibition Assay | IC50=45.4058 μM | SANGER | |||
| NCI-H82 | Growth Inhibition Assay | IC50=45.5758 μM | SANGER | |||
| RPMI-8866 | Growth Inhibition Assay | IC50=46.1873 μM | SANGER | |||
| ACN | Growth Inhibition Assay | IC50=46.434 μM | SANGER | |||
| NCI-H1395 | Growth Inhibition Assay | IC50=46.4756 μM | SANGER | |||
| NCI-H209 | Growth Inhibition Assay | IC50=47.1405 μM | SANGER | |||
| TGW | Growth Inhibition Assay | IC50=49.0791 μM | SANGER | |||
| NCI-H748 | Growth Inhibition Assay | IC50=49.4753 μM | SANGER | |||
| EKVX | Growth Inhibition Assay | IC50=49.6628 μM | SANGER | |||
| MV4-11 | Antiproliferative activity assay | IC50 = 0.00087 μM | 19754199 | |||
| MV4-11 | Cytotoxicity assay | IC50 = 0.00087 μM | 19654408 | |||
| TT | Antiproliferative activity assay | IC50 = 0.0014 μM | 18849971 | |||
| RS4-11 | Function assay | IC50 = 0.002 μM | 19654408 | |||
| MOLM13 | Function assay | IC50 = 0.002 μM | 24641103 | |||
| Sf9 | Function assay | IC50 = 0.003 μM | 21708468 | |||
| RS4-11 | Function assay | IC50 = 0.0032 μM | 19654408 | |||
| MV4-11 | Growth inhibition assay | IC50 = 0.004 μM | 29357250 | |||
| A375 | Function assay | IC50 = 0.0044 μM | 29461827 | |||
| MOLM13 | Antiproliferative activity assay | IC50 = 0.005 μM | 24641103 | |||
| MV4-11 | Function assay | IC50 = 0.007 μM | 23362959 | |||
| MV4-11 | Cytotoxicity assay | IC50 = 0.007 μM | 26342867 | |||
| HEK293 | Function assay | Kd = 0.013 μM | 19754199 | |||
| Kasumi-1 | Antiproliferative activity assay | IC50 = 0.015 μM | 20570526 | |||
| MV4-11 | Antiproliferative activity assay | IC50 = 0.015 μM | 20570526 | |||
| sf9 | Function assay | IC50 = 0.015 μM | 29266937 | |||
| sf9 | Function assay | IC50 = 0.018 μM | 21708468 | |||
| sf9 | Function assay | IC50 = 0.02 μM | 23618709 | |||
| MV4-11 | Antiproliferative activity assay | GI50 = 0.03 μM | 21708468 | |||
| Sf9 | Function assay | IC50 = 0.042 μM | 26081023 | |||
| SF9 | Function assay | IC50 = 0.043 μM | 18473434 | |||
| MV4-11 | Cytotoxicity assay | GI50 = 0.043 μM | 23618709 | |||
| MV4-11 | Function assay | GI50 = 0.043 μM | 26081023 | |||
| SF9 | Function assay | IC50 = 0.044 μM | 26081023 | |||
| SF9 | Function assay | IC50 = 0.054 μM | 21708468 | |||
| SF9 | Function assay | IC50 = 0.054 μM | 23618709 | |||
| MOLM13 | Antiproliferative activity assay | GI50 = 0.056 μM | 21708468 | |||
| MOLM13 | Antiproliferative activity assay | GI50 = 0.056 μM | 22726931 | |||
| MOLM13 | Cytotoxicity assay | GI50 = 0.056 μM | 23618709 | |||
| MOLM-13 | Function assay | GI50 = 0.056 μM | 26081023 | |||
| SF9 | Function assay | IC50 = 0.058 μM | 29266937 | |||
| WM266.4 | Antiproliferative activity assay | IC50 = 0.06 μM | 25496804 | |||
| MDA-MB-231 | Function assay | IC50 = 0.063 μM | 28431342 | |||
| SF9 | Function assay | IC50 = 0.09 μM | 29266937 | |||
| Spodoptera frugiperda | Function assay | IC50 = 0.09 μM | 26922228 | |||
| SF9 | Function assay | IC50 = 0.09 μM | 23562241 | |||
| SF9 | Function assay | IC50 = 0.1 μM | 23442188 | |||
| SF9 | Function assay | IC50 = 0.1 μM | 23442188 | |||
| HEK293 | Function assay | IC50 = 0.12 μM | 26318998 | |||
| MCF7 | Antiproliferative activity assay | IC50 = 0.19 μM | 25496804 | |||
| MV4-11 | Antiproliferative activity human | IC50 = 0.3 μM | 28242553 | |||
| HepG2 | Antiproliferative activity assay | EC50 = 0.302 μM | 27010810 | |||
| THP1 | Antiproliferative activity assay | IC50 = 0.31 μM | 20570526 | |||
| SMMC7721 | Cytotoxicity assay | IC50 = 0.37 μM | 25982075 | |||
| HT-29 | Antiproliferative activity assay | IC50 = 0.39 μM | 25778995 | |||
| sf9 | Function assay | Kd = 0.6 μM | 28109791 | |||
| T47D | Antiproliferative activity assay | IC50 = 0.61 μM | 25778995 | |||
| A549 | Cytotoxicity assay | IC50 = 0.63 μM | 25982075 | |||
| HeLa | Cytotoxicity assay | IC50 = 0.64 μM | 30015070 | |||
| WM3629 | Antiproliferative activity assay | GI50 = 0.65 μM | 20466542 | |||
| SMMC7721 | Antiproliferative activity assay | IC50 = 0.65 μM | 26342134 | |||
| WM3629 | Growth inhibition assay | GI50 = 0.78 μM | 20149658 | |||
| WM3629 | Antiproliferative activity assay | GI50 = 0.78 μM | 26810260 | |||
| MCF7 | Antiproliferative activity assay | IC50 = 0.78 μM | 25778995 | |||
| K562 | Antiproliferative activity assay | IC50 = 0.86 μM | 24315192 | |||
| COLO205 | Antiproliferative activity assay | IC50 = 0.87 μM | 25778995 | |||
| T29 | Cytotoxicity assay | IC50 = 0.9 μM | 26318998 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 0.94 μM | 25086238 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 0.94 μM | 25440879 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 1.06 μM | 29628325 | |||
| BL21(DE3) | Function assay | IC50 = 1.1 μM | 19928858 | |||
| K562 | Antiproliferative activity assay | IC50 = 1.22 μM | 25778995 | |||
| MDA-MB-231 | Growth inhibition assay | GI50 = 1.26 μM | 28088086 | |||
| MDA-MB-231 | Cytotoxicity assay | GI50 = 1.26 μM | 26590508 | |||
| MDA-MB-231 | Antiproliferative activity assay | GI50 = 1.26 μM | 27017549 | |||
| SK-MEL-28 | Antiproliferative activity assay | EC50 = 1.3 μM | 18942827 | |||
| sf9 | Function assay | Kd = 1.3 μM | 28109791 | |||
| F-6-8 | Cytotoxicity assay | IC50 = 1.3 μM | 26318998 | |||
| A549 | Antiproliferative activity assay | IC50 = 1.45 μM | 26342134 | |||
| rhabdomyosarcoma | Antiviral activity assay | EC50 = 1.5 μM | 27288186 | |||
| HL-60(TB) | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| RPMI8226 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| KM12 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| LOXIMVI | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| HCT116 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| HOP92 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| SF539 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| UACC62 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| SF295 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| MDA-MB-435 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| SK-MEL-5 | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| T47D | Growth inhibition assay | GI50 = 1.58 μM | 28088086 | |||
| RPMI8266 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| HL-60(TB) | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| KM12 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| HCT116 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| SF295 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| SF539 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| SK-MEL-2 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| MDA-MB-435 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| SK-MEL-5 | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| LOXIMVI | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| T47D | Cytotoxicity assay | GI50 = 1.58 μM | 26590508 | |||
| SMMC7721 | Antiproliferative activity assay | IC50 = 1.58 μM | 26753815 | |||
| LOXIMVI | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| UACC62 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| SK-MEL-5 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| KM12 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| MDA-MB-435 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| SF539 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| SF295 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| HCT116 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| HOP92 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| RPMI8226 | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| HL-60(TB) | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| T47D | Antiproliferative activity assay | GI50 = 1.58 μM | 27017549 | |||
| 7dF3 | Function assay | IC50 = 1.62 μM | 29266937 | |||
| MDA-MB-435 | Antiproliferative activity assay | IC50 = 1.67 μM | 25778995 | |||
| HL60 | Cytotoxicity assay | IC50 = 1.68 μM | 24858546 | |||
| SW579 | Antiproliferative activity human | IC50 = 1.73 μM | 28242553 | |||
| MCF7 | Antiproliferative activity assay | IC50 = 1.88 μM | 25637123 | |||
| A375P | Antiproliferative activity assay | IC50 = 1.9 μM | 24128410 | |||
| A549 | Cytotoxicity assay | IC50 = 1.92 μM | 24826815 | |||
| HCT116 | Antiproliferative activity assay | IC50 = 2 μM | 22808911 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 2 μM | 23454017 | |||
| NCI-H23 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| NCI-H522 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| HOP62 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| NCI-H226 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| HT-29 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| U251 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| COLO205 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| CCRF-CEM | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| MALME-3M | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| M14 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| UACC257 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| PC3 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| SK-MEL-2 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| MDA-MB-468 | Growth inhibition assay | GI50 = 2 μM | 28088086 | |||
| CCRF-CEM | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| NCI-H23 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| NCI-H522 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| COLO205 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| HT-29 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| SK-MEL-28 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| HOP62 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| UACC257 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| NCI-H226 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| U251 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| PC3 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| M14 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| MDA-MB-468 | Cytotoxicity assay | GI50 = 2 μM | 26590508 | |||
| PC3 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| MALME-3M | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| U251 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| UACC257 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| SK-MEL-2 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| COLO205 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| NCI-H23 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| HT-29 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| M14 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| NCI-H226 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| HOP62 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| MDA-MB-468 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| CCRF-CEM | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| NCI-H522 | Antiproliferative activity assay | GI50 = 2 μM | 27017549 | |||
| A549 | Antiproliferative activity assay | IC50 = 2.02 μM | 26753815 | |||
| MDA-MB-435 | Growth inhibition assay | IC50 = 2.11 μM | 29189002 | |||
| NCI-H460 | Antiproliferative activity assay | IC50 = 2.19 μM | 30216849 | |||
| H460 | Cytotoxicity assay | IC50 = 2.19 μM | 24826815 | |||
| H460 | Cytotoxicity assay | IC50 = 2.19 μM | 25086238 | |||
| H460 | Cytotoxicity assay | IC50 = 2.19 μM | 25440879 | |||
| A375 | Toxicity assay | IC50 = 2.2 μM | 19654408 | |||
| H460 | Antiproliferative activity assay | IC50 = 2.25 μM | 26991938 | |||
| HCT116 | Antiproliferative activity assay | IC50 = 2.3 μM | 23260578 | |||
| MKN45 | Antiproliferative activity assay | IC50 = 2.32 μM | 30216849 | |||
| MKN45 | Cytotoxicity assay | IC50 = 2.32 μM | 24826815 | |||
| MKN45 | Cytotoxicity assay | IC50 = 2.32 μM | 25086238 | |||
| MKN45 | Cytotoxicity assay | IC50 = 2.32 μM | 25440879 | |||
| U937 | Antiproliferative activity assay | GI50 = 2.34 μM | 29459144 | |||
| A375 | Antiproliferative activity assay | IC50 = 2.4 μM | 22808911 | |||
| HeLa | Growth inhibition assay | IC50 = 2.44 μM | 29102175 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 2.5 μM | 22414612 | |||
| MCF7 | Antiproliferative activity assay | IC50 = 2.51 μM | 30216849 | |||
| UO31 | Antiproliferative activity assay | IC50 = 2.51 μM | 30216849 | |||
| NCI-H322M | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| NCI-H460 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| KM12 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| LOXIMVI | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| EKVX | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| HCT116 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| SW620 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SF268 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SF539 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| A498 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| UACC62 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| IGROV1 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| HCT15 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SK-MEL-28 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SK-MEL-28 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| ACHN | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SK-MEL-5 | Growth inhibition assay | TGI = 2.51 μM | 28088086 | |||
| NCI/ADR-RES | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| MCF7 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SN12C | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| Hs 578T | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| SKOV3 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| UO31 | Growth inhibition assay | GI50 = 2.51 μM | 28088086 | |||
| NCI-H460 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| HCT15 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| SW620 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| SF268 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| IGROV1 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| OVCAR8 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| NCI/ADR-RES | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| SKOV3 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| SN12C | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| NCI-H322M | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| MCF7 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| UO31 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| Hs578T | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| A498 | Cytotoxicity assay | GI50 = 2.51 μM | 26590508 | |||
| A498 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| SKOV3 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| UO31 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| NCI-ADR-RES | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| SN12C | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| IGROV1 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| ACHN | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| OVCAR8 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| SK-MEL-28 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| NCI-H322M | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| SF268 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| HCT15 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| NCI-H460 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| SW620 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| EKVX | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| MCF7 | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| Hs 578T | Antiproliferative activity assay | GI50 = 2.51 μM | 27017549 | |||
| HT-29 | Antiproliferative activity assay | IC50 = 2.58 μM | 29886324 | |||
| A375P | Antiproliferative activity assay | GI50 = 2.58 μM | 26810260 | |||
| TPC1 | Antiproliferative activity assay | EC50 = 2.6 μM | 22559926 | |||
| Hep3B | Growth inhibition assay | IC50 = 2.63 μM | 29102175 | |||
| A375P | Antiproliferative activity assay | IC50 = 2.7 μM | 22460030 | |||
| U937 | Antiproliferative activity assay | GI50 = 2.74 μM | 26318067 | |||
| U937 | Cytotoxicity assay | GI50 = 2.74 μM | 24878193 | |||
| MCF7 | Growth inhibition assay | IC50 = 2.78 μM | 29102175 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 2.8 μM | 29202403 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 2.84 μM | 28242553 | |||
| U937 | Growth inhibition assay | GI50 = 2.85 μM | 22014755 | |||
| A549 | Cytotoxicity assay | IC50 = 2.92 μM | 28927801 | |||
| A549 | Cytotoxicity assay | IC50 = 2.92 μM | 27777009 | |||
| ZR75-30 | Growth inhibition assay | IC50 = 2.96 μM | 29102175 | |||
| PC3 | Cytotoxicity assay | IC50 = 3.03 μM | 28340913 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 3.08 μM | 26991938 | |||
| A549 | Growth inhibition assay | IC50 = 3.1 μM | 29102175 | |||
| K562 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| MOLT4 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| A549/ATCC | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| RPMI8226 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| SR | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| HOP62 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| NCI-H226 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| U251 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| COLO205 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| M14 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| HCC2998 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| SNB75 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| 786-0 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| A498 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| HCT15 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| OVCAR3 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| OVCAR4 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| Caki1 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| SNB19 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| OVCAR5 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| DU145 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| OVCAR8 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| SN12C | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| MDA-MB-231 | Growth inhibition assay | TGI = 3.16 μM | 28088086 | |||
| RXF393 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| BT549 | Growth inhibition assay | GI50 = 3.16 μM | 28088086 | |||
| K562 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| MOLT4 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| SNB19 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| SR | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| A549 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| OVCAR3 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| OVCAR4 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| SNB75 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| OVCAR5 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| DU145 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| 786-0 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| BT549 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| ACHN | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| Caki1 | Cytotoxicity assay | GI50 = 3.16 μM | 26590508 | |||
| 786-0 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| LOXIMVI | Antiproliferative activity assay | TGI = 3.16 μM | 27017549 | |||
| Caki1 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| OVCAR5 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| RXF393 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| SNB75 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| OVCAR3 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| SNB19 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| SK-MEL-5 | Antiproliferative activity assay | TGI = 3.16 μM | 27017549 | |||
| OVCAR4 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| SK-MEL-2 | Antiproliferative activity assay | TGI = 3.16 μM | 27017549 | |||
| BT549 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| MOLT4 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| K562 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| HCC2998 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| HCC2998 | Antiproliferative activity assay | TGI = 3.16 μM | 27017549 | |||
| A549/ATCC | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| DU145 | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| SR | Antiproliferative activity assay | GI50 = 3.16 μM | 27017549 | |||
| MCF7 | Cytotoxicity assay | IC50 = 3.18 μM | 28927801 | |||
| PC3 | Cytotoxicity assay | IC50 = 3.18 μM | 28927801 | |||
| MCF7 | Cytotoxicity assay | IC50 = 3.18 μM | 27777009 | |||
| A549 | Antiproliferative activity assay | IC50 = 3.19 μM | 25778995 | |||
| PC3 | Cytotoxicity assay | IC50 = 3.24 μM | 27777009 | |||
| HeLa | Function assay | IC50 = 3.3 μM | 15225706 | |||
| HeLa | Function assay | IC50 = 3.3 μM | 15225706 | |||
| LoVo | Cytotoxicity assay | IC50 = 3.3 μM | 23454017 | |||
| A375 | Antiproliferative activity assay | IC50 = 3.36 μM | 29602674 | |||
| HT-29 | Antiproliferative activity assay | IC50 = 3.37 μM | 26991938 | |||
| FLT3 gene-deficient U937 | Antiproliferative activity assay | GI50 = 3.4 μM | 21708468 | |||
| FLT3 negative U937 | Cytotoxicity assay | GI50 = 3.4 μM | 23618709 | |||
| U937 | Cytotoxicity assay | GI50 = 3.4 μM | 26081023 | |||
| A375P | Antiproliferative activity assay | GI50 = 3.4 μM | 29459144 | |||
| ACHN | Cytotoxicity assay | IC50 = 3.4 μM | 29517908 | |||
| ACHN | Cytotoxicity assay | IC50 = 3.4 μM | 29297688 | |||
| HepG2 | Function assay | IC50 = 3.4 μM | 26071861 | |||
| A375P | Cytotoxicity assay | GI50 = 3.4 μM | 24878193 | |||
| HepG2 | Cytotoxicity assay | IC50 = 3.44 μM | 28927801 | |||
| HepG2 | Cytotoxicity assay | IC50 = 3.44 μM | 27777009 | |||
| ACHN | Cytotoxicity assay | IC50 = 3.5 μM | 29786436 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 3.5 μM | 25462265 | |||
| HT-29 | Cytotoxicity assay | IC50 = 3.61 μM | 24826815 | |||
| HT-29 | Antiproliferative activity assay | IC50 = 3.61 μM | 30216849 | |||
| HT-29 | Cytotoxicity assay | IC50 = 3.61 μM | 25086238 | |||
| HT-29 | Cytotoxicity assay | IC50 = 3.61 μM | 25440879 | |||
| MCF7 | Anticancer activity assay | IC50 = 3.64 μM | 29549841 | |||
| Sf9 | Function assay | IC50 = 3.8 μM | 21708468 | |||
| Sf9 | Function assay | IC50 = 3.8 μM | 23618709 | |||
| Sf9 | Function assay | IC50 = 3.8 μM | 26081023 | |||
| A2058 | Cytotoxicity assay | IC50 = 3.8 μM | 22708987 | |||
| NCI-H460 | Cytotoxicity assay | IC50 = 3.9 μM | 23454017 | |||
| SK-MEL-30 | Cytotoxicity assay | IC50 = 3.9 μM | 29461827 | |||
| HT-29 | Anticancer activity assay | IC50 = 3.97 μM | 29549841 | |||
| SF268 | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| SNB75 | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| OVCAR3 | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| SNB19 | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| Hs 578T | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| TK10 | Growth inhibition assay | GI50 = 3.98 μM | 28088086 | |||
| BT549 | Growth inhibition assay | TGI = 3.98 μM | 28088086 | |||
| Caki1 | Antiproliferative activity assay | TGI = 3.98 μM | 27017549 | |||
| TK10 | Antiproliferative activity assay | GI50 = 3.98 μM | 27017549 | |||
| MDA-MB-435 | Antiproliferative activity assay | TGI = 3.98 μM | 27017549 | |||
| HCT116 | Antiproliferative activity assay | TGI = 3.98 μM | 27017549 | |||
| MDA-MB-231 | Antiproliferative activity assay | TGI = 3.98 μM | 27017549 | |||
| HepG2 | Cytotoxicity assay | IC50 = 4 μM | 23454017 | |||
| HT-29 | Cytotoxicity assay | IC50 = 4 μM | 23454017 | |||
| A549 | Cytotoxicity assay | IC50 = 4 μM | 23454017 | |||
| HuH7 | Cytotoxicity assay | GI50 = 4 μM | 23726028 | |||
| PC3 | Antiproliferative activity assay | IC50 = 4.13 μM | 25778995 | |||
| HeLa | Cytotoxicity assay | IC50 = 4.163 μM | 23362959 | |||
| MDA-MB-436 | Antiproliferative activity assay | IC50 = 4.2 μM | 29202403 | |||
| MCF7 | Cytotoxicity assay | IC50 = 4.21 μM | 27043268 | |||
| MCF7 | Cytotoxicity assay | IC50 = 4.21 μM | 28340913 | |||
| LOXIMVI | Antiproliferative activity assay | IC50 = 4.25 μM | 25778995 | |||
| OVCAR4 | Cytotoxicity assay | TGI = 4.31 μM | 26590508 | |||
| WM1361 | Antiproliferative activity assay | GI50 = 4.345 μM | 20148563 | |||
| WM266.4 | Growth inhibition assay | GI50 = 4.5 μM | 20199087 | |||
| HepG2 | Cytotoxicity assay | GI50 = 4.5 μM | 23726028 | |||
| A549 | Antiproliferative activity assay | IC50 = 4.5 μM | 23260578 | |||
| LS174T | Function assay | IC50 = 4.52 μM | 29266937 | |||
| 8505C | Antiproliferative activity assay | IC50 = 4.7 μM | 29032031 | |||
| OVCAR8 | Cytotoxicity assay | TGI = 4.73 μM | 26590508 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 4.77 μM | 20435479 | |||
| B16-F1 | Antiproliferative activity assay | IC50 = 4.9 μM | 18477505 | |||
| WM164 | Antiproliferative activity assay | IC50 = 4.9 μM | 17561392 | |||
| B16-F1 | Antiproliferative activity assay | IC50 = 4.9 μM | 20056548 | |||
| MDA-MB-468 | Cytotoxicity assay | IC50 = 4.9 μM | 26159483 | |||
| 786-O | Cytotoxicity assay | IC50 = 4.9 μM | 29517908 | |||
| 786-O | Cytotoxicity assay | IC50 = 4.9 μM | 29297688 | |||
| WM266.4 | Antiproliferative activity assay | GI50 = 4.933 μM | 20148563 | |||
| WM266.4 | Growth inhibition assay | IC50 = 5 μM | 19323560 | |||
| WM164 | Antiproliferative activity assay | IC50 = 5 μM | 20056548 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 5 μM | 23260578 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 5 μM | 23932071 | |||
| NCI-H23 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| NCI-H322M | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| NCI-H460 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| NCI-H522 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| HT-29 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| CCRF-CEM | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| SW620 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| HOP92 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| 786-0 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| MDA-MB-435 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| PC3 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| UACC257 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| SKOV3 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| RXF393 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| T47D | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| MDA-MB-468 | Growth inhibition assay | TGI = 5.01 μM | 28088086 | |||
| UACC62 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| UACC257 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| SK-MEL-5 | Antiproliferative activity assay | LC50 = 5.01 μM | 27017549 | |||
| M14 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| NCI-H23 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| SF295 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| HOP92 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| BT549 | Antiproliferative activity assay | TGI = 5.01 μM | 27017549 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 5.1 μM | 26159483 | |||
| DU145 | Cytotoxicity assay | IC50 = 5.1 μM | 22708987 | |||
| A549 | Cytotoxicity assay | IC50 = 5.21 μM | 20181414 | |||
| HeLa | Cytotoxicity assay | IC50 = 5.23 μM | 26342867 | |||
| 786-O | Cytotoxicity assay | IC50 = 5.3 μM | 29786436 | |||
| HCT116 | Antiproliferative activity assay | GI50 = 5.4 μM | 15225706 | |||
| A375 | Antiproliferative activity assay | IC50 = 5.4 μM | 18477505 | |||
| SK-MEL-188 | Antiproliferative activity assay | IC50 = 5.4 μM | 17561392 | |||
| A375 | Antiproliferative activity assay | IC50 = 5.4 μM | 20056548 | |||
| MCF7 | Cytotoxicity assay | IC50 = 5.5 μM | 26159483 | |||
| A375 | Antiproliferative activity assay | IC50 = 5.58 μM | 19464887 | |||
| A375P | Cytotoxicity assay | IC50 = 5.58 μM | 20797858 | |||
| A375P | Antiproliferative activity assay | GI50 = 5.58 μM | 21353571 | |||
| A375P | Growth inhibition assay | GI50 = 5.58 μM | 22014755 | |||
| A375P | Growth inhibition assay | GI50 = 5.58 μM | 20149658 | |||
| A375P | Antiproliferative activity assay | GI50 = 5.58 μM | 20466542 | |||
| A375P | Antiproliferative activity assay | IC50 = 5.6 μM | 19857963 | |||
| A375P | Antiproliferative activity assay | IC50 = 5.6 μM | 21592628 | |||
| A375P | Antiproliferative activity assay | IC50 = 5.6 μM | 22033063 | |||
| A375P | Cytotoxicity assay | IC50 = 5.6 μM | 19897366 | |||
| A375P | Antiproliferative activity assay | IC50 = 5.6 μM | 22647720 | |||
| HCT116 | Cytotoxicity assay | IC50 = 5.65 μM | 24215818 | |||
| SH-SY5Y | Antiproliferative activity assay | IC50 = 5.73 μM | 24315192 | |||
| HepG2 | Cytotoxicity assay | IC50 = 5.74 μM | 25461318 | |||
| Bel7402 | Cytotoxicity assay | IC50 = 5.8 μM | 23454017 | |||
| HT-29 | Cytotoxicity assay | IC50 = 5.9 μM | 28865276 | |||
| melanoma | Growth inhibition assay | IC50 = 6.1 μM | 16392826 | |||
| K562 | Antiproliferative activity assay | GI50 = 6.2 μM | 21708468 | |||
| HepG2 | Cytotoxicity assay | IC50 = 6.2 μM | 28865276 | |||
| A549 | Anticancer activity assay | IC50 = 6.21 μM | 29549841 | |||
| SMMC7721 | Antiproliferative activity assay | IC50 = 6.23 μM | 22721924 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 6.3 μM | 23932071 | |||
| SF295 | Growth inhibition assay | TGI = 6.31 μM | 28088086 | |||
| SK-MEL-2 | Growth inhibition assay | TGI = 6.31 μM | 28088086 | |||
| OVCAR5 | Growth inhibition assay | TGI = 6.31 μM | 28088086 | |||
| UO31 | Growth inhibition assay | TGI = 6.31 μM | 28088086 | |||
| A498 | Antiproliferative activity assay | TGI = 6.31 μM | 27017549 | |||
| MALME-3M | Antiproliferative activity assay | TGI = 6.31 μM | 27017549 | |||
| U251 | Antiproliferative activity assay | TGI = 6.31 μM | 27017549 | |||
| SK-MEL-2 | Antiproliferative activity assay | LC50 = 6.31 μM | 27017549 | |||
| RPMI8226 | Antiproliferative activity assay | TGI = 6.31 μM | 27017549 | |||
| HCC2998 | Antiproliferative activity assay | LC50 = 6.31 μM | 27017549 | |||
| NCI-H522 | Antiproliferative activity assay | TGI = 6.31 μM | 27017549 | |||
| HUVEC | Antiproliferative activity assay | IC50 = 6.42 μM | 23644219 | |||
| A549 | Cytotoxicity assay | IC50 = 6.53 μM | 27043268 | |||
| A549 | Cytotoxicity assay | IC50 = 6.53 μM | 28340913 | |||
| LoVo | Antiproliferative activity assay | IC50 = 6.56 μM | 24315192 | |||
| A375 | Function assay | IC50 = 6.599 μM | 21807507 | |||
| HCT116 | Function assay | GI50 = 6.6 μM | 15225706 | |||
| HepG2 | Cytotoxicity assay | IC50 = 6.7 μM | 26159483 | |||
| BCPAP | Antiproliferative activity assay | IC50 = 6.7 μM | 29032031 | |||
| A549 | Cytotoxicity assay | IC50 = 6.7 μM | 28865276 | |||
| PC3 | Cytotoxicity assay | IC50 = 6.8 μM | 23021967 | |||
| SGC7901 | Cytotoxicity assay | IC50 = 6.9 μM | 23454017 | |||
| OS-RC2 | Cytotoxicity assay | IC50 = 7 μM | 29517908 | |||
| OS-RC2 | Cytotoxicity assay | IC50 = 7 μM | 29297688 | |||
| COLO205 | Antiproliferative activity human | IC50 = 7.04 μM | 28242553 | |||
| rhabdomyosarcoma | Cytotoxicity activity against human | CC50 = 7.05 μM | 27288186 | |||
| HuH7 | Cytotoxicity assay | IC50 = 7.1 μM | 26159483 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 7.18 μM | 22721924 | |||
| FLT3-negative K562 | Cytotoxicity assay | GI50 = 7.3 μM | 23618709 | |||
| K562 | Function assay | GI50 = 7.3 μM | 26081023 | |||
| K562 | Function assay | GI50 = 7.3 μM | 26081023 | |||
| MCF7 | Antiproliferative activity assay | IC50 = 7.33 μM | 23644219 | |||
| A549 | Cytotoxicity assay | TGI = 7.36 μM | 26590508 | |||
| WI38 | Cytotoxicity assay | IC50 = 7.54 μM | 24826815 | |||
| A375 | Cytotoxicity assay | IC50 = 7.56 μM | 24215818 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 7.62 μM | 20181414 | |||
| 786-0 | Cytotoxicity assay | TGI = 7.62 μM | 26590508 | |||
| HS27 | Antiproliferative activity assay | IC50 = 7.8 μM | 19857963 | |||
| HS27 | Cytotoxicity assay | IC50 = 7.8 μM | 19897366 | |||
| HCT116 | Cytotoxicity assay | IC50 = 7.8 μM | 22483592 | |||
| HS27 | Antiproliferative activity assay | IC50 = 7.85 μM | 19464887 | |||
| HS27 | Antiproliferative activity assay | GI50 = 7.85 μM | 21353571 | |||
| A549/ATCC | Growth inhibition assay | TGI = 7.94 μM | 28088086 | |||
| DU145 | Growth inhibition assay | TGI = 7.94 μM | 28088086 | |||
| NCI/ADR-RES | Growth inhibition assay | TGI = 7.94 μM | 28088086 | |||
| MCF7 | Growth inhibition assay | TGI = 7.94 μM | 28088086 | |||
| PC3 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| UO31 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| NCI-ADR-RES | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| LOXIMVI | Antiproliferative activity assay | LC50 = 7.94 μM | 27017549 | |||
| SK-MEL-28 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| KM12 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| COLO205 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| HT-29 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| NCI-H226 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| HOP62 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| T47D | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| MDA-MB-468 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| EKVX | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| MCF7 | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| Hs 578T | Antiproliferative activity assay | TGI = 7.94 μM | 27017549 | |||
| MGC803 | Antiproliferative activity assay | IC50 = 7.99 μM | 26560049 | |||
| HCT116 | Antiproliferative activity assay | IC50 = 8.08 μM | 24440479 | |||
| PC3 | Cytotoxicity assay | IC50 = 8.08 μM | 27043268 | |||
| HCT116 | Antiproliferative activity assay | IC50 = 8.08 μM | 23644219 | |||
| WM266.4 | Antiproliferative activity assay | GI50 = 8.1 μM | 23025996 | |||
| WM266.4 | Antiproliferative activity assay | GI50 = 8.12 μM | 22583669 | |||
| Ketr3 | Antiproliferative activity assay | IC50 = 8.27 μM | 30216849 | |||
| A375 | Antiproliferative activity assay | IC50 = 8.27 μM | 30216849 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 8.27 μM | 30216849 | |||
| MX1 | Antiproliferative activity assay | IC50 = 8.27 μM | 30216849 | |||
| MX1 | Cytotoxicity assay | IC50 = 8.27 μM | 24675135 | |||
| WM266.4 | Growth inhibition assay | GI50 = 8.3 μM | 22361686 | |||
| PLC/PRF/5 | Cytotoxicity assay | IC50 = 8.3 μM | 21531053 | |||
| A375 | Antiproliferative activity assay | IC50 = 8.33 μM | 29886324 | |||
| HCT116 | Cytotoxicity assay | IC50 = 8.41 μM | 29631788 | |||
| WI38 | Cytotoxicity assay | IC50 = 8.42 μM | 26991938 | |||
| HepG2 | Cytotoxicity assay | IC50 = 8.42 μM | 27162123 | |||
| HepG2 | Cytotoxicity assay | IC50 = 8.42 μM | 23871909 | |||
| HT-29 | Antiproliferative activity assay | IC50 = 8.44 μM | 29602674 | |||
| HepG2 | Cytotoxicity assay | IC50 = 8.67 μM | 24675135 | |||
| MCF7 | Cytotoxicity assay | IC50 = 8.83 μM | 20435479 | |||
| MCF7 | Cytotoxicity assay | IC50 = 9.12 μM | 24300920 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 9.14 μM | 26560049 | |||
| A375 | Cytotoxicity assay | IC50 = 9.17 μM | 24675135 | |||
| MGC803 | Antiproliferative activity assay | IC50 = 9.2 μM | 29032031 | |||
| SKOV3 | Cytotoxicity assay | IC50 = 9.25 μM | 24300920 | |||
| RS4:11 | Function assay | GI50 = 9.3 μM | 26081023 | |||
| RS4:11 | Cytotoxicity assay | GI50 = 9.3 μM | 23618709 | |||
| MIAPaCa2 | Antiproliferative activity assay | IC50 = 9.32 μM | 29032031 | |||
| PANC1 | Growth inhibition assay | IC50 = 9.32 μM | 29102175 | |||
| MHCC97H | Growth inhibition assay | IC50 = 9.32 μM | 29102175 | |||
| RS4:11 | Antiproliferative activity assay | GI50 = 9.4 μM | 21708468 | |||
| Hep3B | Cytotoxicity assay | IC50 = 9.4 μM | 28865276 | |||
| LoVo | Growth inhibition assay | IC50 = 9.47 μM | 29102175 | |||
| SMMC7721 | Antiproliferative activity assay | IC50 = 9.96 μM | 25637123 | |||
| MCF7 | Cytotoxicity assay | TGI = 9.97 μM | 26590508 | |||
| IGROV1 | Growth inhibition assay | TGI = 10 μM | 28088086 | |||
| ACHN | Growth inhibition assay | TGI = 10 μM | 28088086 | |||
| TK10 | Growth inhibition assay | TGI = 10 μM | 28088086 | |||
| 786-0 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| SN12C | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| Caki1 | Antiproliferative activity assay | LC50 = 10 μM | 27017549 | |||
| TK10 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| RXF393 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| OVCAR8 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| SNB75 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| NCI-H322M | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| MDA-MB-435 | Antiproliferative activity assay | LC50 = 10 μM | 27017549 | |||
| SF539 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| SF268 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| NCI-H460 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| DU145 | Antiproliferative activity assay | TGI = 10 μM | 27017549 | |||
| K1 | Antiproliferative activity assay | IC50 = 10.2 μM | 29032031 | |||
| Bel7402 | Cytotoxicity assay | IC50 = 10.26 μM | 21504204 | |||
| Bel7402 | Growth inhibition assay | IC50 = 10.31 μM | 29102175 | |||
| OVCAR3 | Cytotoxicity assay | TGI = 10.32 μM | 26590508 | |||
| SGC7901 | Cytotoxicity assay | IC50 = 10.37 μM | 20435479 | |||
| SNB75 | Cytotoxicity assay | TGI = 10.42 μM | 26590508 | |||
| A431 | Antiproliferative activity assay | IC50 = 10.46 μM | 29032031 | |||
| SMMC7721 | Antiproliferative activity assay | IC50 = 10.61 μM | 29032031 | |||
| GES-1 | Cytotoxicity assay | IC50 = 10.68 μM | 27162123 | |||
| H460 | Cytotoxicity assay | IC50 = 10.8 μM | 24300920 | |||
| BGC823 | Antiproliferative activity assay | IC50 = 10.91 μM | 22721924 | |||
| Hep3B | Cytotoxicity assay | IC50 = 11.2 μM | 28342400 | |||
| MCF7 | Cytotoxicity assay | IC50 = 11.34 μM | 27162123 | |||
| MCF7 | Cytotoxicity assay | IC50 = 11.34 μM | 23871909 | |||
| SK-MEL-2 | Antiproliferative activity human | IC50 = 11.35 μM | 28242553 | |||
| HepG2 | Cytotoxicity assay | IC50 = 11.49 μM | 21504204 | |||
| A375P | Antiproliferative activity assay | IC50 = 11.5 μM | 22014559 | |||
| SGC7901 | Cytotoxicity assay | IC50 = 11.5 μM | 24300920 | |||
| HeLa | Antiproliferative activity assay | IC50 = 12.01 μM | 29103873 | |||
| PANC1 | Cytotoxicity assay | IC50 = 12.3 μM | 24300920 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 12.54 μM | 29032031 | |||
| A549 | Antiproliferative activity assay | IC50 = 12.54 μM | 23644219 | |||
| Caki1 | Growth inhibition assay | TGI = 12.59 μM | 28088086 | |||
| OVCAR8 | Growth inhibition assay | TGI = 12.59 μM | 28088086 | |||
| SKOV3 | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| ACHN | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| OVCAR3 | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| OVCAR4 | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| HCT15 | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| SW620 | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| A549/ATCC | Antiproliferative activity assay | TGI = 12.6 μM | 27017549 | |||
| MDA-MB-468 | Cytotoxicity assay | TGI = 13.09 μM | 26590508 | |||
| SK-MEL-28 | Cytotoxicity assay | TGI = 13.39 μM | 26590508 | |||
| A549 | Antiproliferative activity assay | IC50 = 13.64 μM | 26560049 | |||
| A375 | Antiproliferative activity human | IC50 = 13.64 μM | 28242553 | |||
| A375 | Antiproliferative activity assay | IC50 = 13.64 μM | 25462267 | |||
| K562 | Antiproliferative activity assay | IC50 = 13.85 μM | 23644219 | |||
| Hep3B | Cytotoxicity assay | IC50 = 14.08 μM | 21504204 | |||
| T47D | Cytotoxicity assay | TGI = 14.38 μM | 26590508 | |||
| NCI-H226 | Cytotoxicity assay | TGI = 14.42 μM | 26590508 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 14.62 μM | 24440479 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 14.62 μM | 23644219 | |||
| NCI-H522 | Cytotoxicity assay | TGI = 14.82 μM | 26590508 | |||
| SK-MEL-2 | Cytotoxicity assay | TGI = 14.85 μM | 26590508 | |||
| OS-RC2 | Cytotoxicity assay | IC50 = 15 μM | 29786436 | |||
| HepG2 | Antiproliferative activity assay | IC50 = 15 μM | 29103873 | |||
| fibroblast | Antiproliferative activity assay | IC50 = 15.1 μM | 18477505 | |||
| HT-29 | Cytotoxicity assay | IC50 = 15.2 μM | 24675135 | |||
| U87MG | Cytotoxicity assay | IC50 = 15.57 μM | 24826815 | |||
| BT549 | Antiproliferative activity assay | LC50 = 15.8 μM | 27017549 | |||
| GES-1 | Cytotoxicity assay | IC50 = 15.8 μM | 23871909 | |||
| SNB19 | Antiproliferative activity assay | TGI = 15.85 μM | 27017549 | |||
| IGROV1 | Antiproliferative activity assay | TGI = 15.9 μM | 27017549 | |||
| HCT116 | Antiproliferative activity assay | LC50 = 15.9 μM | 27017549 | |||
| OS-RC2 | Cytotoxicity assay | IC50 = 16 μM | 23454017 | |||
| A375 | Cytotoxicity assay | IC50 = 16.24 μM | 25461318 | |||
| SKOV3 | Cytotoxicity assay | TGI = 16.36 μM | 26590508 | |||
| WM1361 | Antiproliferative activity assay | IC50 = 16.44 μM | 29602674 | |||
| MKN28 | Antiproliferative activity assay | IC50 = 17 μM | 29032031 | |||
| SMMC7721 | Cytotoxicity assay | IC50 = 17.3 μM | 21504204 | |||
| UACC257 | Cytotoxicity assay | TGI = 17.33 μM | 26590508 | |||
| EAhy926 | Antiangiogenic activity assay | IC50 = 18.52 μM | 29032031 | |||
| SMMC7721 | Cytotoxicity assay | IC50 = 18.7 μM | 24300920 | |||
| Ketr3 | Cytotoxicity assay | IC50 = 18.8 μM | 24675135 | |||
| NCI-H522 | Cytotoxicity assay | IC50 = 19.26 μM | 25461318 | |||
| A549 | Cytotoxicity assay | IC50 = 19.54 μM | 27162123 | |||
| SH-SY5Y | Cytotoxicity assay | IC50 = 19.54 μM | 23871909 | |||
| MGC803 | Antiproliferative activity assay | IC50 = 19.92 μM | 29103873 | |||
| EKVX | Growth inhibition assay | TGI = 19.95 μM | 28088086 | |||
| OVCAR5 | Antiproliferative activity assay | TGI = 20 μM | 27017549 | |||
| UACC62 | Antiproliferative activity assay | LC50 = 20 μM | 27017549 | |||
| UACC257 | Antiproliferative activity assay | LC50 = 20 μM | 27017549 | |||
| MDA-MB-231 | Antiproliferative activity assay | LC50 = 20 μM | 27017549 | |||
| U87 | Cytotoxicity assay | IC50 = 21.07 μM | 20435479 | |||
| EJ | Cytotoxicity assay | IC50 = 22.9 μM | 24300920 | |||
| HCT116 | Antiproliferative activity assay | IC50 = 23.31 μM | 29602674 | |||
| HOP62 | Cytotoxicity assay | TGI = 23.55 μM | 26590508 | |||
| MDA-MB-231 | Cytotoxicity assay | TGI = 23.93 μM | 26590508 | |||
| PC3 | Antiproliferative activity assay | IC50 = 24.2 μM | 24440479 | |||
| PC3 | Antiproliferative activity assay | IC50 = 24.2 μM | 23644219 | |||
| EAhy926 | Antiproliferative activity assay | IC50 = 24.36 μM | 28068599 | |||
| EAhy926 | Anti-angiogenic activity in human | IC50 = 24.36 μM | 29102175 | |||
| U251 | Cytotoxicity assay | IC50 = 24.71 μM | 20435479 | |||
| NIH/3T3 | Antiproliferative activity assay | IC50 = 24.75 μM | 21592628 | |||
| NIH/3T3 | Antiproliferative activity assay | IC50 = 24.75 μM | 22647720 | |||
| DU145 | Cytotoxicity assay | IC50 = 24.91 μM | 27162123 | |||
| DU145 | Cytotoxicity assay | IC50 = 24.91 μM | 23871909 | |||
| U251 | Antiproliferative activity assay | LC50 = 25.1 μM | 27017549 | |||
| M14 | Antiproliferative activity assay | LC50 = 25.1 μM | 27017549 | |||
| NCI-H522 | Antiproliferative activity assay | LC50 = 25.1 μM | 27017549 | |||
| OVCAR4 | Growth inhibition assay | TGI = 25.12 μM | 28088086 | |||
| SH-SY5Y | Cytotoxicity assay | IC50 = 27.71 μM | 27162123 | |||
| A549 | Cytotoxicity assay | IC50 = 27.71 μM | 23871909 | |||
| PLC/PRF/5 | Cytotoxicity assay | IC50 = 29.9 μM | 28109948 | |||
| SK-MEL-5 | Cytotoxicity assay | TGI = 31.26 μM | 26590508 | |||
| A498 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| SK-MEL-28 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| KM12 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| COLO205 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| NCI-H226 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| SF295 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| HOP62 | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| T47D | Antiproliferative activity assay | LC50 = 31.6 μM | 27017549 | |||
| MHCC97L | Cytotoxicity assay | IC50 = 34.41 μM | 28109948 | |||
| MDA-MB-231 | Antiproliferative activity assay | IC50 = 35 μM | 24315192 | |||
| MDA-MB-231 | Cytotoxicity assay | IC50 = 36 μM | 22483592 | |||
| B16-BL6 | Cytotoxicity assay | IC50 = 36.48 μM | 29631788 | |||
| UO31 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| SN12C | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| RXF393 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| SNB75 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| SF268 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| NCI-H23 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| HT-29 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| HOP92 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| NCI-H460 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| SW620 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| MDA-MB-468 | Antiproliferative activity assay | LC50 = 39.8 μM | 27017549 | |||
| MALME-3M | Growth inhibition assay | TGI = 39.81 μM | 28088086 | |||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Sorafenib is a multikinase inhibitor of Raf-1 and B-Raf with IC50 of 6 nM and 22 nM in cell-free assays, respectively. Sorafenib inhibits VEGFR-2, VEGFR-3, PDGFR-β, Flt-3 and c-KIT with IC50 of 90 nM, 20 nM, 57 nM, 59 nM and 68 nM, respectively. Sorafenib induces autophagy and apoptosis and activates ferroptosis with anti-tumor activity. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | Sorafenib inhibits both wild-type and V599E mutant B-Raf activity with IC50 of 22 nM and 38 nM, respectively. Sorafenib also potently inhibits mVEGFR2 (Flk-1), mVEGFR3, mPDGFRβ, Flt3, and c-Kit with IC50 of 15 nM, 20 nM, 57 nM, 58 nM, and 68 nM, respectively. Sorafenib weakly inhibits FGFR-1 with IC50 of 580 nM. Sorafenib tosylate is not active against ERK-1, MEK-1, EGFR, HER-2, IGFR-1, c-Met, PKB, PKA, cdk1/cyclinB, PKCα, PKCγ, and pim-1. Sorafenib markedly inhibits VEGFR2 phosphorylation in NIH 3T3 cells with IC50 of 30 nM, and Flt-3 phosphorylation in HEK-293 cells with IC50 of 20 nM. Sorafenib potently blocks MEK 1/2 and ERK 1/2 phosphorylation in most cell lines but not in A549 or H460 cells, while having no effect on inhibition of the PKB pathway. Sorafenib inhibits the proliferation of HAoSMC and MDA-MB-231 cells with IC50 of 0.28 μM and 2.6 μM, respectively. [1] In addition to inhibition of the RAF/MEK/ERK signaling pathway, Sorafenib significantly inhibits the phosphorylation of eIF4E and down-regulates Mcl-1 levels in hepatocellular carcinoma (HCC) cells in a MEK/ERK-independent manner. Sorafenib inhibits the proliferation of PLC/PRF/5 and HepG2 cells with IC50 of 6.3 μM and 4.5 μM, respectively, and leads to the significant induction of apoptosis. [2] | |||
|---|---|---|---|---|
| Kinase Assay | Biochemical assays | |||
| Recombinant baculoviruses expressing Raf-1 (residues 305–648) and B-Raf (residues 409–765) are purified as fusion proteins. Full-length human MEK-1 is generated by PCR and purified as a fusion protein from Escherichia coli lysates. Sorafenib tosylate is added to a mixture of Raf-1 (80 ng), or B-Raf (80 ng) with MEK-1 (1 μg) in assay buffer [20 mM Tris (pH 8.2), 100 mM NaCl, 5 mM MgCl2, and 0.15% β-mercaptoethanol] at a final concentration of 1% DMSO. The Raf kinase assay (final volume of 50 μL) is initiated by adding 25 μL of 10 μM γ[33P]ATP (400 Ci/mol) and incubated at 32 °C for 25 minutes. Phosphorylated MEK-1 is harvested by filtration onto a phosphocellulose mat, and 1% phosphoric acid is used to wash away unbound radioactivity. After drying by microwave heating, a β-plate counter is used to quantify filter-bound radioactivity. Human VEGFR2 (KDR) kinase domain is expressed and purified from Sf9 lysates. Time-resolved fluorescence energy transfer assays for VEGFR2 are performed in 96-well opaque plates in the time-resolved fluorescence energy transfer format. Final reaction conditions are as follows: 1 to 10 μM ATP, 25 nM poly GT-biotin, 2 nM Europium-labeled phospho (p)-Tyr antibody (PY20), 10 nM APC, 1 to 7 nM cytoplasmic kinase domain in final concentrations of 1% DMSO, 50 mM HEPES (pH 7.5), 10 mM MgCl2, 0.1 mM EDTA, 0.015% Brij-35, 0.1 mg/mL BSA, and 0.1% β-mercaptoethanol. Reaction volumes are 100 μL and are initiated by addition of enzyme. Plates are read at both 615 and 665 nM on a Perkin-Elmer VictorV Multilabel counter at ~1.5 to 2.0 hours after reaction initiation. Signal is calculated as a ratio: (665 nm/615 nM) × 10,000 for each well. For IC50 generation, Sorafenib tosylate is added before the enzyme initiation. A 50-fold stock plate is made with Sorafenib tosylate serially diluted 1:3 in a 50% DMSO/50% distilled water solution. Final Sorafenib tosylate concentrations range from 10 μM to 4.56 nM in 1% DMSO. | ||||
| Cell Research | Cell lines | MDA-MB-231, and HAoSMC | ||
| Concentrations | Dissolved in DMSO, final concentrations ~10 μM | |||
| Incubation Time | 72 hours | |||
| Method | Cells are exposed to increasing concentrations of Sorafenib tosylate for 72 hours. Cell number is quantitated using the Cell TiterGlo ATP Luminescent assay kit. This assay measures the number of viable cells per well by measurement of luminescent signal based on amount of cellular ATP. |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | LC3-I / LC-3II / ATG5 p-STAT3 / STAT3 / Mcl-1 β-catenin / Survivin / Mcl-1 / PTMA pERK / ERK p-PKM2(y105) / PMK2 / Caspase-9 RET(pY1016) / VEGFR2(pY1214) / MEK1(pT292) / ERK(pY204) Cyclin D1 |
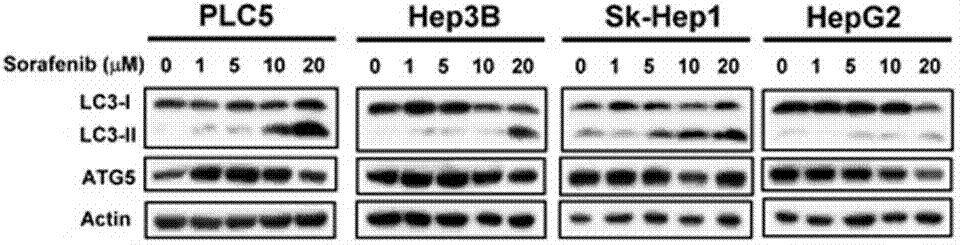
|
23392173 | |
| Growth inhibition assay | Cell viability |
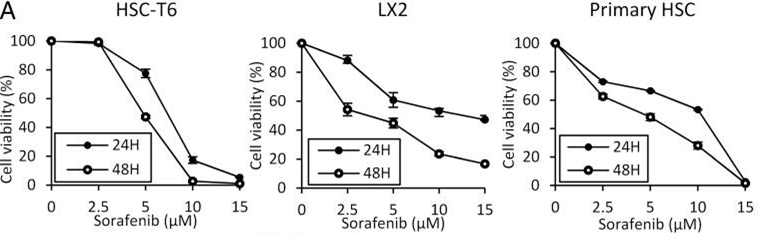
|
26039995 | |
| Immunofluorescence | p65 cytochrome c |
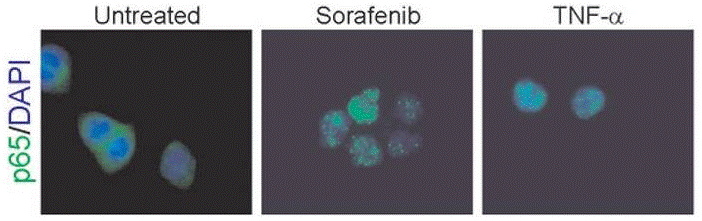
|
22286758 | |
| ELISA | TGF-beta / CD206 Caspase-9 / Caspase-3 |
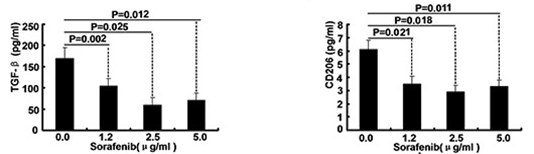
|
26158762 | |
| In Vivo | ||
| In vivo | Oral administration of Sorafenib (~60 mg/kg) demonstrates broad spectrum, dose-dependent anti-tumor activity against a variety of human tumor xenograft models including MDA-MB-231, Colo-205, HT-29, DLD-1, NCI-H460, and A549, with no evidence of toxicity. In association with the anti-tumor efficacy, Sorafenib treatment potently inhibits MEK 1/2 phosphorylation and pERK 1/2 levels in HT-29 and MDA-MB-231 xenografts but not in Colo-205 xenografts, and significantly suppresses tumor microvessel area (MVA) and microvessel density (MVD) in MDA MB-231, HT-29 and Colo-205 tumor xenografts. [1] Sorafenib treatment produces dose-dependent growth inhibition of PLC/PRF/5 tumor xenografts in SCID mice with TGIs of 49% and 78% at 10 mg/kg and 30 mg/kg, respectively, consistent with the inhibition of ERK and eIF4E phosphorylation, reduction of the microvessel area, and induction of tumor cell apoptosis. [2] Sorafenib sensitizes bax-/- cells to TRAIL in a dose-dependent manner, through a mechanism involving down-regulating NF-κB mediated Mcl-1 and cIAP2 expression. Combining Sorafenib (30-60 mg/kg) with TRAIL (5 mg/kg) show dramatic efficacy in TRAIL-resistant HCT116 bax-/- and HT29 tumor xenografts. [3] | |
|---|---|---|
| Animal Research | Animal Models | Female NCr-nu/nu mice implanted s.c. with MDA-MB-231, Colo-205, HT-29, H460, or A549 cells |
| Dosages | ~60 mg/kg | |
| Administration | Orally once daily | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05068752 | Recruiting | Pancreas Cancer |
HonorHealth Research Institute|Bayer|Genentech Inc. |
October 28 2021 | Phase 2 |
| NCT04763408 | Active not recruiting | Carcinoma Hepatocellular |
Eisai Inc. |
April 9 2021 | -- |
Chemical Information & Solubility
| Molecular Weight | 464.82 | Formula | C21H16ClF3N4O3 |
| CAS No. | 284461-73-0 | SDF | Download Sorafenib SDF |
| Smiles | CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 92 mg/mL ( (197.92 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy Sorafenib | Sorafenib supplier | purchase Sorafenib | Sorafenib cost | Sorafenib manufacturer | order Sorafenib | Sorafenib distributor