research use only
Rimonabant (SR141716) Cannabinoid Receptor antagonist
Cat.No.S3021
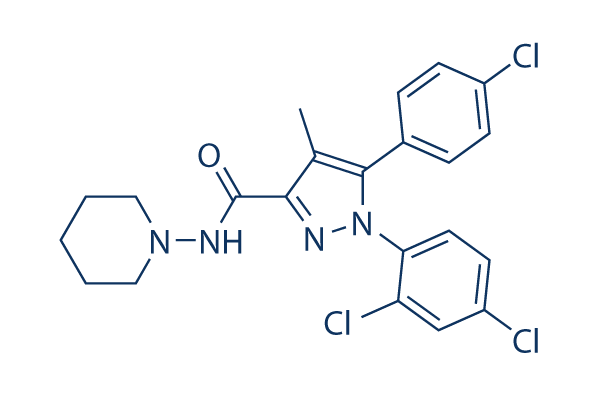
Chemical Structure
Molecular Weight: 463.79
Quality Control
| Related Targets | CXCR Hedgehog/Smoothened PKA Adrenergic Receptor AChR 5-HT Receptor Histamine Receptor Dopamine Receptor Ras KRas |
|---|---|
| Other Cannabinoid Receptor Inhibitors | AM1241 AM251 6-Iodopravadoline (AM630) CID16020046 BML-190 Org 27569 Otenabant (CP-945598) HCl WIN 55, 212-2 mesylate GW842166X Pregnenolone monosulfate sodium |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| CHO cells | Function assay | Ability to displace [3H]-SR- 141716A binding to human CB1 receptor expressed in CHO cell membranes, Ki=8.9 nM | 10465552 | |||
| CHO | Function assay | Antagonistic activity towards cannabinoid receptor 1 expressed as [3H]Arachidonic acid release in CHO cells, Kd=0.002512μM | 14736243 | |||
| CHO | Function assay | Displacement of CP-55940 binding from recombinant human cannabinoid receptor 1 expressed in CHO cells, Ki=0.025μM | 14736243 | |||
| CHO | Function assay | Affinity to displace CP-55940 binding from Cannabinoid receptor 2 of human expressed in CHO cells, Ki=1.58μM | 14736243 | |||
| CHO | Function assay | Inhibitory concentration against human recombinant cannabinoid receptor type 1 expressed in Chinese Hamster Ovary (CHO) cells, IC50=0.006μM | 15713403 | |||
| CHO | Function assay | In vitro cannabinoid receptor 1 antagonism of [3H]arachidonic acid release by CHO cells, Kd=0.002512μM | 15771428 | |||
| CHO | Function assay | In vitro displacement of CP-55940 binding to human CB1 receptor expressed in CHO cells, Ki=0.025μM | 15771428 | |||
| CHO | Function assay | In vitro displacement of CP-55940 binding to human CB2 receptor expressed in CHO cells, Ki=1.58μM | 15771428 | |||
| CHO | Function assay | Inhibition of [3H]SR-141,716A binding to human CB1 receptor expressed in CHO cells, Ki=0.0054μM | 15801840 | |||
| CHO | Function assay | Antagonistic activity against cannabinoid receptor 1 measured by CP-55940 induced arachnoid acid release in CHO cells, Kd=0.002512μM | 16140010 | |||
| CHO | Function assay | Displacement of specific CP-55940 binding in CHO cells stably transfected with human cannabinoid receptor 1, Ki=0.025μM | 16140010 | |||
| CHO | Function assay | Displacement of specific CP-55940 binding in CHO cells stably transfected with human cannabinoid receptor 2, Ki=1.58μM | 16140010 | |||
| CHOK1 | Function assay | Functional activity at human CB1 receptor transfected in CHOK1 cells by [35SGTP]gammaS assay, Ki=0.001μM | 16263283 | |||
| HEK293 | Function assay | Displacement of [3H]SR-141716 from human CB1 receptor transfected in HEK293 cells, Ki=0.0021μM | 16263283 | |||
| CHO | Function assay | Displacement of [3H]SR141716A from human CB1 receptor expressed in CHO cells, Ki=0.0054μM | 16279809 | |||
| HEK293 | Function assay | Displacement of [3H]SR-141716 from human CB1 receptor expressed in HEK293 cells, Kd=0.0018μM | 17004712 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, Ki=0.0071μM | 17004712 | |||
| HEK293 | Function assay | Displacement of [3H]WIN-55212-2 from human CB1 receptor expressed in HEK293 cells, Ki=0.018μM | 17004712 | |||
| CHO | Function assay | Inhibition of [3H]CP-55940 binding to human recombinant CB1 receptor in CHO cells, IC50=0.0061μM | 17181138 | |||
| CHO | Function assay | Inhibition of [3H]CP-55940 binding to human recombinant CB2 receptor in CHO cells, IC50=0.6μM | 17181138 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in CHO cells, IC50=0.0061μM | 17293109 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in CHO cells, IC50=0.6033μM | 17293109 | |||
| HEK293 | Function assay | Displacement of [3H]CP-559440 from human CB1 receptor expressed in HEK293 cells, Ki=0.0011μM | 17383180 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells, Ki=0.016μM | 17942307 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK293 cells, Ki=1.64μM | 17942307 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells, Ki=0.047μM | 17979261 | |||
| CHOK1 | Function assay | Displacement of [3H]WIN-552122 from human recombinant CB2 receptor expressed in CHOK1 cells, Ki=1.99μM | 17979261 | |||
| HEK293 | Function assay | Displacement of radioligand from human CB1 receptor expressed in HEK293 cells, Ki=0.0018μM | 18083560 | |||
| HEK293 EBNA | Function assay | Inverse agonist activity at human CB1 receptor expressed in HEK293 EBNA cells by [35S]GTPgammaS incorporation assay, EC50=0.004μM | 18083560 | |||
| HEK293 | Function assay | Displacement of radioligand from human CB2 receptor expressed in HEK293 cells, Ki=0.554μM | 18083560 | |||
| CHOK1 | Function assay | Antagonist activity at human CB1 receptor expressed in CHOK1 cells by luciferase assay, IC50=0.12μM | 18243711 | |||
| CHOK1 | Function assay | Displacement of [3H]WIN-55212-2 from human CB2 receptor expressed in CHOK1 cells, IC50=1.76μM | 18243711 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK cells, Ki=0.012μM | 18293908 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK cells, Ki=0.79μM | 18293908 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1R expressed in HEK293 cells, Ki=0.006μM | 18335976 | |||
| CHO | Function assay | Displacement of [3H]WIN-55212-2 from human cannabinoid CB2 receptor expressed in CHO cells, IC50=1.76μM | 18337096 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in CHO cells, Ki=0.025μM | 18342403 | |||
| CHO | Function assay | Displacement of [3H]WIN-55212-2 from human CB2 receptor expressed in CHO cells, Ki=1.58μM | 18342403 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, Ki=0.0024μM | 18363352 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in CHO cells, Ki=0.56μM | 18363352 | |||
| SF9 | Function assay | Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding, Ki=0.00031μM | 18448340 | |||
| SF9 | Function assay | Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding, Ki=0.00043μM | 18448340 | |||
| SF9 | Function assay | Inverse agonist at human CB1 receptor expressed in SF9 cells assessed as decrease in GTPgammaS level, IC50=0.00135μM | 18448340 | |||
| SF9 | Function assay | Antagonist activity at human CB2 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding, Ki=0.815μM | 18448340 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 form human recombinant CB1 receptor expressed in HEK293 cells by liquid scintillation counting, Ki=0.04μM | 18511157 | |||
| CHO-K1 | Function assay | Displacement of [3H]SR141716 from human CB1 receptor expressed in CHO-K1 cells, Ki=0.00118μM | 18512901 | |||
| HEK293 | Function assay | Antagonist activity at human CB1 receptor expressed in HEK293 cells assessed as inhibition of CP-55940-stimulated [35S]GTPgammaS binding, Kd=0.00257μM | 18512901 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, Ki=0.00618μM | 18512901 | |||
| CHO-K1 | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in CHO-K1 cells, Ki=0.313μM | 18512901 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK cells, Ki=0.008μM | 18579386 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK cells, Ki=0.79μM | 18579386 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, Ki=0.012μM | 18680276 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in HEK293 cells, Ki=0.79μM | 18680276 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, IC50=0.015μM | 18712856 | |||
| HEK293 | Function assay | Antagonist activity at human CB1 receptor expressed in HEK293 cells assessed as inhibition of Eu-GTP binding, EC50=0.0182μM | 18712856 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in HEK293 cells, IC50=1.9398μM | 18712856 | |||
| HEK293 | Function assay | Inverse agonist activity at human CB1 receptor expressed in HEK293 cells assessed as decrease in [35S]gammaGTP binding relative to control | 18712856 | |||
| CHO | Function assay | Antagonist activity at human CB1 receptor in CHO cells assessed as GTPgammaS binding, Kb=0.000698μM | 18800770 | |||
| CHOK1 | Function assay | Displacement of [3H]WIN-55212-2 from human CB2 receptor expressed in CHOK1 cells by liquid scintillation spectrometry, IC50=1.76μM | 18954042 | |||
| CHO | Function assay | Displacement of [3H]WIN-55212-2 from human CB2 receptor expressed in CHO cells, IC50=1.76μM | 19022666 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in HEK293 cells, IC50=0.0019μM | 19095444 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK293 cells, IC50=0.015μM | 19095444 | |||
| HEK293 | Function assay | Antagonist activity at human CB1 receptor expressed in HEK293 cells assessed as inhibition of Eu-GTP binding, EC50=0.0182μM | 19095444 | |||
| CHO-K1 | Function assay | Antagonist activity against human CB1 receptor expressed in CHO-K1 cells by [35S]GTPgamma binding assay, Ki=0.00019μM | 19102698 | |||
| HEK293 | Function assay | Displacement of [3H]SR141716A from human CB1 receptor expressed in HEK293 cells, Ki=0.0009μM | 19102698 | |||
| CHOK1 | Function assay | Displacement of [3H]WIN-55212-2 from human recombinant cannabinoid CB2 receptor expressed in CHOK1 cells by liquid scintillation spectrometry, IC50=1.76μM | 19269817 | |||
| CHOK1 | Function assay | Antagonist activity at human cannabinoid CB1 receptor expressed in CHOK1 cells assessed as cAMP activity, EC50=0.24μM | 19328683 | |||
| CHOK1 | Function assay | Antagonist activity at human cannabinoid CB2 receptor expressed in CHOK1 cells assessed as cAMP activity, EC50=31.21μM | 19328683 | |||
| HEK293 | Function assay | Displacement of [3H]SR141716 from human recombinant CB1 receptor expressed in HEK293 cells, Ki=0.001995μM | 19338356 | |||
| HEK293 | Function assay | Displacement of [3H]SR141716 from human recombinant CB2 receptor expressed in HEK293 cells, Ki=0.39811μM | 19338356 | |||
| CHOK1 | Function assay | 10 mins | Antagonist activity at human CB1 receptor expressed in CHOK1 cells assessed as inhibition of CP-55940-induced response after 10 mins by GTPgamma[35S] binding assay, Ki=0.0016μM | 19351113 | ||
| HEK293 | Function assay | Displacement of [3H]rimonabant from human CB1 receptor expressed in HEK293 cells by liquid scintillation counting, Ki=0.0019μM | 19520572 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1R expressed in HEK293 cells, IC50=0.0132μM | 19530697 | |||
| HEK293 | Function assay | Inverse agonist activity at human recombinant CB1R expressed in HEK293 cells assessed as inhibition of CP-55940-stimulated Eu-GTP binding, EC50=0.0157μM | 19530697 | |||
| HEK293 | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2R expressed in HEK293 cells, IC50=1.6311μM | 19530697 | |||
| HEK293 | Function assay | 10 uM | Intrinsic activity at human recombinant CB1R expressed in HEK293 cells assessed as Eu-GTP binding at 10 uM relative to basal level | 19530697 | ||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK cells, Ki=0.012μM | 19595596 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK cells, Ki=0.79μM | 19595596 | |||
| CHO-K1 | Function assay | Antagonist activity at human CB1 receptor transfected in CHO-K1cells by GTPgamma[35S] binding assay, Ki=0.0016μM | 19683918 | |||
| CHO | Function assay | Binding affinity to human CB2 receptor expressed in CHO cells by luciferase reporter gene assay, IC50=0.0925μM | 19850473 | |||
| CHOK1 | Function assay | Displacement of [3H]WIN-552122 from human CB2 receptor expressed in CHOK1 cells, IC50=1.76μM | 19850473 | |||
| CHO-K1 | Function assay | Antagonist activity at human recombinant CB1 receptor expressed in CHO-K1 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding, IC50=0.0045μM | 19954978 | |||
| CHO-K1 | Function assay | Inverse agonist activity at human CB1 receptor expressed in CHO-K1 cells by GTPgammaS binding assay, IC50=0.0029μM | 20015647 | |||
| CHO-K1 | Function assay | Antagonist activity at human CB1 receptor expressed in CHO-K1 cells assessed as inhibition of CP-55940-induced GTPgammaS binding, IC50=0.0045μM | 20015647 | |||
| COS7 | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in COS7 cells, IC50=0.0051μM | 20015647 | |||
| CHO | Function assay | Inverse agonist activity at human recombinant CB1 receptor expressed in CHO cells by luciferase reporter gene assay, IC50=0.108μM | 20045337 | |||
| CHO | Function assay | 60 mins | Inverse agonist activity at human cannabinoid CB1 receptor expressed in CHO cells assessed as inhibition of forskolin-stimulated cAMP accumulation after 60 mins, EC50=0.00011μM | 20047779 | ||
| CHO | Function assay | 3 hrs | Displacement of [3H]CP-55940 from human recombinant cannabinoid CB1 receptor expressed in CHO cells after 3 hrs by liquid scintillation counting, Ki=0.00074μM | 20047779 | ||
| CHO | Function assay | 10 mins | Antagonist activity at human cannabinoid CB1 receptor expressed in CHO cells coexpressing Galpha15/16 assessed as inhibition of CP-55940-induced Ca2+ release after 10 mins by micro plate reader, IC50=0.0032μM | 20047779 | ||
| CHO | Function assay | 3 hrs | Displacement of [3H]CP-55940 from human recombinant cannabinoid CB2 receptor expressed in CHO cells after 3 hrs by liquid scintillation counting, Ki=0.126μM | 20047779 | ||
| CHO | Function assay | Antagonist activity at human CB1 receptor expressed in CHO cells assessed as inhibition of CP-55940-induced [3H]arachidonic acid release, Kd=0.002512μM | 20363132 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in CHO cells, Ki=0.025μM | 20363132 | |||
| CHO | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in CHO cells, Ki=1.58μM | 20363132 | |||
| CHO-K1 | Function assay | 1 hr | Displacement of [3H]WIN-55212-2 from human CB2 receptor expressed in CHO-K1 cells after 1 hr by liquid scintillation counting, IC50=1.76μM | 20673729 | ||
| HEK | Function assay | Displacement of [3H]CP-55940 from human CB1 receptor expressed in HEK cells, Ki=0.012μM | 20718492 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human CB2 receptor expressed in HEK cells, Ki=0.79μM | 20718492 | |||
| CHO | Function assay | Agonist activity at human CB1 receptor expressed in CHO cells co-expressing Galpha16 protein assessed as mobilization of intracellular calcium, Ke=0.0011μM | 20845959 | |||
| HEK293 | Function assay | Displacement of [3H]SR141716 from human CB1 receptor expressed in HEK293 cells, Ki=0.00118μM | 20845959 | |||
| HEK293 | Function assay | Displacement of [3H]CP55940 from human CB1 receptor expressed in HEK293 cells, Ki=0.00618μM | 20845959 | |||
| CHOK1 | Function assay | Displacement of [3H]CP55940 from human CB2 receptor expressed in CHOK1 cells, Ki=0.313μM | 20845959 | |||
| HEK | Function assay | Displacement of [3H]CP-55,940 from recombinant human CB1 receptor transfected in HEK cells, Ki=0.012μM | 20943290 | |||
| HEK | Function assay | Displacement of [3H]CP-55,940 from recombinant human CB2 receptor transfected in HEK cells, Ki=0.79μM | 20943290 | |||
| CHO | Function assay | Displacement of [3H]SR141716A from human CB1 receptor expressed in CHO cells, Ki=0.0009333μM | 21334892 | |||
| Sf9 | Function assay | Displacement of [3H]CP55940 from human recombinant CB2 receptor expressed in Sf9 cells, Ki=0.85114μM | 21334892 | |||
| CHO | Function assay | Antagonist activity at CB1 receptor transfected in CHO cells expressing apoaequorin as a reporter for G-protein-coupled receptor-mediated calcium signaling by bioluminescence assay, IC50=0.004μM | 21376588 | |||
| HEK293 | Function assay | Noncompetitive inhibition of MRP1-mediated E2-17betaG transport in human MRP1 expressing HEK293 cells by Dixon plot, Ki=1.4μM | 21511945 | |||
| HEK293 | Function assay | Noncompetitive inhibition of MRP4-mediated E2-17betaG transport in human MRP4 expressing HEK293 cells by Dixon plot, Ki=4μM | 21511945 | |||
| HEK293 | Function assay | Induction of MRP2-mediated E2-17betaG transport in human MRP2 expressing HEK293 cells in presence of 2 uM E2-17betaG | 21511945 | |||
| HEK293 | Function assay | Inhibition of MRP3-mediated E2-17betaG transport in human MRP3 expressing HEK293 cells in presence of 15 uM E2-17betaG | 21511945 | |||
| HEK293 | Function assay | Inhibition of MRP1-mediated E2-17betaG transport in human MRP1 expressing HEK293 cells in presence of 0.6 to 5 uM E2-17betaG | 21511945 | |||
| HEK293 | Function assay | Inhibition of MRP4-mediated E2-17betaG transport in human MRP4 expressing HEK293 cells in presence of 0.6 to 5 uM E2-17betaG | 21511945 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK cells, Ki=0.012μM | 21702498 | |||
| HEK | Function assay | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK cells, Ki=0.79μM | 21702498 | |||
| CHO | Function assay | 1 hr | Displacement of [3H]CP55940 from human cannabinoid CB2 receptor expressed in CHO cells at pH 7.4 after 1 hr by liquid scintillation counting, IC50=1.98μM | 21741835 | ||
| HEK293 | Function assay | 5 mins | Inhibition of human ERG expressed in HEK293 cells assessed as inhibition of potassium channel current after 5 mins by patch clamp assay, IC50=2.79μM | 21741835 | ||
| CHO | Function assay | 90 mins | Displacement of [3H]BMS-725519 from human CB1 receptor expressed in CHO cells after 90 mins by scintillation counting, Ki=0.00073μM | 21962575 | ||
| CHO-K1 | Function assay | Antagonist activity at human CB1 receptor expressed in CHO-K1 cells co-expressing Galphaq16 assessed as inhibition of CP55940-induced intracellular calcium mobilization by fluorometry, Ke=0.0011μM | 22372835 | |||
| CHO-K1 | Function assay | Displacement of [3H]CP55940 from human CB1 receptor expressed in CHO-K1 cells, Ki=0.0062μM | 22372835 | |||
| CHO-K1 | Function assay | Displacement of [3H]CP55940 from CB2 receptor expressed in CHO-K1 cells, Ki=0.313μM | 22372835 | |||
| HEK | Function assay | Displacement of [3H]CP-55,940 from human recombinant CB1 receptor transfected in HEK cells, Ki=0.012μM | 22548457 | |||
| HEK | Function assay | Displacement of [3H]CP-55,940 from human recombinant CB2 receptor transfected in HEK cells, Ki=0.79μM | 22548457 | |||
| CHO | Function assay | 2 hrs | Displacement of [3H]CP55,940 from human CB1 receptor expressed in CHO cells after 2 hrs by liquid scintillation counter, Ki=0.0126μM | 22916707 | ||
| CHO | Function assay | 2 hrs | Displacement of [3H]CP55,940 from human CB2 receptor expressed in CHO cells after 2 hrs by liquid scintillation counter, Ki=0.9μM | 22916707 | ||
| CHO | Function assay | Inverse agonist activity against CB1 receptor expressed in human CHO cells assessed as effect on forskolin-stimulated cAMP level, IC50=0.051μM | 22959249 | |||
| CHO | Function assay | 1 hr | Displacement of [3H]CP55940 from human CB1 receptor expressed in CHO cells incubated for 1 hr, Ki=0.0106μM | 23072339 | ||
| CHO | Function assay | > 0.1 nM | 1 hr | Inverse agonist activity at human recombinant CB1 receptor transfected in CHO cells assessed as reduction of [35S]GTPgammaS binding at > 0.1 nM after 1 hr by liquid scintillation spectrometry | 23357307 | |
| N1E-115 | Function assay | 1 uM | 5 mins | Inverse agonist activity at CB1 receptor in mouse N1E-115 cells assessed as inhibition of WIN 55,212-2-induced ERK1/2 phosphorylation at 1 uM treated 5 mins prior to WIN 55,212-2 challenge by Western blot analysis | 23357307 | |
| CHO | Function assay | Antagonist activity at human GPR18 transfected in CHO cells assessed as delta9-THC-induced beta-arrestin recruitment incubated 60 mins prior to delta9-THC addition by beta-arrestin translocation assay, IC50=10.1μM | 23679955 | |||
| HEK | Function assay | 1 hr | Antagonist activity at human CB1 receptor overexpressed in HEK cells assessed as [35S]GTPgamma binding after 1 hr by liquid scintillation counting analysis, EC50=0.0015μM | 24175572 | ||
| CHOK1 | Function assay | 1 hr | Displacement of [3H]CP55940 from human CB1 receptor expressed in CHOK1 cells after 1 hr by liquid scintillation counting analysis, Ki=0.0059μM | 24175572 | ||
| CHO | Function assay | 45 mins | Antagonist activity at human CB1 receptor stably expressed in CHO cells co-expressing co-expressing Ga15/16 assessed as calcium current after 45 mins by fluo-4 AM assay, IC50=0.013μM | 24445310 | ||
| CHO | Function assay | 45 mins | Antagonist activity at CB2 receptor (unknown origin) stably expressed in CHO cells co-expressing co-expressing Ga15/16 assessed as calcium current after 45 mins by fluo-4 AM assay, IC50=9.8μM | 24445310 | ||
| CHO-K1 | Function assay | 10 mins | Antagonist activity at human CB1 receptor expressed in CHO-K1 cells assessed as inhibition of CP-55940-induced [35S]GTPgammaS binding incubated for 10 mins prior to CP-55940-challenge measured after 1 hr by beta counting, Ki=0.0016μM | 24900484 | ||
| HEK293 | Function assay | 60 mins | Displacement of [3H]SR141716A form human CB1 receptor expressed in HEK293 cells after 60 mins by beta counting, Ki=0.0018μM | 24900484 | ||
| CHO-K1 | Function assay | 60 mins | Displacement of [3H]CP-55940 from human CB2 receptor expressed in CHO-K1 cells after 60 mins by beta counting, Ki=0.522μM | 24900484 | ||
| CHO | Function assay | 2 hrs | Displacement of [3H]CP55,940 from recombinant human CB1 receptor expressed in CHO cells after 2 hrs by liquid scintillation counting, Ki=0.0126μM | 24900561 | ||
| CHO | Function assay | 2 hrs | Displacement of [3H]CP55,940 from recombinant human CB2 receptor expressed in CHO cells after 2 hrs by liquid scintillation counting, Ki=0.9μM | 24900561 | ||
| CHO | Function assay | 90 mins | Agonist activity at GPR55 (unknown origin) expressed in CHO cells assessed as inhibition of LPI-induced beta-arrestin translocation after 90 mins by luminescence assay, EC50=2.01μM | 24900561 | ||
| RD-HGA16 | Function assay | Antagonist activity at CB1-OX1 heterodimer (unknown origin) expressed in RD-HGA16 cells assessed as inhibition of orexin A-induced calcium mobilization by fluorometric imaging plate reader analysis, Ke=0.001μM | 24944734 | |||
| RD-HGA16 | Function assay | Antagonist activity at CB1 receptor (unknown origin) expressed in RD-HGA16 cells assessed as inhibition of CP55940-induced calcium mobilization by fluorometric imaging plate reader analysis, Ke=0.0011μM | 24944734 | |||
| Sf9 | Function assay | 20 mins | Inverse agonist activity at human CB1 receptor expressed in Sf9 cells coexpressing Gbeta1gamma2 and RSG4 assessed as degradation of [gamma-33P]GTP after 20 mins by steady-state GTPase assay, EC50=0.05μM | 25096297 | ||
| Sf9 | Function assay | 20 mins | Inverse agonist activity at human CB1 receptor expressed in Sf9 cells coexpressing Gbeta1gamma2 and RSG4 assessed as degradation of [gamma-33P]GTP after 20 mins by steady-state GTPase assay, EC50=0.05012μM | 25096297 | ||
| CHO | Function assay | Antagonist activity against CB1R (unknown origin) CHO cells stably expressing Galpha16 assessed as inhibition of CP55940-induced increase in intracellular calcium level pre-treated 10 mins before CP55940 stimulation by microplate reader based assay, IC50=0.0135μM | 26151231 | |||
| CHO | Function assay | Antagonist activity against CB2R (unknown origin) CHO cells stably expressing Galpha16 assessed as inhibition of CP55940-induced increase in intracellular calcium level pre-treated 10 mins before CP55940 stimulation by microplate reader based assay, IC50=9.1μM | 26151231 | |||
| HEK293 | Function assay | Immediate antagonist/Inverse agonist activity at hemagglutinin-tagged human CB1 receptor expressed in HEK293 cells assessed as reversal of 1 uM CP55,940-induced inhibition of 5 uM forskolin-induced cAMP accumulation by kinetic cAMP assay | 26203658 | |||
| HEK293 | Function assay | 90 mins | Displacement of [3H]-CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells after 90 mins by Competition binding assay, Kieq=0.012μM | 26756097 | ||
| HEK293 | Function assay | 90 mins | Displacement of [3H]-CP-55940 from human recombinant CB2 receptor expressed in HEK293 cells after 90 mins by Competition binding assay, Kieq=0.79μM | 26756097 | ||
| CHO-K1 | Function assay | Antagonist activity at human CB1 receptor expressed in CHO-K1 cells assessed as inhibition of CP55940-induced intracellular calcium mobilization by Calcein-4 AM-staining based FLIPR assay, Ke=0.0011μM | 26827137 | |||
| CHO | Function assay | 90 secs | Inverse agonist activity at human CB1 receptor expressed in CHO cells assessed as increase in intracellular calcium mobilization after 90 secs by Calcein-4 AM-staining based FLIPR assay, EC50=0.005μM | 26827137 | ||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-MC cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for DAOY cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for TC32 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Saos-2 cells | 29435139 | |||
| CHOK1 | Function assay | 90 mins | Inverse agonist activity at C-terminally prolink-tagged mouse CB2 receptor expressed in CHOK1 cells harboring beta-galactosidase enzyme fused beta-arrestin assessed as increase in beta-arrestin recruitment after 90 mins by chemiluminescent assay, EC50=0.015μM | 29939744 | ||
| HEK293 | Function assay | Antagonist activity at human CB1 receptor expressed in HEK293 cells by GTPgammaS binding assay, Ke=0.0002μM | 30077609 | |||
| HEK293 | Function assay | 90 secs | Inverse agonist activity at human CB1 receptor expressed in HEK293 cells co-expressing Galphaq16 assessed as inhibition of calcium mobilization after 90 secs by calcein-4 AAM dye-based FLIPR assay, EC50=0.586μM | 30077609 | ||
| HEK293 | Function assay | 30 mins | Inverse agonist activity at human CB1 receptor expressed in HEK293 cells assessed as inhibition of forskolin-induced cAMP accumulation after 30 mins by HTRF assay, EC50=0.0041μM | 30339387 | ||
| CHO | Function assay | Displacement of [3H]-CP55940 from human recombinant cannabinoid CB1 receptor expressed in CHO cells by radioligand binding assay, Ki=0.143μM | 31609608 | |||
| CHO | Function assay | Agonist activity at human GPR55 expressed in CHO cells assessed as induction of LPI-induced beta-arrestin recruitment by beta-galactosidase enzyme fragment complementation method, EC50=2.01μM | ChEMBL | |||
| CHO | Function assay | Agonist activity at human GPR18 expressed in CHO cells assessed as induction of beta-arrestin recruitment by beta-galactosidase enzyme fragment complementation method, EC50=10.1μM | ChEMBL | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 92 mg/mL
(198.36 mM)
Ethanol : 50 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 463.79 | Formula | C22H21Cl3N4O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 168273-06-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=C(N(N=C1C(=O)NN2CCCCC2)C3=C(C=C(C=C3)Cl)Cl)C4=CC=C(C=C4)Cl | ||
Mechanism of Action
| Features |
Efficacious to induce weight reduction and improvements in cardiometabolic risk factors, however was withdrawn in 2009 due to severe depressive disorder and anxiety.
|
|---|---|
| Targets/IC50/Ki |
ACAT1
ACAT2
MmpL3
hCB1
(Cell-free assay) 13.6 nM
hCB2
(Cell-free assay) 1.64 μM
|
| In vitro |
Rimonabant (SR141716) dose-dependently reduces ACAT activity in Raw264.7macrophages with IC50 of 2.9 μM and isolated peritoneal macrophages. It inhibits ACATactivity in intact CHO-ACAT1 and CHO-ACAT2 cells and in cell-free assays with approximately equal efficiency with IC50 of 1.5 μM and 2.2 μM for CHO-ACAT1 and CHO-ACAT2, respectively. Consistent with ACAT inhibition, this compound treatment blocks ACAT dependent processes in macrophages, oxysterol-induced apoptosis and acetylated-LDL induced foam cell formation. It antagonizes the inhibitory effects of cannabinoid receptor agonists on both mouse vas deferens contractions and adenylyl cyclase activity in rat brain membranes in a concentration-dependent manner. The compound significantly reduces cell growth and induces cell death of human colorectal cancer cells (DLD-1, CaCo-2 and SW620). It is able to alter cell cycle distribution in all the cell lines tested. Particularly, Rimonabant produces a G2/M cell cycle arrest in DLD-1 cells without inducing apoptosis or necrosis. |
| Kinase Assay |
Radioligand Binding Assay
|
|
Human CB1 and CB2 stably transfect HEK 293 cells and cell membrane is purified. 0.2-8 μg of the purified membrane is incubated with 0.75 nM [3H] CP55,940 and Rimonabant (SR141716) in the incubation buffer (50 mM Tris-HCl, 5 mM MgCl2, 1 mM EDTA, 0.3%BSA, pH 7.4). The non-specific binding is defined in the presence of 1 μM of CP55,940. The reactions are incubated for one and a half hours at 30 °C in Multiscreen. The reactions are terminated by manifold filtration and washed four times with ice-cold wash buffer (50mM Tris, pH 7.4, 0.25% BSA).The radioactivity bound to the filters is measured by Topcount. The IC50 is determined as the concentration of this compound required to inhibit 50% of the binding of [3H] CP55,940 and calculated by non-linear regression.
|
|
| In vivo |
Rimonabant (SR141716) is administered intraperitoneally or orally and potently and dose-dependently antagonizes classical pharmacological and behavioural effects of cannabinoid receptor agonists. In the mouse model of azoxymethane-induced colon carcinogenesis, it significantly decreased aberrant crypt foci (ACF) formation, which precedes colorectal cancer. This compound (10 mg/kg by gavage) is fed for 2 weeks to 3-month-old male obese Zucker rats as an impaired glucose tolerance model and for 10 weeks to 6-month-old male obese Zucker rats as a model of the metabolic syndrome. RANTES (Regulated upon Activation, Normal T cell Expressed, and Secreted) and MCP-1 (monocyte chemotactic protein-1) serum levels are increased in obese vs lean Zucker rats and significantly reduced by long-term treatment with it, which slows weight gain in rats with the metabolic syndrome. Neutrophils and monocytes are significantly increased in young and old obese vs lean Zucker rats and lowered by this compound. Platelet-bound fibrinogen is significantly enhanced in obese vs lean Zucker rats of both ages, and is reduced by it. Platelets from obese rats are more sensitive to thrombin-induced aggregation and adhesion to fibrinogen, which are both attenuated by Rimonabant therapy. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | CD44 / Lgr5 / CD133 / EpCAM β-catenin p-LRP6 / LRP6 / Lgr5 |
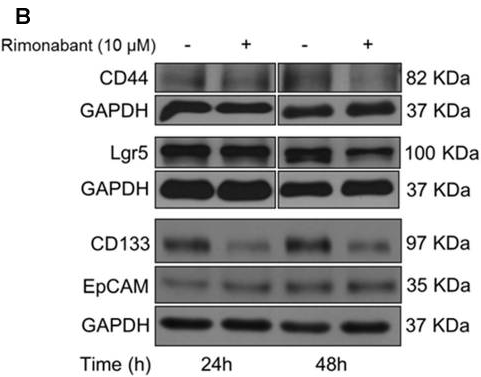
|
29354056 |
| Immunofluorescence | β-catenin Lgr5 |
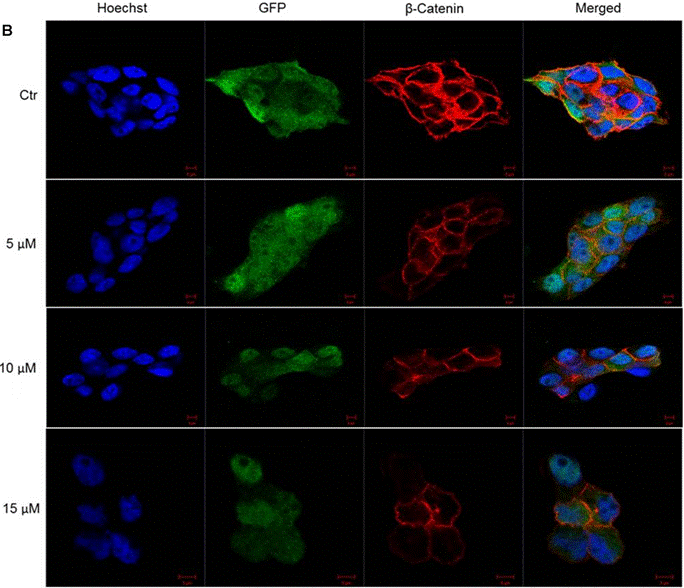
|
29354056 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00750347 | Completed | Pain |
University Hospital Clermont-Ferrand |
September 2008 | Phase 1 |
| NCT00656487 | Completed | Cannabis Dependence|Cannabis Withdrawal |
The Scripps Research Institute|National Institute on Drug Abuse (NIDA) |
April 30 2008 | Phase 2 |
| NCT00546325 | Completed | Diabetes Mellitus Type 2 |
Sanofi |
October 2007 | Phase 3 |
| NCT00525681 | Completed | Renal Transplantation |
University of Oslo School of Pharmacy |
September 2007 | Phase 4 |
| NCT00584389 | Terminated | Obesity |
University of Surrey|European Foundation for the Study of Diabetes|Royal Surrey County Hospital NHS Foundation Trust |
July 2007 | Phase 4 |
| NCT01041170 | Completed | Cannabis|Dependence |
National Institute on Drug Abuse (NIDA)|National Institutes of Health Clinical Center (CC) |
April 16 2006 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































