research use only
PNU-120596 AChR modulator
Cat.No.S2629
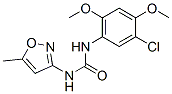
Chemical Structure
Molecular Weight: 311.72
Quality Control
Solubility
|
In vitro |
DMSO
: 62 mg/mL
(198.89 mM)
Ethanol : 1 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 311.72 | Formula | C13H14ClN3O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 501925-31-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Nsc 216666 | Smiles | CC1=CC(=NO1)NC(=O)NC2=CC(=C(C=C2OC)OC)Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
α7 nAChR
216 nM(EC50)
|
|---|---|
| In vitro |
PNU-120596 increases agonist (Ach)-evoked calcium flux mediated by an engineered variant of the human α7 nAChR. This compound increases agonists (choline and ACh)-evoked currents mediated by wild-type receptors and also demonstrates a pronounced prolongation of the evoked response in the continued presence of agonist in Xenopus oocytes. It increases the channel mean open time ofα7 nAChRs but has no effect on ion selectivity and relatively little, if any, effect on unitary conductance. When applied to acute hippocampal slices, this chemical increases the frequency of ACh-evoked GABAergic postsynaptic currents measured in pyramidal neurons; this effect is suppressed by TTX, suggesting that it modulates the function of α7 nAChRs located on the somatodendritic membrane of hippocampal interneurons. Besides the positive modulation to α7 nAChR, this compound induces a profound retardation of the kinetics of desensitization, raising the potential of Ca2+-induced toxicity through excessive stimulation of α7 nAChR. It causes changes in cysteine accessibility at the inner beta sheet, transition zone and agonist binding site while binding to α7 nAChR. Binding sites for this chemical are not in the agonist-binding sites and it enhances agonist-evoked gating of nicotinic receptors by eliciting conformational effects that are similar but nonidentical to the gating conformations promoted by Ach.
|
| Kinase Assay |
Ca2+ Fluorescence Assay
|
|
SH-EP1 human epithelial cells expressing a variant of theα7 nAChR (α7*) are grown in minimal essential medium (MEM) containing nonessential amino acids supplemented with 10% fetal bovine serum, L-glutamine, 100 U/ml penicillin/streptomycin, 250 ng/mL fungizone, 400 μg/mL hygromycin B, and 800 μg/mL geneticin. α7* is a variant of the human α7 nAChR, with two point mutations in the first transmembrane domain (T230P and C241S) that allow for high functional expression in SH-EP1 cells [Groppi VE, Wolfe ML, Berkenpas MB (2003) U.S. Patent 6,693,172 B1]. Cells are grown in a 37 °C incubator with 6% CO2. Cells are trypsinized and plated in 96-well plates with dark side walls and clear bottoms at a density of 2 × 104 cells/well 2 days before analysis. Cells are loaded with a mixture of Calcium reen-1AM in anhydrous dimethylsulfoxide and 20% pluronic F-127. This reagent is added directly to the growth medium of each well to achieve a final concentration of 2 μM Calcium Green-1 AM. Cells are then incubated in the dye for 1 hour at 37 °C and then washed four times with Mark's modified Earle's balanced salt solution (MMEBSS) composed of the following (inmM): 4 CaCl2, 0.8 MgSO4, 20 NaCl, 5.3 KCl, 5.6 D-glucose, 20 Tris-HEPES, and 120 N-methyl-D-glucamine, pH 7.4. After the fourth cycle, the cells are allowed to incubate at 37 °C for at least 10 minutes. The final volume of MMEBSS in each well is 100 μL and atropine is added to all wells for a final concentration of 1 μM. A fluorometric imaging plate reader (FLIPR; Molecular Devices, Union City, CA) is set up to excite Calcium Green at 488 nm using 500 mW of power and reading fluorescence emission of >525 nm. A 0.5 seconds exposure is used to illuminate each well. Fluorescence is detected using an F-stop set of either 2.0 or 1.2. After 30 seconds of baseline recording, test compounds are added to each well of a 96-well plate in 50 μL of a 3 × stock. In each experiment, four wells are used as vehicle (0.2% DMSO) controls.
|
|
| In vivo |
Systemic administration of PNU-120596 (1 mg/kg) to rats improves the auditory gating deficit caused by amphetamine, a model proposed to reflect a circuit level disturbance associated with schizophrenia. When administered prior to Carrageenan, 30 mg/kg of this compound significantly blunts mechanical hyperalgesia and weight bearing deficits for up to 4 hours. This chemical attenuates the carrageenan-induced increase in levels of TNF-α and IL-6 within the hindpaw oedema, diclofenac only attenuated IL-6 levels. Established mechanical hyperalgesia induced by Carrageenan or CFA is also partially reversed by this agent.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































