- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Alisertib (MLN8237)
Alisertib (MLN8237) is a selective Aurora A inhibitor with IC50 of 1.2 nM in a cell-free assay. It has >200-fold higher selectivity for Aurora A than Aurora B. Alisertib induces cell cycle arrest, apoptosis and autophagy. Phase 3.
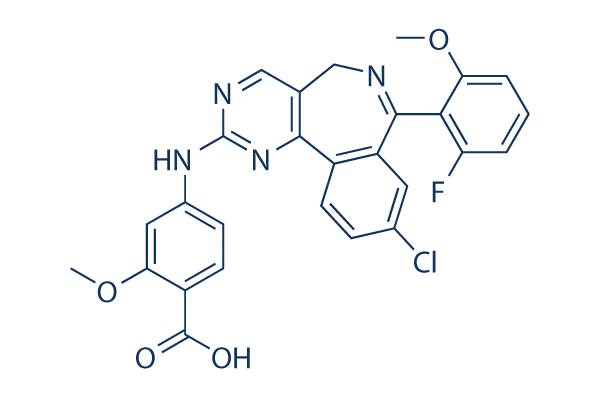
Alisertib (MLN8237) Chemical Structure
CAS: 1028486-01-2
Selleck's Alisertib (MLN8237) has been cited by 338 publications
Purity & Quality Control
Batch:
Purity:
99.64%
99.64
Products often used together with Alisertib (MLN8237)
Alisertib and Barasertib strongly inhibit the growth and proliferation of AML cells.
Alisertib and Volasertib inhibit AURKA and PLK1 and synergistically block cell cycle progression in diffuse midline glioma cells.
Alisertib and Lenvatinib significantly enhance the suppression of tumor growth and induce apoptosis in nude mice.
Alisertib and Irinotecan along with temozolomide show antitumor activity in patients with MYCN nonamplified tumors.
DuBois SG, et al.Clin Cancer Res. 2018 Dec 15;24(24):6142-6149.
Alisertib and Paclitaxel give additive and synergistic antitumor effects in xenograft models of triple-negative breast cancer.
Alisertib (MLN8237) Related Products
| Related Targets | Aurora A Aurora B Aurora C Aurora B | Click to Expand |
|---|---|---|
| Related Compound Libraries | Kinase Inhibitor Library PI3K/Akt Inhibitor Library MAPK Inhibitor Library DNA Damage/DNA Repair compound Library Cell Cycle compound library | Click to Expand |
Signaling Pathway
Choose Selective Aurora Kinase Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HCT116 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=0.04 μM | 26136684 |
| LS174T | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=0.05 μM | 26136684 |
| T84 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=0.09 μM | 26136684 |
| LS180 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=1 μM | 26136684 |
| SW948 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=1 μM | 26136684 |
| HCT15 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50<0.4 μM | 26136684 |
| DLD-1 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50<0.8 μM | 26136684 |
| MIP-101 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=1 μM | 26136684 |
| SNU1544 | Growth Inhibition Assay | 0.5 μM | 72 h | DMSO | IC50=1 μM | 26136684 |
| OCI-Ly10 | Cytotoxic Assay | 72 h | DMSO | IC50=0.058 μM | 25878331 | |
| SU-DHL2 | Cytotoxic Assay | 72 h | DMSO | IC50=0.01 μM | 25878331 | |
| OCI-LY7 | Cytotoxic Assay | 72 h | DMSO | IC50=0.081 μM | 25878331 | |
| SU-DHL6 | Cytotoxic Assay | 72 h | DMSO | IC50=0.482 μM | 25878331 | |
| Jeko-1 | Cytotoxic Assay | 72 h | DMSO | IC50=0.029 μM | 25878331 | |
| JVM-2 | Cytotoxic Assay | 72 h | DMSO | IC50=0.01 μM | 25878331 | |
| Rec-1 | Cytotoxic Assay | 72 h | DMSO | IC50=0.087 μM | 25878331 | |
| Z-138 | Cytotoxic Assay | 72 h | DMSO | IC50=0.013 μM | 25878331 | |
| H9 | Cytotoxic Assay | 72 h | DMSO | IC50=0.6 μM | 25878331 | |
| HH | Cytotoxic Assay | 72 h | DMSO | IC50=0.7 μM | 25878331 | |
| DND41 | Cytotoxic Assay | 72 h | DMSO | IC50=0.1 μM | 25878331 | |
| CCL119 | Cytotoxic Assay | 72 h | DMSO | IC50=0.062 μM | 25878331 | |
| J.Cam 1.6 | Cytotoxic Assay | 72 h | DMSO | IC50=0.105 μM | 25878331 | |
| Sup-T1 | Cytotoxic Assay | 72 h | DMSO | IC50=2.142 μM | 25878331 | |
| Tib 152 | Cytotoxic Assay | 72 h | DMSO | IC50=0.8 μM | 25878331 | |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Induces G2/M arrest | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Induces G3/M arrest | 25834401 |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Decreases the expression level of CDK1/CDC2 | 25834401 |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Decreases the expression level of CDK2 | 25834401 |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Decreases the expression level of cyclin B1 | 25834401 |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Increases the expression level of p21 Waf1/Cip1 | 25834401 |
| MCF7 | Function Assay | 5 μM | 24 h | DMSO | Increases the expression level of p27 Kip1 | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Decreases the expression level of CDK1/CDC2 | 25834401 |
| MDA-MB-231 | Function Assay | 1 μM | 24 h | DMSO | Increases the expression level of CDK2 | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Decreases the expression level of cyclin B1 | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Increases the expression level of p21 Waf1/Cip1 | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Increases the expression level of p27 Kip1 | 25834401 |
| MDA-MB-231 | Function Assay | 5 μM | 24 h | DMSO | Increases the expression level of p53 | 25834401 |
| MCF7 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptotic death | 25834401 |
| MDA-MB-231 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptotic death | 25834401 |
| MCF7 | Function Assay | 1 μM | 72 h | DMSO | Induces autophagic death | 25834401 |
| MDA-MB-231 | Function Assay | 1 μM | 72 h | DMSO | Induces autophagic death | 25834401 |
| U-2 OS | Growth Inhibition Assay | 50 μM | 24 h | DMSO | IC50=16.6 μM | 25792811 |
| MG-63 | Growth Inhibition Assay | 50 μM | 24 h | DMSO | IC50=9.5 μM | 25792811 |
| U-2 OS | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptotic cell death | 25792811 |
| MG-63 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptotic cell death | 25792811 |
| U-2 OS | Function Assay | 5 μM | 24 h | DMSO | Promotes autophagic cell death | 25792811 |
| MG-63 | Function Assay | 5 μM | 24 h | DMSO | Promotes autophagic cell death | 25792811 |
| PANC-1 | Growth Inhibition Assay | 50 μM | 24 h | DMSO | IC50=7.1 μM | 25632225 |
| BxPC-3 | Growth Inhibition Assay | 50 μM | 24 h | DMSO | IC50=6.8 μM | 25632225 |
| PANC-1 | Function Assay | 5 μM | 24 h | DMSO | Induces cell cycle arrest in G2/M phase | 25632225 |
| BxPC-3 | Function Assay | 5 μM | 24 h | DMSO | Induces cell cycle arrest in G2/M phase | 25632225 |
| PANC-1 | Function Assay | 5 μM | 24 h | DMSO | Induces autophagic cell death | 25632225 |
| BxPC-3 | Function Assay | 5 μM | 24 h | DMSO | Induces autophagic cell death | 25632225 |
| SKOV3 | Growth Inhibition Assay | 100 μM | 24 h | DMSO | IC50=20.48 μM | 25624750 |
| OVCAR4 | Growth Inhibition Assay | 100 μM | 24 h | DMSO | IC50=22.13 μM | 25624750 |
| SKOV3 | Function Assay | 5 μM | 72 h | DMSO | Induces G2/M arrest | 25624750 |
| OVCAR4 | Function Assay | 5 μM | 72 h | DMSO | Induces G2/M arrest | 25624750 |
| SKOV3 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptosis | 25624750 |
| OVCAR4 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptosis | 25624750 |
| AGS | Growth Inhibition Assay | 25 μM | 24 h | DMSO | IC50=19.09 μM | 25609923 |
| NCI-N78 | Growth Inhibition Assay | 25 μM | 24 h | DMSO | IC50=26.33 μM | 25609923 |
| AGS | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptosis | 25609923 |
| NCI-N78 | Apoptosis Assay | 5 μM | 24 h | DMSO | Induces apoptosis | 25609923 |
| AGS | Function Assay | 5 μM | 24 h | DMSO | Induces the autophagy | 25609923 |
| NCI-N78 | Function Assay | 5 μM | 24 h | DMSO | Induces the autophagy | 25609923 |
| HSC-3 | Growth Inhibition Assay | 1 μM | 48 h | IC50=0.54 μM | 25366143 | |
| GB30 | Growth Inhibition Assay | 1 μM | 7 d | DMSO | IC50=0.011 μM | 25106428 |
| GB9 | Growth Inhibition Assay | 1 μM | 7 d | DMSO | IC50=0.024 μM | 25106428 |
| GB169 | Growth Inhibition Assay | 1 μM | 7 d | DMSO | IC50=0.032 μM | 25106428 |
| T24 | Function Assay | 1 μM | 48 h | DMSO | Induces cell cycle arrest | 23403633 |
| RT4 | Function Assay | 1 μM | 48 h | DMSO | Induces cell cycle arrest | 23403633 |
| UM-UC-3 | Function Assay | 1 μM | 48 h | DMSO | Induces cell cycle arrest | 23403633 |
| T24 | Apoptosis Assay | 3.16 μM | 96 h | DMSO | IC50=0.0306 μM | 23403633 |
| RT4 | Apoptosis Assay | 3.16 μM | 96 h | DMSO | IC50=0.1198 μM | 23403633 |
| UM-UC-3 | Apoptosis Assay | 3.16 μM | 96 h | DMSO | IC50=0.0449 μM | 23403633 |
| OVCAR-5 | Function Assay | 50 nM | Inhibits cell migration | 23334327 | ||
| SKOV3ip2 | Function Assay | 50 nM | Inhibits cell migration | 23334327 | ||
| S462 | Growth Inhibition Assay | 100 μM | 72 h | DMSO | Attenuates cell growth | 23328114 |
| 2884 | Growth Inhibition Assay | 100 μM | 72 h | DMSO | Attenuates cell growth | 23328114 |
| 2885 | Growth Inhibition Assay | 100 μM | 72 h | DMSO | Attenuates cell growth | 23328114 |
| CRL-2396 | Growth Inhibition Assay | 100 μM | water | IC50=0.092 μM | 23153524 | |
| TIB-48 | Growth Inhibition Assay | 100 μM | water | IC50=0.088 μM | 23153524 | |
| CRL-2396 | Cytotoxic Assay | 1 μM | 48 h | water | Induces apoptosis | 23153524 |
| TIB-48 | Cytotoxic Assay | 1 μM | 48 h | water | Induces apoptosis | 23153524 |
| AGS | Cytotoxic Assay | 0.5 μM | 24 h | DMSO | Decreases cell survival | 22972611 |
| FLO-1 | Cytotoxic Assay | 0.5 μM | 24 h | DMSO | Decreases cell survival | 22972611 |
| OE33 | Cytotoxic Assay | 0.5 μM | 24 h | DMSO | Decreases cell survival | 22972611 |
| SKLMS | Cytotoxic Assay | 75 nM | 96 h | Induces apoptosis | 22821997 | |
| Leio285 | Cytotoxic Assay | 75 nM | 96 h | Induces apoptosis | 22821997 | |
| Mes-Sa | Cytotoxic Assay | 75 nM | 96 h | Induces apoptosis | 22821997 | |
| DAOY | Cytotoxic Assay | 10 μM | 72 h | DMSO | IC50=0.04 μM | 22669335 |
| IMR32 | Cytotoxic Assay | 10 μM | 72 h | DMSO | IC50=0.03 μM | 22669335 |
| Molt-4 | Cytotoxic Assay | 10 μM | 72 h | DMSO | IC50=0.02 μM | 22669335 |
| MOLM-13 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| HL-60 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| MV4-11 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| SKM-1 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| SH2 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| NOMO-1 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| OCL-AML2 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| PL-21 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| KG-1 | Growth Inhibition Assay | 3 μM | 72 h | Diminishes cell viability | 22488249 | |
| A172 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.120 μM | 22274399 |
| U87 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.105 μM | 22274399 |
| U251 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.100 μM | 22274399 |
| T98 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.125 μM | 22274399 |
| LN18 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.210 μM | 22274399 |
| LN443 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.220 μM | 22274399 |
| HF66 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.225 μM | 22274399 |
| HF2303 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.060 μM | 22274399 |
| HF2359 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.060 μM | 22274399 |
| HF2414 | Cytotoxic Assay | 100 μM | 24 h | DMSO | IC50=0.080 μM | 22274399 |
| A-673 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.032 μM | 21448591 |
| TC-32 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.039 μM | 21448591 |
| TC-71 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.102 μM | 21448591 |
| SK-N-MC | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.072 μM | 21448591 |
| CHLA-9 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.018 μM | 21448591 |
| CHLA-10 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.060 μM | 21448591 |
| CHLA-25 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.168 μM | 21448591 |
| CHLA-32 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.136 μM | 21448591 |
| CHLA-56 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=10 μM | 21448591 |
| CHLA-258 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.132 μM | 21448591 |
| COG-E-352 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.043 μM | 21448591 |
| CHLA-90 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.061 μM | 21448591 |
| CHLA-119 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.022 μM | 21448591 |
| CHLA-122 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.019 μM | 21448591 |
| CHLA-136 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.039 μM | 21448591 |
| CHLA-140 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.026 μM | 21448591 |
| LA-N-6 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.054 μM | 21448591 |
| NB-1643 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.037 μM | 21448591 |
| NB-EBc1 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.050 μM | 21448591 |
| SK-N-BE-1 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.028 μM | 21448591 |
| SK-N-BE-2 | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.036 μM | 21448591 |
| SMS-KAN | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.034 μM | 21448591 |
| SMS-KANR | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.026 μM | 21448591 |
| SMS-KCN | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.019 μM | 21448591 |
| SMS-KCNR | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.010 μM | 21448591 |
| SMS-LHN | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.032 μM | 21448591 |
| SMS-MSN | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.022 μM | 21448591 |
| SMS-SAN | Growth Inhibition Assay | 10 μM | 96 h | DMSO | IC50=0.020 μM | 21448591 |
| Granta-4 | Cytotoxic Assay | 10 μM | 7 d | IC50=0.040 μM | 21291867 | |
| DB | Cytotoxic Assay | 10 μM | 7 d | IC50=0.042 μM | 21291867 | |
| RL | Cytotoxic Assay | 10 μM | 7 d | IC50=0.015 μM | 21291867 | |
| K562 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.087 μM | 21091633 | |
| LAMA-84 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.057 μM | 21091633 | |
| MM15 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=0.13 μM | 20382844 |
| OPM1 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=0.03 μM | 20382844 |
| RPM1 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=10.32 μM | 20382844 |
| INA6 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=0.002 μM | 20382844 |
| OPM2 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=4.37 μM | 20382844 |
| MM1R | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=1.68 μM | 20382844 |
| DOX40 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=5.48 μM | 20382844 |
| LR5 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=2.53 μM | 20382844 |
| U266 | Growth Inhibition Assay | 4 μM | 72 h | DMSO | IC50=1.43 μM | 20382844 |
| RD | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.228 μM | 20108338 | |
| Rh41 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.090 μM | 20108338 | |
| Rh30 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.230 μM | 20108338 | |
| BT-12 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.060 μM | 20108338 | |
| CHLA-266 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.072 μM | 20108338 | |
| TC-71 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.102 μM | 20108338 | |
| SJ-GBM2 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.050 μM | 20108338 | |
| NALM-6 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.062 μM | 20108338 | |
| COG-LL-317 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.047 μM | 20108338 | |
| RS4-11 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.018 μM | 20108338 | |
| MOLT-4 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.026 μM | 20108338 | |
| CCRF-CEM | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.094 μM | 20108338 | |
| Kasumi-1 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.103 μM | 20108338 | |
| Karpas-299 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.038 μM | 20108338 | |
| Ramos-RA1 | Growth Inhibition Assay | 10 μM | 96 h | IC50=0.127 μM | 20108338 | |
| GSS | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GSS cells after 72 hrs by WST8 assay, IC50 = 0.039 μM. | 25625617 | ||
| LU99A | Antiproliferative assay | 72 hrs | Antiproliferative activity against human LU99A cells after 72 hrs by WST8 assay, IC50 = 0.062 μM. | 25625617 | ||
| HL60 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HL60 cells after 72 hrs by WST8 assay, IC50 = 0.074 μM. | 25625617 | ||
| LC2/ad | Antiproliferative assay | 72 hrs | Antiproliferative activity against human LC2/ad cells after 72 hrs by WST8 assay, IC50 = 0.077 μM. | 25625617 | ||
| MKN45 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MKN45 cells after 72 hrs by WST8 assay, IC50 = 0.093 μM. | 25625617 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by WST8 assay, IC50 = 0.095 μM. | 25625617 | ||
| Lu116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Lu116 cells after 72 hrs by WST8 assay, IC50 = 0.097 μM. | 25625617 | ||
| NCI-H358 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H358 cells after 72 hrs by WST8 assay, IC50 = 0.1 μM. | 25625617 | ||
| MIAPaCa2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MIAPaCa2 cells after 72 hrs by WST8 assay, IC50 = 0.13 μM. | 25625617 | ||
| PC14 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PC14 cells after 72 hrs by WST8 assay, IC50 = 0.17 μM. | 25625617 | ||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells after 72 hrs by WST8 assay, IC50 = 0.33 μM. | 25625617 | ||
| HCT15 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT15 cells after 72 hrs by WST8 assay, IC50 = 0.74 μM. | 25625617 | ||
| Sf9 | Function assay | Competitive inhibition of recombinant mouse aurora kinase A expressed in insect Sf9 cells in presence of ATP, Ki = 0.0003 μM. | 26101564 | |||
| Sf9 | Function assay | Inhibition of recombinant mouse aurora kinase A expressed in insect Sf9 cells using biotin-GLRRASLG as substrate in presence of [gamma-33P]ATP, IC50 = 0.001 μM. | 26101564 | |||
| HCT116 | Function assay | Inhibition of aurora kinase A autophosphorylation at T288 in human HCT116 cells by immunofluorescence analysis, IC50 = 0.007 μM. | 26101564 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells assessed as inhibition of cell proliferation by BrdU incorporation assay, GI50 = 0.03 μM. | 26101564 | |||
| HCT116 | Function assay | Inhibition of aurora kinase B in human HCT116 cells assessed as inhibition of histone H3 phosphorylation by immunofluorescence analysis, IC50 = 1.5 μM. | 26101564 | |||
| BL21 (DE3) Rosetta | Function assay | 30 mins | Inhibition of His-tagged human Aurora A kinase (122 to 40 residues) expressed in Escherichia coli BL21 (DE3) Rosetta cells using biotinylated STK2 substrate incubated for 30 mins by HTRF assay, IC50 = 0.00004 μM. | 27391133 | ||
| HeLa Kyoto | Function assay | 20 hrs | Inhibition of Aurora B kinase in human HeLa Kyoto cells assessed as effect on distribution of phspho-histone H3 ser10 level incubated for 20 hrs, IC50 = 0.0015 μM. | 27391133 | ||
| HeLa Kyoto | Function assay | 20 hrs | Inhibition of Aurora A kinase autophosphorylation at Thr288 in human HeLa Kyoto cells incubated for 20 hrs, IC50 = 0.0067 μM. | 27391133 | ||
| multiple myeloma | Function assay | Suppression of cell mitosis in human multiple myeloma cells, IC50 = 0.003 μM. | 28918096 | |||
| Calu6 | Antitumor assay | 20 mg/kg | 21 days | Antitumor activity against human Calu6 cells xenografted in mouse assessed as tumor growth inhibition at 20 mg/kg, po bid administered for 21 days | 26101564 | |
| HeLa Kyoto | Function assay | 0.25 uM | 20 hrs | Inhibition of Aurora A kinase localization at spindle microtubules in human HeLa Kyoto cells at 0.25 uM incubated for 20 hrs | 27391133 | |
| MDA-MB-231 | Function assay | 1 uM | 48 hrs | Induction of chromosome alignment defects in human MDA-MB-231 cells at 1 uM after 48 hrs by DAPI staining based immunofluorescence assay | 29358147 | |
| MDA-MB-231 | Function assay | 1 uM | 48 hrs | Induction of aberrant spindle formation with tripolar and tetrapolar occurrence in human MDA-MB-231 cells at 1 uM after 48 hrs by DAPI staining based immunofluorescence assay | 29358147 | |
| MDA-MB-231 | Function assay | 0.5 uM | 48 hrs | Inhibition of alpha-tubulin in human MDA-MB-231 cells assessed as abolishment of regular location of protein at 0.5 uM after 48 hrs by DAPI staining based immunofluorescence assay | 29358147 | |
| MDA-MB-231 | Function assay | 0.5 uM | 48 hrs | Inhibition of AURKA in human MDA-MB-231 cells assessed as abolishment of regular location of protein at 0.5 uM after 48 hrs by DAPI staining based immunofluorescence assay | 29358147 | |
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB-EBc1 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for MG 63 (6-TG R) cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-MC cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for TC32 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Saos-2 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Alisertib (MLN8237) is a selective Aurora A inhibitor with IC50 of 1.2 nM in a cell-free assay. It has >200-fold higher selectivity for Aurora A than Aurora B. Alisertib induces cell cycle arrest, apoptosis and autophagy. Phase 3. | ||
|---|---|---|---|
| Features | First orally available inhibitor of Aurora A. | ||
| Targets |
|
| In vitro | ||||
| In vitro | MLN8237 shows >200-fold higher selectivity for Aurora A than the structurally related Aurora B with an IC50 of 396.5 nM, and does not have any significant activity against 205 other kinases. [1] MLN8237 (0.5 μM) treatment inhibits the phosphorylation of Aurora A in MM1.S and OPM1 cells, without affecting the Aurora B mediated histone H3 phosphorylation. MLN8237 significantly inhibits cell proliferation in multiple myeloma (MM) cell lines with IC50 values of 0.003-1.71 μM. MLN8237 displays more potent anti-proliferation activity against primary MM cells and MM cell lines in the presence of BM stroma cells, as well as IL-6 and IGF-1 than against MM cells alone. MLN8237 (0.5 μM) induces 2- to 6-fold increase in G2/M phase in primary MM cells and cell lines, as well as significant apoptosis and senescence, involving the up-regulation of p53, p21 and p27, as well as PARP, caspase 3, and caspase 9 cleavage. In addition, MLN8237 shows strong synergistic anti-MM effect with dexamethasone, as well as additive effect with doxorubicin and bortezomib. [2] MLN8237 (0.5 μM) treatment causes the inhibition of colony formation of FLO-1, OE19, and OE33 esophageal adenocarinoma cell lines, and induces a significant increase in the percentage of polyploid cells, and subsequently an increase in the percentage of cells in the sub-G1 phase, which can be further enhanced in combination with cisplatin (2.5 μM), involving the higher induction of TAp73β, PUMA, NOXA, cleaved caspase-3, and cleaved PARP as compared with a single-agent treatment. [3] | |||
|---|---|---|---|---|
| Kinase Assay | Aurora A radioactive Flashplate enzyme assay | |||
| Aurora A radioactive Flashplate enzyme assay is conducted to determine the nature and degree of MLN8237-mediated inhibition in vitro. Recombinant Aurora A is expressed in Sf9 cells and purified with GST affinity chromatography. The peptide substrate for Aurora A is conjugated with biotin (Biotin-GLRRASLG). Aurora A kinase (5 nM) is assayed in 50 mM Hepes (pH 7.5), 10 mM MgCl2, 5 mM DTT, 0.05% Tween 20, 2 μM peptide substrate, 3.3 μCi/mL [γ-33P]ATP at 2 μM, and increasing concentrations of MLN8237 by using Image FlashPlates. | ||||
| Cell Research | Cell lines | MM1.S, MM.1R, LR5, RPMI 8226, DOX40, OPM1, OPM2, INA6, and U266 | ||
| Concentrations | Dissolved in DMSO, final concentrations ~10 μM | |||
| Incubation Time | 24, 48, and 72 hours | |||
| Method | Cells are exposed to various concentrations of MLN8237 for 24, 48, and 72 hours. Cells viability is measured using MTT assay, and cell proliferation is measured using 3[H]-thymidine incorporation. For cell cycle analysis, cells are permeabilized by 70% ethanol at -20 °C, and incubated with 50 μg/mL PI and 20 units/mL RNase-A. DNA content is analyzed by flow cytometry using BDFACS-Canto II and FlowJo software. For the detection of apoptosis and senescence, cells are stained with fluorescein isothiocyanate-annexin V and PI. Apoptotic cells are determined by flow cytometric analysis using BDFACS-Canto II and FlowJo software. | |||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | p-AURKA(T288) / p-EIF4E(S209) / c-Myc phospho-Aurora A / Aurora B H3S10P / H3K27me2 / H3K27me3 / H3K9me2 / H3AcK / H4K16Ac |
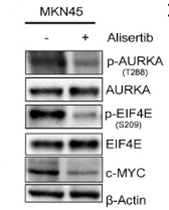
|
28073841 | |
| Growth inhibition assay | Cell viability |
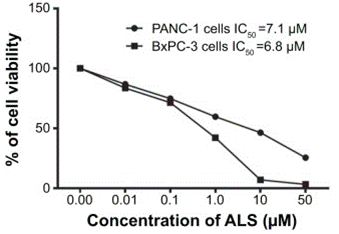
|
25632225 | |
| Immunofluorescence | acetylated α-tubulin / γ-tubulin E-cadherin / β-catenin / vimentin / p-SMAD5 Centrin-2 / tubulin phospho-Aurora A(T288) |
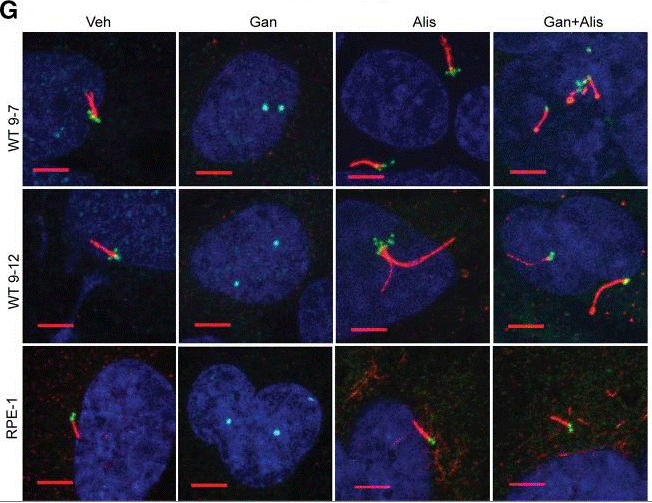
|
29401581 | |
| In Vivo | ||
| In vivo | MLN8237 significantly reduces the tumor burden with tumor growth inhibition (TGI) of 42% and 80% at 15 mg/kg and 30 mg/kg, respectively, and prolongs the survival of mice compared with the control. [2] | |
|---|---|---|
| Animal Research | Animal Models | Severe combined immune-deficient (SCID) mice inoculated subcutaneously with MM1.S cells |
| Dosages | ~30 mg/kg/day | |
| Administration | Orally | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04479306 | Completed | Recurrent Lung Non-Small Cell Carcinoma|Stage IIIB Lung Cancer AJCC v8|Stage IV Lung Cancer AJCC v8|Stage IVA Lung Cancer AJCC v8|Stage IVB Lung Cancer AJCC v8 |
M.D. Anderson Cancer Center |
June 18 2020 | Phase 1 |
| NCT02812056 | Withdrawn | Malignant Neoplasms of Digestive Organs|Malignant Neoplasms of Female Genital Organs|Malignant Neoplasms of Lip Oral Cavity and Pharynx|Malignant Neoplasms of Male Genital Organs |
M.D. Anderson Cancer Center|Millennium Pharmaceuticals Inc. |
September 2016 | Phase 1 |
| NCT02719691 | Completed | Metastatic Breast Cancer|Solid Tumors |
University of Colorado Denver |
May 13 2016 | Phase 1 |
| NCT02367352 | Terminated | Advanced Solid Tumors|Ovarian Cancer|Small Cell Lung Cancer |
Millennium Pharmaceuticals Inc.|Takeda |
March 19 2015 | Phase 1 |
Chemical Information & Solubility
| Molecular Weight | 518.92 | Formula | C27H20ClFN4O4 |
| CAS No. | 1028486-01-2 | SDF | Download Alisertib (MLN8237) SDF |
| Smiles | COC1=C(C(=CC=C1)F)C2=NCC3=CN=C(N=C3C4=C2C=C(C=C4)Cl)NC5=CC(=C(C=C5)C(=O)O)OC | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 50 mg/mL ( (96.35 mM); Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Frequently Asked Questions
Question 1:
What is the suggested formulation of this compound for mouse injection(i.p.)?
Answer:
It can be dissolved in 6% DMSO/50% PEG 300/5% Tween 80/ddH2O at 10 mg/ml as a clear solution.
Tags: buy Alisertib (MLN8237) | Alisertib (MLN8237) supplier | purchase Alisertib (MLN8237) | Alisertib (MLN8237) cost | Alisertib (MLN8237) manufacturer | order Alisertib (MLN8237) | Alisertib (MLN8237) distributor