research use only
Capecitabine DNA/RNA Synthesis inhibitor
Cat.No.S1156
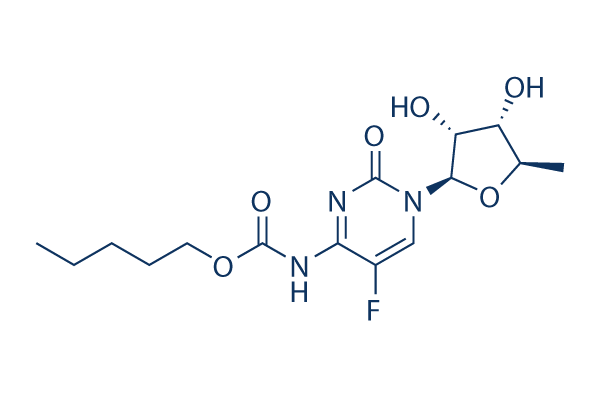
Chemical Structure
Molecular Weight: 359.35
Quality Control
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) SCR7 Favipiravir (T-705) EED226 RK-33 BMH-21 Carmofur Triapine (3-AP) YK-4-279 Tegafur (FT-207) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human RKOp27 cells | Cytotoxic assay | 3 days | Cytotoxicity against human RKOp27 cells after 3 days by MTT assay, IC50=4.33 μM | 20356655 | ||
| RKOp27 | Cytotoxicity assay | Cytotoxicity against human RKOp27 cells by MTT assay, IC50 = 0.00433 μM. | 19345581 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 72 mg/mL
(200.36 mM)
Ethanol : 72 mg/mL Water : 6 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 359.35 | Formula | C15H22FN3O6 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 154361-50-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | RO 09-1978 | Smiles | CCCCCOC(=O)NC1=NC(=O)N(C=C1F)C2C(C(C(O2)C)O)O | ||
Mechanism of Action
| Features |
A tumor-selective fluoropyrimidine carbamate.
|
|---|---|
| Targets/IC50/Ki |
Thymidine phosphorylase
|
| In vitro |
Both LS174T WT and LS174T-c2 cells show significantly greater sensitivity to Capecitabine when cultivated in the same plates as HepG2 hepatoma with IC50 values of 890 and 630 μM in LS174T WT alone and cultivated with HepG2, respectively. In addition, for the LS174T-C2 subline, the IC50 falls from 330 ± 4 down to 89 ± 6 μm when cultivated in the same plates as hepatoma cells. Furthermore, this compound induces apoptosis in a Fas-dependent manner, and shows a 7-fold higher cytotoxicity and markedly stronger apoptotic potential in thymidine phosphorylase (TP)-transfected LS174T-c2 cells.
|
| In vivo |
In the human cancer xenograft models studied, Capecitabine is more effective in a wider dose range and has a broader spectrum of antitumor activity than 5-FU, UFT or its intermediate metabolite 5'-DFUR, which can be correlated with tumor dThdPase levels. This compound inhibits tumor growth and metastatic recurrence after resection of human hepatocellular carcinoma (HCC) in highly metastatic nude mice model which is attributed to the high expression of platelet-derived endothelial cell growth factor in tumors.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06247995 | Not yet recruiting | Breast Cancer |
Novartis Pharmaceuticals|Novartis |
May 16 2024 | Phase 1|Phase 2 |
| NCT06085742 | Recruiting | Breast Cancer |
University of Illinois at Chicago |
November 22 2023 | Phase 2 |
| NCT05929885 | Recruiting | Metastatic Pancreatic Cancer |
National Cancer Centre Singapore|National Medical Research Council (NMRC) Singapore |
August 30 2023 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































