
- Inhibitors
- By product type
- Natural Products
- Inducing Agents
- Peptides
- Antibiotics
- Antibody-drug Conjugates(ADC)
- PROTAC
- Hydrotropic Agents
- Dyes
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
By research - Antibodies
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-DYKDDDDK Tag magnetic beads
- Anti-DYKDDDDK Tag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- Featured Products
- MRTX1133
- Nab-Paclitaxel
- KP-457
- IAG933
- RMC-6236 (Daraxonrasib)
- RMC-7977
- Zoldonrasib (RMC-9805)
- GsMTx4
- Navitoclax (ABT-263)
- TSA (Trichostatin A)
- Y-27632 Dihydrochloride
- SB431542
- SB202190
- MK-2206 Dihydrochloride
- LY294002
- Alisertib (MLN8237)
- XAV-939
- CHIR-99021 (Laduviglusib)
- Bafilomycin A1 (Baf-A1)
- Thiazovivin (TZV)
- CP-673451
- Verteporfin
- DAPT
- Galunisertib (LY2157299)
- MG132
- SBE-β-CD
- Tween 80
- Bavdegalutamide (ARV-110)
- Z-VAD-FMK
- Wnt-C59 (C59)
- IWR-1-endo
- (+)-JQ1
- 3-Deazaneplanocin A (DZNep) Hydrochloride
- RepSox (E-616452)
- Erastin
- Q-VD-Oph
- Puromycin Dihydrochloride
- Cycloheximide
- Telaglenastat (CB-839)
- A-83-01
- Ceralasertib (AZD6738)
- Liproxstatin-1
- Emricasan (IDN-6556)
- PMA (Phorbol 12-myristate 13-acetate)
- Dibutyryl cAMP (Bucladesine) sodium
- Nedisertib (M3814)
- PLX5622
- IKE (Imidazole Ketone Erastin)
- STM2457
- Saruparib (AZD5305)
- New Products
- Contact Us
HCV Protease
Proteases
- All (29)
- HCV Protease Inhibitors (29)
- New HCV Protease Products
| Cat.No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
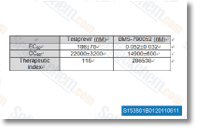
| S1482 | Daclatasvir (BMS-790052) | Daclatasvir (BMS-790052, EBP883) is a highly selective inhibitor of HCV NS5A with EC50 of 9-50 pM, for a broad range of HCV replicon genotypes and the JFH-1 genotype 2a infectious virus in cell culture. Phase 3. |
|
|
| S1538 | Telaprevir | Telaprevir is an HCV NS3-4A serine protease inhibitor with IC50 of 0.35 μM. |
|
|
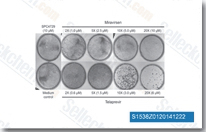
| S1480 | Lomibuvir (VX-222) | Lomibuvir (VX-222, VCH-222) is a novel, potent and selective inhibitor of HCV polymerase with IC50 of 0.94-1.2 μM. This compound is 15.3-fold less effective for mutant M423T, and 108-fold less effective for mutant I482L. Phase 2. |
|
|
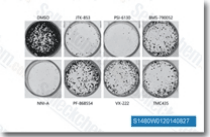
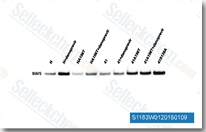
| S1183 | Danoprevir | Danoprevir is a peptidomimetic inhibitor of the NS3/4A protease of hepatitis C virus (HCV) with IC50 of 0.2-3.5 nM, inhibition effect for HCV genotypes 1A/1B/4/5/6 is ~10-fold higher than 2B/3A. Phase 2. |
|
|
| S3733 | Boceprevir | Boceprevir (EBP 520, SCH 503034) is an oral, direct acting hepatitis C virus (HCV) protease inhibitor with Ki value of 14 nM for NS3. It is used in combination with other antiviral agents in the treatment of chronic hepatitis C, genotype 1. | ||
| S5062 | Daclatasvir Dihydrochloride | Daclatasvir is an orally available antiviral agent that inhibits the NS5A region of the hepatitis C virus (HCV) used for the treatment of chronic HCV genotype 1 and 3 infection. | ||
| S7579 | Ledipasvir (GS5885) | Ledipasvir (GS5885) is a HCV NS5A polymerase inhibitor, used for the treatment of hepatitis C virus infection. |
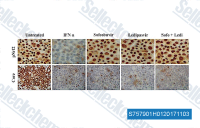
|
|
| S5015 | Simeprevir | Simeprevir is a competitive, reversible, macrocyclic, noncovalent hepatitis C virus (HCV) NS3/4A protease inhibitor that acts directly against the hepatitis C virus. It has a medium inhibitory concentration (IC50) <13 nM for all HCV NS3/4A enzymes(genotypes 1a, 1b, 2, 4, 5, and 6), but has an IC50 value of 37 nM for genotype 3. | ||
| S4935 | Asunaprevir | Asunaprevir is an orally bioavailable inhibitor of the hepatitis C virus enzyme serine protease NS3 that is necessary for protein processing required for viral replication. | ||
| S3728 | Grazoprevir | Grazoprevir anhydrous (MK5172) is a Hepatitis C Virus NS3/4A Protease inhibitor with IC50 values of 7pM, 4pM, and 62pM for HCV genotype 1a, 1B, and 4 respectively. | ||
| S3724 | Velpatasvir | Velpatasvir (GS-5816,VEL) is a second-generation NS5A inhibitor that inhibits hepatitis C viral replication through acting on the crucial "membranous web" that facilitates RNA replication. | ||
| S5403 | Ombitasvir (ABT-267) | Ombitasvir (ABT-267) is an inhibitor of the HCV non-structural protein 5A with antiviral activity. | ||
| S5402 | Dasabuvir (ABT-333) | Dasabuvir (ABT-333) is a non-nucleoside inhibitor of NS5B viral RNA-dependent RNA polymerase that inhibits recombinant NS5B polymerases derived from HCV genotype 1a and 1b clinical isolates with IC50 values between 2.2 and 10.7 nM. It is at least 7,000-fold selective for the inhibition of HCV genotype 1 polymerases over human/mammalian polymerases. | ||
| S5720 | Glecaprevir | Glecaprevir(ABT-493) is a hepatitis C virus (HCV) nonstructural (NS) protein 3/4A protease inhibitor that targets the the viral RNA replication. It displays IC50 values ranging from 3.5 to 11.3 nM for clinical isolates of HCV genotypes 1a, 1b, 2a, 2b, 3a, 4a, 5a, and 6a. | ||
| S5404 | Paritaprevir (ABT-450) | ABT-450 (Paritaprevir) is a nonstructural (NS) protein 3/4A protease inhibitor. | ||
| S5307 | PSI-6206 (GS-331007) | PSI-6206 (RO-2433, GS-331007) is a potent inhibitor of the hepatitis C virus RNA-dependent RNA polymerase, targeting NS5B polymerase. | ||
| E6045New | 2'-C-Methylcytidine | 2'-C-Methylcytidine (NM107) is a potent and selective inhibitor of Chronic hepatitis C virus (HCV) NS5B polymerase with an EC50 of 1.85 μM in wild-type replicon cells. It also effectively inhibits foot-and-mouth disease virus(FMDV) replication with an EC50 of 6.4 μM. Therefore it is a potent antiviral that targets both viral and host factors, making it a promising option for HCV therapy and a potential prophylactic or therapeutic agent against FMDV outbreaks. | ||
| S3395 | Tegobuvir | Tegobuvir (TGV, GS 333126, GS-9190) is a non-nucleoside inhibitor of HCV RNA replication with proven antiviral activity in HCV-infected patients. | ||
| E6002 | Ledipasvir D-tartrate | Ledipasvir D-tartrate (GS-5885 D-tartrate), the D-tartrate salt form of ledipasvir, is an inhibitor of the HCV NS5A, which is essential for the replication of the virus. It exhibits EC50 of 31 pM against GT1a and 4 pM against GT1b replicons. | ||
| S9641 | Pibrentasvir (ABT-530) | Pibrentasvir (ABT-530) is a novel and pan-genotypic hepatitis C virus (HCV) NS5A inhibitor, with this compound exhibiting EC50 values ranging from 1.4 pM to 5.0 pM against HCV replicons containing NS5A from genotypes 1 to 6. | ||
| S5394 | Tizoxanide | Tizoxanide (Desacetyl-nitazoxanide), a thiazolide anti-infective, is active against anaerobic bacteria, protozoa, and a range of viruses in cell culture models, and is currently in phase II clinical development for treating chronic hepatitis C. | ||
| S5652 | Elbasvir | Elbasvir (MK8742) is an NS5A inhibitor, preventing hepatitis C viral RNA replication and virion assembly. Median EC50 values range from 0.2 to 3600 pmol/L, based on genotype. | ||
| E7964 | 2'-C-Methyladenosine | 2’-C-Methyladenosine is an inhibitor of hepatitis C virus (HCV) replication, with an IC50 of 0.3 µM in Huh-7 cells. It is converted intracellularly to its triphosphate form (2’-C-methyladenosine triphosphate), which acts as a chain terminator and directly inhibits the HCV RNA-dependent RNA polymerase (NS5B) with an IC50 of 1.9 µM in enzymatic assays. | ||
| E3597 | Herba taxilli Extract | Herba Taxilli Extract is extracted from Taxillus sutchuenensis, which can be used as inhibitors of HCV NS3 serine protease. | ||
| S2074 | Mecarbinate | Mecarbinate (Dimecarbin) is a chemical intermediate of arbidol hydrochloride. | ||
| S2794 | Sofosbuvir | Sofosbuvir is a HCV NS5B polymerase inhibitor for the treatment of chronic hepatitis C virus (HCV) infection. |
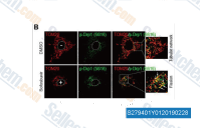
|
|
| S3800 | Lycorine hydrochloride | Lycorine (Galanthidine, Amarylline, Narcissine, Licorine), a natural alkaloid extracted from the Amaryllidaceae plant family, has been reported to exhibit a wide range of physiological effects, including the potential effect against cancer. It is an HCV inhibitor with strong activity. | ||
| E0097 | Mericitabine | Mericitabine (RG 7128, R-7128, PSI 6130 diisobutyrate) is a nucleoside inhibitor of the HCV NS5B polymerase that acts as an RNA chain terminator and prevents elongation of RNA transcripts during replication. | ||
| E1243 | Azvudine | Azvudine (RO-0622) is a novel nucleoside reverse transcriptase inhibitor with antiviral activity against human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV), potently inhibits HIV-1 (EC50 range 0.03 to 6.92 nM) and HIV-2 (EC50s ranging from 0.018 to 0.025 nM). | ||
| S1482 | Daclatasvir (BMS-790052) | Daclatasvir (BMS-790052, EBP883) is a highly selective inhibitor of HCV NS5A with EC50 of 9-50 pM, for a broad range of HCV replicon genotypes and the JFH-1 genotype 2a infectious virus in cell culture. Phase 3. |

|
|
| S1538 | Telaprevir | Telaprevir is an HCV NS3-4A serine protease inhibitor with IC50 of 0.35 μM. |

|
|
| S1480 | Lomibuvir (VX-222) | Lomibuvir (VX-222, VCH-222) is a novel, potent and selective inhibitor of HCV polymerase with IC50 of 0.94-1.2 μM. This compound is 15.3-fold less effective for mutant M423T, and 108-fold less effective for mutant I482L. Phase 2. |

|
|
| S1183 | Danoprevir | Danoprevir is a peptidomimetic inhibitor of the NS3/4A protease of hepatitis C virus (HCV) with IC50 of 0.2-3.5 nM, inhibition effect for HCV genotypes 1A/1B/4/5/6 is ~10-fold higher than 2B/3A. Phase 2. |

|
|
| S3733 | Boceprevir | Boceprevir (EBP 520, SCH 503034) is an oral, direct acting hepatitis C virus (HCV) protease inhibitor with Ki value of 14 nM for NS3. It is used in combination with other antiviral agents in the treatment of chronic hepatitis C, genotype 1. | ||
| S5062 | Daclatasvir Dihydrochloride | Daclatasvir is an orally available antiviral agent that inhibits the NS5A region of the hepatitis C virus (HCV) used for the treatment of chronic HCV genotype 1 and 3 infection. | ||
| S7579 | Ledipasvir (GS5885) | Ledipasvir (GS5885) is a HCV NS5A polymerase inhibitor, used for the treatment of hepatitis C virus infection. |

|
|
| S5015 | Simeprevir | Simeprevir is a competitive, reversible, macrocyclic, noncovalent hepatitis C virus (HCV) NS3/4A protease inhibitor that acts directly against the hepatitis C virus. It has a medium inhibitory concentration (IC50) <13 nM for all HCV NS3/4A enzymes(genotypes 1a, 1b, 2, 4, 5, and 6), but has an IC50 value of 37 nM for genotype 3. | ||
| S4935 | Asunaprevir | Asunaprevir is an orally bioavailable inhibitor of the hepatitis C virus enzyme serine protease NS3 that is necessary for protein processing required for viral replication. | ||
| S3728 | Grazoprevir | Grazoprevir anhydrous (MK5172) is a Hepatitis C Virus NS3/4A Protease inhibitor with IC50 values of 7pM, 4pM, and 62pM for HCV genotype 1a, 1B, and 4 respectively. | ||
| S3724 | Velpatasvir | Velpatasvir (GS-5816,VEL) is a second-generation NS5A inhibitor that inhibits hepatitis C viral replication through acting on the crucial "membranous web" that facilitates RNA replication. | ||
| S5403 | Ombitasvir (ABT-267) | Ombitasvir (ABT-267) is an inhibitor of the HCV non-structural protein 5A with antiviral activity. | ||
| S5402 | Dasabuvir (ABT-333) | Dasabuvir (ABT-333) is a non-nucleoside inhibitor of NS5B viral RNA-dependent RNA polymerase that inhibits recombinant NS5B polymerases derived from HCV genotype 1a and 1b clinical isolates with IC50 values between 2.2 and 10.7 nM. It is at least 7,000-fold selective for the inhibition of HCV genotype 1 polymerases over human/mammalian polymerases. | ||
| S5720 | Glecaprevir | Glecaprevir(ABT-493) is a hepatitis C virus (HCV) nonstructural (NS) protein 3/4A protease inhibitor that targets the the viral RNA replication. It displays IC50 values ranging from 3.5 to 11.3 nM for clinical isolates of HCV genotypes 1a, 1b, 2a, 2b, 3a, 4a, 5a, and 6a. | ||
| S5404 | Paritaprevir (ABT-450) | ABT-450 (Paritaprevir) is a nonstructural (NS) protein 3/4A protease inhibitor. | ||
| S5307 | PSI-6206 (GS-331007) | PSI-6206 (RO-2433, GS-331007) is a potent inhibitor of the hepatitis C virus RNA-dependent RNA polymerase, targeting NS5B polymerase. | ||
| E6045New | 2'-C-Methylcytidine | 2'-C-Methylcytidine (NM107) is a potent and selective inhibitor of Chronic hepatitis C virus (HCV) NS5B polymerase with an EC50 of 1.85 μM in wild-type replicon cells. It also effectively inhibits foot-and-mouth disease virus(FMDV) replication with an EC50 of 6.4 μM. Therefore it is a potent antiviral that targets both viral and host factors, making it a promising option for HCV therapy and a potential prophylactic or therapeutic agent against FMDV outbreaks. | ||
| S3395 | Tegobuvir | Tegobuvir (TGV, GS 333126, GS-9190) is a non-nucleoside inhibitor of HCV RNA replication with proven antiviral activity in HCV-infected patients. | ||
| E6002 | Ledipasvir D-tartrate | Ledipasvir D-tartrate (GS-5885 D-tartrate), the D-tartrate salt form of ledipasvir, is an inhibitor of the HCV NS5A, which is essential for the replication of the virus. It exhibits EC50 of 31 pM against GT1a and 4 pM against GT1b replicons. | ||
| S9641 | Pibrentasvir (ABT-530) | Pibrentasvir (ABT-530) is a novel and pan-genotypic hepatitis C virus (HCV) NS5A inhibitor, with this compound exhibiting EC50 values ranging from 1.4 pM to 5.0 pM against HCV replicons containing NS5A from genotypes 1 to 6. | ||
| S5394 | Tizoxanide | Tizoxanide (Desacetyl-nitazoxanide), a thiazolide anti-infective, is active against anaerobic bacteria, protozoa, and a range of viruses in cell culture models, and is currently in phase II clinical development for treating chronic hepatitis C. | ||
| S5652 | Elbasvir | Elbasvir (MK8742) is an NS5A inhibitor, preventing hepatitis C viral RNA replication and virion assembly. Median EC50 values range from 0.2 to 3600 pmol/L, based on genotype. | ||
| E7964 | 2'-C-Methyladenosine | 2’-C-Methyladenosine is an inhibitor of hepatitis C virus (HCV) replication, with an IC50 of 0.3 µM in Huh-7 cells. It is converted intracellularly to its triphosphate form (2’-C-methyladenosine triphosphate), which acts as a chain terminator and directly inhibits the HCV RNA-dependent RNA polymerase (NS5B) with an IC50 of 1.9 µM in enzymatic assays. | ||
| E3597 | Herba taxilli Extract | Herba Taxilli Extract is extracted from Taxillus sutchuenensis, which can be used as inhibitors of HCV NS3 serine protease. | ||
| S2074 | Mecarbinate | Mecarbinate (Dimecarbin) is a chemical intermediate of arbidol hydrochloride. | ||
| S2794 | Sofosbuvir | Sofosbuvir is a HCV NS5B polymerase inhibitor for the treatment of chronic hepatitis C virus (HCV) infection. |

|
|
| S3800 | Lycorine hydrochloride | Lycorine (Galanthidine, Amarylline, Narcissine, Licorine), a natural alkaloid extracted from the Amaryllidaceae plant family, has been reported to exhibit a wide range of physiological effects, including the potential effect against cancer. It is an HCV inhibitor with strong activity. | ||
| E0097 | Mericitabine | Mericitabine (RG 7128, R-7128, PSI 6130 diisobutyrate) is a nucleoside inhibitor of the HCV NS5B polymerase that acts as an RNA chain terminator and prevents elongation of RNA transcripts during replication. | ||
| E1243 | Azvudine | Azvudine (RO-0622) is a novel nucleoside reverse transcriptase inhibitor with antiviral activity against human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV), potently inhibits HIV-1 (EC50 range 0.03 to 6.92 nM) and HIV-2 (EC50s ranging from 0.018 to 0.025 nM). | ||
| E6045New | 2'-C-Methylcytidine | 2'-C-Methylcytidine (NM107) is a potent and selective inhibitor of Chronic hepatitis C virus (HCV) NS5B polymerase with an EC50 of 1.85 μM in wild-type replicon cells. It also effectively inhibits foot-and-mouth disease virus(FMDV) replication with an EC50 of 6.4 μM. Therefore it is a potent antiviral that targets both viral and host factors, making it a promising option for HCV therapy and a potential prophylactic or therapeutic agent against FMDV outbreaks. |





































