- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
research use only
Telaprevir HCV Protease inhibitor
Telaprevir is an HCV NS3-4A serine protease inhibitor with IC50 of 0.35 μM.
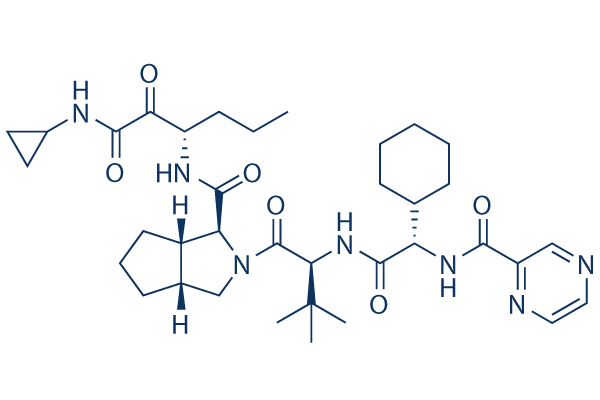
Chemical Structure
Molecular Weight: 679.85
Purity & Quality Control
Batch:
Purity:
99.82%
99.82
Related Products
| Related Products | Lomibuvir (VX-222) Danoprevir Asunaprevir PSI-6206 (GS-331007) | Click to Expand |
|---|---|---|
| Related Compound Libraries | FDA-approved Drug Library Natural Product Library Bioactive Compound Library-I Protease Inhibitor Library Ubiquitination Compound Library | Click to Expand |
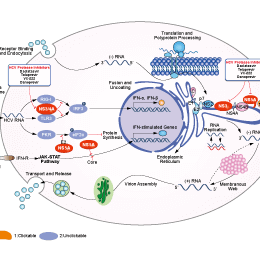
Signaling Pathway
Mechanism of Action
| Features | Telaprevir is a covalent, reversible inhibitor of the NS3-4A protease (unlike BILN 2061which is a noncovalent inhibitor), with a slow-binding and slow-dissociation mechanism. | ||
|---|---|---|---|
| Targets |
|
In vitro |
||||
| In vitro | Telaprevir inhibits the hepatitis C virus NS3-4A serine protease, leading to the block of viral polyprotein processing and subsequently decrease of viral RNA replication, total HCV RNA levels and protein levels in the Con1 (genotype 1b) subgenomic HCV replicon cells in a time- and dose-dependent manner. This compound displays a significant time-dependent increase in inhibitory effect on the replication of HCV RNA with IC50 values of 0.574 μM, 0.488 μM, 0.210 μM and 0.139 μM for 24, 48, 72 and 120 hours incubation, respectively. It displays an average IC50 of 0.354 μM and an average IC90 of 0.830 μM, respectively, from three independent experiments using the 48 hours incubation. This chemical has no significant cytotoxicity to HCV replicon cells, parental Huh-7 and HepG2 cells after 48 hours incubation. It (17.5 μM) completely eradicates HCV RNA from replicon cells after 13 days incubation without rebound after this compound is withdrawn. It displays an additive to moderate synergistic effect on reduction of HCV RNA replication and suppression of resistance mutations without significant increase in cytotoxicity when in combination with IFN-α, compared to treatment with each agent alone. [1] | |||
|---|---|---|---|---|
| Kinase Assay | Determination of anti-HCV activity | |||
| Stable Huh-7 cells containing the self-replicating, subgenomic HCV replicon, which is identical in sequence to the I377neo/NS3-3'/wt replicon are used for anti-HCV assays. Replicon cells are incubated at 37 °C for the indicated period of time with this compound serially diluted in DMEM plus 2% FBS and 0.5% dimethyl sulfoxide (DMSO). Total cellular RNA is extracted using an RNeasy-96 kit, and the copy number of HCV RNA is determined using a quantitative RTPCR (QRT-PCR) assay for the assessment of 50% inhibitory concentration (IC50 | ||||
| Cell Research | Cell lines | Huh-7, HepG2, and peripheral blood mononuclear cells (PBMC) | ||
| Concentrations | Dissolved in DMSO, final concentration ~1 mM | |||
| Incubation Time | 48 hours | |||
| Method | Cells are incubated with various concentrations of Telaprevir for 48 hours. Cell viability is determined by using a tetrazolium (MTS)-based cell viability assay. | |||
In Vivo |
||
| In vivo | Oral administration of Telaprevir reduces HCV protease-dependent cleavage and subsequent secretion of SEAP from the liver into the blood in the mice model to 18.7% and 18.4% at dosage of 10 and 25 mg/kg, respectively. [2] Administration of this compound at 200 mg/kg for 1 week results in a 1.9 log reduction of HCV RNA in genotype 1b HCV-infected human hepatocyte chimeric mice, and when treatment in combination with MK-0608 (50 mg/kg) for 4 weeks, viruses are eliminated from mice. [3] | |
|---|---|---|
| Animal Research | Animal Models | SCID mice injected with recombinant adenovirus (Ad-WT-HCVpro-SEAP or Ad-MT-HCVpro-SEAP) |
| Dosages | ~300 mg/kg | |
| Administration | Oral gavage | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02881034 | Completed | Hepatitis C |
Hospices Civils de Lyon |
February 2014 | -- |
| NCT01994486 | Completed | Hepatitis C Chronic |
University of Florida|Vertex Pharmaceuticals Incorporated |
December 2013 | Phase 2 |
| NCT01980290 | Completed | Hepatitis Chronic |
Janssen Pharmaceutica N.V. Belgium |
May 2013 | -- |
| NCT01841502 | Terminated | Hepatitis C Infection|Depression |
Radboud University Medical Center|Janssen LP |
May 2013 | Phase 2 |
| NCT01766167 | Completed | Chronic Hepatitis C |
Mitsubishi Tanabe Pharma Corporation |
February 2013 | Phase 1 |
References |
|
Chemical Information
| Molecular Weight | 679.85 | Formula | C36H53N7O6 |
| CAS No. | 402957-28-2 | SDF | Download SDF |
| Synonyms | LY-570310, MP-424,VX-950 | ||
| Smiles | CCCC(C(=O)C(=O)NC1CC1)NC(=O)C2C3CCCC3CN2C(=O)C(C(C)(C)C)NC(=O)C(C4CCCCC4)NC(=O)C5=NC=CN=C5 | ||
Storage and Stability
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 136 mg/mL ( (200.04 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field